Rare Hemophilia Factors Market Size, Share, Growth and Forecast (2026 - 2036)
The Rare Hemophilia Factors Market is segmented by Factor Type (Factor I, Factor II, Factor V, Factor VII, Factor X, Factor XI, and Factor XIII), Application (Fresh Frozen Plasma, Factor Concentrates, Cryoprecipitate, Plasma-Derived Products, and Recombinant Therapies), Treatment Approach (On-Demand Treatment, Prophylaxis, Perioperative Bleeding Management, Emergency Bleeding Control, and Long-Term Monitoring), End User (Hospitals, Hemophilia Treatment Centers, Specialty Clinics, Blood Banks, and Research Institutes), Sales Channel (Direct Institutional Sales, Specialty Pharmacy Distribution, Hospital Pharmacy, Government Supply Programs, and Online Medical Supply), and Region. Forecast for 2026 to 2036.
Fact.MR analysis indicates that the rare hemophilia factors market is moving through a treatment-access and factor-specific care phase. Hematologists, hospitals, and treatment centers are evaluating therapies through availability, bleeding control, safety profile, and patient monitoring needs. Demand is gaining depth where rare factor deficiencies need faster diagnosis, better factor replacement access, and structured prophylaxis support.
Rare Hemophilia Factors Market Forecast and Outlook By Fact.MR
- In 2025, the rare hemophilia factors market was valued at USD 345.4 million.
- Based on revised Fact.MR analysis, demand for rare hemophilia factors is estimated to grow to USD 366.1 million in 2026 and USD 693.7 million by 2036.
- Fact.MR projects a CAGR of 6.6% during the forecast period.
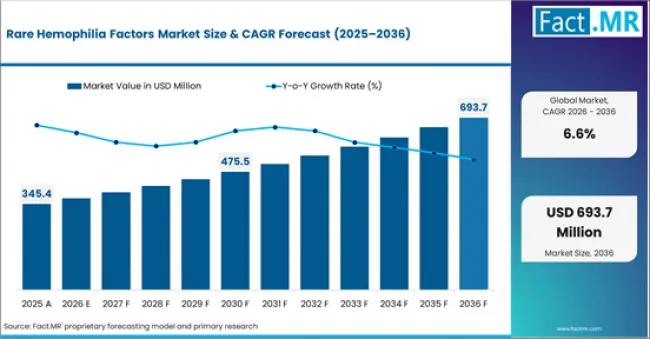
| Metric | Value |
|---|---|
| Estimated Value in 2026 | USD 366.1 million |
| Forecast Value in 2036 | USD 693.7 million |
| Forecast CAGR (2026 to 2036) | 6.6% |
Summary of Rare Hemophilia Factors Market
- Market Definition
- The market includes treatment products used to manage rare coagulation factor deficiencies across on-demand bleeding control, prophylaxis, and perioperative care.
- Demand Drivers
- Hemophilia treatment centers are improving rare factor deficiency diagnosis through structured bleeding disorder evaluation and patient registries.
- Hospitals need available plasma and factor therapies because severe bleeding episodes require fast replacement of missing clotting factors.
- Specialty pharmacies support long-term therapy access where patients need recurring factor supply and clinical monitoring.
- Key Segments Analyzed
- By Factor Type: Factor VII is expected to hold approximately 26.0% share in 2026, supported by available therapeutic options and use in bleeding management.
- By Application: Fresh frozen plasma leads in 2026 with 40.0% share, as it remains widely used where specific factor concentrates are limited.
- By Treatment Approach: On-demand treatment accounts for around 45.0% share in 2026, driven by acute bleeding control needs.
- By End User: Hemophilia treatment centers hold nearly 38.0% share in 2026, due to specialist-led diagnosis and long-term patient monitoring.
- By Sales Channel: Specialty pharmacy distribution is projected to lead with 34.0% share in 2026, as rare disease therapies require controlled access and patient support.
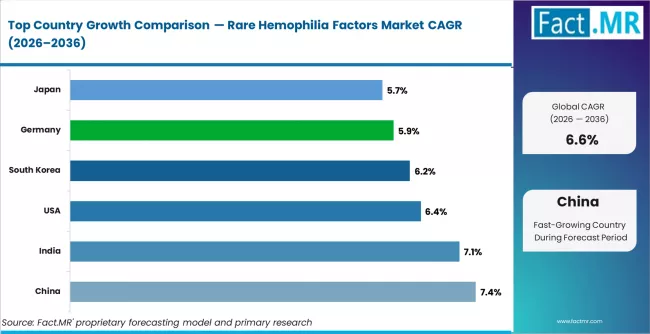
- By Geography: China leads growth through 2036 at 7.4% CAGR, supported by wider diagnosis and improving specialty care access.
- Analyst Opinion at Fact.MR
- Shambhu Nath Jha, Senior Analyst at Fact.MR, opines, “Rare hemophilia factor demand is no longer measured only by bleeding episode treatment. The stronger shift is toward accurate factor diagnosis, dependable therapy access, and care models that reduce emergency dependence. Suppliers that can support specialist centers, patient monitoring, and reliable factor availability will hold a better position than companies competing only on product presence.”
- Strategic Implications
- Manufacturers should prioritize therapy access programs because rare factor deficiencies need dependable supply despite small patient pools.
- Treatment centers need stronger diagnostic pathways to match patients with the correct factor replacement.
- Specialty pharmacies should improve patient support and cold-chain reliability because therapy continuity affects bleeding control.
- Methodology
- Market sizing uses diagnosed rare factor deficiency prevalence, treatment utilization, product availability, prophylaxis adoption, and specialty pharmacy demand.
- Analysis includes CDC resources, WFH guidance, FDA orphan drug information, company therapy data, and hospital treatment references.
- Forecasts account for diagnosis expansion, therapy access programs, factor concentrate availability, plasma product use, and regional hematology infrastructure.
The market is projected to generate USD 327.6 million in absolute opportunity between 2026 and 2036. Growth is supported by wider bleeding disorder diagnosis, better availability of factor concentrates, prophylaxis adoption, and hospital-based management of severe bleeding episodes. CDC resources identify hemophilia as a bleeding disorder requiring treatment and monitoring, which supports clinical demand for replacement therapy. World Federation of Hemophilia guidance also reinforces structured diagnosis and care across bleeding disorders [3].
Japan is projected to grow at 5.7% CAGR through 2036, supported by genetic research and established specialty care. The United States records 6.4%, with demand anchored in reimbursement support, treatment centers, and new therapy approvals. South Korea advances at 6.2%, backed by rising awareness and improved treatment access. Germany grows at 5.9%, supported by structured hemophilia care and specialty therapy use. China posts 7.4%, helped by wider diagnosis and hospital access. India advances at 7.1%, supported by expanding hematology care and plasma therapy availability.
Segmental Analysis
Rare Hemophilia Factors Market Analysis by Factor Type
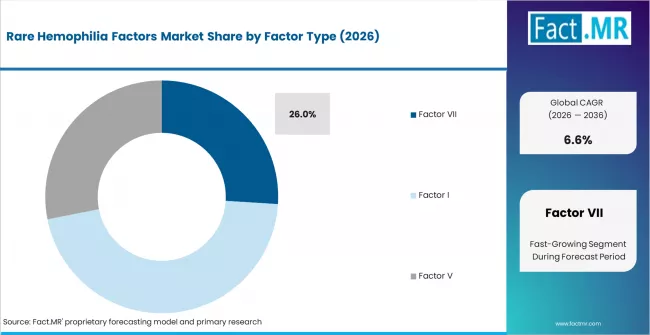
Factor VII is expected to hold 26.0% share in 2026 as treatment options are more established than several other rare factor deficiencies. Hematologists use Factor VII replacement or bypassing therapy where patients face bleeding risk linked to Factor VII deficiency. Fact.MR identifies Factor VII as an important type due to available therapeutic choices such as recombinant factor VIIa. FDA orphan drug resources support the wider context of therapy development for rare conditions where small patient populations require dedicated pathways. Factor I and Factor XIII therapies also gain attention, but Factor VII retains a stronger commercial base. Delayed factor-specific diagnosis can lead to inappropriate treatment and higher bleeding risk.
- Therapy Availability: Factor VII benefits from recognized therapeutic options and specialist familiarity.
- Diagnosis Fit: Correct factor testing helps clinicians separate Factor VII deficiency from other bleeding disorders.
- Bleeding Control: Factor-specific treatment supports better management during surgery and acute bleeding episodes.
Rare Hemophilia Factors Market Analysis by Application
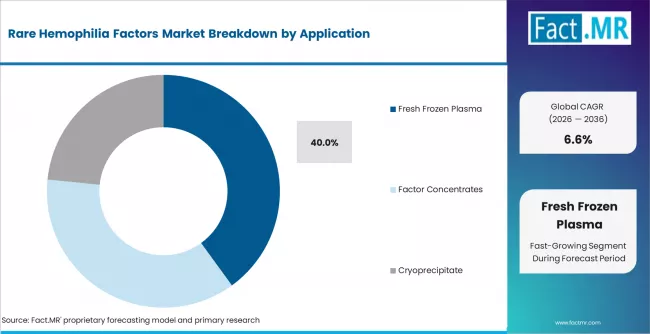
Fresh frozen plasma remains widely used because it contains multiple coagulation factors and can support patients where specific concentrates are not accessible. Fresh frozen plasma accounts for 40.0% share in 2026. Hospitals often use it to manage bleeding episodes or prevent bleeding before urgent procedures when rare factor concentrate availability is limited. Fact.MR states that fresh frozen plasma is widely used in rare hemophilia deficits due to restricted factor concentrate availability. AABB resources also support transfusion medicine practices and blood product quality management [5]. Factor concentrates are gaining use where access and diagnosis improve. Fresh frozen plasma stays important because it remains practical in emergency and resource-limited treatment environments.
- Broad Factor Coverage: Fresh frozen plasma provides multiple coagulation factors when exact concentrate access is limited.
- Urgent Procedure Use: Hospitals use plasma support before invasive procedures when bleeding risk is high.
- Blood Bank Link: Availability depends on transfusion service readiness and safe plasma handling.
Rare Hemophilia Factors Market Analysis by Treatment Approach
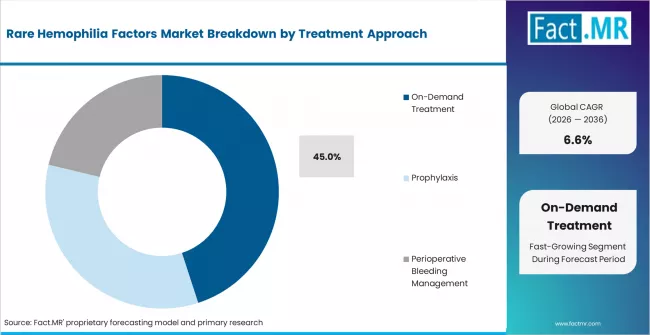
Acute bleeding control keeps on-demand treatment ahead because many rare factor deficiency cases are treated when bleeding occurs. This approach is estimated to account for 45.0% share in 2026. Patients with mild or moderate deficiencies may not require continuous prophylaxis, but they need fast treatment during trauma, surgery, dental procedures, or spontaneous bleeding. CDC resources describe hemophilia care through treatment and prevention of bleeding complications, which supports both on-demand and preventive care needs. Prophylaxis is growing for severe cases, yet on-demand use remains dominant because diagnosis timing and therapy access vary widely. Weak emergency treatment protocols can increase complication risk.
- Acute Response: On-demand therapy supports fast bleeding control when symptoms or injuries occur.
- Procedure Planning: Patients may need factor replacement before surgery or dental intervention.
- Access Variation: On-demand use stays high where prophylaxis coverage is limited or diagnosis happens late.
Rare Hemophilia Factors Market Analysis by End User
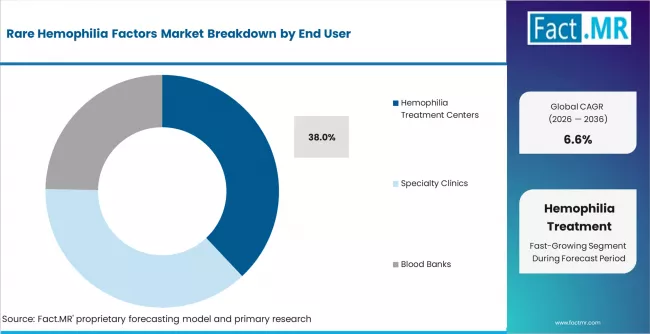
Hemophilia treatment centers hold a strong position because rare factor deficiencies need specialist diagnosis, laboratory interpretation, therapy planning, and patient monitoring. Treatment centers are expected to hold 38.0% share in 2026. These centers coordinate hematologists, nurses, pharmacists, laboratory staff, and genetic counseling resources. WFH guidance emphasizes comprehensive care and specialized treatment structures for bleeding disorders. Hospitals remain important for emergency bleeding and surgery, while blood banks support plasma availability. Hemophilia treatment centers lead because they manage long-term care pathways rather than only single bleeding events. Weak referral pathways can delay diagnosis and reduce therapy access.
- Specialist Care: Treatment centers support factor testing, therapy selection, and follow-up monitoring.
- Patient Registry: Structured centers improve data capture and continuity of care.
- Therapy Coordination: Multidisciplinary care helps align treatment with bleeding severity and procedure risk.
Rare Hemophilia Factors Market Analysis by Sales Channel
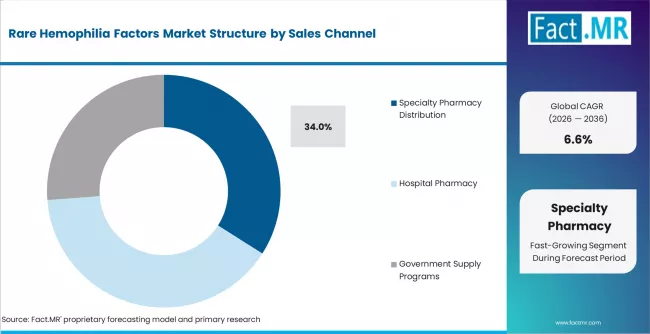
Specialty pharmacy distribution leads because rare hemophilia factor therapies require controlled handling, patient support, reimbursement navigation, and reliable supply coordination. Specialty pharmacy distribution is expected to account for 34.0% share in 2026. These channels support home delivery, patient education, and therapy adherence for patients who need recurring factor access. HRSA hemophilia program resources highlight the importance of care networks and services for bleeding disorder populations. Hospital pharmacies remain important for acute and inpatient therapy, while government programs support access in selected countries. Weak distribution can create treatment gaps during bleeding episodes or planned procedures.
- Access Management: Specialty pharmacies help patients obtain rare factor therapies through controlled supply pathways.
- Patient Support: Education and refill coordination reduce therapy interruption risk.
- Cold-Chain Control: Proper storage and delivery are essential for plasma-derived and recombinant products.
Drivers, Restraints, and Opportunities
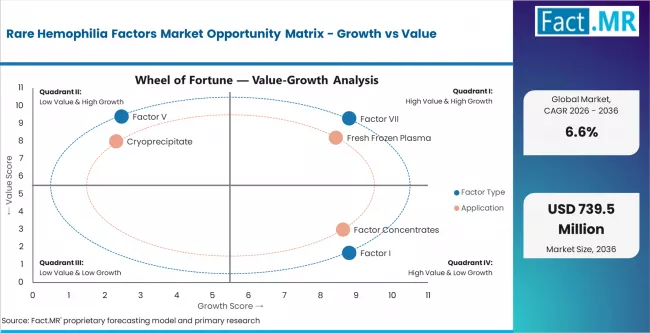
Rare hemophilia factor demand is rising as diagnosis improves and more patients gain access to specialist care. Better awareness of rare coagulation disorders helps clinicians identify deficiencies beyond hemophilia A and B. WFH care guidance supports structured treatment, patient education, and factor access for bleeding disorders. Growth improves where hospitals and treatment centers connect laboratory diagnosis with factor-specific therapy. Fresh frozen plasma remains important, but factor concentrates and recombinant therapies create stronger long-term management options where access is available.
Small patient populations and high therapy cost restrain faster growth. Rare factor deficiencies are difficult to diagnose because symptoms may overlap with other bleeding disorders. Limited availability of specific factor concentrates keeps many patients dependent on plasma products. Reimbursement gaps and cold-chain needs can restrict access in lower-income markets. Clinical evidence can also be limited because patient numbers are small. Suppliers must prove patient support, safety, and reliable availability before wider use expands.
- Factor Concentrate Access: Improved availability can reduce dependence on broad plasma-based treatment.
- Prophylaxis Expansion: Severe rare factor deficiency cases create opportunities for preventive therapy models.
- Diagnostic Pathway Growth: Wider coagulation testing can identify patients who currently remain untreated or misclassified.
Regional Analysis
The rare hemophilia factors market is assessed across North America, Europe, Asia Pacific, Latin America, and Middle East and Africa, covering 40+ countries with demand profiles shaped by diagnosis access, treatment centers, reimbursement systems, plasma supply, specialty pharmacy networks, and rare disease policy support.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 7.4% |
| India | 7.1% |
| USA | 6.4% |
| South Korea | 6.2% |
| Germany | 5.9% |
| Japan | 5.7% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research
Asia Pacific Rare Hemophilia Factors Market Analysis
Asia Pacific records the strongest growth because diagnosis access, hospital hematology services, and rare disease awareness are improving together. China and India are expanding hematology care across urban hospitals, while Japan and South Korea maintain more structured specialty care. Fact.MR notes that Japan is expected to develop at 5.7% CAGR over the forecast period, supported by genetic research and awareness of bleeding disorders. WFH global programs support better diagnosis and treatment access across bleeding disorder communities. The region still includes major access gaps, especially for specific factor concentrates. Growth depends on referral pathways, plasma product availability, reimbursement, and specialist training.
- China: China leads the country outlook as wider diagnosis and improving specialty care access strengthen rare hemophilia factor demand. The country is projected to grow at 7.4% CAGR by 2036. Large hospitals are expanding hematology services and coagulation testing. Plasma products remain important, while factor concentrates gain use in advanced treatment centers. Physicians weigh therapy availability, bleeding severity, and patient affordability before treatment selection.
- India: Expanding hematology care and plasma therapy availability support rare hemophilia factor treatment in India. Urban hospitals are improving access to coagulation testing and specialist consultation. India is forecast to grow at 7.1% CAGR from 2026 to 2036. Cost sensitivity remains important, but severe cases require dependable replacement therapy. Treatment access improves when patient support groups and hospitals connect diagnosis with affordable therapy pathways. [2]
- South Korea: Rising awareness and better treatment access support rare hemophilia factor demand in South Korea. Specialty centers and reimbursement systems improve continuity for diagnosed patients. South Korea is projected to post 6.2% CAGR through 2036. Hematologists focus on correct factor identification and therapy planning. Suppliers with reliable product access and education support can gain stronger clinical acceptance.
- Japan: Genetic research and established specialty care support rare hemophilia factor demand in Japan. Hospitals focus on diagnosis, long-term monitoring, and safe therapy use for rare bleeding disorders. Japan is projected to grow at 5.7% CAGR over the assessment period. Growth is steady because care infrastructure is already structured. Suppliers with strong clinical evidence and stable factor availability can maintain demand through specialist channels.
North America Rare Hemophilia Factors Market Analysis
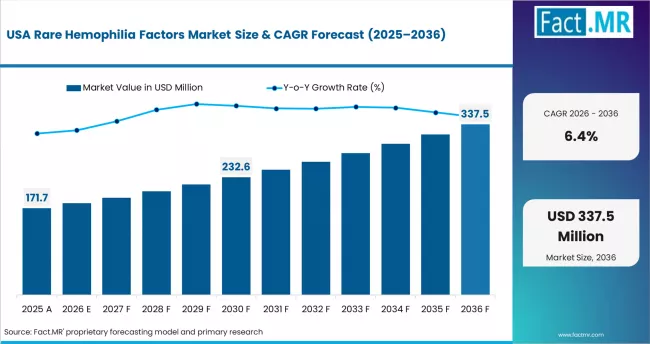
North America remains a high-value region because specialized treatment centers, reimbursement support, and rare disease programs improve access to therapy. The United States anchors regional demand through hemophilia treatment centers, specialty pharmacies, and FDA-approved factor therapies. Fact.MR notes that U.S. demand benefits from reimbursement policies and FDA approvals of coagulation factor concentrates. CDC hemophilia resources support ongoing care, complication prevention, and treatment education. The region has stronger access to factor concentrates than many markets, but high therapy cost remains a challenge. Suppliers must support patient assistance, payer documentation, and treatment continuity.
- USA: The United States has a strong base of hemophilia treatment centers, specialty pharmacies, and rare disease care programs. Demand is supported by diagnosis access, therapy availability, and reimbursement pathways. The country is projected to record 6.4% CAGR by 2036. Factor concentrates gain share where patients receive specialist-led care. Payers and clinicians examine safety, dosing, and long-term bleeding control before expanding therapy use.
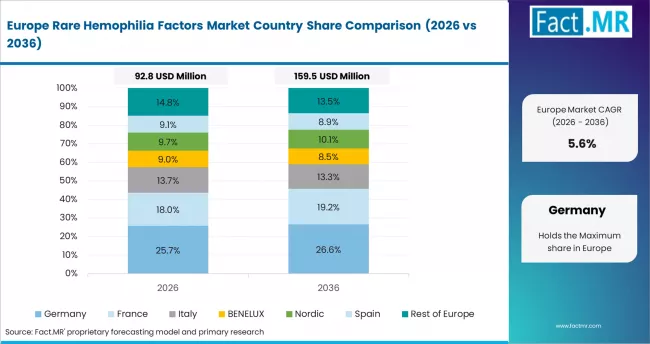
Europe Rare Hemophilia Factors Market Analysis
Europe shows steady demand because specialty care networks, plasma product oversight, and rare disease policies support diagnosed patients. Germany leads regional growth through structured hematology care and access to advanced therapies. European Medicines Agency rare disease and orphan medicine pathways support therapy development for small patient populations. Hospitals and treatment centers emphasize factor-specific diagnosis, safety monitoring, and therapy continuity. Demand growth is moderate because many markets already have established care systems. Suppliers compete through evidence, supply reliability, and patient support programs.
- Germany: Germany benefits from structured hemophilia care, specialty therapy use, and strong hospital networks. Demand is linked to accurate diagnosis, perioperative bleeding management, and long-term monitoring. Germany is forecast to grow at 5.9% CAGR through 2036. Treatment centers tend to favor suppliers with consistent factor availability and clinical support. Reimbursement and specialist confidence influence adoption more than broad product claims.
Fact.MR analysis of regional demand covers Asia Pacific, North America, Europe, Latin America, and Middle East and Africa. The study assesses diagnosis access, specialty care networks, blood product systems, payer coverage, rare disease policies, and supplier support across major markets.
Competitive Aligners for Market Players
The rare hemophilia factors market is concentrated because plasma-derived and recombinant therapies require high manufacturing standards, regulatory strength, and specialist channel access. Large biopharmaceutical companies hold advantages because factor therapy development, safety monitoring, and specialty distribution create entry barriers. Smaller firms often compete through niche rare disease programs, gene therapy research, or region-specific access partnerships.
Competitive advantage depends on factor-specific portfolio depth, product safety, half-life profile, patient support, and distribution reliability. CSL Behring, Novo Nordisk, Pfizer, Takeda, BioMarin, Bayer, Amgen, Bio Products Laboratory, and Alexion represent supplier groups where clinical trust and therapy continuity matter. Treatment centers are less likely to shift therapy when patient stability and dosing familiarity are established. [4]
The market is divided between plasma-based products used for broader access and specialized factor concentrates or recombinant therapies used in diagnosed patients. Suppliers that combine reliable product availability with patient assistance and specialist education can protect institutional demand. Regional access programs can gain importance where diagnosis improves but therapy affordability remains limited.
Key Players
- Amgen Inc.
- BioMarin Pharmaceutical Inc.
- Bio Products Laboratory Ltd.
- Biogen Inc.
- CSL Behring
- Baxter International Inc.
- Bayer AG
- Novo Nordisk A/S
- Pfizer Inc.
- Takeda Pharmaceutical Company Limited
- Alexion Pharmaceuticals, Inc.
- Shire plc
- ICON plc
Bibliography
- [1] Centers for Disease Control and Prevention. (2024). Hemophilia. CDC.
- [2] National Heart, Lung, and Blood Institute. (2024). Hemophilia. NHLBI.
- [3] World Federation of Hemophilia. (2024). Guidelines for the Management of Hemophilia. WFH.
- [4] National Organization for Rare Disorders. (2024). Rare Coagulation Factor Deficiencies. NORD.
- [5] AABB. (2024). Blood Transfusion and Patient Blood Management Resources. AABB.
This Report Addresses
- Strategic intelligence on rare hemophilia factor demand across factor deficiencies, plasma products, factor concentrates, prophylaxis, and emergency bleeding control globally.
- Market forecast from USD 366.1 million in 2026 to USD 693.7 million by 2036 at a CAGR of 6.60%.
- Growth opportunity mapping across China diagnosis expansion, India hematology care growth, U.S. reimbursement support, South Korea treatment access, and Japan genetic research.
- Segment analysis by factor type, application, treatment approach, end user, sales channel, and region.
- Regional outlook covering Asia Pacific diagnosis growth, North America specialty therapy access, and Europe structured rare disease care.
- Competitive analysis of Amgen, BioMarin, Bio Products Laboratory, Biogen, CSL Behring, Baxter International, Bayer, Novo Nordisk, Pfizer, Takeda, Alexion Pharmaceuticals, and Shire.
- Therapy adoption analysis covering factor-specific diagnosis, plasma access, prophylaxis, specialty pharmacy support, reimbursement, and patient monitoring.
- Report delivered with market sizing, segment outlook, regional analysis, company profiling, and forecast assumptions.
Rare Hemophilia Factors Market Definition
The market covers therapies and replacement products used to manage rare hemophilia factor deficiencies involving Factor I, Factor II, Factor V, Factor VII, Factor X, Factor XI, and Factor XIII. It includes fresh frozen plasma, cryoprecipitate, factor concentrates, plasma-derived products, and recombinant therapies used to control bleeding, prevent surgical bleeding, and support long-term patient management.
Rare Hemophilia Factors Market Inclusions
The study includes global and regional forecasts from 2026 to 2036. It covers rare coagulation factor replacement products used in hospitals, hemophilia treatment centers, specialty clinics, blood banks, and research institutions. It includes on-demand therapy, prophylaxis, perioperative bleeding control, emergency bleeding management, and follow-up care supported by hematology specialists.
Rare Hemophilia Factors Market Exclusions
The scope excludes common hemophilia A and hemophilia B therapy when not linked to rare factor deficiencies. It also excludes general anemia drugs, platelet products used outside coagulation factor deficiency care, anticoagulant reversal agents, and blood banking services without rare hemophilia factor treatment use. The focus stays on therapies used for rare coagulation factor deficiencies.
Rare Hemophilia Factors Market Research Methodology
- Primary Research
- Interviews with hematologists, hemophilia treatment center directors, hospital pharmacy heads, transfusion medicine specialists, rare disease program managers, and specialty pharmacy distributors across major consuming regions.
- Desk Research
- Uses bleeding disorder treatment guidance, rare disease resources, regulatory approvals, company therapy information, and blood product safety documentation. [1]
- Market-Sizing and Forecasting
- Hybrid model using diagnosed patient pools, treatment frequency, factor product availability, prophylaxis adoption, hospital therapy use, and average selling prices by therapy format.
- Data Validation and Update Cycle
- Forecasts were validated through specialty pharmacy inputs, hospital treatment patterns, product availability, therapy access programs, and regional rare disease care indicators.
Scope of Report
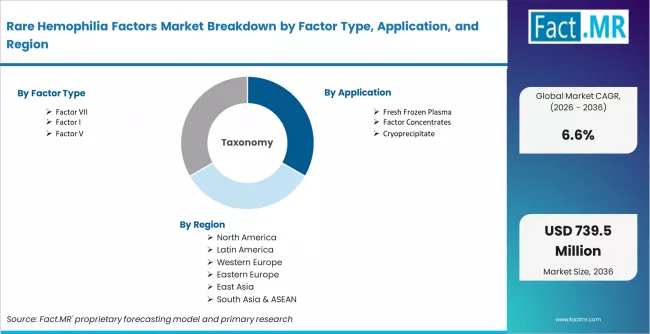
| Attribute | Details |
|---|---|
| Quantitative Units | USD 366.1 million (2026) to USD 693.7 million (2036), at a CAGR of 6.6% |
| Market Definition | Therapies and replacement products used to manage rare coagulation factor deficiencies involving Factor I, Factor II, Factor V, Factor VII, Factor X, Factor XI, and Factor XIII |
| Forecast Period | 2026 to 2036 |
| Historical Reference | 2025 |
| Factor Type Covered | Factor I, Factor II, Factor V, Factor VII, Factor X, Factor XI, Factor XIII |
| Application Covered | Fresh frozen plasma, factor concentrates, cryoprecipitate, plasma-derived products, recombinant therapies |
| Treatment Approach Covered | On-demand treatment, prophylaxis, perioperative bleeding management, emergency bleeding control, long-term monitoring |
| End User Covered | Hospitals, hemophilia treatment centers, specialty clinics, blood banks, research institutes |
| Sales Channel Covered | Direct institutional sales, specialty pharmacy distribution, hospital pharmacy, government supply programs, online medical supply |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Canada, Mexico, Germany, UK, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, ASEAN, Australia and New Zealand, Brazil, Argentina, Chile, Saudi Arabia, GCC, Turkey, South Africa, and Rest of MEA |
| Key Companies Profiled | Amgen, BioMarin, Bio Products Laboratory, Biogen, CSL Behring, Baxter International, Bayer, Novo Nordisk, Pfizer, Takeda, Alexion Pharmaceuticals, Shire, ICON |
| Approach | Hybrid top-down and bottom-up model using diagnosed rare factor deficiency pools, therapy utilization, product availability, prophylaxis adoption, specialty distribution patterns, and primary interviews with hematology care leaders |
Rare Hemophilia Factors Market by Segments
-
By Factor Type:
- Factor I
- Factor II
- Factor V
- Factor VII
- Factor X
- Factor XI
- Factor XIII
-
By Application:
- Fresh Frozen Plasma
- Factor Concentrates
- Cryoprecipitate
- Plasma-Derived Products
- Recombinant Therapies
-
By Treatment Approach:
- On-Demand Treatment
- Prophylaxis
- Perioperative Bleeding Management
- Emergency Bleeding Control
- Long-Term Monitoring
-
By End User:
- Hospitals
- Hemophilia Treatment Centers
- Specialty Clinics
- Blood Banks
- Research Institutes
-
By Sales Channel:
- Direct Institutional Sales
- Specialty Pharmacy Distribution
- Hospital Pharmacy
- Government Supply Programs
- Online Medical Supply
-
By Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Western Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Rest of Middle East and Africa
- North America
- Frequently Asked Questions -
How large is the global rare hemophilia factors market in 2025?
The global rare hemophilia factors market was valued at USD 345.4 million in 2025.
What will the market size be in 2026?
Based on revised Fact.MR analysis, demand for rare hemophilia factors is estimated to grow to USD 366.1 million in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 693.7 million by 2036, generating USD 327.6 million in absolute dollar opportunity.
What is the expected CAGR from 2026 to 2036?
Fact.MR projects a CAGR of 6.60% for the global rare hemophilia factors market during the forecast period.
Which factor type is poised to lead the market?
Factor VII leads with approximately 26.0% share in 2026, supported by available therapeutic options and specialist use.
Which application holds the highest share?
Fresh frozen plasma leads with 40.0% share in 2026 because it remains widely used where specific factor concentrates are limited.
Which treatment approach accounts for the largest demand?
On-demand treatment accounts for around 45.0% share in 2026 due to acute bleeding control needs.
Which end user leads the market?
Hemophilia treatment centers hold nearly 38.0% share in 2026 because rare factor deficiencies need specialist diagnosis and monitoring.
Which sales channel leads demand?
Specialty pharmacy distribution holds around 34.0% share in 2026 because rare disease therapies need controlled access and patient support.
Which country shows the fastest growth?
China leads at 7.4% CAGR through 2036, supported by wider diagnosis and improving specialty care access.
What is driving demand for rare hemophilia factors?
Demand is driven by improved diagnosis, factor concentrate access, prophylaxis adoption, plasma therapy use, and specialist treatment centers.
What is the key challenge in this market?
Small patient pools, high therapy cost, limited concentrate availability, and reimbursement gaps restrict faster adoption across several markets.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Thoracic Drainage Systems
- Pleural Drainage Catheters
- Secured Needles
- Thoracic Drainage Kits
- Thoracic Drainage Systems
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Thoracic Surgeries & Pulmonology
- Cardiac Surgeries
- General Intensive Care & Emergency Medicine
- Infectious Diseases
- Others
- Thoracic Surgeries & Pulmonology
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Others
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Usability
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Usability, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Usability, 2026 to 2036
- Disposable Devices
- Reusable Devices
- Disposable Devices
- Y to o to Y Growth Trend Analysis By Usability, 2021 to 2025
- Absolute $ Opportunity Analysis By Usability, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drainage Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drainage Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drainage Type, 2026 to 2036
- Dry Suction Systems
- Heimlich Valves
- Wet Suction Systems
- Dry Suction Systems
- Y to o to Y Growth Trend Analysis By Drainage Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Drainage Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- By End User
- By Usability
- By Drainage Type
- Competition Analysis
- Competition Deep Dive
- Medtronic plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Teleflex Incorporated
- Becton Dickinson (BD)
- Getinge AB
- Smiths Group plc
- Cook Group Incorporated
- Merit Medical Systems
- Vygon SA
- Medela AG
- Medtronic plc
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Usability, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Drainage Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Usability, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Drainage Type, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Usability, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Drainage Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Usability, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Drainage Type, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Usability, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Drainage Type, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Usability, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Drainage Type, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Usability, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Drainage Type, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Usability, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Drainage Type, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value Share and BPS Analysis by Usability, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Usability, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Usability
- Figure 15: Global Market Value Share and BPS Analysis by Drainage Type, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Drainage Type, 2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Drainage Type
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Product
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by End User
- Figure 38: North America Market Value Share and BPS Analysis by Usability, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Usability, 2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by Usability
- Figure 41: North America Market Value Share and BPS Analysis by Drainage Type, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Drainage Type, 2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Drainage Type
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Product
- Figure 48: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by Application
- Figure 51: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by End User
- Figure 54: Latin America Market Value Share and BPS Analysis by Usability, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Usability, 2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Usability
- Figure 57: Latin America Market Value Share and BPS Analysis by Drainage Type, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Drainage Type, 2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by Drainage Type
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product
- Figure 64: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 66: Western Europe Market Attractiveness Analysis by Application
- Figure 67: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 69: Western Europe Market Attractiveness Analysis by End User
- Figure 70: Western Europe Market Value Share and BPS Analysis by Usability, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Usability, 2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Usability
- Figure 73: Western Europe Market Value Share and BPS Analysis by Drainage Type, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Drainage Type, 2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Drainage Type
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Application
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by End User
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Usability, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Usability, 2026 to 2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Usability
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Drainage Type, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Drainage Type, 2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Drainage Type
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 95: East Asia Market Attractiveness Analysis by Product
- Figure 96: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 98: East Asia Market Attractiveness Analysis by Application
- Figure 99: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 101: East Asia Market Attractiveness Analysis by End User
- Figure 102: East Asia Market Value Share and BPS Analysis by Usability, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Usability, 2026 to 2036
- Figure 104: East Asia Market Attractiveness Analysis by Usability
- Figure 105: East Asia Market Value Share and BPS Analysis by Drainage Type, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Drainage Type, 2026 to 2036
- Figure 107: East Asia Market Attractiveness Analysis by Drainage Type
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Usability, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Usability, 2026 to 2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Usability
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Drainage Type, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Drainage Type, 2026 to 2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Drainage Type
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Usability, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Usability, 2026 to 2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Usability
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Drainage Type, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Drainage Type, 2026 to 2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Drainage Type
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis