Liver Cancer Diagnostics Market Size, Share, Growth and Forecast (2026 - 2036)
Liver Cancer Diagnostics Market is segmented by Test Type (Blood Biomarker Tests, Imaging Tests including CT, MRI, and Ultrasound, Tissue Biopsy, Liquid Biopsy, and Molecular Diagnostics), Cancer Type (Hepatocellular Carcinoma (HCC), Cholangiocarcinoma, and Others including metastatic liver cancer and rare hepatic tumors), End Use (Hospitals and Diagnostic Centers, Cancer Research Institutes, Pathology Labs, and Others), and Region. Forecast for 2026 to 2036.
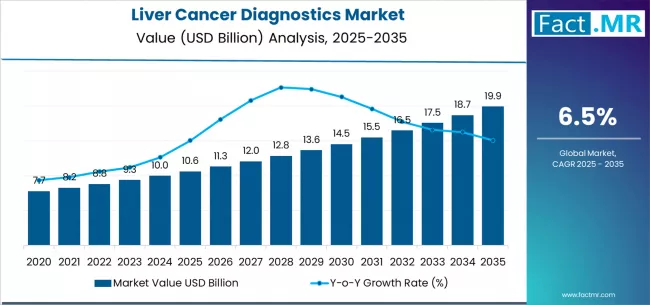
Fact.MR estimates that the liver cancer diagnostics market was valued at USD 10.3 billion in 2025, reaching USD 11.1 billion in 2026 and USD 24.9 billion by 2036. Blood biomarker tests lead with about 40% share, while hepatocellular carcinoma accounts for roughly 45% of diagnostic demand.
Liver Cancer Diagnostics Market Forecast and Outlook 2026 to 2036
In 2025, the liver cancer diagnostics market was valued at approximately USD 10.3 billion. Based on Fact.MR analysis, demand for liver cancer diagnostics is estimated to reach around USD 11.1 billion in 2026 and expand to nearly USD 24.9 billion by 2036, registering a CAGR of 8.5% during the forecast period. As Dr. Paul Limburg, Chief Medical Officer for Screening at Exact Sciences, stated in relation to ALTUS study findings, “These new data from the ALTUS study show that we can reliably detect liver cancer earlier, which is key to improving outcomes.”
Fact.MR analysis interprets this as reflective of a broader industry shift toward earlier-stage detection using blood biomarkers, imaging integration, and molecular platforms that support improved survival outcomes.
The market is projected to add roughly USD 14.6 billion in incremental value over the forecast window, reflecting transition from late-stage confirmation toward structured screening and recurrence monitoring. Early-period expansion is supported by wider adoption of ultrasound and alpha-fetoprotein testing across at-risk cohorts. Mid-period growth aligns with increased CT and MRI utilization for lesion characterization and staging accuracy. In the later years, value realization is increasingly shaped by liquid biopsy and molecular diagnostics, which support therapy selection, minimal residual disease monitoring, and longitudinal disease management.
The United States leads growth at 9.4% CAGR, supported by advanced imaging infrastructure and structured screening pathways. Germany follows at 8.9%, driven by standardized oncology diagnostics. Japan records 8.6%, reflecting established HCC surveillance programs and ageing demographics. China posts 8.1%, supported by expanding diagnostic capacity and a large high-risk population, while South Korea at 7.8% remains driven by hospital-based screening integration and early detection initiatives.
Summary of Liver Cancer Diagnostics Market
- Liver Cancer Diagnostics Market Definition
- The liver cancer diagnostics market comprises imaging modalities, biomarker assays, biopsy procedures, and molecular tests used to detect, stage, and monitor primary and metastatic liver malignancies in clinical practice.
- Demand Drivers
- Structured surveillance programs for high-risk populations with chronic hepatitis and cirrhosis increase routine use of ultrasound and alpha-fetoprotein testing.
- Rising incidence of hepatocellular carcinoma linked to viral hepatitis and metabolic liver disease expands diagnostic procedure volumes across hospital networks.
- Clinical integration of molecular diagnostics and liquid biopsy supports adoption of higher-sensitivity tests for early detection and therapy monitoring.
- Key Segments Analyzed
- Test Type: Blood biomarker tests lead due to non-invasive sampling, suitability for repeat monitoring, and established use in surveillance pathways.
- Cancer Type: Hepatocellular carcinoma accounts for the largest share, reflecting higher incidence and continuous diagnostic follow-up requirements.
- Geography: North America and East Asia drive growth because of screening infrastructure, disease prevalence, and access to advanced imaging.
- Analyst Opinion at Fact.MR
- Shambhu Nath Jha, Principal Consultant at Fact.MR, opines, ‘CXOs will find that adoption of liver cancer diagnostics depends on validated sensitivity thresholds, pathway integration, and reimbursement alignment rather than availability of individual test formats.’
- Strategic Implications
- Diagnostic manufacturers should align product validation with established surveillance guidelines for high-risk patient groups.
- Healthcare providers benefit from combining imaging and biomarker data to support earlier and more consistent detection.
- Investment decisions should prioritize platforms embedded within national screening and reimbursement frameworks.
- Methodology
- Market sizing was derived using liver cancer incidence data, diagnostic procedure volumes, and repeat testing patterns observed in clinical practice.
- Regulatory oversight and diagnostic usage assumptions were validated using guidance. [1]
- Screening and epidemiology context was cross-checked against public health guidance.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 11.1 billion |
| Forecast Value (2036) | USD 24.9 billion |
| Forecast CAGR (2026-2036) | 8.5% |
Market Definition
Liver Cancer Diagnostics Market Definition
The liver cancer diagnostics market covers test and imaging methods used to detect, confirm, and monitor malignant tumors in the liver. These diagnostics help identify cancer presence, assess disease stage, and guide treatment decisions. Common diagnostic approaches include blood based biomarkers, imaging techniques, and tissue sampling. They are used in hospitals, diagnostic laboratories, and specialized cancer centers for patients at risk of or suspected with liver cancer. Regulation, clinical validation, and use of diagnostic devices and tests are overseen by health authorities such as the US Food and Drug Administration and international health agencies. [2]
Liver Cancer Diagnostics Market Inclusions
The report covers global and regional market size estimates in volume and value with a forecast from 2026 to 2036. Segmentation includes diagnostic method such as imaging, biomarker tests, and biopsy based diagnostics, application across screening and disease monitoring, and end use in hospitals and diagnostic laboratories. Regional demand patterns, pricing trends, and diagnostic test adoption are included.
Liver Cancer Diagnostics Market Exclusions
The scope excludes liver cancer treatment products such as drugs, surgical devices, and radiotherapy systems. General liver function tests used for non-cancer conditions are not included unless used specifically in cancer diagnosis. Research use only assays, experimental diagnostic tools without regulatory clearance, and clinical trial services are excluded. Cancer care services, physician consultations, and treatment monitoring software are also outside the scope.
Liver Cancer Diagnostics Market Research Methodology
- Primary Research: Interviews were conducted with diagnostic test manufacturers, oncologists, radiologists, laboratory managers, and hospital procurement teams.
- Desk Research: Desk research used diagnostic device guidance, approvals, and safety communications issued by the US Food and Drug Administration and cancer epidemiology and screening guidance published by the World Health Organization.
- Market Sizing and Forecasting: Estimates were developed using cancer incidence data, diagnostic procedure volumes, test utilization rates, and replacement or repeat testing patterns.
- Data Validation and Update Cycle: Data was cross checked against regulatory approvals, hospital diagnostic volumes, public cancer statistics, and manufacturer disclosures before updates were finalized.
Segmental Analysis
Liver Cancer Diagnostics Market Analysis by Test Type
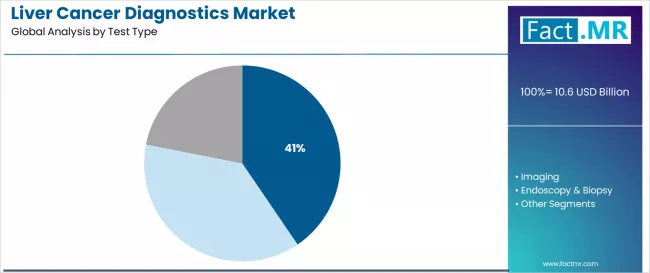
Based on Fact.MR analysis, consumption of blood biomarker tests is estimated to hold 40% share of the global liver cancer diagnostics market. Fact.MR analysts note blood-based testing leads due to non-invasive sampling, repeat testing suitability, and role in early screening and disease monitoring. As per Fact.MR, this test type addresses clinical needs for alpha-fetoprotein measurement and emerging biomarker panels used alongside imaging, supporting surveillance of high-risk populations and ongoing assessment of treatment response across hospital and outpatient diagnostic settings worldwide today.
- Diagnostic oversight: In vitro diagnostic tests are regulated under national medical device and laboratory frameworks.[3]
- Screening use: Fact.MR analysts note blood biomarkers support routine monitoring in at-risk patients.
- Testing access: Blood tests are widely available across primary and specialty care facilities.
Liver Cancer Diagnostics Market Analysis by Cancer Type
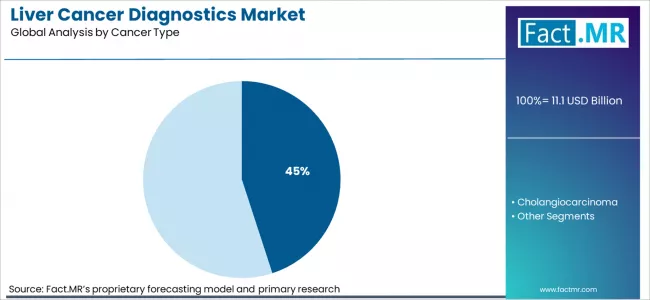
Based on Fact.MR analysis, consumption of liver cancer diagnostics for hepatocellular carcinoma is estimated to hold 45% share of the global market. Fact.MR analysts note HCC leads due to its higher incidence compared with other primary liver cancers and need for regular diagnostic surveillance. As per Fact.MR, this cancer type addresses healthcare system focus on early detection, staging, and recurrence monitoring, supporting use of combined imaging, biomarker testing, and biopsy confirmation across oncology care pathways worldwide today.
- Epidemiology reference: National cancer surveillance programs document liver cancer incidence and subtype distribution.[4]
- Clinical focus: Fact.MR analysts note HCC drives most diagnostic testing volumes.
- Care pathways: HCC management requires repeated diagnostic evaluation.
Liver Cancer Diagnostics Market Drivers, Restraints, and Opportunities
Fact.MR analysis indicates that the liver cancer diagnostics market exists as a specialised segment within oncology diagnostics where technologies such as imaging, biomarker assays, and molecular tests are used to detect primary liver cancer and metastatic lesions at early stages. As per Fact.MR assessment, clinical workflows and diagnostic pathways for liver cancer are shaped by healthcare quality and screening recommendations from public health authorities, such as those outlined in the U.S. Centers for Disease Control and Prevention (CDC) guidelines on viral hepatitis screening and liver cancer risk, which inform provider practices for surveillance in high-risk populations. Fact.MR analysts observe that the current market scale is influenced by rising incidence of liver cancer associated with chronic hepatitis B and C, non-alcoholic fatty liver disease (NAFLD), and demographic shifts in many regions, driving demand for sensitive and specific diagnostic modalities.
Fact.MR is of the opinion that current market dynamics reflect a transition from conventional imaging and basic biomarker testing toward multi-parameter and genomic diagnostic solutions that enable earlier detection, risk stratification, and therapeutic monitoring. Based on Fact.MR assessment, traditional diagnostics such as ultrasound and alpha-fetoprotein (AFP) tests continue to be widely used because of established clinical protocols and cost considerations, but growth is occurring in segments where advanced imaging (MRI/CT), liquid biopsy assays, and molecular signature tests are specified to improve diagnostic accuracy and clinical decision support. Advanced diagnostic solutions generally carry higher per-unit pricing because of complexity, validation requirements, and integration with clinical workflows, meaning realised market value can grow even where overall procedural volumes expand at a measured pace. Fact.MR analysis suggests that this mix of technological progression and demand for precision diagnostics will shape near-term market evolution.
- National screening and risk guidelines: Fact.MR analysts note that public health frameworks such as the CDC liver cancer risk and screening guidance influence clinician adoption of diagnostic pathways for high-risk populations, affecting selection of imaging and biomarker tests.
- Shift to advanced diagnostic technologies: Based on Fact.MR assessment, increasing use of multi-modal imaging, circulating tumour DNA assays, and molecular diagnostic platforms supports demand for solutions that improve sensitivity and specificity relative to basic tests.
- Asia Pacific disease burden impact: Fact.MR opines that higher liver cancer incidence in China, India, Southeast Asia, and Japan-driven by prevalence of viral hepatitis and NAFLD-sustains regional demand for diagnostics across screening, early detection, and monitoring stages.
Regional Analysis
Based on regional assessment, the liver cancer diagnostics sector is analyzed across North America, Europe, East Asia, and other major healthcare markets, spanning more than 40 countries. Regional performance varies according to cancer screening penetration, availability of advanced diagnostic technologies, healthcare expenditure levels, and adoption of early detection protocols for hepatocellular carcinoma.
Liver Cancer Diagnostics Market CAGR Analysis by Country (2026-2036)
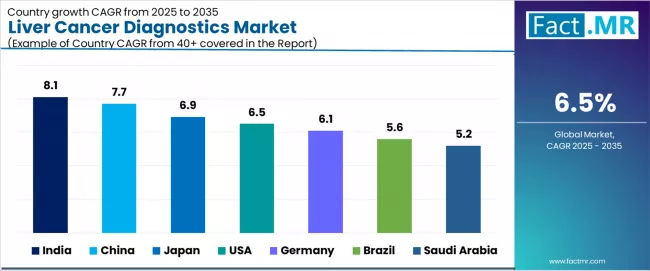
| Country | CAGR |
|---|---|
| United States | 9.4% |
| Germany | 8.9% |
| Japan | 8.6% |
| China | 8.1% |
| South Korea | 7.8% |
Source: Fact.MR analysis, based on proprietary forecasting models and primary research.
Demand for Liver Cancer Diagnostics in Asia Pacific
Asia Pacific reflects rising demand for liver cancer diagnostics supported by high disease prevalence expanding screening programs and hospital infrastructure growth. Diagnostic uptake is shaped by hepatitis related cancer incidence government led early detection initiatives and increased availability of imaging and biomarker tests. China Japan and South Korea anchor regional volumes through public hospital networks and oncology centers. Market expansion reflects wider use of ultrasound CT MRI and AFP testing integration within routine liver disease monitoring across tertiary and secondary care facilities regionwide.
- China: Demand for liver cancer diagnostics in China is projected to rise at 8.1% CAGR through 2036. Growth reflects high hepatocellular carcinoma incidence and expanded use of imaging and serum biomarker testing in public hospitals, as per Fact.MR.
- Japan: Demand for liver cancer diagnostics in Japan is projected to rise at 8.6% CAGR through 2036. Usage reflects established screening protocols and routine surveillance of high risk liver disease populations, according to Fact.MR.
- South Korea: Demand for liver cancer diagnostics in South Korea is projected to rise at 7.8% CAGR through 2036. Adoption is supported by national cancer screening programs and hospital based imaging diagnostics, as per Fact.MR.
Fact.MR’s analysis of the Liver Cancer Diagnostics Market in Asia Pacific consists of country-wise assessment that includes China, Japan, and South Korea. Readers can find screening coverage trends and diagnostic adoption insights.
Analysis On Liver Cancer Diagnostics Sales in North America
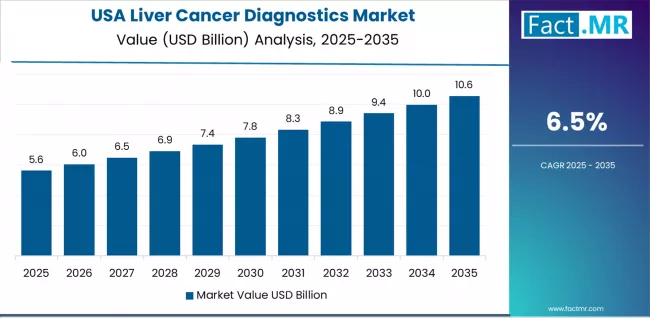
North America demonstrates strong sales of liver cancer diagnostics supported by advanced healthcare infrastructure and high diagnostic awareness. Demand is driven by imaging based detection biomarker testing and monitoring of chronic liver disease patients. The United States anchors regional consumption through oncology centers academic hospitals and outpatient diagnostic facilities. Procurement emphasizes accuracy early stage detection and integration with electronic health systems. Market growth reflects rising screening among at risk populations and continued investment in diagnostic imaging and laboratory services nationwide.
- United States: Demand for liver cancer diagnostics in the United States is projected to rise at 9.4% CAGR through 2036. Sales are supported by widespread use of imaging diagnostics biomarker assays and routine surveillance of chronic liver disease patients, as per Fact.MR.
Fact.MR’s analysis of the Liver Cancer Diagnostics Market in North America consists of country-wise assessment that includes the United States. Readers can find diagnostic utilization patterns and healthcare infrastructure references.
Outlook On Liver Cancer Diagnostics Demand In Europe
Europe reflects structured demand for liver cancer diagnostics shaped by public healthcare coverage and standardized cancer care pathways. Sales are influenced by early detection programs imaging utilization and biomarker testing within hospital settings. Germany anchors regional demand through large oncology networks and diagnostic laboratories. Procurement emphasizes guideline based testing and reimbursement alignment. Market expansion reflects demographic aging rising chronic liver disease burden and integration of diagnostic imaging and laboratory tests within national cancer control frameworks across European healthcare systems regionwide.
- Germany: Demand for liver cancer diagnostics in Germany is projected to rise at 8.9% CAGR through 2036. Adoption reflects standardized cancer screening pathways and strong hospital based diagnostic imaging capacity, according to Fact.MR.
Fact.MR’s analysis of the Liver Cancer Diagnostics Market in Europe consists of country-wise assessment that includes Germany. Readers can find reimbursement alignment and diagnostic protocol insights.
Competitive Landscape for Market Players
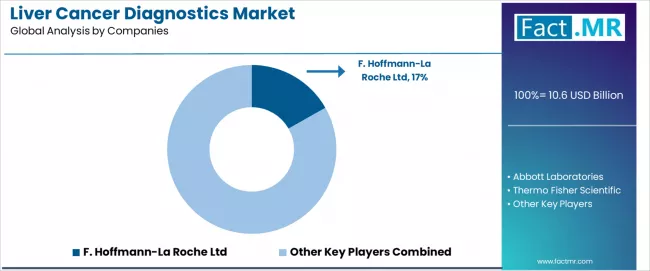
As per Fact.MR analysis, the 2026 liver cancer diagnostics market is defined by validated assay accuracy, system integration, and regulatory compliance, which determine competitive advantage. Abbott Laboratories and Roche Diagnostics lead with high-precision immunoassays and molecular testing platforms, which strengthens adoption in hospitals and clinical laboratories. Siemens Healthineers and GE Healthcare provide integrated imaging and diagnostic solutions with documented sensitivity and specificity, which improves early detection and patient outcomes. Philips Healthcare and Sysmex Corporation focus on validated workflow integration and multi-parameter monitoring, which enhances laboratory efficiency and reduces diagnostic errors. Hitachi Medical Systems and Canon Medical Systems emphasize high-resolution imaging combined with validated detection algorithms, which increases clinical confidence and supports multi-modality diagnostics. Bayer AG and Thermo Fisher Scientific provide assay kits and diagnostic platforms with verified regulatory compliance, which expands adoption across global healthcare networks. Across the market, validated diagnostic accuracy, system-level integration, and adherence to regulatory standards form enduring competitive moats, reducing reliance on pricing or isolated product claims.
Recent Industry Development
- In October 2025, GE Healthcare teamed up with two leading USA health systems, The Queen’s Health Systems in Honolulu, HI and Duke Health in Durham, NC, to help advance the development of GE HealthCare’s new AI-driven hospital operations software. [5]
- In January 2025, Bayer acquired Camelina Assets from Smart Earth Camelina Corp. to advance biofuels. [6]
Key Players in Liver Cancer Diagnostics Market
- Abbott Laboratories
- Roche Diagnostics
- Siemens Healthineers
- GE Healthcare
- Philips Healthcare
- Sysmex Corporation
- Hitachi Medical Systems
- Canon Medical Systems
- Bayer AG
- Thermo Fisher Scientific
Bibliography
- [1] Investing News Network. (2023). Low-carbon, high-grade nickel projects positioned to supply the EV market: Interview with Stuart Harshaw, CEO of Nickel Creek Platinum. Investing News Network.
- [2] U.S. Geological Survey. (2024). Nickel-Statistics and information. National Minerals Information Center, U.S. Department of the Interior; U.S. Department of the Interior. (2024). Mining laws and regulations. Office of Congressional and Legislative Affairs.
- [3] Mine Safety and Health Administration. (2024). Mine safety and health laws and regulations. U.S. Department of Labor.
- [4] U.S. Geological Survey. (2024). Nickel statistics and information. National Minerals Information Center, U.S. Department of the Interior.
- [5] GE HealthCare partners with industry players to expand healthcare solutions. (2025). Bing Search Results.
- [6] Bayer acquires camelina assets from Smart Earth Camelina Corp to advance biofuels. (2024). Bayer AG.
This Report Addresses
- Market intelligence to support structured strategic decision-making in oncology diagnostics, where surveillance protocols, clinical validation, and pathway integration shape adoption more than standalone test availability
- Global market size assessment and 10-year revenue forecasts from 2026 to 2036, developed using liver cancer incidence trends, diagnostic procedure volumes, and repeat-testing dynamics in high-risk populations
- Growth opportunity mapping across diagnostic test types, including blood biomarkers, imaging modalities, tissue biopsy, liquid biopsy, and molecular diagnostics, with emphasis on sensitivity, non-invasiveness, and clinical workflow fit
- Cancer-type-specific demand analysis covering hepatocellular carcinoma, cholangiocarcinoma, and other hepatic malignancies, linking disease prevalence to diagnostic intensity and follow-up frequency
- End-use benchmarking across hospitals and diagnostic centers, pathology laboratories, cancer research institutes, and other care settings, highlighting procurement drivers tied to screening programs and oncology care pathways
- Competitive landscape assessment benchmarking diagnostic manufacturers on assay accuracy, imaging performance, regulatory clearance, and integration with hospital information and oncology systems
- Technology roadmap tracking covering multi-modal imaging, blood-based biomarker panels, liquid biopsy platforms, and molecular diagnostics that support earlier detection and longitudinal disease monitoring
- Regulatory and clinical framework review aligned with diagnostic device oversight, screening guidance, and cancer control recommendations issued by the U.S. FDA and global health authorities
- Regional and country-level growth mapping across North America, Europe, and East Asia, highlighting differentiated adoption trajectories in the United States, Germany, Japan, China, and South Korea
- Strategic implications for diagnostic developers and healthcare providers focused on alignment with surveillance guidelines, reimbursement structures, and integrated diagnostic pathways
- Multi-format market report delivery in PDF, Excel datasets, PPT formats, and interactive dashboards for executives, clinical leaders, laboratory managers, and healthcare strategy teams
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 11.1 billion (2026) to USD 24.9 billion (2036), CAGR 8.5% |
| Market Definition | The liver cancer diagnostics market covers test and imaging methods for detecting, confirming, and monitoring malignant liver tumors. Diagnostics include blood biomarker assays, imaging tests (CT, MRI, Ultrasound), tissue biopsy, liquid biopsy, and molecular diagnostics. These are used in hospitals, diagnostic centers, and research institutes to support early detection, staging, therapy selection, and recurrence monitoring. Products are regulated by authorities such as the US FDA and WHO. |
| Test Type Segmentation | Blood Biomarker Tests, Imaging Tests (CT, MRI, Ultrasound), Tissue Biopsy, Liquid Biopsy, Molecular Diagnostics |
| Cancer Type Segmentation | Hepatocellular Carcinoma (HCC), Cholangiocarcinoma, Others (metastatic liver cancer, rare hepatic tumors) |
| End Use Segmentation | Hospitals and Diagnostic Centers, Cancer Research Institutes, Pathology Labs, Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Japan, South Korea, Indonesia, Australia & New Zealand, Germany, Italy, France, United Kingdom, Spain, Benelux, Nordics, Central & Eastern Europe, United States, Canada, Mexico, Brazil, Argentina, Chile, Kingdom of Saudi Arabia, United Arab Emirates, South Africa, Turkey |
| Key Companies Profiled | Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, GE Healthcare, Philips Healthcare, Sysmex Corporation, Hitachi Medical Systems, Canon Medical Systems, Bayer AG, Thermo Fisher Scientific |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up modeling validated through interviews with diagnostic manufacturers, oncologists, laboratory managers, and hospital procurement teams. Cross-checked with regulatory approvals, diagnostic procedure volumes, and public cancer statistics. |
Liver Cancer Diagnostics Market Analysis by Segments
-
Test Type :
- Blood Biomarker Tests
- Imaging Tests (CT, MRI, Ultrasound)
- Tissue Biopsy
- Liquid Biopsy
- Molecular Diagnostics
-
Cancer Type :
- Hepatocellular Carcinoma (HCC)
- Cholangiocarcinoma
- Others (Metastatic liver cancer, rare hepatic tumors)
-
End Use :
- Hospitals and Diagnostic Centers
- Cancer Research Institutes
- Pathology Labs
- Others
-
Region :
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
- Asia Pacific
- Frequently Asked Questions -
How large is the liver cancer diagnostics market in 2026?
The liver cancer diagnostics market is estimated at USD 11.1 billion in 2026, supported by expanded surveillance and imaging utilization in high-risk cohorts.
What will the liver cancer diagnostics market be worth by 2036?
By 2036, the market is projected to reach USD 24.9 billion, driven by higher adoption of advanced diagnostics and structured monitoring pathways.
What is the CAGR for the liver cancer diagnostics market from 2026 to 2036?
The market is forecast to grow at 8.5% CAGR during 2026–2036.
How much absolute value will the market add over the forecast period?
The market is expected to add approximately USD 14.6 billion in incremental value from 2026 to 2036.
What is structurally governing demand in liver cancer diagnostics?
Demand is structurally governed by surveillance protocols for high-risk populations, along with clinical validation, sensitivity thresholds, and pathway integration.
Which test type leads the liver cancer diagnostics market?
Blood biomarker tests lead with an estimated 40% share, driven by non-invasive sampling and suitability for repeat surveillance.
Why do blood biomarker tests hold the leading share?
They are easier to deploy at scale for routine monitoring and are commonly used alongside imaging in high-risk patient surveillance.
What is the largest cancer type segment in this market?
Hepatocellular carcinoma (HCC) leads with an estimated 45% share, reflecting higher incidence and recurring diagnostic follow-up needs.
Why does HCC drive the largest diagnostic demand?
HCC typically requires repeated testing for detection, staging, and recurrence monitoring, sustaining high procedure volumes.
What is driving early-period growth (2026 onward) in this market?
Early growth is supported by broader adoption of ultrasound and alpha-fetoprotein (AFP) testing in at-risk cohorts.
What factors support mid-period growth in liver cancer diagnostics?
Mid-period expansion aligns with increased CT and MRI utilization for lesion characterization and staging accuracy.
What technologies shape value realization in the later years of the forecast?
Later-year value is shaped by liquid biopsy and molecular diagnostics, supporting therapy selection and longitudinal monitoring.
Which country is projected to grow the fastest in liver cancer diagnostics?
The United States leads growth at 9.4% CAGR, supported by advanced imaging infrastructure and structured screening pathways.
What is Germany’s growth outlook for liver cancer diagnostics?
Germany is projected to grow at 8.9% CAGR, supported by standardized oncology diagnostics and pathway-driven testing.
How fast is Japan expected to grow in this market?
Japan is projected to expand at 8.6% CAGR, reflecting established HCC surveillance programs and ageing demographics.
What is the growth outlook for China in liver cancer diagnostics?
China is projected to grow at 8.1% CAGR, supported by expanding diagnostic capacity and a large high-risk population.
What is the growth outlook for South Korea?
South Korea is expected to grow at 7.8% CAGR, driven by hospital-based screening integration and early detection initiatives.
Which end users account for the bulk of diagnostic utilization?
Most utilization occurs in hospitals and diagnostic centers, supported by imaging access, oncology care pathways, and biopsy capability.
What is the key commercial adoption barrier in liver cancer diagnostics?
Adoption depends more on validated sensitivity thresholds, pathway integration, and reimbursement alignment than on simple availability of test formats.
What is the overall long-term outlook for the liver cancer diagnostics market?
The outlook remains strongly positive, with growth anchored in structured surveillance, rising diagnostic volumes, and increasing use of liquid biopsy and molecular platforms.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Desk Research Programme (Secondary Evidence)
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type , 2026 to 2036
- Blood Biomarker Tests
- Imaging Tests (CT, MRI, Ultrasound)
- Tissue Biopsy
- Liquid Biopsy
- Blood Biomarker Tests
- Y to o to Y Growth Trend Analysis By Test Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Test Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Cancer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Cancer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Cancer Type, 2026 to 2036
- Hepatocellular Carcinoma (HCC)
- Cholangiocarcinoma
- Hepatocellular Carcinoma (HCC)
- Y to o to Y Growth Trend Analysis By Cancer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Cancer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Type
- By Cancer Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Cancer Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Cancer Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Type
- By Cancer Type
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Roche Diagnostics
- Siemens Healthineers
- GE Healthcare
- Philips Healthcare
- Sysmex Corporation
- Hitachi Medical Systems
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Test Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Cancer Type, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Test Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Test Type
- Figure 6: Global Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Cancer Type, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Cancer Type
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Test Type , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Test Type
- Figure 23: North America Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Cancer Type, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Cancer Type
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Test Type , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Test Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Cancer Type, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Cancer Type
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Test Type , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Test Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Cancer Type, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Cancer Type
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Test Type , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Test Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Cancer Type, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Cancer Type
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Test Type , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Test Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Cancer Type, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Cancer Type
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Test Type , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Test Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Cancer Type, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Cancer Type
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Test Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Test Type , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Test Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Cancer Type, 2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Cancer Type, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Cancer Type
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis