Omeprazole Sulfone Market Size, Share, Growth and Forecast (2026 - 2036)
Omeprazole Sulfone Market is segmented by Purity (≥ 99%, < 99%), Application (API Intermediate, Research), Grade (Pharma, Lab), and Region. Forecast for 2026 to 2036.
Fact.MR opines the omeprazole sulfone market was valued at USD 380.00 million in 2025. Sales are expected to reach USD 391.40 million in 2026 and USD 526.01 million by 2036. ≥ 99% is poised to lead by Purity with 70.0% share in 2026. API Intermediate is estimated to command by Application in 2026.
Omeprazole Sulfone Market Size, Market Forecast and Outlook By Fact.MR
The omeprazole sulfone market was valued at USD 380.00 million in 2025, projected to reach USD 391.40 million in 2026, and is forecast to expand to USD 526.01 million by 2036 at a 3.0% CAGR. is a stable pharmaceutical intermediate segment where demand is anchored by the global consumption of proton pump inhibitor (PPI) medications, with omeprazole remaining among the most prescribed drugs worldwide and India serving as the primary manufacturing hub for both the sulfone intermediate and the finished API.
The absolute dollar expansion between 2026 and 2036 amounts to approximately USD 134.61 million. Growth reflects sustained global PPI prescription volumes, expanding generic omeprazole production in India and China, and the use of omeprazole sulfone as a reference standard in pharmaceutical quality control. The mature nature of omeprazole as an off-patent molecule and pricing pressure from generic competition constrain revenue growth despite steady volume demand.
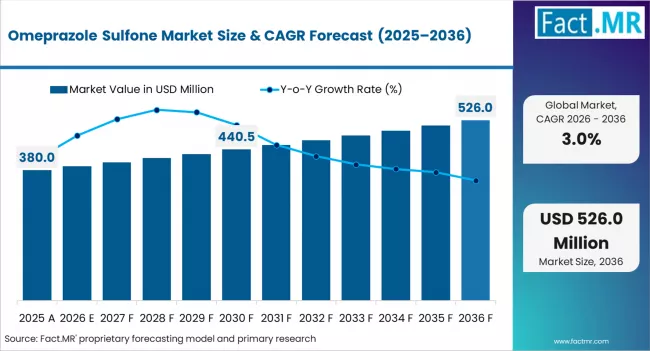
Omeprazole Sulfone Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 391.40 million |
| Industry Value (2036) | USD 526.01 million |
| CAGR (2026 to 2036) | 3.0% |
API procurement managers at generic pharmaceutical companies are consolidating omeprazole sulfone sourcing with vertically integrated Indian manufacturers who control the synthesis from pyridine intermediates through to finished sulfone. Suppliers unable to maintain Drug Master File (DMF) registrations with the U.S. FDA and EU EMA face progressive exclusion from qualified vendor lists at major generic drug producers.
All major regional markets reflect differentiated growth parameters. India sets the pace at 4.0% CAGR. China registers at 3.4% CAGR. Italy registers at 2.5% CAGR. Germany registers at 2.4% CAGR. South Korea registers at 2.3% CAGR. USA registers at 2.2% CAGR. Japan registers at 1.8% CAGR.
Summary of Omeprazole Sulfone Market
- Omeprazole Sulfone Market Definition
- Omeprazole sulfone is a chemical intermediate and metabolite of omeprazole, the proton pump inhibitor used to treat gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. Chemically, it is the sulfone oxidation product of omeprazole (5-methoxy-2-[[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]sulfonyl]-1H-benzimidazole).
- Demand Drivers in the Market
- Global PPI prescription volume: The IQVIA Institute reports proton pump inhibitors remain among the most dispensed drug classes globally, with omeprazole accounting for over 50 million prescriptions annually in the United States alone.
- Indian API manufacturing expansion: India's pharmaceutical export revenue exceeded USD 28 billion in FY2024 according to the Pharmaceuticals Export Promotion Council (Pharmexcil).
- Reference standard demand: Pharmacopoeia reference standards for omeprazole impurity profiling (including sulfone) are consumed by quality control laboratories at every omeprazole manufacturing site globally.
- Key Segments Analyzed in the Fact.MR Report
- ≥ 99% purity: 70.0% share in 2026.
- API Intermediate application: 80.0% share in 2026.
- India: 4.0% compound growth through 2036.
- Analyst Opinion at Fact.MR
- India dominates production with over 60% of global omeprazole API output concentrated in Hyderabad and Gujarat. The purity level of 99% and above holds 70% share because pharmaceutical-grade omeprazole sulfone must meet ICH Q3A impurity limits for use in API synthesis. API intermediate is the primary application at 80% share. Dr.
- Strategic Implications / Executive Takeaways
- Procurement teams must establish multi-year supply agreements with qualified producers to secure allocation ahead of anticipated demand increases.
- Product development resources should prioritize formulations that meet tightening regulatory specifications in key markets.
- Regional expansion strategies must account for divergent growth rates and regulatory environments across major consuming nations.
Why is the Omeprazole Sulfone Market Growing?
Market expansion is being supported by the increasing demand for high-purity pharmaceutical intermediates and the corresponding need for high-efficiency production systems in omeprazole sulfone applications across global pharmaceutical and research operations. Modern pharmaceutical manufacturers are increasingly focused on advanced intermediate technologies that can improve quality control, reduce production costs, and enhance manufacturing efficiency while meeting stringent purity requirements. The proven efficacy of omeprazole sulfone in various pharmaceutical applications makes it an essential component of comprehensive API manufacturing strategies and specialized production programs.
The growing focus on pharmaceutical industry transformation and advanced quality optimization is driving demand for ultra-efficient intermediate systems that meet stringent performance specifications and manufacturing requirements for pharmaceutical applications. Pharmaceutical manufacturers' preference for reliable, high-performance production systems that can ensure consistent quality standards is creating opportunities for innovative omeprazole sulfone technologies and customized manufacturing solutions. The rising influence of regulatory requirements and pharmaceutical protocols is also contributing to increased adoption of premium-grade intermediate systems across different pharmaceutical applications and manufacturing systems requiring advanced purity technology.
Opportunity Pathways - Omeprazole Sulfone Market
The omeprazole sulfone market represents a specialized growth opportunity, expanding from USD 380.0 million in 2025 to USD 510.0 million by 2035 at a 3.0% CAGR. As manufacturers prioritize quality control, regulatory compliance, and manufacturing performance in complex pharmaceutical processes, intermediate systems have evolved from a niche pharmaceutical technology to an essential component enabling quality assurance, production optimization, and multi-stage manufacturing production across pharmaceutical operations and specialized research applications.
The convergence of pharmaceutical expansion, increasing intermediate adoption, specialized manufacturing organization growth, and purity requirements creates momentum in demand. High-purity formulations offering superior performance characteristics, cost-effective ≥ 99% systems balancing performance with economics, and specialized pharmaceutical-grade variants for premium applications will capture market premiums, while geographic expansion into high-growth Asian pharmaceutical markets and emerging market penetration will drive volume leadership. Manufacturing focus on quality assurance and regulatory reliability provides structural support.
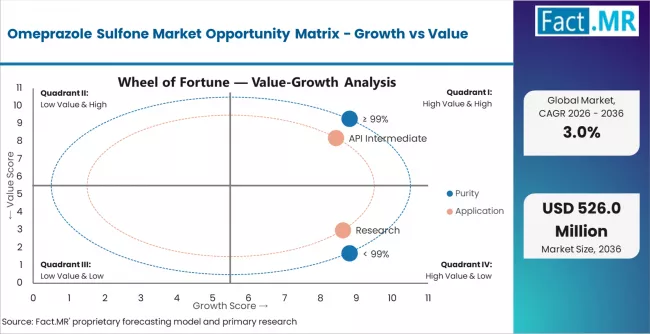
- Pathway A - High Purity Dominance: Leading with 70.0% market share, ≥ 99% purity applications drive primary demand through complex pharmaceutical workflows requiring comprehensive intermediate systems for specialized manufacturing. Advanced formulations enabling improved quality control, reduced impurity levels, and enhanced manufacturing efficiency command premium pricing from manufacturers requiring stringent purity specifications and regulatory compliance. Expected revenue pool: USD 266.0-357.0 million.
- Pathway B - API Intermediate Leadership: Dominating with 80.0% market share through an optimal balance of performance and cost-effectiveness, API intermediate serves most pharmaceutical applications while meeting manufacturing requirements. This application addresses both performance standards and economic considerations, making it the preferred choice for pharmaceutical operations seeking reliable intermediate performance. Opportunity: USD 304.0-408.0 million.
- Pathway C - Asian Market Acceleration: India (4.0% CAGR) and China (3.4% CAGR) lead global growth through pharmaceutical infrastructure expansion, manufacturing capability development, and domestic intermediate demand. Strategic partnerships with local manufacturers, regulatory compliance expertise, and supply chain localization enable the expansion of intermediate technology in major pharmaceutical hubs. Geographic expansion upside: USD 80.0-120.0 million.
- Pathway D - Pharmaceutical Grade Premium Segment: Pharmaceutical grade serves specialized applications requiring exceptional purity specifications for critical manufacturing processes. Pharmaceutical formulations supporting consistent quality requirements, complex API applications, and regulatory-sensitive processes command significant premiums from advanced pharmaceutical organizations and specialized manufacturing facilities. Revenue potential: USD 342.0-459.0 million.
- Pathway E - Advanced Manufacturing & Technology Systems: Companies investing in sophisticated intermediate technologies, specialized production systems, and automated manufacturing processes gain competitive advantages through consistent system performance and quality reliability. Advanced capabilities enabling customized specifications and rapid deployment capture premium pharmaceutical partnerships. Technology premium: USD 50.0-80.0 million.
- Pathway F - Supply Chain Optimization & Reliability: Specialized distribution networks, strategic inventory management, and reliable supply chain systems create competitive differentiation in pharmaceutical markets requiring consistent intermediate availability. Companies offering guaranteed supply security, technical support, and regulatory documentation gain preferred supplier status with compliance-focused manufacturers. Supply chain value: USD 40.0-70.0 million.
- Pathway G - Emerging Applications & Market Development: Beyond traditional API intermediates, omeprazole sulfone in specialty research, advanced pharmaceutical formulations, and novel manufacturing systems represent growth opportunities. Companies developing new applications, supporting R&D initiatives, and expanding into adjacent pharmaceutical markets capture incremental demand while diversifying revenue streams. Emerging opportunity: USD 30.0-60.0 million.
Segmental Analysis
The market is segmented by purity, application, grade, and region. By purity, the market is divided into ≥ 99% and < 99%. Based on application, the market is categorized into API intermediate and research. By grade, the market is divided into pharma and lab. Regionally, the market is divided into Asia Pacific, North America, Europe, Latin America, Middle East & Africa.
By Purity, ≥ 99% Segment Accounts for 70.0% Market Share
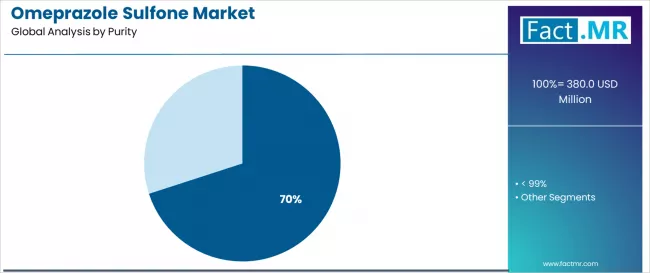
The ≥ 99% purity segment is projected to account for 70.0% of the market in 2025, reaffirming its position as the category's dominant purity level. Manufacturers increasingly recognize the optimal balance of performance and regulatory compliance offered by ≥ 99% purity for most pharmaceutical applications, particularly in API intermediate production and manufacturing processes. This purity level addresses both performance requirements and regulatory considerations while providing reliable quality across diverse pharmaceutical applications.
This purity level forms the foundation of most pharmaceutical protocols for intermediate applications, as it represents the most widely accepted and commercially viable level of omeprazole sulfone technology in the industry. Quality control standards and extensive regulatory testing continue to strengthen confidence in ≥ 99% purity formulations among pharmaceutical and manufacturing providers. With increasing recognition of the quality-performance optimization requirements in pharmaceutical manufacturing, ≥ 99% purity systems align with both operational efficiency and regulatory compliance goals, making them the central growth driver of comprehensive pharmaceutical manufacturing strategies.
By Application, API Intermediate Segment Accounts for 80.0% Market Share
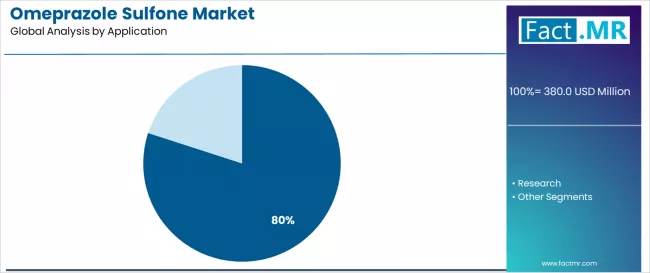
API intermediate is projected to represent 80.0% of omeprazole sulfone demand in 2025, highlighting its role as the primary application segment driving market adoption and growth. Manufacturers recognize that API intermediate requirements, including complex pharmaceutical processes, specialized manufacturing needs, and multi-stage production systems, often require advanced intermediate services that standard pharmaceutical technologies cannot adequately provide. Omeprazole sulfone offers enhanced manufacturing efficiency and regulatory compliance in API intermediate pharmaceutical applications.
The segment is supported by the growing nature of API intermediate adoption, requiring sophisticated production systems, and the increasing recognition that advanced intermediate technologies can improve pharmaceutical performance and manufacturing outcomes. Manufacturers are increasingly adopting evidence-based pharmaceutical guidelines that recommend specific intermediate services for optimal production efficiency. As understanding of pharmaceutical complexity advances and manufacturing requirements become more stringent, intermediate services will continue to play a crucial role in comprehensive pharmaceutical strategies within the API intermediate market.
What are the Drivers, Restraints, and Key Trends of the Omeprazole Sulfone Market?
The omeprazole sulfone market is advancing steadily due to increasing recognition of pharmaceutical intermediate technologies' importance and growing demand for high-purity production systems across the pharmaceutical and research sectors. The market faces challenges, including complex regulatory processes, potential for purity variations during production and storage, and concerns about supply chain consistency for specialized pharmaceutical equipment. Innovation in intermediate technologies and customized pharmaceutical protocols continues to influence product development and market expansion patterns.
Expansion of Advanced Manufacturing Facilities and Intermediate Technologies
The growing adoption of advanced manufacturing facilities is enabling the development of more sophisticated intermediate production and quality control systems that can meet stringent pharmaceutical requirements. Specialized manufacturing facilities offer comprehensive intermediate services, including advanced purification and quality processes that are particularly important for achieving high-purity requirements in pharmaceutical applications. Advanced manufacturing channels provide access to premium services that can optimize production efficiency and reduce manufacturing costs while maintaining cost-effectiveness for large-scale pharmaceutical operations.
Integration of Digital Manufacturing Systems and Quality Management Systems
Modern pharmaceutical organizations are incorporating digital technologies such as real-time quality monitoring, automated purification systems, and supply chain integration to enhance intermediate production and distribution processes. These technologies improve production performance, enable continuous quality monitoring, and provide better coordination between suppliers and manufacturers throughout the intermediate cycle. Advanced digital platforms also enable customized purity specifications and early identification of potential quality deviations or supply disruptions, supporting reliable pharmaceutical production.
Analysis of the Omeprazole Sulfone Market by Key Country
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| India | 4.0% |
| China | 3.4% |
| Italy | 2.5% |
| Germany | 2.4% |
| South Korea | 2.3% |
| USA | 2.2% |
| Japan | 1.8% |
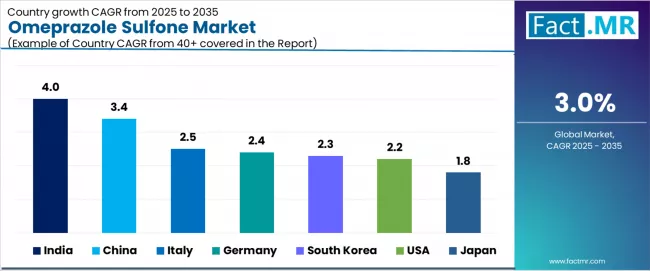
The market is experiencing varied growth globally, with India leading at a 4.0% CAGR through 2036, driven by the expansion of pharmaceutical infrastructure development, increasing manufacturing capability capabilities, and growing domestic demand for high-purity intermediate systems. China follows at 3.4%, supported by pharmaceutical expansion, growing recognition of advanced intermediate technology importance, and expanding manufacturing capacity. Italy records 2.5% growth, while Germany shows 2.4% growth, both with focus on developing the pharmaceutical technology and manufacturing industries. South Korea demonstrates 2.3% growth with focus on pharmaceutical manufacturing adoption. The USA shows 2.2% growth, representing a mature market with established manufacturing patterns and regulatory frameworks. Japan demonstrates 1.8% growth, focusing pharmaceutical infrastructure expansion and systematic manufacturing approaches.
India Demonstrates Growing Market Potential with Pharmaceutical Infrastructure Development
India is projected to exhibit robust growth with a CAGR of 4.0% through 2036, driven by ongoing pharmaceutical expansion and increasing recognition of high-purity intermediate systems as essential manufacturing components for complex pharmaceutical processes. The country's expanding pharmaceutical infrastructure and growing availability of specialized manufacturing capabilities are creating significant opportunities for intermediate adoption across both domestic and export-oriented pharmaceutical facilities. Major international and domestic pharmaceutical companies are establishing comprehensive production and distribution networks to serve the growing population of manufacturers and pharmaceutical facilities requiring high-performance intermediate systems across API and research applications throughout India's major pharmaceutical hubs.
The Indian government's strategic focus on pharmaceutical manufacturing modernization and industry advancement is driving substantial investments in specialized production capabilities. This policy support, combined with the country's large domestic pharmaceutical market and expanding manufacturing requirements, creates a favorable environment for the omeprazole sulfone market development. Indian manufacturers are increasingly focusing on high-value pharmaceutical technologies to improve manufacturing capabilities, with intermediate services representing a key component in this pharmaceutical transformation.
- Government initiatives supporting pharmaceutical development and manufacturing modernization are driving demand for high-purity intermediate systems throughout major pharmaceutical and manufacturing centers, including Mumbai, Hyderabad, and Bangalore regions.
- Manufacturing capacity expansion and production system development are supporting appropriate utilization of intermediate services among manufacturers and pharmaceutical facilities nationwide, with particular growth in API manufacturing operations and research engagement services.
China Demonstrates Exceptional Market Potential with Manufacturing Growth
China is expanding at a CAGR of 3.4%, supported by increasing pharmaceutical accessibility, growing manufacturing awareness, and developing technology market presence across the country's major pharmaceutical clusters. The country's large pharmaceutical sector and increasing recognition of advanced intermediate systems are driving demand for effective high-purity manufacturing solutions in both API production and research applications. International pharmaceutical companies and domestic providers are establishing comprehensive distribution channels to serve the growing demand for quality intermediate systems while supporting the country's position as an emerging pharmaceutical technology market.
China's pharmaceutical sector continues to benefit from favorable manufacturing policies, expanding pharmaceutical capabilities, and cost-competitive production infrastructure development. The country's focus on becoming a global pharmaceutical technology hub is driving investments in specialized intermediate technology and manufacturing infrastructure. This development is particularly important for intermediate applications, as manufacturers seek reliable domestic sources for critical production technologies to reduce import dependency and improve supply chain security.
- Rising awareness about advanced manufacturing options and improving pharmaceutical capabilities are creating opportunities for specialized intermediate systems across API production and research settings in major hubs like Beijing, Shanghai, and Guangzhou.
- Growing pharmaceutical infrastructure development and technology adoption are supporting increased access to high-purity intermediate services among organizations requiring comprehensive manufacturing capabilities, particularly in API production and pharmaceutical research organizations.
USA Maintains Technology Leadership
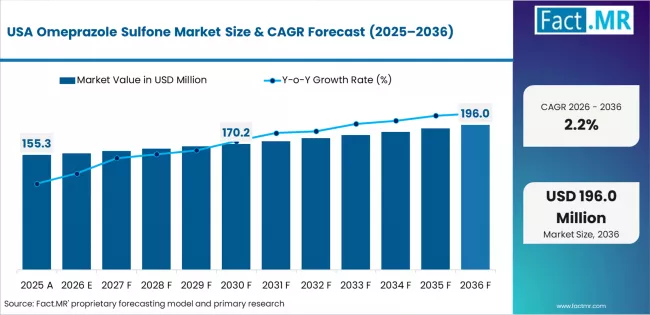
The USA's advanced pharmaceutical technology market demonstrates sophisticated manufacturing deployment with documented intermediate effectiveness in pharmaceutical departments and research centers through integration with existing manufacturing systems and production infrastructure. The country leverages manufacturing expertise in pharmaceutical technology and production systems integration to maintain a 2.2% CAGR through 2036. Manufacturing centers, including major metropolitan areas, showcase premium installations where intermediate systems integrate with comprehensive pharmaceutical information systems and production platforms to optimize manufacturing accuracy and operational workflow effectiveness.
American manufacturers prioritize system reliability and regulatory compliance in pharmaceutical development, creating demand for premium systems with advanced features, including quality validation and integration with US pharmaceutical standards. The market benefits from established pharmaceutical industry infrastructure and a willingness to invest in advanced intermediate technologies that provide long-term operational benefits and compliance with regulatory requirements.
Germany Shows Strong Regional Leadership
Germany's market expansion benefits from diverse pharmaceutical demand, including manufacturing modernization in major cities, production development programs, and government pharmaceutical programs that increasingly incorporate intermediate solutions for manufacturing enhancement applications. The country maintains a 2.4% CAGR through 2036, driven by rising manufacturing awareness and increasing adoption of production benefits, including superior manufacturing capabilities and reduced complexity.
Market dynamics focus on cost-effective intermediate solutions that balance advanced manufacturing features with affordability considerations important to German pharmaceutical operators. Growing pharmaceutical infrastructure creates demand for modern production systems in new manufacturing facilities and pharmaceutical equipment modernization projects.
Strategic Market Considerations:
- Pharmaceutical and API manufacturing segments leading growth with focus on production enhancement and operational efficiency applications
- Regional manufacturing requirements are driving a diverse product portfolio from basic intermediate systems to advanced production platforms
- Import dependency challenges offset by potential local development partnerships with international manufacturing companies
- Government pharmaceutical initiatives beginning to influence procurement standards and manufacturing requirements
Italy Shows Steady Market Growth
Italy demonstrates steady market development with a 2.5% CAGR through 2036, distinguished by pharmaceutical operators' preference for high-quality intermediate systems that integrate seamlessly with existing manufacturing equipment and provide reliable long-term operation in specialized pharmaceutical applications. The market prioritizes advanced features, including precision production algorithms, quality validation, and integration with comprehensive pharmaceutical platforms that reflect Italian manufacturing expectations for technological advancement and operational excellence.
Italian manufacturers focus on system reliability and precision in intermediate development, creating demand for premium systems with advanced features including automated quality monitoring and comprehensive technical support. The market benefits from established pharmaceutical research infrastructure and investment in production technology that provides long-term manufacturing benefits.
Strategic Market Indicators:
- Premium focus on precision systems with advanced production algorithms and high-reliability capabilities
- Integration requirements with existing pharmaceutical information systems and manufacturing management platforms
- Focus on intermediate reliability and long-term performance in pharmaceutical applications
South Korea Emphasizes Digital Innovation
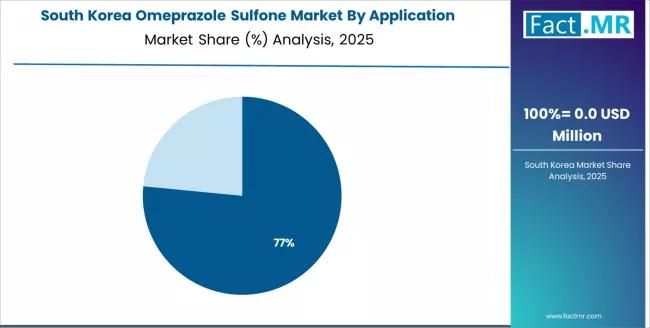
South Korea demonstrates strong market development with a 2.3% CAGR through 2036, driven by advanced digital infrastructure and manufacturing preference for technology-integrated intermediate services. The country's sophisticated pharmaceutical ecosystem and high technology penetration rates are creating significant opportunities for intermediate adoption across both domestic and technology-driven pharmaceutical facilities. Major international and domestic pharmaceutical companies are establishing comprehensive digital production and distribution networks to serve the growing population of tech-savvy manufacturers and pharmaceutical facilities requiring high-performance intermediate systems across specialized and pharmaceutical applications throughout South Korea's major pharmaceutical hubs.
The South Korean government's strategic focus on digital transformation and pharmaceutical advancement is driving substantial investments in specialized production capabilities. This policy support, combined with the country's digitally advanced manufacturing base and expanding technology requirements, creates a favorable environment for the omeprazole sulfone market development. South Korean manufacturers are increasingly focusing on high-value digital pharmaceutical technologies to improve manufacturing capabilities, with intermediate services representing a key component in this pharmaceutical transformation.
- Government initiatives supporting digital pharmaceutical development and technology modernization are driving demand for high-purity intermediate systems throughout major pharmaceutical and manufacturing centers, including Seoul, Busan, and Incheon regions.
- Technology capacity expansion and digital production system development are supporting appropriate utilization of intermediate services among manufacturers and pharmaceutical facilities nationwide, with particular growth in automated operations and digital manufacturing engagement services.
Japan Emphasizes Precision and Integration Excellence
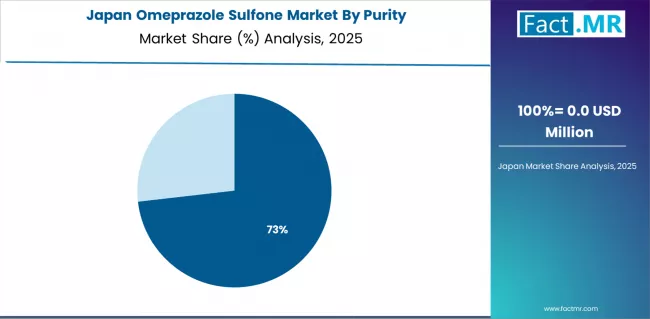
Japan demonstrates steady market development with a 1.8% CAGR through 2036, distinguished by pharmaceutical operators' preference for high-quality intermediate systems that integrate seamlessly with existing manufacturing equipment and provide reliable long-term operation in specialized pharmaceutical applications. The market prioritizes advanced features, including precision production algorithms, quality validation, and integration with comprehensive pharmaceutical platforms that reflect Japanese manufacturing expectations for technological advancement and operational excellence.
Japanese manufacturers focus on system reliability and precision in intermediate development, creating demand for premium systems with advanced features including automated quality monitoring and comprehensive technical support. The market benefits from established pharmaceutical research infrastructure and investment in production technology that provides long-term manufacturing benefits.
Strategic Market Indicators:
- Premium focus on precision systems with advanced production algorithms and high-reliability capabilities
- Integration requirements with existing pharmaceutical information systems and manufacturing management platforms
- Focus on intermediate reliability and long-term performance in pharmaceutical applications
Europe Market Split by Country
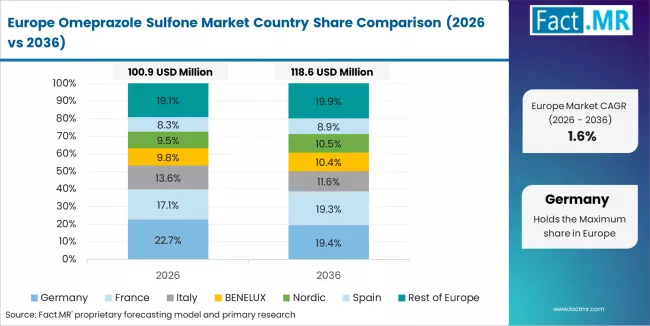
The omeprazole sulfone market in Europe is projected to show steady growth, with individual country performance varying across the region. Germany is expected to maintain its leadership position with a market value of USD 85.0 million in 2026, supported by its advanced pharmaceutical infrastructure, precision manufacturing capabilities, and strong research presence throughout major pharmaceutical regions.
UK follows with USD 60.0 million in 2026, driven by advanced production protocols, pharmaceutical innovation integration, and expanding specialty manufacturing networks serving both domestic and international markets. France holds USD 50.0 million in 2026, supported by pharmaceutical infrastructure expansion and growing adoption of high-purity intermediate systems. Italy commands USD 40.0 million in 2026, while Spain accounts for USD 30.0 million in 2026. The Rest of Europe region, including Nordic countries, Eastern Europe, and smaller Western European markets, holds USD 115.0 million in 2026, representing diverse market opportunities with established pharmaceutical and manufacturing capabilities.
Competitive Landscape of the Omeprazole Sulfone Market
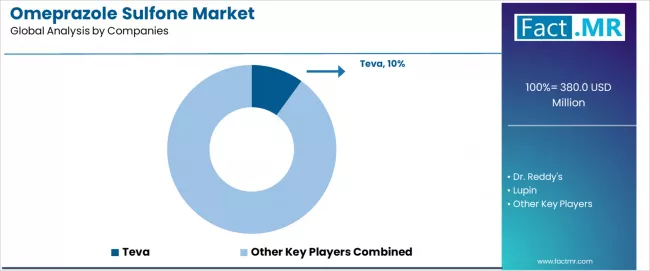
The market is characterized by competition among established pharmaceutical companies, specialty intermediate companies, and manufacturing technology suppliers focused on delivering high-purity, consistent, and reliable intermediate systems. Companies are investing in production technology advancement, quality control enhancement, strategic partnerships, and customer technical support to deliver effective, efficient, and reliable intermediate solutions that meet stringent pharmaceutical and research requirements. Quality optimization, purity validation protocols, and supply chain strategies are central to strengthening product portfolios and market presence.
Teva leads the market with comprehensive high-purity intermediate system offerings with a focus on quality consistency and production reliability for pharmaceutical applications, holding a 10.0% market share. Dr. Reddy's provides specialized manufacturing systems with focus on API intermediate applications and comprehensive technical support services. Lupin focuses on advanced intermediate technologies and customized pharmaceutical solutions for production systems serving global markets. Sandoz delivers established manufacturing systems with strong quality control systems and customer service capabilities.
Aurobindo operates with a focus on bringing innovative intermediate technologies to specialized pharmaceutical applications and emerging markets. Sun Pharma provides comprehensive production system portfolios, including advanced intermediate services, across multiple pharmaceutical applications and manufacturing processes. Cipla specializes in customized manufacturing solutions and quality management systems for pharmaceutical systems with focus on regulatory compliance. Hetero provides reliable supply chain solutions and technical expertise to enhance market accessibility and customer access to essential intermediate systems.
Key Players in the Omeprazole Sulfone Market
- Teva
- Dr. Reddy's
- Lupin
- Sandoz
- Aurobindo
- Sun Pharma
- Cipla
- Hetero
- Zhejiang Huahai
- Amneal
- Torrent
- Alkem
- Zydus
- Jubilant
- Apotex
Bibliography
- IQVIA Institute. (2024). Global medicine spending and usage trends. IQVIA.
- Pharmaceuticals Export Promotion Council (Pharmexcil), India. (2024). Pharmaceutical export statistics FY2024. Pharmexcil.
- United States Pharmacopeia (USP). (2024). Omeprazole related compound specifications. USP.
- Dr. Reddy's Laboratories Ltd. (2024). Annual report 2024: API manufacturing capabilities. Dr. Reddy's.
- Aurobindo Pharma Ltd. (2024). Annual report 2024: Formulations and API business. Aurobindo.
- European Pharmacopoeia (EP). (2024). Omeprazole monograph: Related substances specifications. EDQM.
This bibliography is provided for reader reference. The full Fact.MR report contains the complete reference list with primary research documentation.
This Report Addresses
- Market sizing and quantitative forecast metrics detailing the precise expenditure dedicated to omeprazole sulfone across major industrial sectors through 2036.
- Segmentation analysis mapping the adoption patterns of specific purity categories and evaluating the technical and economic factors driving segment share distribution.
- Regional deployment intelligence comparing growth trajectories across Asia Pacific, North America, Europe, Latin America, and Middle East and Africa.
- Regulatory compliance assessment analyzing how evolving standards and policies affect product specifications and market access in key consuming regions.
- Competitive posture evaluation tracking the positioning of leading producers and the structural factors determining competitive advantage.
- Capital investment strategic guidance defining the procurement and capacity planning considerations for industry participants.
- Supply chain risk analysis identifying the specific feedstock, logistics, and regulatory variables that constrain market expansion.
- Custom data delivery formats encompassing interactive dashboards, raw Excel datasets, and PDF narrative reports.
Omeprazole Sulfone Market Definition
Omeprazole sulfone is a chemical intermediate and metabolite of omeprazole, the proton pump inhibitor used to treat gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. Chemically, it is the sulfone oxidation product of omeprazole (5-methoxy-2-[[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]sulfonyl]-1H-benzimidazole). It is used primarily as an intermediate in omeprazole API synthesis, as a pharmaceutical reference standard for impurity profiling, and as a research compound in drug metabolism studies.
Omeprazole Sulfone Market Inclusions
Market scope covers global and regional revenue for omeprazole sulfone across purity levels of 99% and above and below 99%. Application segments include API intermediate and research. Grade segments span pharmaceutical and laboratory. The forecast period is 2026 to 2036.
Omeprazole Sulfone Market Exclusions
The scope excludes finished omeprazole drug products (capsules, tablets, injectables). Other PPI intermediates (lansoprazole sulfone, pantoprazole sulfone) are excluded. Omeprazole API in its final form falls outside the boundary.
Omeprazole Sulfone Market Research Methodology
- Primary Research: Analysts engaged with procurement specialists, process engineers, and supply chain directors across producing and consuming regions to map the specific decision criteria triggering purchasing and specification changes.
- Desk Research: Data collection aggregated regulatory filings, trade body publications, standards body documentation, and company disclosures relevant to the product category.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of production volumes and trade flows, applying region-specific demand curves to project adoption velocity through 2036.
- Data Validation and Update Cycle: Projections are tested against publicly reported industry expenditure guidance and cross-referenced with primary interview data to ensure internal consistency.
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 391.40 million to USD 526.01 million, at a CAGR of 3.0% |
| Market Definition | Omeprazole sulfone is a chemical intermediate and metabolite of omeprazole, the proton pump inhibitor used to treat gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. Chemically, it is the sulfone oxidation product of omeprazole (5-methoxy-2-[[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]sulfonyl]-1H-benzimidazole). |
| Purity Segmentation | ≥ 99%, < 99% |
| Application Segmentation | API Intermediate, Research |
| Grade Segmentation | Pharma, Lab |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | India, China, Italy, Germany, South Korea, USA, Japan, and 40 plus countries |
| Key Companies Profiled | Teva, Dr. Reddy's, Lupin, Sandoz, Aurobindo, Sun Pharma, Cipla, Hetero, Zhejiang Huahai, Amneal, Torrent, Alkem, Zydus, Jubilant, Apotex |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up methodology reconciling production, trade, and consumption data with primary research validation. |
Omeprazole Sulfone Market by Segments
-
Purity :
- ≥ 99%
- < 99%
-
Application :
- API Intermediate
- Research
-
Grade :
- Pharma
- Lab
-
Region :
-
Asia Pacific
- China
- India
- Japan
- South Korea
- Australia & New Zealand
- Thailand
- Singapore
- Malaysia
- Indonesia
- Philippines
- Vietnam
- Taiwan
- Hong Kong
- ASEAN
- Rest of Asia Pacific
-
North America
- United States
- Canada
- Mexico
-
Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Belgium
- Switzerland
- Austria
- Sweden
- Norway
- Denmark
- Finland
- Poland
- Czech Republic
- Hungary
- Romania
- Portugal
- Ireland
- Greece
- Nordic
- BENELUX
- Rest of Europe
-
Latin America
- Brazil
- Argentina
- Chile
- Mexico
- Colombia
- Peru
- Venezuela
- Ecuador
- Uruguay
- Rest of Latin America
-
Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- Other GCC Countries
- Turkey
- South Africa
- Egypt
- Nigeria
- Morocco
- Israel
- Kenya
- Other African Countries
- Rest of Middle East & Africa
-
- Frequently Asked Questions -
How large is the demand for Omeprazole Sulfone in the global market in 2026?
Demand for Omeprazole Sulfone in the global market is estimated to be valued at USD 391.40 million in 2026.
What will be the market size of Omeprazole Sulfone in the global market by 2036?
Market size for Omeprazole Sulfone is projected to reach USD 526.01 million by 2036.
What is the expected demand growth for Omeprazole Sulfone in the global market between 2026 and 2036?
Demand for Omeprazole Sulfone is expected to grow at a CAGR of 3.0% between 2026 and 2036.
Which Purity is poised to lead global sales by 2026?
≥ 99% accounts for 70.0% share in 2026.
How is the Application segment structured in this report?
API Intermediate holds a 80.0% share in 2026 within the Application segment.
What is the growth outlook for India in this report?
India is projected to grow at a CAGR of 4.0% during 2026 to 2036.
What is Omeprazole Sulfone and what is it mainly used for?
Omeprazole sulfone is a chemical intermediate and metabolite of omeprazole, the proton pump inhibitor used to treat gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome. Chemically, it is the sulfone oxidation product of omeprazole (5-methoxy-2-[[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]sulfonyl]-1H-benzimidazole).
How does Fact.MR build and validate the Omeprazole Sulfone forecast?
Forecasting models apply a hybrid top-down and bottom-up methodology starting with production and trade data, and cross-validate projections against primary research findings and quarterly industry data releases.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Purity
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Purity, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Purity, 2026 to 2036
- ≥ 99%
- < 99%
- ≥ 99%
- Y to o to Y Growth Trend Analysis By Purity, 2021 to 2025
- Absolute $ Opportunity Analysis By Purity, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- API Intermediate
- Research
- API Intermediate
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Grade
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Grade, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Grade, 2026 to 2036
- Pharma
- Lab
- Pharma
- Y to o to Y Growth Trend Analysis By Grade, 2021 to 2025
- Absolute $ Opportunity Analysis By Grade, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Purity
- By Application
- By Grade
- By Country
- Market Attractiveness Analysis
- By Country
- By Purity
- By Application
- By Grade
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Purity
- By Application
- By Grade
- By Country
- Market Attractiveness Analysis
- By Country
- By Purity
- By Application
- By Grade
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Purity
- By Application
- By Grade
- By Country
- Market Attractiveness Analysis
- By Country
- By Purity
- By Application
- By Grade
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Purity
- By Application
- By Grade
- By Country
- Market Attractiveness Analysis
- By Country
- By Purity
- By Application
- By Grade
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Purity
- By Application
- By Grade
- By Country
- Market Attractiveness Analysis
- By Country
- By Purity
- By Application
- By Grade
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Purity
- By Application
- By Grade
- By Country
- Market Attractiveness Analysis
- By Country
- By Purity
- By Application
- By Grade
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Purity
- By Application
- By Grade
- By Country
- Market Attractiveness Analysis
- By Country
- By Purity
- By Application
- By Grade
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Purity
- By Application
- By Grade
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Purity
- By Application
- By Grade
- Competition Analysis
- Competition Deep Dive
- Teva
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Dr. Reddy's
- Lupin
- Sandoz
- Aurobindo
- Sun Pharma
- Cipla
- Hetero
- Zhejiang Huahai
- Amneal
- Torrent
- Alkem
- Zydus
- Jubilant
- Apotex
- Teva
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Grade, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Grade, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Grade, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Grade, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Grade, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Grade, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Grade, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Grade, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Purity
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by Grade, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Grade, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Grade
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Purity
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by Grade, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Grade, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Grade
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Purity
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by Grade, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Grade, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by Grade
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Purity
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by Grade, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Grade, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by Grade
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Purity
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Grade, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Grade, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Grade
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Purity
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by Grade, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Grade, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by Grade
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Purity
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Grade, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Grade, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Grade
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Purity
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Grade, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Grade, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Grade
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis