Immune Checkpoint Inhibitor Market Size, Share, Growth and Forecast (2026 - 2036)
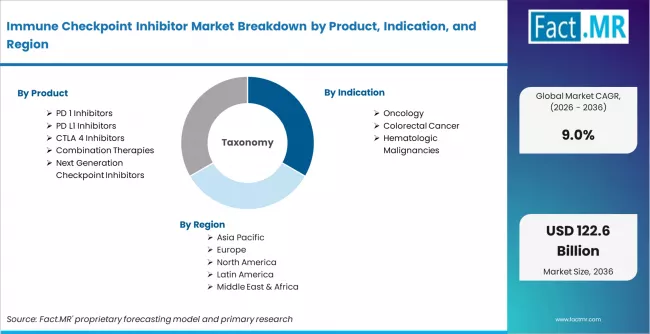
Immune Checkpoint Inhibitor Market is segmented by Product (PD 1 Inhibitors, PD L1 Inhibitors, CTLA 4 Inhibitors, Combination Therapies, Next Generation Checkpoint Inhibitors), Indication (Oncology, Colorectal Cancer, Hematologic Malignancies), Therapy Line (First Line Therapy, Second Line Therapy, Third Line and Beyond), and Region, with forecasts covering the period from 2026 to 2036.
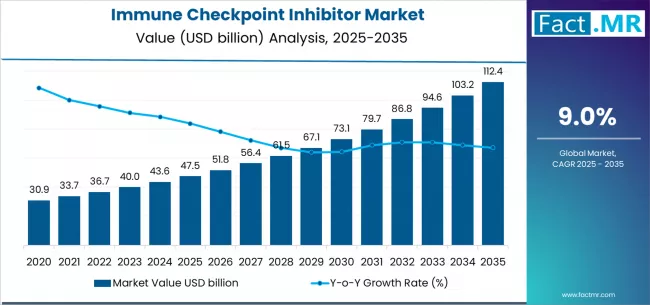
According to Fact.MR estimates, the global immune checkpoint inhibitor market market was valued at USD 47.5 billion in 2025. The market is projected to reach USD 51.8 billion in 2026 and is expected to grow to USD 122.6 billion by 2036, expanding at a CAGR of 9.0%. PD 1 Inhibitors is anticipated to account for 42.6% of the product segment in 2026, while Oncology is expected to remain the leading application with around 68.9% share.
Immune Checkpoint Inhibitor Market Analysis and Forecast by Fact.MR
- The global immune checkpoint inhibitor market is estimated at USD 47.5 billion in 2025 and is forecast to reach USD 51.8 billion in 2026 before expanding to USD 122.6 billion by 2036, progressing at a CAGR of 9.0% over the 2026 to 2036 forecast period.
- PD-1 Inhibitors is anticipated to account for 42.6% share of the product segment in 2026, supported by first-mover advantage, broadest approved indication coverage, and dominant positioning across first-line oncology treatment protocols globally.
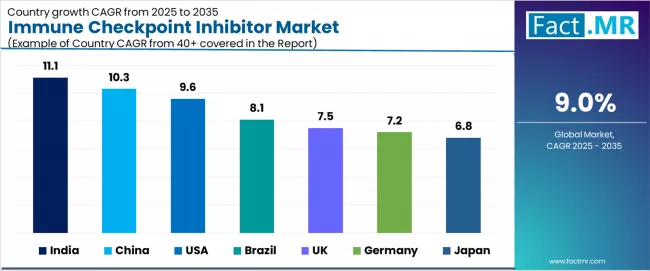
- India (11.1%) and China (10.3%) are the fastest growing country markets through 2036.
Summary of Immune Checkpoint Inhibitor Market
- Market Snapshot
- In 2025, the global Immune Checkpoint Inhibitor Market was valued at approximately USD 47.5 billion.
- The market is estimated to reach USD 51.8 billion in 2026 and is projected to attain USD 122.6 billion by 2036.
- The immune checkpoint inhibitor market is likely to expand at a CAGR of 9.0% during the forecast period.
- The market is anticipated to create an absolute dollar opportunity of USD 70.8 billion between 2026 and 2036.
- PD-1 inhibitors account for 42.6% of market share in 2026.
- India (11.1%) and China (10.3%) are the key growth markets during the forecast period.
- Demand and Growth Drivers
- Continued indication expansion for approved PD-1 and PD-L1 inhibitors across new tumor types and earlier treatment lines is broadening the eligible patient population and increasing per-indication treatment volumes.
- Shift of checkpoint immunotherapy from second-line and third-line to first-line treatment positioning in major cancer types is increasing the proportion of newly diagnosed patients receiving checkpoint inhibitors.
- Combination regimen development pairing checkpoint inhibitors with chemotherapy, targeted agents, and other immunotherapy modalities is improving clinical outcomes and supporting treatment protocol adoption.
- Rising institutional procurement volumes across hospitals, academic facilities, and government programs create a sustained base-level demand that is less sensitive to short-term economic cycles.
- Expanding distribution through specialized channels and direct-to-institution sales models is improving product accessibility across underpenetrated geographies.
- Product and Segment View
- PD-1 Inhibitors accounts for 42.6% of the product segment in 2026, maintaining the leading position due to established usage patterns and broad commercial adoption.
- Monoclonal Antibodies represents 73.2% of the drug class segment in 2026, supported by the largest concentration of end-use demand and procurement volumes.
- The immune checkpoint inhibitor market is segmented by product, indication, therapy line, mechanism target, drug class, and region.
- Geography and Competitive Outlook
- India leads growth at 11.1%, supported by infrastructure expansion and increasing institutional adoption.
- North America reflects a mature but steady demand profile, with the USA at 9.6% supported by replacement demand and established clinical protocols.
- Europe maintains stable growth, with Germany at 7.2% supported by regulatory standards and established institutional procurement frameworks.
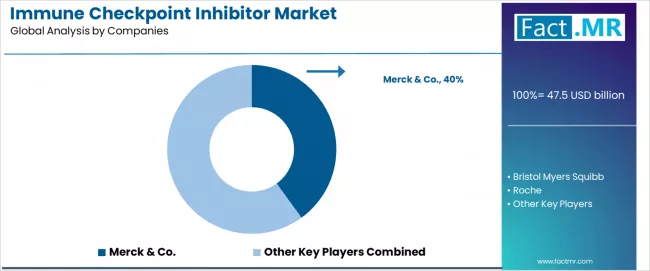
- Merck & Co., Bristol Myers Squibb, Roche hold strong positions through broad product portfolios and established distribution networks.
- Analyst Opinion
- Shambhu Nath Jha, Principal Consultant at Fact.MR, says 'The immune checkpoint inhibitor market is one of the largest and fastest-growing therapeutic categories in oncology. Growth reflects the continued expansion of approved indications for PD-1 and PD-L1 inhibitors, the shift of checkpoint immunotherapy from second-line to first-line treatment positioning, and the growing adoption of combination regimens that pair checkpoint inhibitors with chemotherapy, targeted therapy, or other immunotherapy agents. Merck's Keytruda (pembrolizumab) holds the dominant commercial position, supported by the broadest approved indication portfolio across solid tumor types. Bristol Myers Squibb's Opdivo (nivolumab) competes strongly in specific indications and combination regimens. Chinese domestic biosimilar and me-too checkpoint inhibitor developers, including BeiGene and Innovent Biologics, are creating pricing pressure in Asia Pacific and positioning for global market entry. The market's structural growth constraint is affordability. In emerging markets, the high cost of branded checkpoint inhibitors limits access to a fraction of eligible patients, creating a significant treatment gap that biosimilar and domestic manufacturer entry is beginning to address.'
Key Growth Drivers, Constraints, and Opportunities
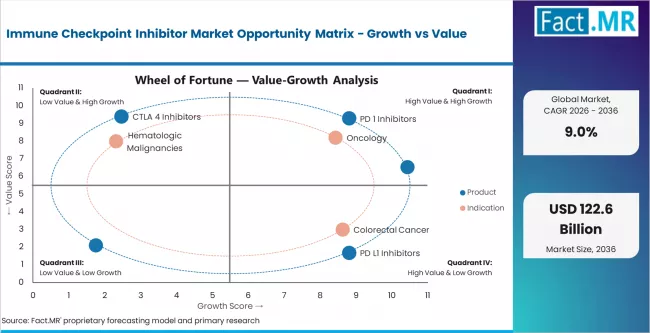
Key Factors Driving Growth
- Continued indication expansion for approved PD-1 and PD-L1 inhibitors across new tumor types and earlier treatment lines is broadening the eligible patient population and increasing per-indication treatment volumes.
- Shift of checkpoint immunotherapy from second-line and third-line to first-line treatment positioning in major cancer types is increasing the proportion of newly diagnosed patients receiving checkpoint inhibitors.
- Combination regimen development pairing checkpoint inhibitors with chemotherapy, targeted agents, and other immunotherapy modalities is improving clinical outcomes and supporting treatment protocol adoption.
Key Market Constraints
- High per-patient treatment costs limit access in emerging markets and create payer pressure in established markets, particularly for long-duration therapy regimens.
- Primary and acquired resistance to checkpoint inhibitors reduces treatment effectiveness in a significant proportion of patients, limiting long-term response rates.
- Immune-related adverse events require specialized management expertise and create additional healthcare costs that affect treatment economics and clinical adoption patterns.
Key Opportunity Areas
- Biosimilar and domestic checkpoint inhibitor entry in China, India, and other emerging markets is reducing per-dose costs and expanding patient access beyond current penetration levels.
- Next-generation checkpoint inhibitors targeting novel immune co-stimulatory and co-inhibitory pathways create pipeline opportunities for differentiated therapy positioning.
- Biomarker-guided patient selection using PD-L1 expression, tumor mutational burden, and microsatellite instability testing is improving treatment response rates and supporting evidence-based prescribing.
Segment-wise Analysis of the Immune Checkpoint Inhibitor Market
- PD-1 Inhibitors holds 42.6% of the product segment in 2026.
- Monoclonal Antibodies represents 73.2% of the drug class segment in 2026.
The immune checkpoint inhibitor market is segmented by product, indication, therapy line, mechanism target, drug class, and region.
Which Product Segment Leads the Immune Checkpoint Inhibitor Market?
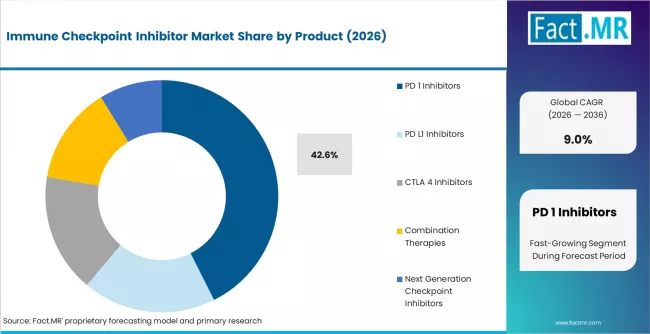
PD-1 Inhibitors accounts for 42.6% of the product segment in 2026. This position reflects first-mover advantage, broadest approved indication coverage, and dominant positioning across first-line oncology treatment protocols globally. The segment benefits from an established base, broad geographic adoption, and consistent demand from institutional and individual buyer channels.
Which Drug Class Segment Leads the Immune Checkpoint Inhibitor Market?
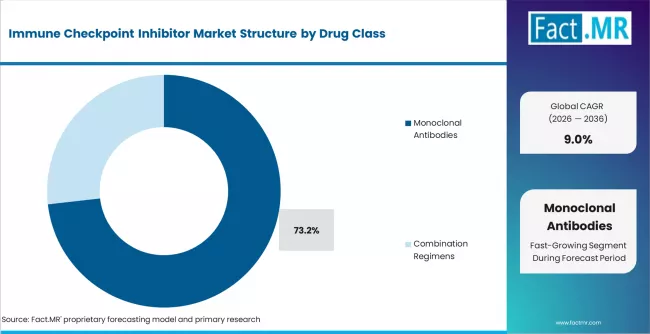
Monoclonal Antibodies is expected to account for 73.2% of the drug class segment in 2026. This segment represents the largest concentration of purchasing activity and maintains its position through established procurement cycles and direct alignment with the primary use cases.
Which Product Trend is Shaping the Next Phase of Growth in the Immune Checkpoint Inhibitor Market?
Combination therapy regimens that pair PD-1 or PD-L1 inhibitors with chemotherapy, targeted agents, or other immunotherapy modalities represent the primary growth vector in the checkpoint inhibitor market. Clinical trials are consistently demonstrating improved progression-free and overall survival when checkpoint inhibitors are combined with complementary mechanisms of action compared to monotherapy. Combination protocols are now standard of care in multiple first-line solid tumor indications, including non-small cell lung cancer, renal cell carcinoma, and head and neck squamous cell carcinoma. The commercial impact is significant because combination regimens increase per-patient drug costs, extend treatment duration, and expand the proportion of newly diagnosed patients receiving checkpoint immunotherapy as part of their initial treatment protocol.
Regional Outlook Across Key Markets
.webp)
- India leads growth at 11.1%, supported by infrastructure expansion and rising adoption.
- North America shows steady growth, with the USA at 9.6% reflecting a mature market with established procurement.
- Europe maintains consistent demand, with Germany at 7.2%.
- Asia Pacific is the fastest growing region overall.
Country CAGR Table
| Country | CAGR (%) |
|---|---|
| India | 11.1% |
| China | 10.3% |
| USA | 9.6% |
| Brazil | 8.1% |
| UK | 7.5% |
| Germany | 7.2% |
| Japan | 6.8% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research.
Immune Checkpoint Inhibitor Market in India
India leads global market expansion with a projected CAGR of 11.1% through 2036. Growth reflects rising cancer incidence, expanding oncology treatment infrastructure, entry of domestic biosimilar checkpoint inhibitors that improve affordability, and increasing inclusion of immunotherapy in national treatment guidelines.
- Rising cancer incidence creates growing patient demand for immunotherapy treatment options.
- Domestic biosimilar checkpoint inhibitor production improves affordability and expands patient access.
- Inclusion of immunotherapy in national treatment guidelines supports institutional adoption.
Immune Checkpoint Inhibitor Market in China
China is projected to grow at a CAGR of 10.3% through 2036. Demand reflects a large cancer patient population, strong domestic checkpoint inhibitor development by BeiGene and Innovent Biologics, and national health insurance inclusion of select checkpoint inhibitor products.
- Large cancer patient population creates one of the world's biggest addressable oncology markets.
- Domestic checkpoint inhibitor manufacturers provide cost-competitive alternatives to imported branded products.
- National health insurance inclusion improves patient access and supports treatment adoption.
Immune Checkpoint Inhibitor Market in USA
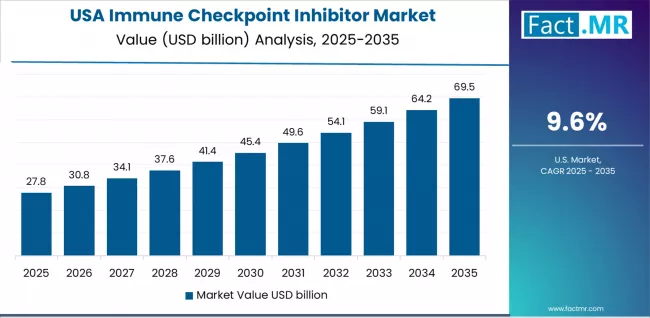
The United States is projected to grow at a CAGR of 9.6% through 2036. The market benefits from the broadest approved indication portfolio globally, high adoption of combination regimens, insurance coverage for immunotherapy, and a strong clinical trial pipeline.
- Broadest approved indication portfolio supports high per-patient treatment revenue.
- Combination regimen adoption in first-line settings increases immunotherapy utilization rates.
- Insurance coverage and patient assistance programs support treatment access for eligible patients.
Immune Checkpoint Inhibitor Market in Brazil
Brazil is projected to grow at a CAGR of 8.1% through 2036. Expansion reflects rising cancer incidence, improving access to immunotherapy through public and private healthcare channels, and growing adoption of checkpoint inhibitors in major oncology centers.
- Rising cancer incidence expands the immunotherapy-eligible patient population.
- Public and private healthcare channel development improves checkpoint inhibitor access.
- Major oncology centers adopt immunotherapy protocols consistent with international treatment guidelines.
Immune Checkpoint Inhibitor Market in UK
The United Kingdom is projected to grow at a CAGR of 7.5% through 2036. Growth reflects NICE appraisal-based access to checkpoint inhibitors, NHS cancer treatment investment, and clinical adoption of combination immunotherapy regimens.
- NICE appraisal processes provide structured access to cost-effective checkpoint inhibitor treatments.
- NHS cancer treatment investment supports immunotherapy adoption across hospital cancer centers.
- Combination regimen adoption aligns with international treatment guideline updates.
Immune Checkpoint Inhibitor Market in Germany
Germany is projected to grow at a CAGR of 7.2% through 2036. Demand reflects established oncology infrastructure, early access to new checkpoint inhibitor approvals through the AMNOG process, and strong clinical trial participation.
- Early access through AMNOG regulatory process supports rapid checkpoint inhibitor adoption.
- Established oncology infrastructure maintains high treatment volumes.
- Strong clinical trial participation provides early access to emerging checkpoint inhibitor combinations.
Immune Checkpoint Inhibitor Market in Japan
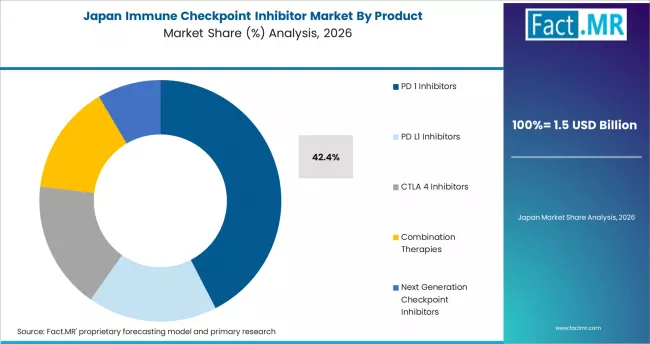
Japan is projected to grow at a CAGR of 6.8% through 2036. The market benefits from established oncology care standards, national health insurance coverage for approved checkpoint inhibitors, and domestic pharmaceutical development activity.
- National health insurance coverage supports patient access to approved checkpoint inhibitor therapies.
- Established oncology care standards drive consistent treatment adoption.
- Domestic pharmaceutical development contributes to locally adapted immunotherapy options.
Competitive Benchmarking and Company Positioning
Immune Checkpoint Inhibitor Market Analysis By Company
- Merck & Co., Bristol Myers Squibb, Roche hold strong positions through broad portfolios and established distribution networks.
- Regeneron, Sanofi, AstraZeneca participate meaningfully across specific segments and regional markets.
Merck & Co. holds the dominant position in the global immune checkpoint inhibitor market through Keytruda (pembrolizumab), which commands the broadest approved indication portfolio and the largest commercial revenue base among all checkpoint inhibitor products globally.
Bristol Myers Squibb competes strongly through Opdivo (nivolumab) and the ipilimumab-nivolumab combination regimen, particularly in renal cell carcinoma, melanoma, and hepatocellular carcinoma indications.
Chinese domestic manufacturers including BeiGene (tislelizumab) and Innovent Biologics (sintilimab) are creating significant competitive pressure through cost-effective PD-1 inhibitors that are gaining national health insurance coverage in China and pursuing global regulatory submissions.
The competitive landscape is concentrated at the branded level but increasingly dynamic as biosimilar pembrolizumab candidates advance through regulatory pathways and next-generation checkpoint inhibitors targeting novel immune targets enter clinical development.
Recent Industry Developments
- Merck & Co. received expanded indication approvals for Keytruda in early 2026 across multiple solid tumor types, further strengthening its position as the most widely prescribed checkpoint inhibitor globally.
- BeiGene expanded global registration for tislelizumab in 2025, with regulatory submissions in Europe and the United States targeting multiple first-line oncology indications.
- Bristol Myers Squibb advanced its next-generation combination immunotherapy program in 2025, with clinical data supporting novel checkpoint inhibitor pairings beyond traditional PD-1 and CTLA-4 combinations.
Leading Companies in the Immune Checkpoint Inhibitor Market
Major Players
- Merck & Co.
- Bristol Myers Squibb
- Roche
- Regeneron
- Sanofi
- AstraZeneca
- Novartis
- GSK
Emerging Players
- BeiGene
- Innovent Biologics
- Junshi Biosciences
- Hengrui Medicine
- Ono Pharmaceutical
- MacroGenics
- Incyte Corporation
Sources and Research References
- World Health Organization (WHO). Global Cancer Observatory: Cancer Incidence and Mortality Data.
- National Comprehensive Cancer Network (NCCN). Clinical Practice Guidelines for Immune Checkpoint Inhibitor Use in Oncology.
- Merck & Co. Product Portfolio, Keytruda Indication Approvals, and Clinical Development Updates.
- U.S. Food and Drug Administration (FDA). Approved Biologic Products for Oncology Indications.
- Primary interviews with oncologists, hospital pharmacy directors, pharmaceutical distributors, and clinical trial investigators.
This bibliography is provided for reader reference and is not exhaustive.
Key Questions This Report Addresses
- What is the current and future size of the immune checkpoint inhibitor market?
- How fast is the market expected to grow between 2026 and 2036?
- Which product is likely to lead by 2026?
- Which drug class segment accounts for the highest demand?
- What factors are driving demand globally?
- Which countries show the fastest growth?
- Who are the key companies?
- How does Fact.MR validate the forecast?
Immune Checkpoint Inhibitor Market Definition
The immune checkpoint inhibitor market covers PD-1, PD-L1, and CTLA-4 targeting monoclonal antibodies and combination therapy regimens used across oncology indications including solid tumors, colorectal cancer, and hematologic malignancies in first-line, second-line, and subsequent therapy settings.
Immune Checkpoint Inhibitor Market Inclusions
The scope covers global and regional market size and forecasts for 2026 to 2036 across product, indication, therapy line, mechanism target, drug class, and region. It includes regional demand analysis, pricing and cost analysis, technology trends, market drivers and constraints, and the competitive landscape.
Immune Checkpoint Inhibitor Market Exclusions
The scope excludes general-purpose devices, products, or services not purpose-built for the applications defined within this market. It excludes unbranded products with low market visibility, rental or leasing services without hardware sales, and custom-built systems without commercial sales distribution.
Immune Checkpoint Inhibitor Market Research Methodology
- Primary research includes interviews with manufacturers, distributors, end users, and institutional procurement teams across key geographies.
- Secondary research draws on industry publications, regulatory filings, company reports, and trade databases.
- Market sizing follows a demand-side model supported by top-down validation across 30 plus countries.
- Final estimates are validated through trade-flow checks, pricing trend analysis, and sensitivity testing.
Scope of Analysis
| Parameter | Details |
|---|---|
| Quantitative Units | USD 51.8 billion to USD 122.6 billion, at a CAGR of 9.0% |
| Market Definition | The immune checkpoint inhibitor market covers PD-1, PD-L1, and CTLA-4 targeting monoclonal antibodies and combination therapy regimens used across oncology indications including solid tumors, colorectal cancer, and hematologic malignancies in first-line, second-line, and subsequent therapy settings. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, India, China, Brazil, UK, Germany, Japan, and 30 plus countries |
| Key Companies | Merck & Co., Bristol Myers Squibb, Roche, Regeneron, Sanofi, AstraZeneca, Novartis, GSK, BeiGene, Innovent Biologics |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid demand-side and top-down methodology built on country-level oncology treatment volumes, drug pricing analysis, indication expansion tracking, and primary interviews across oncologists, hospital procurement teams, pharmaceutical distributors, and clinical trial investigators |
Market Segmentation Analysis
-
Immune Checkpoint Inhibitor Market Market Segmented by Product:
- PD 1 Inhibitors
- Nivolumab
- Pembrolizumab
- Cemiplimab
- PD L1 Inhibitors
- Atezolizumab
- Durvalumab
- Avelumab
- CTLA 4 Inhibitors
- Ipilimumab
- Tremelimumab
- Combination Therapies
- PD 1 Plus CTLA 4 Combination
- PD 1 Plus Chemotherapy Combination
- PD L1 Plus Targeted Therapy Combination
- Next Generation Checkpoint Inhibitors
- LAG 3 Inhibitors
- TIGIT Inhibitors
- TIM 3 Inhibitors
- Other Emerging Immune Checkpoints
- PD 1 Inhibitors
-
Immune Checkpoint Inhibitor Market Market Segmented by Indication:
- Oncology
- Non Small Cell Lung Cancer
- Melanoma
- Renal Cell Carcinoma
- Urothelial Carcinoma
- Hodgkin Lymphoma
- Liver Cancer
- Head and Neck Cancer
- Gastric Cancer
- Breast Cancer
- Colorectal Cancer
- MSI High Colorectal Cancer
- Endometrial Cancer
- Hematologic Malignancies
- Blood Cancers
- Lymphomas
- Oncology
-
Immune Checkpoint Inhibitor Market Market Segmented by Therapy Line:
- First Line Therapy
- Monotherapy First Line
- Combination First Line
- Second Line Therapy
- Second Line Monotherapy
- Second Line Combination
- Third Line and Beyond
- Refractory Cancer Treatment
- Advanced Stage Therapy
- First Line Therapy
-
Immune Checkpoint Inhibitor Market Market Segmented by Mechanism Target:
- PD 1 Pathway
- PD 1 Receptor Blockade
- PD L1 Ligand Blockade
- CTLA 4 Pathway
- T Cell Activation Enhancement
- Immune Co Stimulatory Pathways
- LAG 3 Blockade
- TIGIT Blockade
- TIM 3 Blockade
- PD 1 Pathway
-
Immune Checkpoint Inhibitor Market Market Segmented by Drug Class:
- Monoclonal Antibodies
- Humanized Antibodies
- Fully Human Antibodies
- Combination Regimens
- Biologic Plus Chemotherapy
- Biologic Combination Immunotherapy
- Monoclonal Antibodies
-
Immune Checkpoint Inhibitor Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
How big is the immune checkpoint inhibitor market in 2025?
The global immune checkpoint inhibitor market is estimated at USD 47.5 billion in 2025.
What will be the market size in 2036?
The immune checkpoint inhibitor market is projected to reach USD 122.6 billion by 2036.
What is the expected CAGR?
The market is expected to grow at a 9.0% CAGR between 2026 and 2036.
What are the key product types?
Key types include pd-1 inhibitors, pd-l1 inhibitors, ctla-4 inhibitors.
Which drug class segment leads?
Monoclonal Antibodies is expected to account for 73.2% share in 2026.
Which countries grow fastest?
India (11.1%) and China (10.3%) lead through 2036.
What is the market definition?
The immune checkpoint inhibitor market covers PD-1, PD-L1, and CTLA-4 targeting monoclonal antibodies and combination therapy regimens used across oncology indications including solid tumors, colorectal cancer, and hematologic malignancies in first-line, second-line, and subsequent therapy settings.
What methodology is used?
The methodology combines secondary research, primary interviews, and forecast modelling across 30 plus countries.
How big is the immune checkpoint inhibitor market in 2025?
The global immune checkpoint inhibitor market is estimated at USD 47.5 billion in 2025.
What will be the market size in 2036?
The immune checkpoint inhibitor market is projected to reach USD 122.6 billion by 2036.
What is the expected CAGR?
The market is expected to grow at a 9.0% CAGR between 2026 and 2036.
What are the key product types?
Key types include pd-1 inhibitors, pd-l1 inhibitors, ctla-4 inhibitors.
Which drug class segment leads?
Monoclonal Antibodies is expected to account for 73.2% share in 2026.
Which countries grow fastest?
India (11.1%) and China (10.3%) lead through 2036.
What is the market definition?
The immune checkpoint inhibitor market covers PD-1, PD-L1, and CTLA-4 targeting monoclonal antibodies and combination therapy regimens used across oncology indications including solid tumors, colorectal cancer, and hematologic malignancies in first-line, second-line, and subsequent therapy settings.
What methodology is used?
The methodology combines secondary research, primary interviews, and forecast modelling across 30 plus countries.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- PD 1 Inhibitors
- Nivolumab
- Pembrolizumab
- Cemiplimab
- PD L1 Inhibitors
- Atezolizumab
- Durvalumab
- Avelumab
- CTLA 4 Inhibitors
- Ipilimumab
- Tremelimumab
- Combination Therapies
- PD 1 Plus CTLA 4 Combination
- PD 1 Plus Chemotherapy Combination
- PD L1 Plus Targeted Therapy Combination
- Next Generation Checkpoint Inhibitors
- LAG 3 Inhibitors
- TIGIT Inhibitors
- TIM 3 Inhibitors
- Other Emerging Immune Checkpoints
- PD 1 Inhibitors
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Oncology
- Non Small Cell Lung Cancer
- Melanoma
- Renal Cell Carcinoma
- Urothelial Carcinoma
- Hodgkin Lymphoma
- Liver Cancer
- Head and Neck Cancer
- Gastric Cancer
- Breast Cancer
- Colorectal Cancer
- MSI High Colorectal Cancer
- Endometrial Cancer
- Hematologic Malignancies
- Blood Cancers
- Lymphomas
- Oncology
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapy Line
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapy Line, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapy Line, 2026 to 2036
- First Line Therapy
- Monotherapy First Line
- Combination First Line
- Second Line Therapy
- Second Line Monotherapy
- Second Line Combination
- Third Line and Beyond
- Refractory Cancer Treatment
- Advanced Stage Therapy
- First Line Therapy
- Y to o to Y Growth Trend Analysis By Therapy Line, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapy Line, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Mechanism Target
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Mechanism Target, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Mechanism Target, 2026 to 2036
- PD 1 Pathway
- PD 1 Receptor Blockade
- PD L1 Ligand Blockade
- CTLA 4 Pathway
- T Cell Activation Enhancement
- Immune Co Stimulatory Pathways
- LAG 3 Blockade
- TIGIT Blockade
- TIM 3 Blockade
- PD 1 Pathway
- Y to o to Y Growth Trend Analysis By Mechanism Target, 2021 to 2025
- Absolute $ Opportunity Analysis By Mechanism Target, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Class, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Class, 2026 to 2036
- Monoclonal Antibodies
- Humanized Antibodies
- Fully Human Antibodies
- Combination Regimens
- Biologic Plus Chemotherapy
- Biologic Combination Immunotherapy
- Monoclonal Antibodies
- Y to o to Y Growth Trend Analysis By Drug Class, 2021 to 2025
- Absolute $ Opportunity Analysis By Drug Class, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Indication
- By Therapy Line
- By Mechanism Target
- By Drug Class
- Competition Analysis
- Competition Deep Dive
- Merck & Co.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Bristol Myers Squibb
- Roche
- Regeneron
- Sanofi
- AstraZeneca
- Novartis
- GSK
- BeiGene
- Innovent Biologics
- Merck & Co.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Therapy Line, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Mechanism Target, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Therapy Line, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Mechanism Target, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Therapy Line, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Mechanism Target, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Therapy Line, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Mechanism Target, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Therapy Line, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Mechanism Target, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Therapy Line, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Mechanism Target, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Therapy Line, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Mechanism Target, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Therapy Line, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Mechanism Target, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product,2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Indication,2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Indication,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value Share and BPS Analysis by Therapy Line,2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Therapy Line,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Therapy Line
- Figure 12: Global Market Value Share and BPS Analysis by Mechanism Target,2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Mechanism Target,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Mechanism Target
- Figure 15: Global Market Value Share and BPS Analysis by Drug Class,2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Drug Class,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Drug Class
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region,2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 22: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 28: North America Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product,2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Product
- Figure 32: North America Market Value Share and BPS Analysis by Indication,2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Indication,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Indication
- Figure 35: North America Market Value Share and BPS Analysis by Therapy Line,2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Therapy Line,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Therapy Line
- Figure 38: North America Market Value Share and BPS Analysis by Mechanism Target,2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Mechanism Target,2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by Mechanism Target
- Figure 41: North America Market Value Share and BPS Analysis by Drug Class,2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Drug Class,2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Drug Class
- Figure 44: Latin America Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product,2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Product
- Figure 48: Latin America Market Value Share and BPS Analysis by Indication,2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Indication,2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by Indication
- Figure 51: Latin America Market Value Share and BPS Analysis by Therapy Line,2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Therapy Line,2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Therapy Line
- Figure 54: Latin America Market Value Share and BPS Analysis by Mechanism Target,2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Mechanism Target,2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Mechanism Target
- Figure 57: Latin America Market Value Share and BPS Analysis by Drug Class,2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Drug Class,2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by Drug Class
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product,2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product
- Figure 64: Western Europe Market Value Share and BPS Analysis by Indication,2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Indication,2026 to 2036
- Figure 66: Western Europe Market Attractiveness Analysis by Indication
- Figure 67: Western Europe Market Value Share and BPS Analysis by Therapy Line,2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Therapy Line,2026 to 2036
- Figure 69: Western Europe Market Attractiveness Analysis by Therapy Line
- Figure 70: Western Europe Market Value Share and BPS Analysis by Mechanism Target,2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Mechanism Target,2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Mechanism Target
- Figure 73: Western Europe Market Value Share and BPS Analysis by Drug Class,2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Drug Class,2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Drug Class
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product,2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Indication,2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Indication,2026 to 2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Therapy Line,2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Therapy Line,2026 to 2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Therapy Line
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Mechanism Target,2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Mechanism Target,2026 to 2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Mechanism Target
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Drug Class,2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Drug Class,2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Drug Class
- Figure 92: East Asia Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product,2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 95: East Asia Market Attractiveness Analysis by Product
- Figure 96: East Asia Market Value Share and BPS Analysis by Indication,2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Indication,2026 to 2036
- Figure 98: East Asia Market Attractiveness Analysis by Indication
- Figure 99: East Asia Market Value Share and BPS Analysis by Therapy Line,2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Therapy Line,2026 to 2036
- Figure 101: East Asia Market Attractiveness Analysis by Therapy Line
- Figure 102: East Asia Market Value Share and BPS Analysis by Mechanism Target,2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Mechanism Target,2026 to 2036
- Figure 104: East Asia Market Attractiveness Analysis by Mechanism Target
- Figure 105: East Asia Market Value Share and BPS Analysis by Drug Class,2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Drug Class,2026 to 2036
- Figure 107: East Asia Market Attractiveness Analysis by Drug Class
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product,2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Indication,2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication,2026 to 2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Therapy Line,2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Therapy Line,2026 to 2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Therapy Line
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Mechanism Target,2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Mechanism Target,2026 to 2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Mechanism Target
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Drug Class,2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug Class,2026 to 2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Drug Class
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product,2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Indication,2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Indication,2026 to 2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Therapy Line,2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Therapy Line,2026 to 2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Therapy Line
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Mechanism Target,2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Mechanism Target,2026 to 2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Mechanism Target
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Drug Class,2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Drug Class,2026 to 2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Drug Class
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis