Pepsin Market Size, Share, Growth and Forecast (2026 - 2036)
Pepsin Market is segmented by Grade (Pharmaceutical, Food, Industrial), Source (Porcine, Microbial/Fermentation), Application (Digestive Preparations, Rennet Substitutes, Diagnostics & Others), and Region. Forecast for 2026 to 2036.
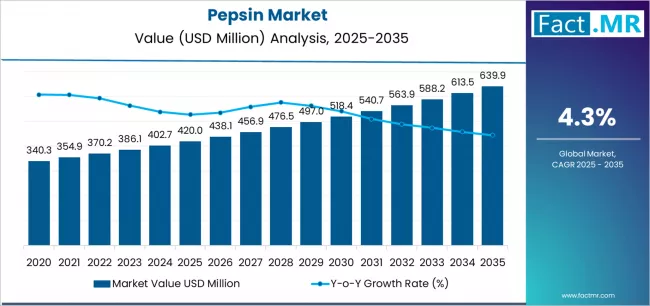
According to Fact.MR estimates, the Pepsin market stood at USD 420.0 million in 2025. The market is projected to reach USD 438.1 million in 2026 and climb to USD 667.4 million by 2036, progressing at a CAGR of 4.3%. Pharmaceutical grade pepsin is anticipated to hold around 46% share, while digestive preparations are likely to remain the leading application segment with nearly 48% share.
Pepsin Market Size, Market Forecast and Outlook By Fact.MR
The pepsin market was valued at USD 420.00 million in 2025, projected to reach USD 438.06 million in 2026, and is forecast to expand to USD 667.39 million by 2036 at a 4.3% CAGR. Sustained demand from pharmaceutical digestive aid formulations and food-grade rennet substitute applications anchors consumption volumes, while the emergence of microbial fermentation-derived pepsin creates a parallel supply channel that reduces dependency on porcine-sourced material. Regulatory affairs teams at pharmaceutical excipient suppliers now evaluate pepsin sourcing strategies against halal and kosher certification requirements that constrain traditional animal-derived supply routes.
Summary of Pepsin Market
- Market Snapshot
- The pepsin market is valued at USD 420.00 million in 2025 and is projected to reach USD 667.39 million by 2036.
- The industry is expected to grow at a 4.3% CAGR from 2026 to 2036, creating an incremental opportunity of USD 229.33 million.
- The market operates as a sourcing-sensitive, pharmaceutical-and-food-grade enzyme category where porcine supply availability and halal/kosher certification requirements shape procurement decisions.
- Demand and Growth Drivers
- Demand is sustained by pharmaceutical digestive aid formulations that require pepsin as an active proteolytic enzyme for gastric supplement products.
- Food-grade rennet substitute applications in cheese and dairy processing expand consumption as manufacturers replace traditional calf rennet with pepsin-based coagulants.
- Microbial fermentation-derived pepsin gains traction as buyers seek non-animal-sourced alternatives to satisfy halal, kosher, and vegetarian certification mandates.
- Among key countries, the USA leads at 4.9% CAGR, followed by Mexico at 4.6%, Germany at 4.2%, France at 4.0%, the UK at 3.8%, South Korea at 3.7%, and Japan at 3.5%.
- Product and Segment View
- Pepsin is an aspartic protease enzyme extracted from porcine gastric mucosa or produced through microbial fermentation, used in pharmaceutical digestive preparations, food-grade rennet substitutes, and diagnostic reagent applications.
- Pharmaceutical Grade accounts for 46.0% of Grade share in 2026, serving as the primary specification for digestive enzyme supplement and excipient applications.
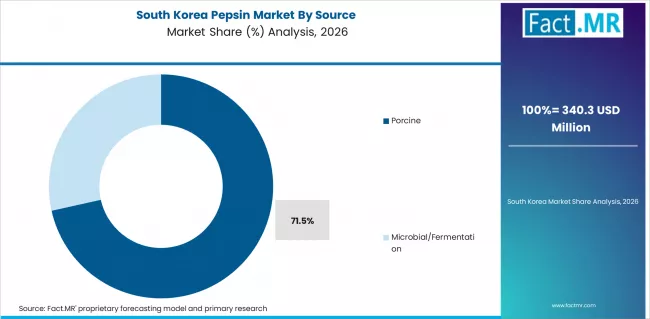
- Porcine-sourced pepsin commands 74.0% of Source share in 2026, reflecting established extraction infrastructure from slaughterhouse by-product processing.
- Digestive Preparations leads Application with 48.0% share in 2026, driven by over-the-counter gastric supplement demand in ageing consumer demographics.
- The scope includes pharmaceutical, food, and industrial grade pepsin from porcine and microbial sources, while excluding other proteolytic enzymes such as trypsin, chymotrypsin, and plant-derived papain.
- Geography and Competitive Outlook
- The USA and Mexico are the fastest-growing markets due to digestive supplement demand and cheese processing expansion, while Germany and France represent mature pharmaceutical-grade procurement hubs.
- Competition is shaped by sourcing reliability and pharmacopeial compliance, with key players including BIOFAC, A. Constantino, Amano Enzyme, Vivatis Pharma, and Sichuan Deebio.
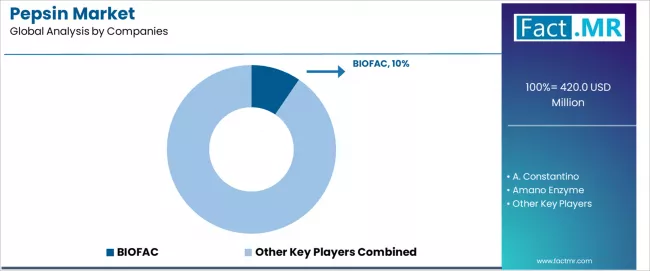
- BIOFAC holds approximately 10% market share, leveraging its integrated porcine enzyme extraction platform and pharmaceutical-grade quality management systems.
Pepsin Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 438.06 million |
| Industry Value (2036) | USD 667.39 million |
| CAGR (2026 to 2036) | 4.30% |
Source: Fact.MR, 2026
The forecast period projects an absolute dollar expansion of approximately USD 229.33 million between 2026 and 2036. This growth trajectory reflects incremental expansion in digestive enzyme supplement demand across ageing populations and steady replacement of calf rennet with pepsin-based coagulants in cheese production. Porcine raw material pricing remains subject to swine herd health cycles and slaughterhouse throughput fluctuations, creating procurement timing challenges for pharmaceutical-grade buyers requiring consistent activity specifications.
The USA leads at 4.9% CAGR through 2036, supported by expanding digestive supplement retail distribution and pharmaceutical excipient demand. Mexico follows at 4.6%, driven by growing processed cheese production volumes. Germany registers 4.2% growth through pharmaceutical manufacturing requirements. France tracks at 4.0%, benefiting from dairy processing modernization. The UK maintains 3.8% expansion, South Korea records 3.7%, and Japan registers 3.5%, both anchored by pharmaceutical and food-grade enzyme procurement cycles.
Why is the Pepsin Market Growing?
Market expansion is being supported by the increasing global prevalence of digestive disorders driven by lifestyle changes and aging populations, alongside the corresponding need for effective enzyme replacement therapies that can improve digestive function, support gastrointestinal health, and maintain therapeutic consistency across various pharmaceutical, nutraceutical, and medical applications. Modern pharmaceutical companies and healthcare providers are increasingly focused on implementing pepsin-based solutions that can provide therapeutic benefits, support patient outcomes, and ensure regulatory compliance while meeting comprehensive safety and efficacy requirements.
The growing emphasis on personalized medicine and precision therapeutics is driving demand for pepsin that can support individualized treatment approaches, enable targeted therapy development, and ensure comprehensive therapeutic optimization. Healthcare manufacturers' preference for enzyme solutions that combine therapeutic effectiveness with safety profiles and regulatory acceptance is creating opportunities for innovative pepsin implementations. The rising influence of biotechnology research and regenerative medicine is also contributing to increased adoption of pepsin solutions that can provide research capabilities without compromising therapeutic potential or regulatory compliance.
Segmental Analysis
The market is segmented by grade, source, and application. By grade, the market is divided into pharmaceutical, food, and industrial categories. Based on source, the market is categorized into porcine and microbial/fermentation sources. By application, the market includes digestive preparations, rennet substitutes, and diagnostics & others. Regionally, the market is divided into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa.
By Grade, the Pharmaceutical Segment Leads the Market

The pharmaceutical segment is projected to maintain its leading position in the pepsin market in 2026 with a 46.0% market share, reaffirming its role as the preferred quality grade for therapeutic applications and medical formulations. Pharmaceutical manufacturers increasingly utilize pharmaceutical-grade pepsin for its proven therapeutic effectiveness, stringent quality specifications, and established safety profile in digestive health applications while supporting regulatory compliance and patient safety requirements. Pharmaceutical pepsin technology's proven efficacy and regulatory acceptance directly address the industry requirements for therapeutic enzyme solutions and comprehensive medical applications across diverse healthcare and pharmaceutical sectors.
This grade segment forms the foundation of modern therapeutic enzyme applications, as it represents the quality category with the greatest contribution to patient outcomes and established clinical acceptance across multiple therapeutic applications and regulatory environments. Pharmaceutical industry investments in enzyme-based therapies continue to strengthen adoption among healthcare providers and pharmaceutical developers. With growing demands for digestive health solutions and therapeutic enzyme optimization, pharmaceutical-grade pepsin aligns with both therapeutic effectiveness requirements and safety standards, making it the central component of comprehensive enzyme therapy strategies.
By Application, the Digestive Preparations Segment Dominates Market Demand
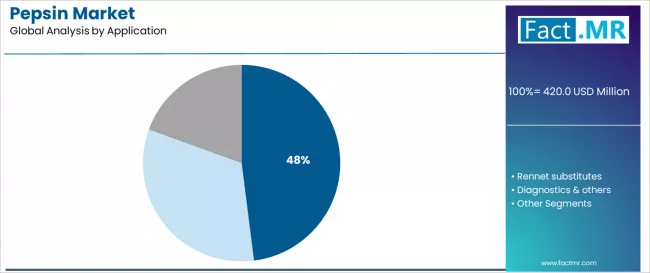
The digestive preparations application segment is projected to represent the largest share of pepsin demand in 2026 with a 48.0% market share, underscoring its critical role as the primary driver for pepsin adoption across digestive aid formulations, enzyme replacement therapies, and gastrointestinal health products. Pharmaceutical companies prefer pepsin for digestive applications due to its proven digestive enhancement capabilities, established therapeutic benefits, and ability to improve patient outcomes while supporting regulatory compliance and safety standards. Positioned as essential enzymes for modern digestive health management, pepsin offers both therapeutic advantages and patient benefits.
The segment is supported by continuous innovation in digestive health technologies and the growing availability of standardized pepsin formulations that enable superior therapeutic performance with enhanced bioavailability and patient tolerance. Pharmaceutical companies are investing in comprehensive digestive health programs to support increasingly sophisticated patient care requirements and healthcare provider expectations for effective therapeutic solutions. As digestive health awareness increases and therapeutic enzyme applications expand, the digestive preparations segment will continue to dominate the market while supporting advanced pepsin utilization and therapeutic optimization strategies.
What are the Drivers, Restraints, and Key Trends of the Pepsin Market?
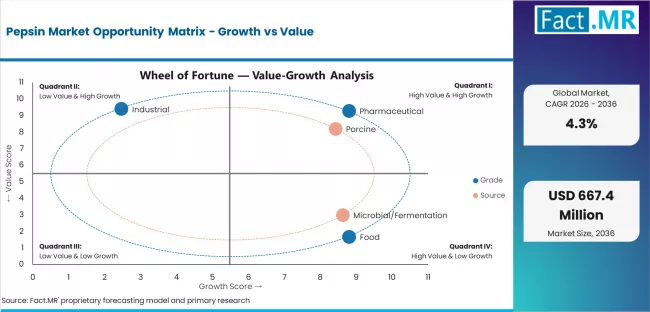
The pepsin market is advancing steadily due to increasing demand for digestive health solutions driven by rising gastrointestinal disorder prevalence and growing adoption of enzyme replacement therapies that require effective proteolytic enzymes providing therapeutic benefits, safety assurance, and regulatory compliance across diverse pharmaceutical, nutraceutical, and medical applications. The market faces challenges, including source-related supply constraints and ethical considerations, competition from synthetic alternatives and microbial enzymes, and regulatory complexity related to therapeutic enzyme approval and quality standardization. Innovation in microbial production technologies and purification methods continues to influence product development and market expansion patterns.
Expansion of Personalized Medicine and Precision Therapeutics
The growing adoption of personalized medicine is driving demand for pepsin applications that address individual patient needs, support customized therapeutic approaches, and provide tailored digestive health solutions while maintaining therapeutic effectiveness and safety compliance. Precision medicine applications require pepsin with consistent activity profiles, proven bioavailability, and documented therapeutic outcomes that enable individualized treatment optimization. Healthcare companies are increasingly recognizing the competitive advantages of pepsin integration for personalized digestive health programs and precision therapeutic development, creating opportunities for specialized formulations designed for individual patient requirements and therapeutic customization approaches.
Integration of Biotechnology Production and Sustainable Sourcing
Modern pepsin producers are incorporating biotechnology platforms and microbial fermentation systems to enhance supply chain sustainability, reduce animal-derived sourcing dependency, and support comprehensive ethical manufacturing through optimized production processes and renewable feedstock utilization. Leading companies are developing recombinant production methods, implementing sustainable manufacturing practices, and advancing fermentation technologies that provide consistent enzyme supply while minimizing ethical concerns and regulatory complexity. These technologies improve supply reliability while enabling new market opportunities, including vegan-compatible applications, sustainable pharmaceutical development, and ethical healthcare solutions. Advanced biotechnology integration also allows producers to support comprehensive sustainability objectives and ethical compliance beyond traditional animal-derived sourcing methods.
Development of Advanced Diagnostic and Research Applications
The expansion of biotechnology research and advanced diagnostic applications is driving demand for pepsin with enhanced purity specifications, specialized activity profiles, and research-grade quality that exceed traditional therapeutic applications. These advanced applications require ultra-pure pepsin with precise characterization and validated performance specifications, creating premium market segments with differentiated value propositions. Manufacturers are investing in advanced purification technologies and analytical capabilities to serve emerging research applications while supporting innovation in biotechnology development, diagnostic advancement, and therapeutic research.
Analysis of the Pepsin Market by Key Countries
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| USA | 4.9% |
| Mexico | 4.6% |
| Germany | 4.2% |
| France | 4.0% |
| UK | 3.8% |
| South Korea | 3.7% |
| Japan | 3.5% |

The pepsin market is experiencing solid growth globally, with the USA leading at a 4.9% CAGR through 2036, driven by expanding pharmaceutical industry, comprehensive digestive health awareness, and increasing investment in enzyme-based therapeutics supported by advanced healthcare infrastructure and regulatory frameworks. Mexico follows at 4.6%, supported by growing pharmaceutical manufacturing, expanding healthcare access, and increasing adoption of digestive health solutions. Germany shows growth at 4.2%, emphasizing pharmaceutical excellence, enzyme technology leadership, and advanced biotechnology development capabilities. France demonstrates 4.0% growth, supported by pharmaceutical innovation, healthcare system strength, and enzyme research capabilities. The UK records 3.8%, focusing on biotechnology advancement, pharmaceutical research excellence, and healthcare innovation programs. South Korea exhibits 3.7% growth, emphasizing biotechnology development, pharmaceutical manufacturing capabilities, and healthcare modernization initiatives. Japan shows 3.5% growth, supported by pharmaceutical industry excellence, precision biotechnology, and advanced healthcare applications.
The report covers an in-depth analysis of 40+ countries, with top-performing countries highlighted below.
USA Leads Global Market Growth with Pharmaceutical Innovation and Healthcare Excellence
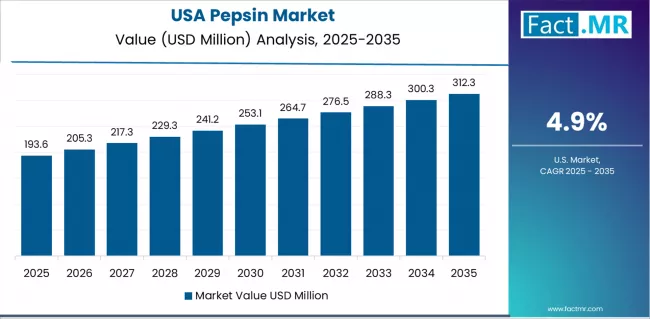
Revenue from pepsin in the USA is projected to exhibit exceptional growth with a CAGR of 4.9% through 2036, driven by expanding pharmaceutical industry capabilities and comprehensive digestive health awareness supported by advanced healthcare infrastructure and increasing investment in enzyme-based therapeutic development. The country's robust pharmaceutical sector and healthcare innovation ecosystem are creating substantial demand for high-quality pepsin solutions. Major pharmaceutical companies and biotechnology firms are establishing comprehensive pepsin sourcing and application capabilities to serve both domestic healthcare needs and global therapeutic markets.
- Growing digestive health awareness and gastrointestinal disorder prevalence are driving demand for pepsin-based therapeutics throughout major healthcare networks and pharmaceutical companies across research centers, clinical facilities, and therapeutic development organizations.
- Strong pharmaceutical industry development and enzyme therapy innovation are supporting the adoption of pepsin technologies among companies seeking therapeutic differentiation and patient outcome improvement in competitive healthcare markets.
- Advanced regulatory framework and FDA approval pathways are enabling pepsin market expansion through comprehensive safety assessment, therapeutic validation, and quality assurance programs throughout major pharmaceutical development centers.
- Expanding biotechnology research and precision medicine initiatives are driving demand for research-grade pepsin supporting drug discovery, therapeutic development, and personalized medicine applications across major research institutions.
Mexico Demonstrates Strong Market Potential with Pharmaceutical Growth and Healthcare Development
Demand for pepsin in Mexico is expanding at a CAGR of 4.6%, supported by growing pharmaceutical manufacturing capabilities, expanding healthcare access, and increasing adoption of digestive health solutions supporting pepsin integration across diverse healthcare and pharmaceutical applications. The country's developing healthcare infrastructure and pharmaceutical sector growth are driving sophisticated pepsin utilization throughout medical and manufacturing sectors. Leading pharmaceutical companies and healthcare providers are establishing extensive pepsin sourcing and application capabilities.
- Growing pharmaceutical manufacturing and healthcare infrastructure development are creating opportunities for pepsin adoption throughout major manufacturing centers and healthcare facilities serving domestic and export markets.
- Expanding healthcare access and digestive health awareness are driving adoption of enzyme-based therapies among healthcare providers seeking improved patient outcomes and therapeutic effectiveness.
- Strategic geographic advantages and trade relationship benefits are supporting pepsin market development including efficient supply chains, competitive cost structures, and access to North American pharmaceutical expertise and technologies.
- Rising healthcare investment and pharmaceutical sector modernization programs are enabling advanced pepsin applications including therapeutic development and healthcare delivery improvement throughout major metropolitan and industrial regions.
Germany Emphasizes Pharmaceutical Excellence with Enzyme Technology and Biotechnology Leadership
Revenue from pepsin in Germany is anticipated to grow at a CAGR of 4.2%, supported by the country's pharmaceutical industry excellence, established enzyme technology capabilities, and comprehensive biotechnology development supporting high-quality pepsin applications. Germany's pharmaceutical expertise and biotechnology focus are supporting investment in sophisticated pepsin solutions. Major pharmaceutical companies and biotechnology firms are establishing comprehensive development and quality assurance programs for pepsin applications.
- Advanced pharmaceutical industry capabilities and enzyme technology leadership are creating demand for high-quality pepsin throughout major pharmaceutical centers and biotechnology facilities emphasizing therapeutic excellence and regulatory compliance.
- Strong biotechnology development and enzyme research capabilities are driving the advancement of pepsin applications including novel therapeutic formulations, production optimization, and quality enhancement throughout major research and development facilities.
- Comprehensive regulatory framework and pharmaceutical quality standards are supporting pepsin development meeting stringent European requirements for therapeutic enzymes and pharmaceutical applications.
- Export market leadership and international pharmaceutical reputation are enabling German pepsin solutions to access global markets while maintaining premium positioning and technological excellence advantages.
France Shows Healthcare Innovation with Pharmaceutical Research and Enzyme Development
Demand for pepsin in France is expected to grow at a CAGR of 4.0%, driven by pharmaceutical innovation excellence, established healthcare system capabilities, and comprehensive enzyme research programs supporting pepsin development in therapeutic and biotechnology applications. The country's healthcare heritage and research focus are creating opportunities for sophisticated pepsin utilization. Leading pharmaceutical companies and research institutions are establishing comprehensive enzyme development programs incorporating therapeutic innovation and scientific excellence.
- Strong pharmaceutical innovation and healthcare research capabilities are creating demand for advanced pepsin applications throughout major pharmaceutical centers and research institutions emphasizing therapeutic development and clinical excellence.
- Established healthcare system and digestive health expertise are supporting pepsin integration including therapeutic applications, patient care optimization, and clinical research throughout major healthcare networks and medical centers.
- Comprehensive research capabilities and academic collaboration networks are advancing pepsin science including fundamental research, therapeutic optimization, and application development throughout major research institutions and pharmaceutical companies.
- Healthcare excellence culture and pharmaceutical quality expectations are driving demand for superior pepsin solutions supporting premium therapeutic applications and patient care optimization throughout major medical and pharmaceutical centers.
UK Demonstrates Biotechnology Leadership with Research Excellence and Pharmaceutical Innovation
Revenue from pepsin in the UK is expanding at a CAGR of 3.8%, supported by comprehensive biotechnology advancement, established pharmaceutical research capabilities, and healthcare innovation programs emphasizing pepsin integration for therapeutic and research applications. The country's biotechnology leadership and research excellence are driving pepsin adoption throughout pharmaceutical and research sectors. Leading biotechnology companies and research institutions are establishing advanced pepsin research and application capabilities.
- Comprehensive biotechnology leadership and pharmaceutical research excellence are creating systematic market opportunities for pepsin throughout major research centers and biotechnology companies emphasizing innovation and therapeutic development.
- Strong healthcare innovation and enzyme research programs are supporting pepsin development including novel applications, therapeutic optimization, and research advancement throughout major pharmaceutical and academic institutions.
- Advanced research capabilities and academic excellence are advancing pepsin technology including production optimization, purification enhancement, and application innovation throughout major universities and research facilities.
- Innovation ecosystem development and technology transfer programs are facilitating pepsin commercialization through industry collaboration, startup support, and therapeutic advancement initiatives.
South Korea Emphasizes Biotechnology Development with Healthcare Modernization and Pharmaceutical Growth
Demand for pepsin in South Korea is growing at a CAGR of 3.7%, supported by comprehensive biotechnology development, healthcare system modernization, and expanding pharmaceutical manufacturing capabilities emphasizing pepsin integration for therapeutic and industrial applications. The country's technology focus and healthcare advancement are driving sophisticated pepsin utilization in pharmaceutical and biotechnology applications. Leading pharmaceutical companies and biotechnology firms are establishing advanced pepsin processing and application capabilities.
- Comprehensive biotechnology development and pharmaceutical manufacturing capabilities are creating demand for pepsin solutions throughout major biotechnology centers and pharmaceutical facilities emphasizing production efficiency and therapeutic effectiveness.
- Healthcare modernization and digestive health awareness are supporting pepsin integration in diverse therapeutic applications including enzyme replacement therapy, digestive aids, and specialized medical treatments.
- Technology innovation focus and research investment are advancing pepsin applications including production optimization, quality enhancement, and therapeutic development throughout major technology and research centers.
- Export pharmaceutical capabilities and international market development are enabling South Korean pepsin applications to serve global markets while maintaining quality standards and competitive positioning.
Japan Shows Quality Excellence with Pharmaceutical Precision and Healthcare Innovation

Revenue from pepsin in Japan is expanding at a CAGR of 3.5%, driven by exceptional pharmaceutical industry standards, established healthcare excellence, and comprehensive biotechnology capabilities emphasizing superior pepsin quality and therapeutic effectiveness. The country's commitment to pharmaceutical precision and healthcare innovation are creating opportunities for ultra-high-quality pepsin applications. Leading pharmaceutical companies and healthcare organizations are establishing comprehensive quality control and therapeutic optimization programs.
- Exceptional pharmaceutical industry standards and healthcare excellence are creating demand for ultra-high-quality pepsin meeting Japanese requirements for therapeutic effectiveness and safety assurance.
- Advanced biotechnology capabilities and enzyme technology expertise are supporting pepsin optimization including purification enhancement, activity standardization, and therapeutic validation throughout major pharmaceutical and biotechnology facilities.
- Comprehensive quality control systems and rigorous healthcare standards are ensuring superior pepsin consistency and therapeutic reliability throughout major pharmaceutical manufacturing regions and healthcare applications.
- Healthcare innovation culture and continuous improvement focus are driving demand for pepsin solutions that combine therapeutic excellence with safety assurance and comprehensive quality optimization.
Europe Market Split by Countries
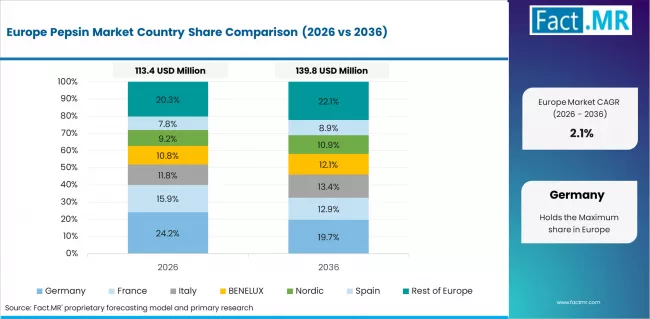
The pepsin market in Europe is projected to grow from USD 126.0 million in 2026 to USD 192.0 million by 2036, registering a CAGR of 4.3% over the forecast period. Germany is expected to maintain leadership with a 28.5% market share in 2026, moderating to 28.2% by 2036, supported by pharmaceutical industry excellence, enzyme technology capabilities, and comprehensive biotechnology development.
France follows with 21.0% in 2026, projected at 21.3% by 2036, driven by pharmaceutical innovation, healthcare research capabilities, and enzyme development programs. The United Kingdom holds 18.5% in 2026, expected to reach 18.7% by 2036 on the back of biotechnology leadership and pharmaceutical research excellence. Italy commands 13.0% in 2026, rising slightly to 13.1% by 2036, while Spain accounts for 9.5% in 2026, reaching 9.7% by 2036 aided by healthcare development and pharmaceutical growth. The Netherlands maintains 4.5% in 2026, up to 4.6% by 2036 due to biotechnology capabilities and pharmaceutical expertise. The Rest of Europe region, including Nordic countries, Central & Eastern Europe, and other markets, is anticipated to hold 5.0% in 2026 and 4.4% by 2036, reflecting steady growth in healthcare modernization and pharmaceutical development.
Competitive Landscape of the Pepsin Market
The pepsin market is characterized by competition among established enzyme manufacturers, pharmaceutical ingredient suppliers, and biotechnology companies. Companies are investing in production technology development, quality assurance systems, regulatory compliance programs, and application-specific solution development to deliver high-purity, therapeutically effective, and cost-competitive pepsin solutions. Innovation in microbial production methods, purification technologies, and therapeutic formulation is central to strengthening market position and competitive advantage.
BIOFAC leads the market with a 9.5% share, offering comprehensive pepsin solutions with a focus on pharmaceutical applications, quality excellence, and regulatory compliance across diverse therapeutic, diagnostic, and research applications. The company has announced major production capacity expansions and quality enhancement programs, with plans to incorporate advanced purification technologies and sustainable production systems, with facility upgrades initiated in 2024 and enhanced capabilities planned for 2026. A. Constantino provides specialized enzyme solutions with emphasis on pharmaceutical-grade quality, having advanced their pepsin strategy by expanding production capabilities and developing customized formulations for specific therapeutic applications.
Amano Enzyme delivers comprehensive enzyme solutions including pepsin products with focus on pharmaceutical and industrial applications. Vivatis Pharma offers pharmaceutical-grade pepsin with emphasis on therapeutic applications and regulatory compliance. Sichuan Deebio provides pepsin manufacturing and processing capabilities for diverse market segments. Linzhang Hengyi focuses on enzyme production and quality assurance for pharmaceutical applications. Nitta Gelatin specializes in protein-derived ingredients including pepsin solutions. Renco offers enzyme products for pharmaceutical and industrial markets. Enzymology Research Center provides specialized enzyme research and development services. Pangbo Biological delivers enzyme manufacturing and application support for diverse industries.
Pepsin Market - Stakeholder Contribution Framework
Pepsin represents a specialized enzyme segment within pharmaceutical, biotechnology, and industrial applications, projected to grow from USD 420.0 million in 2025 to USD 640.0 million by 2035 at a 4.3% CAGR. This proteolytic enzyme primarily sourced from porcine and microbial fermentation, serves as a critical therapeutic and industrial ingredient in digestive preparations, pharmaceutical formulations, and biotechnology applications where enzymatic activity, therapeutic efficacy, and quality assurance are essential. Market expansion is driven by increasing digestive health awareness, growing pharmaceutical applications, expanding biotechnology research, and rising demand for enzyme-based therapeutic solutions across diverse healthcare and industrial segments.
How Healthcare Regulatory Bodies Could Strengthen Quality Standards and Therapeutic Applications
- Pharmaceutical Grade Standards and Quality Assurance: Establish comprehensive quality specifications for pharmaceutical-grade pepsin including purity requirements, activity standards, and contamination limits that ensure therapeutic effectiveness while supporting safe clinical applications and regulatory compliance across diverse healthcare environments.
- Therapeutic Application Guidelines and Safety Protocols: Develop systematic clinical guidelines for pepsin therapeutic applications including dosage recommendations, safety monitoring, and efficacy assessment that support evidence-based treatment while ensuring patient safety and optimal therapeutic outcomes.
- Manufacturing Standards and Good Manufacturing Practice: Create stringent production requirements for pepsin manufacturing including facility standards, process controls, and quality management systems that ensure consistent product quality while supporting therapeutic reliability and regulatory compliance.
- Source Material Validation and Traceability: Implement comprehensive source material requirements including supplier qualification, traceability systems, and contamination prevention that ensure raw material quality while supporting supply chain integrity and therapeutic safety.
- Clinical Research and Evidence Development: Facilitate clinical studies and therapeutic research for pepsin applications including efficacy validation, safety assessment, and optimal application development that provide scientific foundation for therapeutic use and regulatory approval.
How Pharmaceutical Industry Associations Could Advance Therapeutic Standards and Clinical Applications
- Clinical Application Guidelines and Best Practices: Develop comprehensive technical resources for pepsin therapeutic applications including clinical protocols, dosage optimization, and patient monitoring that enhance therapeutic effectiveness while ensuring safety and regulatory compliance across diverse healthcare applications.
- Quality Assurance and Manufacturing Excellence: Create industry-wide quality standards and certification programs including analytical methods, specification requirements, and manufacturing protocols that ensure consistent therapeutic quality while supporting clinical effectiveness and patient safety.
- Research and Development Collaboration: Facilitate partnerships between pepsin suppliers, pharmaceutical companies, research institutions, and clinical organizations to advance therapeutic applications while accelerating innovation development and clinical validation programs.
- Regulatory Engagement and Approval Support: Provide systematic regulatory guidance and clinical development assistance including regulatory pathway navigation, clinical trial support, and approval strategy development that help companies achieve market authorization effectively.
- Professional Education and Clinical Training: Create specialized training programs for healthcare professionals covering pepsin therapeutic applications, clinical benefits, and patient management that enhance clinical competency while supporting appropriate therapeutic utilization.
How Pepsin Manufacturers Could Drive Innovation and Market Leadership
- Advanced Production Technology and Quality Excellence: Invest in state-of-the-art manufacturing systems including fermentation optimization, purification enhancement, and quality control technologies that improve product consistency while reducing costs and ensuring therapeutic effectiveness for competitive positioning.
- Therapeutic Formulation Development: Create specialized pepsin formulations including enhanced bioavailability, targeted delivery systems, and combination therapies that address specific therapeutic needs while maintaining safety profiles and regulatory compliance requirements.
- Sustainable Production and Ethical Sourcing: Implement environmentally responsible and ethically sound production practices including microbial fermentation development, sustainable sourcing, and animal welfare considerations that address ethical concerns while maintaining product quality and market access.
- Regulatory Compliance and Quality Assurance: Establish comprehensive regulatory and quality management systems including analytical testing, documentation, and traceability that ensure therapeutic compliance while supporting market authorization and clinical acceptance.
- Customer Partnership and Technical Support: Provide comprehensive support services including formulation assistance, regulatory guidance, clinical data support, and technical expertise that strengthen customer relationships while enabling successful therapeutic development and market success.
How Healthcare Providers Could Optimize Pepsin Therapeutic Applications and Patient Outcomes
- Evidence-Based Treatment Protocols: Develop systematic clinical approaches for pepsin therapy including patient selection criteria, dosage optimization, and outcome monitoring that maximize therapeutic benefits while ensuring patient safety and treatment effectiveness.
- Patient Education and Compliance Support: Create comprehensive patient education programs covering pepsin benefits, proper usage, and adherence strategies that enhance treatment compliance while supporting optimal therapeutic outcomes and patient satisfaction.
- Clinical Monitoring and Safety Assessment: Implement systematic monitoring protocols including efficacy evaluation, adverse event tracking, and therapy optimization that ensure patient safety while enabling treatment adjustment and outcome improvement.
- Multidisciplinary Care Coordination: Establish collaborative treatment approaches including gastroenterology, pharmacy, and nutrition services that provide comprehensive patient care while optimizing pepsin therapy effectiveness and patient outcomes.
- Quality Assurance and Product Selection: Develop systematic evaluation criteria for pepsin products including quality specifications, supplier assessment, and therapeutic validation that ensure optimal product selection while maintaining treatment effectiveness and patient safety.
How Research Institutions Could Advance Pepsin Science and Therapeutic Development
- Fundamental Enzyme Research and Mechanism Studies: Conduct comprehensive research investigating pepsin biochemistry, therapeutic mechanisms, and physiological effects that advance scientific understanding while informing therapeutic development and clinical optimization across diverse healthcare applications.
- Clinical Research and Therapeutic Validation: Design and execute clinical studies including efficacy trials, safety assessment, and optimal application research that provide evidence foundation for therapeutic use while supporting regulatory approval and clinical adoption.
- Production Technology Innovation: Develop advanced manufacturing methods including microbial fermentation optimization, purification enhancement, and quality improvement that advance production capabilities while reducing costs and improving therapeutic availability.
- Diagnostic and Biotechnology Applications: Investigate novel pepsin applications including diagnostic development, biotechnology research, and analytical methods that expand market opportunities while supporting scientific advancement and technology development.
- Technology Transfer and Commercialization: Facilitate innovation development through industry partnerships, licensing programs, and collaborative research that accelerate therapeutic development while strengthening industry-academia cooperation and clinical application advancement.
How Investment Community Could Support Market Growth and Therapeutic Innovation
- Therapeutic Development and Clinical Research Funding: Provide capital for pepsin therapeutic research, clinical trials, and product development programs that advance therapeutic applications while creating competitive advantages and patient benefits for long-term market success.
- Manufacturing Infrastructure and Capacity Investment: Finance production facility development, equipment acquisition, and quality system implementation that address growing therapeutic demand while improving operational efficiency and product quality standards.
- Biotechnology Innovation and Sustainable Production: Support development of microbial production technologies, sustainable manufacturing processes, and biotechnology advancement that address ethical concerns while creating market opportunities and competitive advantages.
- Market Development and Healthcare Access: Fund therapeutic education programs, clinical development initiatives, and market access programs that build demand for pepsin therapies while supporting patient access and healthcare outcome improvement.
- Strategic Partnership and Technology Acquisition: Provide capital for strategic alliances, technology acquisitions, and collaborative development programs that strengthen market position while enabling access to advanced capabilities and technologies for comprehensive business growth.
Key Players in the Pepsin Market
- BIOFAC
- A. Constantino
- Amano Enzyme
- Vivatis Pharma
- Sichuan Deebio
- Linzhang Hengyi
- Nitta Gelatin
- Renco
- Enzymology Research Center
- Pangbo Biological
Bibliography
- United States Pharmacopeia. (2024). USP-NF Monograph: Pepsin. USP.
- European Pharmacopoeia Commission. (2024). Ph. Eur. 11.0: Pepsin Powder. EDQM.
- Food and Agriculture Organization. (2024). Codex Alimentarius: Enzyme Preparations Used in Food Processing. FAO/WHO.
- U.S. Food and Drug Administration. (2024). GRAS Notice Inventory: Enzyme Preparations. FDA.
- BIOFAC A/S. (2024). Company Profile and Product Catalogue. BIOFAC.
- Amano Enzyme Inc. (2024). Annual Report 2024. Amano Enzyme Inc.
This bibliography is provided for reader reference. The full Fact.MR report contains the complete reference list with primary research documentation.
This Report Addresses
- Market sizing and quantitative forecast metrics detailing precise enzyme expenditure dedicated to pepsin across pharmaceutical and food processing sectors through 2036.
- Segmentation analysis mapping the sourcing velocity of specific pepsin grades and evaluating the certification requirements driving porcine versus microbial supply selection.
- Regional deployment intelligence comparing the pharmaceutical excipient demand in North America against the dairy processing enzyme requirements prevalent in European markets.
- Regulatory compliance assessment analyzing how pharmacopeial monograph updates and food-grade enzyme GRAS notifications affect sourcing and specification decisions.
- Competitive posture evaluation tracking the integrated extraction advantages of established porcine processors and the emerging position of microbial fermentation suppliers.
- Supply chain vulnerability analysis identifying the specific swine herd health cycles and slaughterhouse throughput fluctuations that constrain pharmaceutical-grade pepsin availability.
- Custom data delivery formats encompassing interactive dashboards, raw Excel datasets, and comprehensive PDF narrative reports.
Pepsin Market Definition
Pepsin is an aspartic protease enzyme produced primarily from porcine gastric mucosa or through microbial fermentation processes. It functions as a proteolytic agent in pharmaceutical digestive preparations, as a rennet substitute in cheese manufacturing, and as a reagent in diagnostic and laboratory applications. The market serves pharmaceutical manufacturers, food processing companies, and diagnostic reagent suppliers.
Pepsin Market Inclusions
Market scope includes pharmaceutical-grade, food-grade, and industrial-grade pepsin sourced from porcine and microbial/fermentation origins. Regional and segment-level market sizes, forecast values from 2026 to 2036, and sourcing trend analysis are covered.
Pepsin Market Exclusions
The scope excludes other proteolytic enzymes such as trypsin, chymotrypsin, and plant-derived papain. Downstream finished pharmaceutical dosage forms and finished dairy products are not covered.
Pepsin Market Research Methodology
- Primary Research: Analysts engaged with pharmaceutical excipient procurement managers, dairy processing enzyme buyers, and porcine by-product processors to map sourcing volumes and specification requirements.
- Desk Research: Data collection aggregated pharmacopeial monograph updates, food-grade enzyme regulatory filings, and published slaughterhouse throughput statistics from major pork-producing regions.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of enzyme shipment volumes by grade and application, applying region-specific pharmaceutical and food processing activity indices to project demand velocity.
- Data Validation and Update Cycle: Projections are tested against publicly reported production volumes from listed enzyme manufacturers and cross-referenced with pharmaceutical excipient trade flow records.
Scope of the Report

| Items | Values |
|---|---|
| Quantitative Units (2025) | USD 420.0 million |
| Grade | Pharmaceutical, Food, Industrial |
| Source | Porcine, Microbial/fermentation |
| Application | Digestive preparations, Rennet substitutes, Diagnostics & others |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | USA, Mexico, Germany, France, UK, South Korea, Japan, and 40+ countries |
| Key Companies Profiled | BIOFAC, A. Constantino, Amano Enzyme, Vivatis Pharma, Sichuan Deebio, Linzhang Hengyi |
| Additional Attributes | Dollar sales by grade and application category, regional demand trends, competitive landscape, technological advancements in enzyme production, therapeutic development, biotechnology innovation, and healthcare application optimization |
Pepsin Market by Segments
-
Grade :
- Pharmaceutical
- Food
- Industrial
-
Source :
- Porcine
- Microbial/fermentation
-
Application :
- Digestive preparations
- Rennet substitutes
- Diagnostics & others
-
Region :
-
North America
- United States
- Canada
- Mexico
-
Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- Rest of Europe
-
Asia Pacific
- Japan
- South Korea
- China
- India
- Australia
- Rest of Asia Pacific
-
Latin America
- Brazil
- Argentina
- Rest of Latin America
-
Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- South Africa
- Rest of Middle East & Africa
-
- Frequently Asked Questions -
How large is the demand for Pepsin in the global market in 2026?
Demand for pepsin in the global market is estimated to be valued at USD 438.06 million in 2026.
What will be the market size of Pepsin in the global market by 2036?
Market size for pepsin is projected to reach USD 667.39 million by 2036.
What is the expected demand growth for Pepsin in the global market between 2026 and 2036?
Demand for pepsin is expected to grow at a CAGR of 4.3% between 2026 and 2036.
Which Grade is poised to lead global sales by 2026?
Pharmaceutical Grade accounts for 46.0% in 2026 as digestive enzyme supplement formulations require pepsin meeting pharmacopeial activity specifications.
How significant is the role of Porcine sourcing in the Pepsin market in 2026?
Porcine-sourced pepsin represents 74.0% of supply share as established extraction infrastructure from slaughterhouse by-products maintains cost and volume advantages.
What is driving demand in the USA?
Expanding digestive supplement retail distribution and pharmaceutical excipient procurement cycles support the USA's 4.9% CAGR through 2036.
What is Germany's growth outlook in this report?
Germany is projected to grow at a CAGR of 4.2% during 2026 to 2036.
What is Japan's growth outlook in this report?
Japan is projected to grow at a CAGR of 3.5% during 2026 to 2036.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Grade
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Grade , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Grade , 2025 to 2035
- Pharmaceutical
- Food
- Industrial
- Y to o to Y Growth Trend Analysis By Grade , 2020 to 2024
- Absolute $ Opportunity Analysis By Grade , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2025 to 2035
- Digestive preparations
- Rennet substitutes
- Diagnostics & others
- Y to o to Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Grade
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Grade
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Grade
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Grade
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Grade
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Grade
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Grade
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Grade
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Grade
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Grade
- By Application
- Competition Analysis
- Competition Deep Dive
- BIOFAC
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- A. Constantino
- Amano Enzyme
- Vivatis Pharma
- Sichuan Deebio
- Linzhang Hengyi
- Nitta Gelatin
- Renco
- Enzymology Research Center
- Pangbo Biological
- BIOFAC
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Global Market Value (USD Million) Forecast by Grade , 2020 to 2035
- Table 3: Global Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 4: North America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: North America Market Value (USD Million) Forecast by Grade , 2020 to 2035
- Table 6: North America Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 8: Latin America Market Value (USD Million) Forecast by Grade , 2020 to 2035
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 11: Western Europe Market Value (USD Million) Forecast by Grade , 2020 to 2035
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Grade , 2020 to 2035
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 17: East Asia Market Value (USD Million) Forecast by Grade , 2020 to 2035
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Grade , 2020 to 2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Grade , 2020 to 2035
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2020 to 2035
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value Share and BPS Analysis by Grade , 2025 and 2035
- Figure 4: Global Market Y to o to Y Growth Comparison by Grade , 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Grade
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 7: Global Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2025-2035
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: North America Market Value Share and BPS Analysis by Grade , 2025 and 2035
- Figure 21: North America Market Y to o to Y Growth Comparison by Grade , 2025-2035
- Figure 22: North America Market Attractiveness Analysis by Grade
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 24: North America Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 27: Latin America Market Value Share and BPS Analysis by Grade , 2025 and 2035
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Grade , 2025-2035
- Figure 29: Latin America Market Attractiveness Analysis by Grade
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 34: Western Europe Market Value Share and BPS Analysis by Grade , 2025 and 2035
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Grade , 2025-2035
- Figure 36: Western Europe Market Attractiveness Analysis by Grade
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Grade , 2025 and 2035
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Grade , 2025-2035
- Figure 43: Eastern Europe Market Attractiveness Analysis by Grade
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 48: East Asia Market Value Share and BPS Analysis by Grade , 2025 and 2035
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Grade , 2025-2035
- Figure 50: East Asia Market Attractiveness Analysis by Grade
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Grade , 2025 and 2035
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Grade , 2025-2035
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Grade
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Grade , 2025 and 2035
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Grade , 2025-2035
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Grade
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Application, 2025-2035
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis