Needle Protection Systems Market Size, Share, Growth and Forecast (2026 - 2036)
Needle Protection Systems Market Size and Share Forecast Outlook 2026 to 2036
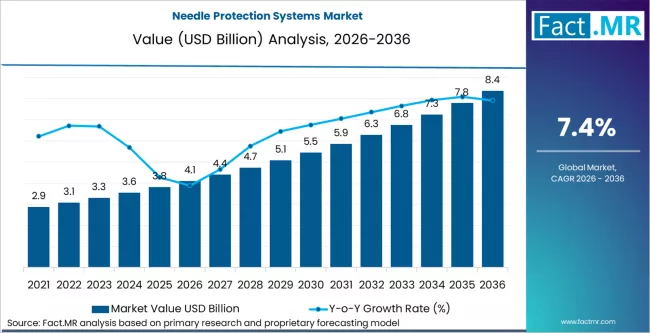
Needle protection systems market is projected to grow from USD 4.1 billion in 2026 to USD 8.4 billion by 2036, at a CAGR of 7.4%. Safety Syringes (Passive/Active) will dominate with a 34.0% market share, while medication administration (im/sc/id) will lead the clinical use cases segment with a 30.0% share.
Needle Protection Systems Market Forecast and Outlook 2026 to 2036
The needle protection systems market is projected to grow from USD 4.1 billion in 2026 to USD 8.4 billion by 2036, reflecting a 7.40% CAGR.
Needle Protection Systems Market Key Takeaways
- Needle Protection Systems Market Value (2026): USD 4.1 billion
- Needle Protection Systems Market Forecast Value (2036): USD 8.4 billion
- Needle Protection Systems Market Forecast CAGR: 7.4%
- Leading Type in Needle Protection Systems Market: Safety syringes (passive or active)
- Key Growth Regions in Needle Protection Systems Market: North America, Europe, Asia Pacific, Latin America, Middle East & Africa
- Top Players in Needle Protection Systems Market: Becton, Dickinson and Company; ICU Medical; Terumo Corporation; Smiths Medical; B. Braun Melsungen AG
This segment serves hospitals, clinics, and laboratories where prevention of needle stick injuries is a routine operational requirement rather than a discretionary feature. Product selection focuses on reliability of activation, compatibility with existing syringes or devices, and ease of use during high-volume procedures.
Once a protection system is adopted within an institution, change tends to be limited since training programs, waste handling protocols, and purchasing contracts are built around fixed device types. Procurement teams value consistent performance, stable supply, and clear compliance with safety standards. Usage volumes follow procedure counts and staffing levels rather than changes in device design. Growth is supported by wider enforcement of safety regulations, expansion of healthcare services, and continued emphasis on workplace safety practices rather than rapid turnover of protection technologies.
Needle Protection Systems Market
| Metric | Value |
|---|---|
| Market Value (2026) | USD 4.1 billion |
| Forecast Value (2036) | USD 8.4 billion |
| Forecast CAGR (2026-2036) | 7.4% |
Category
| Category | Segments |
|---|---|
| Device Categories | Safety syringes (passive or active); Safety IV catheters and blood collection sets; Needleless connectors and closed IV systems; Sharps containers and disposal systems; Training, compliance and monitoring services |
| Clinical Use Cases | Medication administration (IM/SC/ID); Phlebotomy and blood collection; IV therapy and vascular access; Surgical suturing (blunt-tip adoption); Home care or self-injection safety |
| End User or Channel | Hospitals; Ambulatory and outpatient clinics; Home healthcare providers; Blood banks or labs; Others |
| Region | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
Needle Protection Systems Market Segmental Analysis
Which Device Category Holds the Leading Share of the Needle Protection Systems Market?
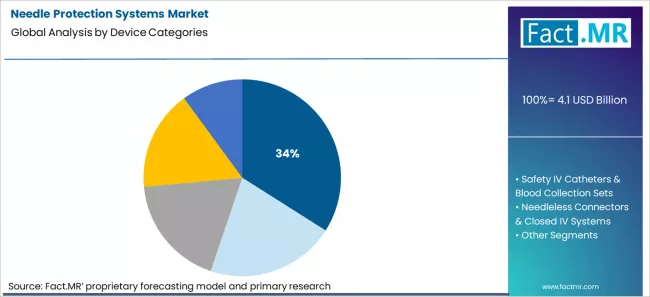
Fact.MR analysis indicates that safety syringes, including passive and active designs, hold the leading share at 34% of total device category allocation in the needle protection systems market. This position reflects where injection volume concentrates across hospitals, clinics, and outpatient settings. Medication delivery by injection remains one of the most frequent clinical acts in healthcare systems. Budget ownership for these devices sits within central procurement and safety compliance programmes rather than within individual departments. Standardisation policies and occupational safety mandates focus first on the highest-volume exposure points. Replacement cycles follow consumption rather than capital planning. This category sits at the centre of daily clinical workflow, which concentrates unit volumes and secures its position as the largest device segment in the overall market structure.
Which Clinical Use Case Accounts for The Largest Share of Market Activity?
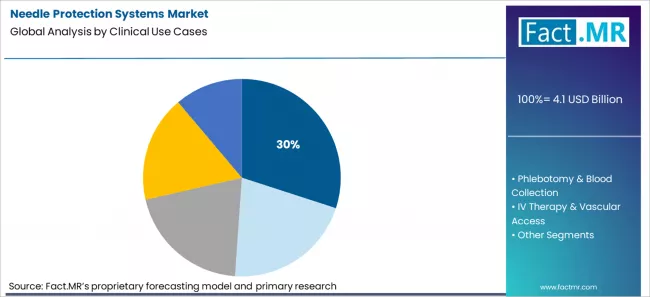
Fact.MR assessment shows that medication administration by intramuscular, subcutaneous, and intradermal routes represents the leading clinical use case at 30% of total activity in the needle protection systems market. This reflects the structure of care delivery rather than procedure complexity. Injections occur across almost every specialty, across inpatient, outpatient, and community settings. Each administration event carries a compliance and exposure control requirement, which multiplies the number of protected devices consumed. Budget responsibility for this activity sits with hospital operations and safety governance functions. The sheer frequency of routine injections concentrates demand here, which secures this use case the largest share of total market volume.
What are the Drivers, Restraints, and Key Trends of the Needle Protection Systems Market?
- Drivers: Clinical and regulatory emphasis on reducing needlestick injuries and occupational exposure risk in hospitals, clinics, and long-term care facilities is increasing specification of mechanical needle protection systems that shield sharps after use to safeguard health care workers.
- Restraint: Variability in reimbursement for safety devices and cost pressures in budget-limited facilities can delay widespread replacement of traditional devices and slow acceptance among price-constrained purchasers.
- Trend 1: Expansion of passive safety designs that automatically activate protection without additional user steps, supporting consistent compliance with safety protocols.
- Trend 2: Increased integration of needle protection with routine procedural workflows, including prefilled syringes and needle-integrated injection systems, to streamline handling and disposal.
Country
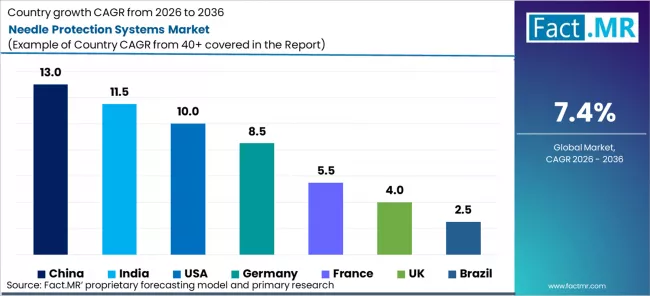
| Country | CAGR (2026-2036) |
|---|---|
| USA | 10.0% |
| China | 13.0% |
| India | 11.5% |
| Germany | 8.5% |
| France | 5.5% |
| UK | 4.0% |
| Brazil | 2.5% |
The report covers an in-depth analysis of 40+ countries; top-performing countries are highlighted below.
What Drives Institutional Adoption in the Needle Protection Systems Market in USA?
Workplace injury liability and hospital compliance programs explain why the needle protection systems market in USA is advancing near 10% per year through 2036. Healthcare providers face sustained pressure to reduce sharps injuries and documentation exposure, which makes safety-engineered devices a procurement requirement rather than a clinical preference. Adoption is led by hospital systems and group purchasing organizations, not individual departments. Once included in standard kits, usage becomes routine. Growth therefore comes from protocol enforcement and audit requirements, not from discretionary product switching. Expansion follows regulatory scrutiny and litigation risk management, which keeps demand stable and replacement cycles predictable across acute care and outpatient settings nationwide.
Why Does Scale and Policy Enforcement Shape the Needle Protection Systems Market in China?
Centralized hospital administration and workforce safety mandates explain why a 13% annual trajectory through 2036 is shaping the needle protection systems market in China. Large public hospitals manage high procedure volumes, which makes injury reduction a throughput and staffing issue rather than only a compliance topic. Once safety devices are specified in provincial purchasing lists, adoption spreads quickly. Growth therefore follows policy enforcement and capacity expansion, not local clinical advocacy. The category scales because hospital networks standardize consumables, not because individual facilities experiment. Long-term volume depends on continued expansion of regulated care settings and tightening occupational safety supervision.
How Do System Formalization and Training Influence the Needle Protection Systems Market in India?
Gradual formalization of hospital practices explains why the needle protection systems market in India is progressing near 11.5% per year through 2036. Many facilities are shifting from cost-first procurement to protocol-driven purchasing as accreditation and training requirements spread. Safety devices are adopted when they reduce training burden and incident reporting risk. Growth therefore comes from institutional upgrading rather than from price competition alone. Private hospital chains and diagnostic networks lead adoption, then standards diffuse outward. The category expands because organized care is expanding, not because disposable usage is rising uniformly. Penetration remains uneven, tracking where audit and certification pressure is strongest.
What Role Does Compliance Culture Play in the Needle Protection Systems Market in Germany?
A strong occupational safety framework explains why an 8.5% growth path through 2036 is steadily widening use of the needle protection systems market in Germany. Hospitals and clinics operate under strict inspection regimes, which makes safety-engineered devices part of baseline procurement. Adoption is conservative but persistent, driven by documentation and liability management rather than cost savings. Growth therefore comes from gradual replacement of legacy products and tightening enforcement standards. The category expands because compliance expectations rise, not because procedure volumes change rapidly. Once specified in tenders, usage is stable. Market development follows regulatory interpretation and audit cycles more than supplier-driven innovation.
How Does Public System Structure Affect the Needle Protection Systems Market in France?
Centralized healthcare governance explains why the needle protection systems market in France is moving near 5.5% per year through 2036. Safety devices are adopted through national and regional procurement decisions, not through department-level choice. Hospitals implement changes when standards are updated or contracts renewed. Growth therefore follows policy cycles and budget approvals rather than incident-driven urgency. The category expands because compliance frameworks evolve, not because usage intensity increases. Once included in standard supply lists, penetration becomes stable. Development remains incremental, shaped by administrative processes and inspection regimes more than by supplier competition or rapid product differentiation.
How Do Capacity and Budget Limits Shape the Needle Protection Systems Market in UK?
System-wide cost control explains why the needle protection systems market in UK is advancing near 4% per year through 2036. Adoption is driven by minimum compliance requirements rather than proactive upgrading. Hospitals prioritize meeting safety standards at lowest acceptable cost. Growth therefore follows replacement cycles and audit findings, not expansion of device usage. Central procurement frameworks slow change but ensure uniformity. The category expands because risk cannot be ignored, not because funding allows broad upgrades. Penetration remains conservative, shaped by staffing pressure and budget ceilings. Long-term growth depends on inspection intensity and policy enforcement rather than supplier-led innovation or clinical demand shifts.
Why Needle Protection Systems Are Becoming a Procurement Standard Rather Than a Product Choice?
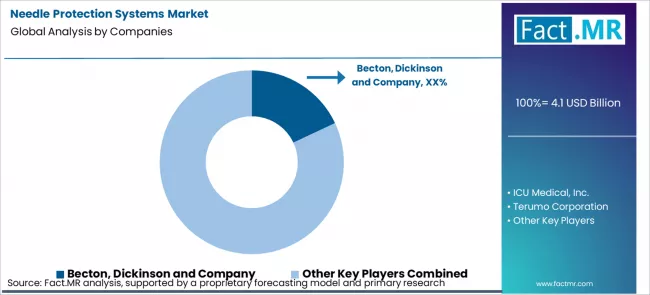
As per Fact.MR observations, the needle protection systems market is being shaped by institutional safety mandates rather than by discretionary purchasing behaviour. Hospitals and health systems now define sharps injury prevention at policy level, which fixes demand before any supplier engagement begins. Becton, Dickinson and B. Braun benefit from broad device portfolios that allow safety mechanisms to be standardised across syringes, catheters, and infusion lines. ICU Medical and Smiths Medical compete through integration into closed medication delivery and IV therapy workflows, where needle protection becomes part of a larger safety architecture. Terumo holds positions in regions where proprietary device platforms and long standing clinical relationships influence standard setting. Purchasing decisions are governed by training burden, activation reliability, and compatibility with existing consumables rather than by unit price. Once a protection system is written into protocol, switching costs rise because staff retraining and inventory replacement become operational risks. In this structure, competitive advantage sits with suppliers that control procedure level specifications rather than with those offering isolated safety features. This underpins Fact.MR’s long term view of the segment.
Key Players in Needle Protection Systems Market
- Becton, Dickinson and Company
- ICU Medical, Inc.
- Terumo Corporation
- Smiths Medical
- B. Braun Melsungen AG
References
Centers for Disease Control and Prevention. (2023). Sharps safety for healthcare settings. CDC.
International Organization for Standardization. (2023). ISO 23908:2023 Sharps injury protection - Requirements and test methods -Sharps protection features for single-use hypodermic needles, introducers for catheters and needles used for blood sampling. ISO.
Occupational Safety and Health Administration. (2024). Bloodborne pathogens and needlestick prevention. U.S. Department of Labor, OSHA.
Centers for Disease Control and Prevention. (2024). Workbook for designing, implementing, and evaluating a sharps injury prevention program. CDC.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Device Categories | Safety syringes (passive or active), safety IV catheters and blood collection sets, needleless connectors and closed IV systems, sharps containers and disposal systems, training, compliance and monitoring services |
| Clinical Use Cases | Medication administration (IM/SC/ID), phlebotomy and blood collection, IV therapy and vascular access, surgical suturing (blunt-tip adoption), home care or self-injection safety |
| End User or Channel | Hospitals, ambulatory and outpatient clinics, home healthcare providers, blood banks or labs, others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Becton, Dickinson and Company, ICU Medical, Terumo Corporation, Smiths Medical, B. Braun Melsungen AG |
| Additional Attributes | Dollar by sales by device category, clinical use case, and end user; focus on compliance-driven procurement and safety standard adoption; demand tied to procedure volumes, training protocols, and regulatory enforcement |
Needle Protection Systems Market Segmentation
-
Device Categories :
- Safety Syringes (Passive/Active)
- Safety IV Catheters & Blood Collection Sets
- Needleless Connectors & Closed IV Systems
- Sharps Containers & Disposal Systems
- Training, Compliance & Monitoring Services
-
Clinical Use Cases :
- Medication Administration (IM/SC/ID)
- Phlebotomy & Blood Collection
- IV Therapy & Vascular Access
- Surgical Suturing (Blunt-Tip Adoption)
- Home Care or Self-Injection Safety
-
End User or Channel :
- Hospitals
- Ambulatory & Outpatient Clinics
- Home Healthcare Providers
- Blood Banks or Labs
- Others
-
Region :
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
- Asia Pacific
- Frequently Asked Questions -
How big is the needle protection systems market in 2026?
The global needle protection systems market is estimated to be valued at USD 4.1 billion in 2026.
What will be the size of needle protection systems market in 2036?
The market size for the needle protection systems market is projected to reach USD 8.4 billion by 2036.
How much will be the needle protection systems market growth between 2026 and 2036?
The needle protection systems market is expected to grow at a 7.4% CAGR between 2026 and 2036.
What are the key product types in the needle protection systems market?
The key product types in needle protection systems market are safety syringes (passive/active), safety iv catheters & blood collection sets, needleless connectors & closed iv systems, sharps containers & disposal systems and training, compliance & monitoring services.
Which clinical use cases segment to contribute significant share in the needle protection systems market in 2026?
In terms of clinical use cases, medication administration (im/sc/id) segment to command 30.0% share in the needle protection systems market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Device Categories
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Device Categories, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Device Categories, 2026 to 2036
- Safety Syringes (Passive/Active)
- Safety IV Catheters & Blood Collection Sets
- Needleless Connectors & Closed IV Systems
- Sharps Containers & Disposal Systems
- Training, Compliance & Monitoring Services
- Safety Syringes (Passive/Active)
- Y to o to Y Growth Trend Analysis By Device Categories, 2021 to 2025
- Absolute $ Opportunity Analysis By Device Categories, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Clinical Use Cases
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Clinical Use Cases, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Clinical Use Cases, 2026 to 2036
- Medication Administration (IM/SC/ID)
- Phlebotomy & Blood Collection
- IV Therapy & Vascular Access
- Surgical Suturing (Blunt-Tip Adoption)
- Home Care or Self-Injection Safety
- Medication Administration (IM/SC/ID)
- Y to o to Y Growth Trend Analysis By Clinical Use Cases, 2021 to 2025
- Absolute $ Opportunity Analysis By Clinical Use Cases, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User or Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User or Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User or Channel, 2026 to 2036
- Hospitals
- Ambulatory & Outpatient Clinics
- Home Healthcare Providers
- Blood Banks or Labs
- Others
- Hospitals
- Y to o to Y Growth Trend Analysis By End User or Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By End User or Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Device Categories
- By Clinical Use Cases
- By End User or Channel
- Competition Analysis
- Competition Deep Dive
- Becton, Dickinson and Company
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- ICU Medical, Inc.
- Terumo Corporation
- Smiths Medical
- B. Braun Melsungen AG
- Others
- Becton, Dickinson and Company
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Device Categories, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Clinical Use Cases, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User or Channel, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Device Categories, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Clinical Use Cases, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User or Channel, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Device Categories, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Clinical Use Cases, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User or Channel, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Device Categories, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Clinical Use Cases, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User or Channel, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Device Categories, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Clinical Use Cases, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User or Channel, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Device Categories, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Clinical Use Cases, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User or Channel, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Device Categories, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Clinical Use Cases, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User or Channel, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Device Categories, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Clinical Use Cases, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User or Channel, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Device Categories, 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Device Categories, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Device Categories
- Figure 6: Global Market Value Share and BPS Analysis by Clinical Use Cases, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Clinical Use Cases, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Clinical Use Cases
- Figure 9: Global Market Value Share and BPS Analysis by End User or Channel, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by End User or Channel, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User or Channel
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Device Categories, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Device Categories, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Device Categories
- Figure 26: North America Market Value Share and BPS Analysis by Clinical Use Cases, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Clinical Use Cases, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Clinical Use Cases
- Figure 29: North America Market Value Share and BPS Analysis by End User or Channel, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by End User or Channel, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User or Channel
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Device Categories, 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Device Categories, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Device Categories
- Figure 36: Latin America Market Value Share and BPS Analysis by Clinical Use Cases, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Clinical Use Cases, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Clinical Use Cases
- Figure 39: Latin America Market Value Share and BPS Analysis by End User or Channel, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End User or Channel, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User or Channel
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Device Categories, 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Device Categories, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Device Categories
- Figure 46: Western Europe Market Value Share and BPS Analysis by Clinical Use Cases, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Clinical Use Cases, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Clinical Use Cases
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User or Channel, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End User or Channel, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User or Channel
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Device Categories, 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Device Categories, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Device Categories
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Clinical Use Cases, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Clinical Use Cases, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Clinical Use Cases
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User or Channel, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End User or Channel, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User or Channel
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Device Categories, 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Device Categories, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Device Categories
- Figure 66: East Asia Market Value Share and BPS Analysis by Clinical Use Cases, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Clinical Use Cases, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Clinical Use Cases
- Figure 69: East Asia Market Value Share and BPS Analysis by End User or Channel, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End User or Channel, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User or Channel
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Device Categories, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Device Categories, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Device Categories
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Clinical Use Cases, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Clinical Use Cases, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Clinical Use Cases
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User or Channel, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End User or Channel, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User or Channel
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Device Categories, 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Device Categories, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Device Categories
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Clinical Use Cases, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Clinical Use Cases, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Clinical Use Cases
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User or Channel, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End User or Channel, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User or Channel
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis