GMP Storage Market Size, Share, Growth and Forecast (2026 - 2036)
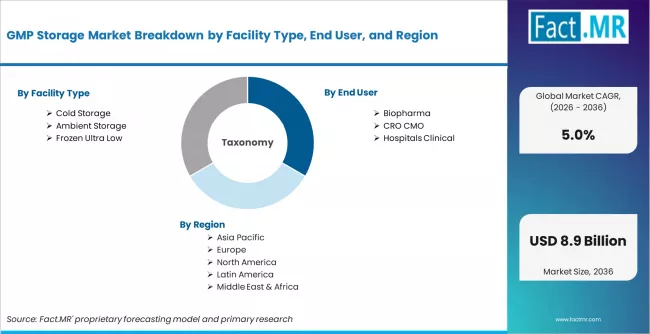
GMP Storage Market is segmented by Facility Type (Cold Storage, Ambient Storage, Frozen Ultra Low), End User (Biopharma, CRO CMO, Hospitals Clinical), Application (Pharmaceutical Storage, Biologics Storage, Clinical Trial Storage, Medical Device Storage, Raw Material Storage), and Region, with forecasts covering the period from 2026 to 2036.
According to Fact.MR estimates, the global gmp storage market market was valued at USD 5.2 billion in 2025. The market is projected to reach USD 5.5 billion in 2026 and is expected to grow to USD 8.9 billion by 2036, expanding at a CAGR of 5.0%. Cold Storage is anticipated to account for 50.0% of the product segment in 2026, while Biopharma is expected to remain the leading application with around 55.0% share.
GMP Storage Market Analysis and Forecast by Fact.MR
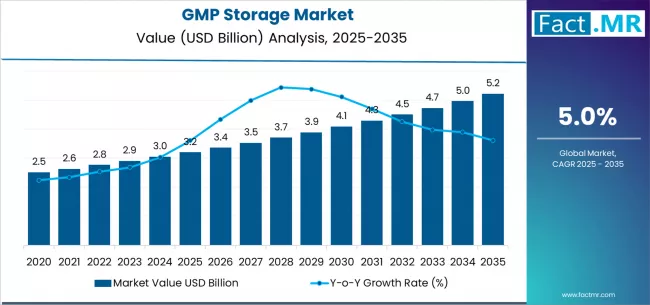
The GMP storage market, valued at USD 5.21 billion in 2025, is expanding at a 5.0% CAGR to reach USD 8.911 billion by 2036. GMP (Good Manufacturing Practice) storage encompasses temperature-controlled warehousing, ambient storage, and ultra-low frozen storage facilities that comply with pharmaceutical regulatory requirements for storing drugs, biologics, clinical trial materials, medical devices, and raw materials. Growth reflects the expanding global biopharmaceutical production base, increasing clinical trial activity, and tightening cold chain integrity requirements for biologics and cell/gene therapies. The USD 3.44 billion incremental opportunity between 2026 and 2036 is concentrated in cold and ultra-low storage for biologics across Asia, North America, and Europe.
Summary of GMP Storage Market
- Market Snapshot
- Market value in 2025: USD 5.21 billion
- Projected value in 2026: USD 5.471 billion; forecast to reach USD 8.911 billion by 2036
- CAGR from 2026 to 2036: 5.0%
- Incremental opportunity (2026 to 2036): USD 3.44 billion
- Cold storage leads facility type demand with 50.0% share in 2025
- India and China lead country-level growth, followed by Brazil and Germany
- Demand and Growth Drivers
- The biopharmaceutical industry is producing an expanding portfolio of temperature-sensitive biologics, biosimilars, and cell/gene therapies that require validated GMP cold chain storage from manufacturing through distribution.
- Clinical trial activity is increasing globally, with each trial requiring GMP-compliant storage for investigational products, comparator drugs, and biological samples throughout the study period.
- Regulatory inspections and audit requirements are tightening worldwide, mandating validated storage conditions with continuous temperature monitoring, deviation management, and documented quality systems.
- Pharmaceutical companies are outsourcing storage to specialized third-party logistics providers to reduce capital expenditure and access validated GMP storage capacity without self-build investment.
- Product and Segment View
- Cold storage holds 50.0% of facility type demand, driven by the growing proportion of temperature-sensitive biologics requiring 2 to 8 degrees C storage.
- Biopharma accounts for 55.0% of end-user demand, reflecting the concentration of GMP storage requirements in biologic drug product and substance storage.
- Pharmaceutical storage holds 45.2% of application share, covering finished drug product storage under validated GMP conditions.
- Key segmentation includes:
- Facility Type: Cold Storage, Ambient Storage, Frozen Ultra Low
- End User: Biopharma, CRO/CMO, Hospitals/Clinical
- Application: Pharmaceutical Storage, Biologics Storage, Clinical Trial Storage, Medical Device Storage, Raw Material Storage
- Distribution Channel: Direct Contracts, Third Party Logistics, Digital Platforms
- Geography and Competitive Outlook
- North America is the largest revenue region, with the USA contributing USD 2.023 billion in 2025.
- South Asia is the fastest-growing region, with India recording the highest country CAGR driven by biopharmaceutical manufacturing expansion.
- Europe maintains strong growth, with Germany leading through pharmaceutical manufacturing density.
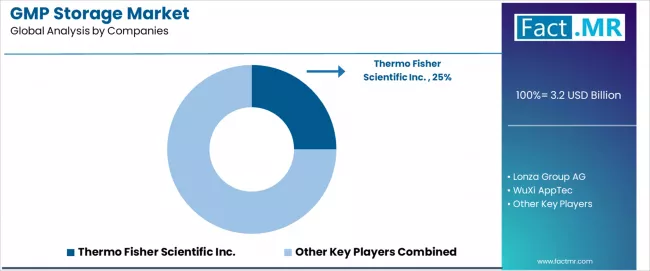
- Thermo Fisher Scientific Inc. leads the competitive field with a 25.0% market share, followed by Lonza Group, WuXi AppTec, and Catalent.
- Analyst Opinion
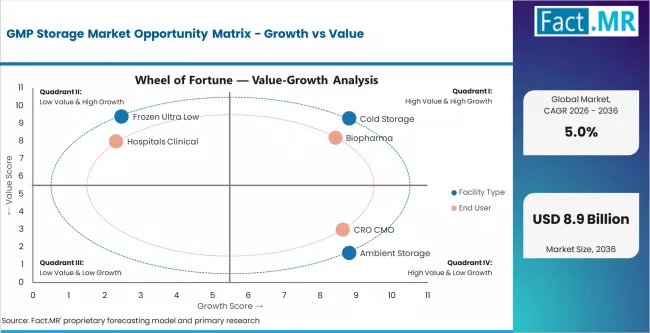
- The GMP storage market is an infrastructure category where growth is tied to the expanding volume and complexity of pharmaceutical products requiring validated storage conditions. The shift toward biologics, cell therapies, and mRNA-based products is increasing the proportion of the pharmaceutical portfolio requiring cold and ultra-low storage, creating capacity demand that exceeds ambient storage growth rates. Fact.MR expects the fastest growth in ultra-low frozen storage for cell/gene therapy materials and in cold storage expansion across India and China, where biopharmaceutical manufacturing capacity is scaling rapidly. The outsourcing trend toward third-party GMP logistics providers is creating a commercial model where storage capacity is purchased as a service rather than built as owned infrastructure.
Key Growth Drivers, Constraints, and Opportunities
Key Factors Driving Growth
- Biologics portfolio growth: The expanding global portfolio of biologic drugs requiring 2 to 8 degrees C storage creates proportional cold chain storage demand that grows with each new product approval and market entry.
- Clinical trial material storage: Increasing global clinical trial activity, particularly multi-center international trials, generates distributed GMP storage requirements across multiple countries and regions.
- Regulatory tightening: FDA, EMA, and other regulatory agencies are increasing inspection frequency and data integrity requirements for pharmaceutical storage facilities, driving investment in validated storage systems.
Key Market Constraints
- Capital intensity: GMP-compliant cold storage facility construction requires significant capital investment in temperature control, monitoring systems, backup power, and regulatory validation.
- Energy costs: Cold and ultra-low storage facilities consume substantial energy for continuous temperature maintenance, creating operating cost pressure that is amplified by energy price volatility.
- Regulatory complexity: Storage facilities must maintain compliance across multiple regulatory jurisdictions with varying GMP requirements, creating compliance management complexity for global storage operations.
Key Opportunity Areas
- Cell/gene therapy storage: The emergence of cell and gene therapies requiring ultra-low (minus 80 degrees C) and cryogenic storage creates a premium storage tier with high per-unit pricing and specialized infrastructure requirements.
- Emerging market storage capacity: Biopharmaceutical manufacturing expansion in India, China, and Southeast Asia is creating demand for local GMP storage infrastructure that currently requires import from established markets.
- Digital storage management: IoT-connected monitoring, blockchain-based chain of custody documentation, and predictive analytics for storage condition management are creating technology-differentiated service offerings.
Segment-wise Analysis of the GMP Storage Market
- Cold storage leads facility type demand at 50.0%, driven by biologic product storage requirements.
- Biopharma holds 55.0% of end-user share, reflecting the concentration of GMP storage in biologic drug storage.
- Pharmaceutical storage accounts for 45.2% of application demand.
Which facility type dominates the GMP Storage Market?
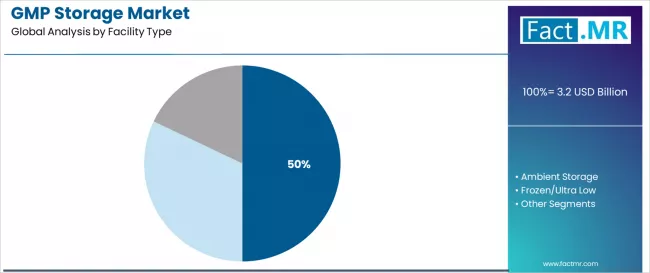
Cold storage holds a 50.0% share of facility type demand in 2025. The share reflects the growing proportion of pharmaceutical products requiring 2 to 8 degrees C storage, driven by the expanding biologics portfolio. Cold storage facilities require continuous temperature monitoring, validated HVAC systems, backup power infrastructure, and regulatory documentation that create higher capital and operating costs than ambient alternatives.
Ambient storage accounts for the second-largest share, serving stable pharmaceutical products, medical devices, and raw materials that do not require refrigeration. Ultra-low and frozen storage is the fastest-growing segment, driven by cell/gene therapy materials.
Which e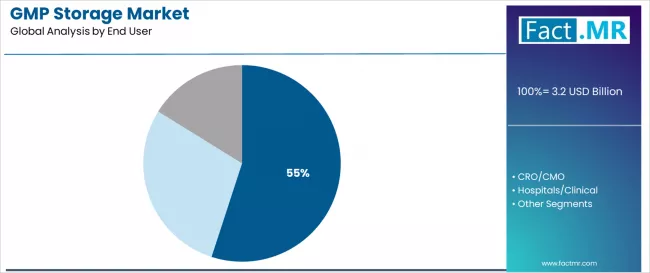 nd user dominates the GMP Storage Market?
nd user dominates the GMP Storage Market?
Biopharma accounts for 55.0% of end-user demand. Biopharmaceutical companies require validated GMP storage for drug substances, drug products, reference standards, and stability samples throughout product lifecycles. The outsourcing trend is shifting biopharma storage from owned warehouses to third-party providers that offer flexible capacity.
CRO/CMO organizations represent the second-largest end user, managing clinical trial material storage and contract manufacturing intermediate storage. Hospitals and clinical settings contribute a smaller share for investigational product storage.
Which application dominates the GMP Storage Market?
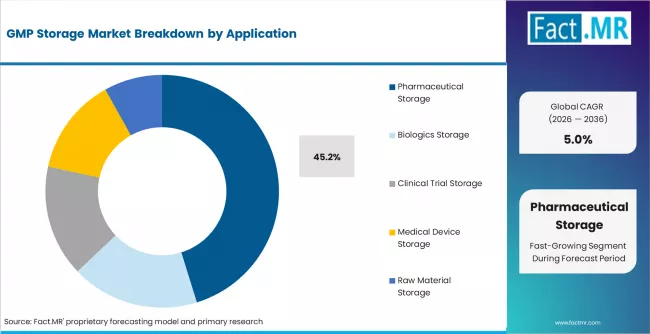
Pharmaceutical storage holds 45.2% of application demand. This category covers finished drug product storage under validated conditions, including temperature-controlled warehousing between manufacturing and distribution. The volume of pharmaceutical products requiring GMP storage grows proportionally with global pharmaceutical production output.
Biologics storage is the fastest-growing application, driven by the temperature sensitivity of monoclonal antibodies, vaccines, and cell/gene therapy products. Clinical trial storage is a specialized application requiring multi-country storage coordination.
Which Product Trend is Shaping the Next Phase of Growth in the GMP Storage Market?
Cell and gene therapy storage requirements are shaping the next capacity investment phase. Cell therapies (CAR-T, TCR-T) and gene therapies require ultra-low storage at minus 80 degrees C or cryogenic storage in liquid nitrogen, creating specialized infrastructure requirements that exceed conventional pharmaceutical cold chain capability. Each cell therapy product lot requires patient-specific storage with chain of custody documentation, creating per-patient storage demand at manufacturing, transit, and clinical sites. The commercial expansion of approved cell therapies (Kymriah, Yescarta, Abecma) and gene therapies is generating growing demand for ultra-low storage capacity.
The outsourcing of GMP storage to specialized third-party logistics providers is accelerating as pharmaceutical companies seek to reduce fixed infrastructure costs and access validated capacity on demand. Providers including DHL Life Sciences, Cryoport, and Marken are expanding GMP cold chain storage networks across North America, Europe, and Asia to serve multinational pharmaceutical clients. Fact.MR projects that third-party GMP storage will grow faster than in-house storage, reaching over 40% of total GMP storage capacity by 2036 as pharmaceutical companies optimize asset-light supply chain models.
Regional Outlook Across Key Markets
.webp)
- North America is the largest revenue region, with the USA contributing USD 2.023 billion in 2025.
- South Asia is the fastest-growing region, with India recording the highest country CAGR.
- East Asia records strong growth, with China expanding biopharmaceutical manufacturing capacity.
- Europe maintains steady growth, shaped by pharmaceutical manufacturing density.
CAGR Table
| Country | CAGR (%) |
|---|---|
| India | 7.2% |
| China | 6.1% |
| Brazil | 5.0% |
| Germany | 4.8% |
| USA | 4.6% |
| South Korea | 4.4% |
| Japan | 4.3% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research.
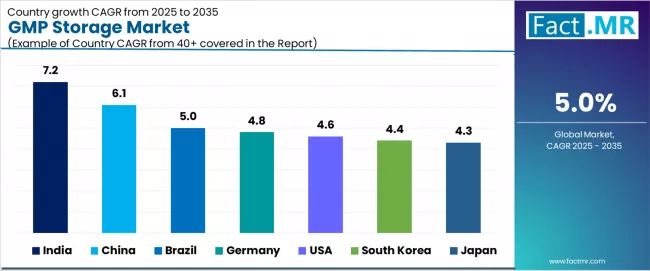
India: biopharmaceutical manufacturing and storage capacity gap
India records the highest country-level CAGR driven by rapid biopharmaceutical and vaccine manufacturing expansion that is creating demand for GMP storage infrastructure. The country is a major vaccine producer and biosimilar manufacturer, and the growth of contract manufacturing for global pharmaceutical companies is generating storage requirements that exceed existing capacity. The government Pharma Vision 2030 initiative is supporting pharmaceutical infrastructure development.
- Growth anchor: Biopharmaceutical manufacturing expansion is creating GMP storage capacity demand that exceeds current infrastructure.
USA: biologics portfolio and cell therapy storage demand
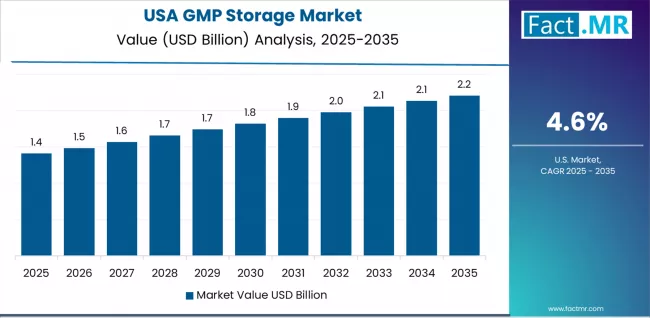
The USA contributes USD 2.023 billion in 2025. Growth reflects the concentration of biologic drug production, cell/gene therapy manufacturing, and clinical trial activity that generates ongoing GMP storage demand. The expansion of ultra-low storage capacity for cell therapy products is a near-term investment priority for storage providers. Third-party GMP logistics providers are expanding US cold chain capacity.
- Growth anchor: Biologics production growth and cell/gene therapy storage requirements drive capacity investment.
China: biopharmaceutical scale-up and cold chain modernization
China records strong growth driven by the rapid scale-up of domestic biopharmaceutical manufacturing, the expansion of clinical trial activity under NMPA modernization, and government investment in pharmaceutical cold chain infrastructure. Chinese biopharmaceutical companies are building GMP storage capacity both domestically and internationally.
- Growth anchor: Biopharmaceutical manufacturing scale-up and cold chain modernization create structured storage demand.
Germany: pharmaceutical manufacturing hub
Germany leads European GMP storage growth supported by its position as a major pharmaceutical and biopharmaceutical manufacturing hub. The concentration of drug substance and drug product manufacturing creates co-located GMP storage demand. German 3PL providers serve as European distribution hubs for multinational pharmaceutical companies.
- Growth anchor: Manufacturing hub concentration and European distribution role create steady storage demand.
Competitive Benchmarking and Company Positioning
GMP Storage Market Analysis By Company
- Thermo Fisher Scientific leads with 25.0% market share, leveraging its integrated pharmaceutical services portfolio including storage, distribution, and clinical trial logistics through its Fisher BioServices and Patheon divisions.
- Lonza Group and WuXi AppTec compete in the CDMO-linked storage tier, where manufacturing and storage services are bundled within contract manufacturing relationships.
- DHL Life Sciences, Cryoport, and Marken maintain positions as specialized pharmaceutical third-party logistics providers offering GMP storage as a service across global networks.
The competitive field combines integrated pharmaceutical services companies (Thermo Fisher, Catalent), CDMOs with co-located storage (Lonza, WuXi AppTec, Samsung Biologics), and specialized pharmaceutical 3PL providers (DHL Life Sciences, Cryoport, Marken). Thermo Fisher leads through its integrated platform approach.
Competition in the 3PL tier is intensifying as pharmaceutical cold chain logistics providers invest in GMP storage capacity expansion across major pharmaceutical manufacturing corridors. Cryoport has emerged as a specialist in cell/gene therapy cold chain, commanding premium pricing for ultra-low and cryogenic storage services.
Regional storage providers in India and China are gaining share by offering GMP-compliant storage at cost points below multinational providers, serving domestic pharmaceutical manufacturers and contract research organizations.
Key Companies in the GMP Storage Market
Thermo Fisher Scientific Inc., Lonza Group AG, WuXi AppTec, Catalent Inc., Samsung Biologics, Charles River Laboratories, DHL Life Sciences, Cryoport Inc., Marken Ltd., and BioLife Solutions Inc. are the principal companies operating in the global GMP storage market.
Recent Industry Developments
- Thermo Fisher: GMP cold storage capacity expansion (2025)
- Thermo Fisher Scientific expanded its GMP cold storage capacity in 2025, commissioning new temperature-controlled warehouse facilities in the USA and Singapore to serve growing biopharmaceutical client demand.
- Cryoport: cell therapy storage network expansion (2025)
- Cryoport expanded its ultra-low temperature storage network in 2025, adding cryogenic storage capacity at facilities across North America, Europe, and Asia to serve the growing cell and gene therapy manufacturing and distribution market.
- DHL Life Sciences: India GMP storage facility commissioning (2025)
- DHL Life Sciences commissioned a new GMP-compliant pharmaceutical storage facility in India in 2025, targeting the growing storage demand from Indian biopharmaceutical manufacturers and multinational companies with Indian manufacturing operations.
- Samsung Biologics: integrated CDMO storage expansion (2026)
- Samsung Biologics expanded integrated GMP storage capacity at its Incheon manufacturing campus in early 2026, co-locating cold chain storage with biologic drug substance and drug product manufacturing lines.
Leading Companies Shaping the GMP Storage Market
-
Global Players
- Thermo Fisher Scientific Inc.
- Lonza Group AG
- WuXi AppTec
- Catalent, Inc.
- Samsung Biologics
- Charles River Laboratories
-
Regional Players
- DHL Life Sciences
- Cryoport, Inc.
- Marken Ltd.
- BioLife Solutions, Inc.
-
Emerging / Start-up Players
- FedEx Life Sciences
- UPS Healthcare
- Biostor
Sources and Research References
- Thermo Fisher Scientific Annual Report 2024 and Pharma Services Division Presentations
- Lonza Group Annual Report 2024
- FDA GMP Inspection and Compliance Reports
- EMA GMP Guidelines for Storage and Distribution
- Cryoport Inc. Annual Report 2024
- DHL Life Sciences Global Network Documentation
All numerical data in this report is derived from Fact.MR proprietary research. Source references are provided for contextual and directional validation only.
Key Questions This Report Addresses
- What is the projected size of the GMP storage market by 2036?
- Which facility type holds the largest share?
- What CAGR is expected from 2026 to 2036?
- Which country is growing fastest?
- Who is the leading company?
- What is the incremental opportunity between 2026 and 2036?
- How is cell/gene therapy affecting storage demand?
- What role does outsourcing play in GMP storage growth?
GMP Storage Market Definition
The GMP storage market covers regulatory-compliant warehousing and storage facilities that maintain pharmaceutical products, biologics, clinical trial materials, and raw materials under validated temperature and environmental conditions in accordance with Good Manufacturing Practice requirements.
GMP Storage Market Inclusions
- Cold storage facilities (2 to 8 degrees C) for biologics and temperature-sensitive pharmaceuticals
- Ambient storage (15 to 25 degrees C) for stable pharmaceutical products and medical devices
- Ultra-low and frozen storage (minus 20 to minus 80 degrees C) for cell/gene therapy materials and biological samples
- Clinical trial material storage services provided by CROs, CMOs, and third-party logistics providers
GMP Storage Market Exclusions
- Pharmaceutical manufacturing equipment and production facilities
- Drug distribution and last-mile delivery logistics
- Laboratory storage equipment (freezers, refrigerators) as capital goods
- Non-pharmaceutical cold chain storage (food, industrial)
GMP Storage Market Research Methodology
- Bottom-up revenue modeling using GMP storage capacity data, storage pricing per pallet/volume, and pharmaceutical production volumes across 15+ countries
- Primary interviews with pharmaceutical supply chain directors, GMP warehouse operators, and third-party logistics providers
- Cross-referencing pharmaceutical manufacturing facility databases and regulatory inspection records
- Validation through 3PL service pricing, warehouse capacity utilization data, and pharmaceutical outsourcing contract values
- Forecasting calibrated against biopharmaceutical pipeline projections, clinical trial activity, and cold chain investment announcements
- Company-level revenue triangulation using annual reports, investor presentations, and logistics division disclosures
Scope of Analysis
| Parameter | Details |
|---|---|
| Market size (2025) | USD 5.21 billion |
| Forecast period | 2026 to 2036 |
| CAGR | 5.0% |
| Base year | 2025 |
| Units | USD Billion |
| Segments covered | Facility Type (Cold, Ambient, Frozen/Ultra-Low); End User (Biopharma, CRO/CMO, Hospitals/Clinical); Application (Pharmaceutical, Biologics, Clinical Trial, Medical Device, Raw Material); Distribution Channel (Direct Contracts, 3PL, Digital) |
| Regions covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries profiled | USA, India, China, Germany, Brazil, South Korea, Japan |
| Companies profiled | Thermo Fisher Scientific, Lonza Group, WuXi AppTec, Catalent, Samsung Biologics, DHL Life Sciences |
| Forecasting approach | Bottom-up revenue modeling validated through primary research, pharmaceutical production analysis, and company-level financial triangulation |
Market Segmentation Analysis
-
GMP Storage Market Market Segmented by Facility Type:
- Cold Storage
- Refrigerated Storage
- Controlled Temperature Storage
- Ambient Storage
- Standard Warehouse Storage
- Controlled Room Temperature Storage
- Frozen Ultra Low
- Deep Freezing Storage
- Cryogenic Storage
- Cold Storage
-
GMP Storage Market Market Segmented by End User:
- Biopharma
- Biologics Manufacturers
- Vaccine Manufacturers
- CRO CMO
- Clinical Research Organizations
- Contract Manufacturing Organizations
- Hospitals Clinical
- Hospitals
- Clinical Trial Sites
- Biopharma
-
GMP Storage Market Market Segmented by Application:
- Pharmaceutical Storage
- Drug Products
- Finished Drugs
- Intermediate Drugs
- Biologics Storage
- Biopharmaceuticals
- Vaccines
- Cell and Gene Therapies
- Clinical Trial Storage
- Investigational Products
- Phase One
- Phase Two and Three
- Medical Device Storage
- Device Components
- Sterile Devices
- Non Sterile Devices
- Raw Material Storage
- Active Ingredients
- Excipients
- Packaging Materials
- Pharmaceutical Storage
-
GMP Storage Market Market Segmented by Distribution Channel:
- Direct Contracts
- Long Term Agreements
- Dedicated Storage
- Integrated Services
- Third Party Logistics
- Specialty Logistics Providers
- Pharma Logistics
- Biologics Logistics
- Digital Platforms
- Booking Platforms
- On Demand Storage
- Inventory Management
- Direct Contracts
-
GMP Storage Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
What is the current size of the GMP storage market?
The global GMP storage market is valued at USD 5.21 billion in 2025.
What growth rate is expected?
The market is projected to grow at a CAGR of 5.0% from 2026 to 2036.
What will the market be worth by 2036?
The market is forecast to reach USD 8.911 billion by 2036.
Which facility type leads?
Cold storage leads with 50.0% of facility type demand in 2025.
Which country is growing fastest?
India records the highest country-level CAGR.
Who is the market leader?
Thermo Fisher Scientific leads with a 25.0% market share in 2025.
What is the USA market value?
The USA contributes USD 2.023 billion in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Facility Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Facility Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Facility Type , 2026 to 2036
- Cold Storage
- Refrigerated Storage
- Controlled Temperature Storage
- Ambient Storage
- Standard Warehouse Storage
- Controlled Room Temperature Storage
- Frozen Ultra Low
- Deep Freezing Storage
- Cryogenic Storage
- Cold Storage
- Y to o to Y Growth Trend Analysis By Facility Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Facility Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Biopharma
- Biologics Manufacturers
- Vaccine Manufacturers
- CRO CMO
- Clinical Research Organizations
- Contract Manufacturing Organizations
- Hospitals Clinical
- Hospitals
- Clinical Trial Sites
- Biopharma
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Pharmaceutical Storage
- Drug Products
- Finished Drugs
- Intermediate Drugs
- Biologics Storage
- Biopharmaceuticals
- Vaccines
- Cell and Gene Therapies
- Clinical Trial Storage
- Investigational Products
- Phase One
- Phase Two and Three
- Medical Device Storage
- Device Components
- Sterile Devices
- Non Sterile Devices
- Raw Material Storage
- Active Ingredients
- Excipients
- Packaging Materials
- Pharmaceutical Storage
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct Contracts
- Long Term Agreements
- Dedicated Storage
- Integrated Services
- Third Party Logistics
- Specialty Logistics Providers
- Pharma Logistics
- Biologics Logistics
- Digital Platforms
- Booking Platforms
- On Demand Storage
- Inventory Management
- Direct Contracts
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Facility Type
- By End User
- By Application
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Thermo Fisher Scientific Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Lonza Group AG
- WuXi AppTec
- Catalent, Inc.
- Samsung Biologics
- Charles River Laboratories International, Inc.
- DHL Life Sciences
- Cryoport, Inc.
- Marken Ltd.
- BioLife Solutions, Inc.
- FedEx Life Sciences
- UPS Healthcare
- Fisher Bioservices
- Biostor
- Q2 Solutions
- Thermo Fisher Scientific Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Facility Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Facility Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Facility Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Facility Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Facility Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Facility Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Facility Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Facility Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Facility Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Facility Type,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Facility Type
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Distribution Channel
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 19: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 22: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Facility Type, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Facility Type,2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Facility Type
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Distribution Channel
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Facility Type, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Facility Type,2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by Facility Type
- Figure 42: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 44: Latin America Market Attractiveness Analysis by End User
- Figure 45: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Application
- Figure 48: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Facility Type, 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Facility Type,2026 to 2036
- Figure 54: Western Europe Market Attractiveness Analysis by Facility Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 57: Western Europe Market Attractiveness Analysis by End User
- Figure 58: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 60: Western Europe Market Attractiveness Analysis by Application
- Figure 61: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Facility Type, 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Facility Type,2026 to 2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Facility Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by End User
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Application
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Facility Type, 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Facility Type,2026 to 2036
- Figure 80: East Asia Market Attractiveness Analysis by Facility Type
- Figure 81: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 83: East Asia Market Attractiveness Analysis by End User
- Figure 84: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 86: East Asia Market Attractiveness Analysis by Application
- Figure 87: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 89: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Facility Type, 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Facility Type,2026 to 2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Facility Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Facility Type, 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Facility Type,2026 to 2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Facility Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis