Orphan Disease Biomarkers Market Size, Share, Growth and Forecast (2026 - 2036)
Orphan Disease Biomarkers Market is segmented by Product (Biomarker Kits, Reagents & Assays, Software & Bioinformatics Tools, Services), Application (Drug Development, Diagnostic Applications, Personalized Medicine, Research Applications), End Use (Pharmaceutical & Biotechnology Companies, Academic & Research Institutes, Diagnostic Laboratories, Contract Research Organizations (CROs)), and Region, with forecasts covering the period from 2026 to 2036.
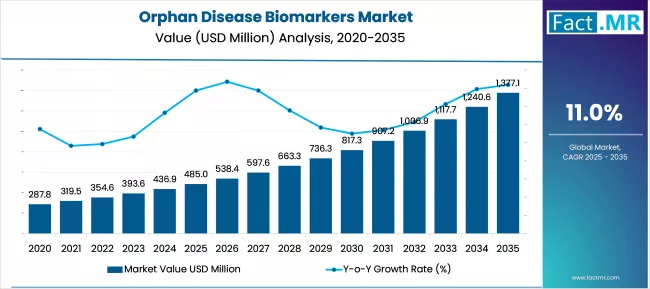
According to Fact.MR estimates, the global orphan disease biomarkers market market was valued at USD 485.0 million in 2025. The market is projected to reach USD 538.4 million in 2026 and is expected to grow to USD 1,528.6 million by 2036, expanding at a CAGR of 11.0%. Biomarker Kits is anticipated to account for 46.0% of the product segment in 2026, while Drug Development is expected to remain the leading application with around 42.0% share.
Orphan Disease Biomarkers Market Analysis and Forecast by Fact.MR
According to Fact.MR estimates, the global orphan disease biomarkers market market was valued at USD 485.0 million in 2025. The market is projected to reach USD 538.4 million in 2026 and is expected to grow to USD 1,528.6 million by 2036, expanding at a CAGR of 11.0%. Biomarker Kits is anticipated to account for 46.0% of the product segment in 2026, while Drug Development is expected to remain the leading application with around 42.0% share.
Summary of Orphan Disease Biomarkers Market
- Market Snapshot
- The Orphan Disease Biomarkers market was valued at USD 485 million in 2025.
- By 2036, the Orphan Disease Biomarkers market is expected to be worth USD 1,528.6 million.
- From 2026 to 2036, the market is projected to expand at a CAGR of 11.0%.
- The market is projected to create an incremental opportunity of USD 990.3 million between 2026 and 2036.
- In 2026, Biomarker Kits are expected to account for 46% of the product segment, driven by rising demand for ready-to-use rare disease diagnostic panels in pharma R&D and clinical validation workflows.
- The United States (12.8%) and Germany (12.2%) are two of the fastest growing markets in the world.
- Demand and Growth Drivers
- Demand is shaped by expanding orphan drug pipelines, where pharmaceutical and biotechnology sponsors need validated biomarkers to stratify small rare disease patient populations, reduce trial failure rates, and support regulatory submissions for accelerated approval pathways.
- Growth reflects the steady rise of companion diagnostics tied to orphan therapies, as payers and regulators increasingly require biomarker-based patient selection to justify premium pricing for high-cost rare disease treatments.
- Adoption is increasing due to wider deployment of next-generation sequencing, multiplex immunoassays, and mass spectrometry platforms across specialty diagnostic labs, which lowers per-sample testing costs and brings rare disease biomarker workflows into routine clinical use.
- Product and Segment View
- Biomarker Kits hold 46% share of the product segment in 2026, anchored by ready-to-use panels for genetic testing, PCR-based detection, and NGS-ready library preparation used by orphan drug developers and specialty labs.
- Drug Development accounts for 42% of application demand in 2026, with target identification and clinical trial stratification workflows driving repeat-volume consumption of kits, reagents, and bioinformatics services.
- Pharmaceutical and Biotechnology Companies represent 48% of end-user demand in 2026, reflecting concentrated procurement by orphan drug developers and biotech research firms that run structured biomarker discovery and validation programs.
- Geography and Competitive Outlook
- North America leads the market, with the United States contributing the largest single-country revenue pool in 2026 and expanding at a 12.8% CAGR on the back of FDA orphan designation incentives and dense biotech clustering around Boston, San Francisco, and Research Triangle Park.
- Europe remains a structurally important region, led by Germany at 12.2% CAGR and the United Kingdom at 11.8% CAGR, supported by EMA orphan drug programs, national rare disease plans, and growing academic-industry co-development.
- Asia Pacific is a rising growth pocket where Japan (11.4%) and South Korea (11.0%) are scaling precision medicine programs and building out rare disease registries. Leading players include Roche Holdings, Thermo Fisher Scientific, Abbott Laboratories, Qiagen N.V., Bio-Rad Laboratories, Agilent Technologies, PerkinElmer, Illumina Inc., Danaher Corporation, and Merck KGaA.
- Analyst Opinion
- The orphan disease biomarkers market is no longer a niche pocket of clinical diagnostics. It is emerging as a core dependency for the orphan drug industry. Growth is shifting from one-off discovery projects toward repeat-volume contracts tied to clinical trials, companion diagnostic co-development, and long-term biomarker stewardship programs. Kit-based formats and NGS-driven workflows are consolidating spend with diagnostic majors that can combine assay development, regulatory expertise, and global distribution. The winners over the next decade will be firms that can package biomarker discovery, validation, and commercial diagnostic rollout into a single commercial offering rather than selling reagents and services in isolation.
Why is the Orphan Disease Biomarkers Market Growing?
- The orphan drug pipeline has expanded materially, and sponsors need validated biomarkers to enroll, stratify, and retain small rare disease patient cohorts, which directly drives repeat demand for kits, assays, and services.
- Regulatory pathways for rare disease therapies increasingly favor biomarker-anchored trial designs and companion diagnostic co-approvals, shifting biomarker spend from optional to structurally required in orphan drug development programs.
- Falling unit costs in NGS, digital PCR, and mass spectrometry are making rare disease biomarker testing economically viable in specialty labs, which widens the addressable user base beyond large pharmaceutical research centers.
Pharmaceutical and biotechnology sponsors carry the largest share of demand. Orphan drug programs are high-cost, long-duration investments where a single clinical failure can erase the economics of the entire program. Biomarker-guided enrollment and stratification reduces trial risk, shortens timelines for proof of concept, and supports regulatory submissions that require molecularly defined patient populations. That commercial pressure is turning biomarker procurement into a line item that sits alongside manufacturing, clinical operations, and CRO spend.
A second demand engine is the commercial diagnostic layer. Companion diagnostics and rare disease confirmatory panels are being built out alongside approved orphan therapies, which creates a post-launch revenue stream that sits behind the drug for as long as it is prescribed. This moves biomarker revenue from episodic trial use into recurring clinical use, which is a more attractive revenue profile for diagnostic majors and drives premium valuations across the segment.
Adoption is widening geographically. North America remains dominant, but Europe and Asia Pacific are scaling national rare disease strategies, reimbursement frameworks, and registry infrastructure that pull biomarker testing into routine clinical pathways. India, China, and Southeast Asia are still early, but they represent structural upside as local pharmaceutical sectors invest in orphan drug discovery and as sequencing infrastructure matures.
Segment-wise Analysis of Orphan Disease Biomarkers Market
- Biomarker Kits lead the product segment with 46% share in 2026, anchored by ready-to-use panels for genetic, PCR-based, and NGS applications in rare disease workflows.
- Drug Development dominates the application segment with 42% share in 2026, with target identification and clinical trial stratification as the highest-volume use cases.
- Pharmaceutical and Biotechnology Companies are the leading end user at 48% share in 2026, followed by academic and research institutes and contract research organizations.
The orphan disease biomarkers market segments across product, application, end use, technology, distribution channel, and packaging. Product coverage includes biomarker kits, reagents and assays, software and bioinformatics tools, and services. Application coverage spans drug development, diagnostic applications, personalized medicine, and research applications. Technology coverage includes genomics-based platforms, proteomics, immunoassays, and bioinformatics and AI. Distribution runs through direct sales, third-party distributors, and service providers, with kit-based and cold-chain packaging dominating the packaging view.
Biomarker Kits Lead the Product Segment
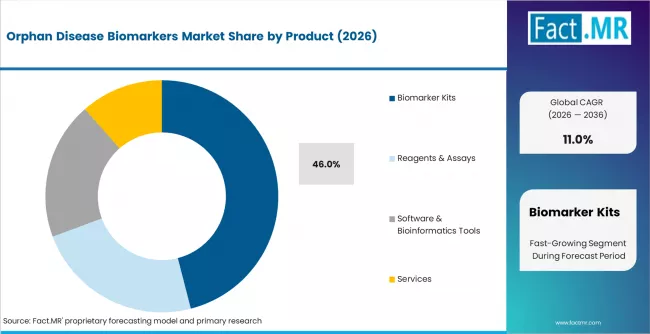
Biomarker kits account for 46% of product revenue in 2026, driven by consistent demand from pharmaceutical sponsors, biotech research firms, and specialty diagnostic labs that need standardized, regulatory-grade assay formats for rare disease workflows. Genetic testing kits, PCR-based detection kits, and NGS-based panels make up the core of the subsegment, while immunoassay kits capture ELISA and multiplex workflows used in protein biomarker applications.
The advantage of the kits subsegment is commercial leverage. Kits carry higher gross margins than individual reagents, they lock customers into validated workflows, and they support recurring volume across discovery, validation, and clinical use phases. That economic profile is why diagnostic majors such as Roche, Thermo Fisher, Qiagen, and Illumina are concentrating product investment on kit-based formats rather than on open-channel reagent sales.
Drug Development Leads the Application Segment
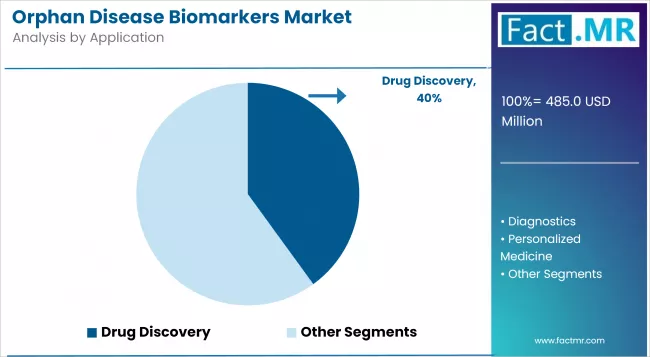
Drug development accounts for 42% of application demand in 2026. Within that, target identification and clinical trial stratification are the two largest pull-through use cases. Orphan drug sponsors rely on biomarker panels to identify molecular subtypes of rare diseases, select trial participants, and demonstrate molecular response during trials. That makes biomarker spend a structural input to the orphan drug cost stack rather than a discretionary line item.
Diagnostic applications make up the second-largest pool, led by rare disease diagnosis and early detection workflows in specialty labs. Personalized medicine and research applications together round out the rest of the segment, with personalized medicine gaining faster share as companion diagnostics become more common across approved orphan therapies.
Key Growth Drivers, Constraints, and Market Scope
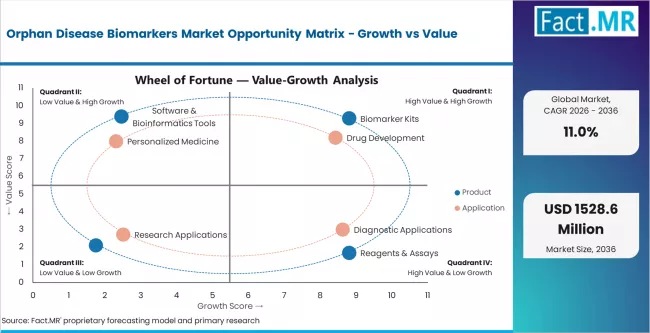
- Orphan drug pipeline expansion and regulatory preference for biomarker-anchored trials are creating structural demand growth across kits, reagents, and services.
- High development and validation costs, combined with small patient populations, limit the speed at which new biomarker assays reach commercial scale.
- Falling NGS and mass spectrometry unit costs, plus expanding rare disease registries, are opening up new adoption pockets in specialty labs and emerging markets.
The orphan disease biomarkers market is shaped by a mix of pull-through demand from the orphan drug industry, structural constraints from rare disease economics, and tailwinds from falling testing costs. Together, these forces set the pace at which biomarker products and services scale across clinical and commercial settings.
Growth Drivers
Growth is shaped by three reinforcing factors. First, the expansion of orphan drug pipelines is pulling structured biomarker demand into every phase of drug development, from target identification through post-market companion diagnostics. Second, regulatory frameworks in the United States, Europe, and Japan increasingly treat biomarker-guided trials and companion diagnostic submissions as standard practice for rare disease approvals. Third, platform maturation in NGS, mass spectrometry, and multiplex immunoassays is lowering per-sample costs and enabling higher testing volumes across a broader set of labs. These three forces together have shifted biomarker spend from episodic to recurring.
Constraints
The main constraint is economics. Rare disease biomarker development is expensive to validate against small patient populations, and reimbursement frameworks for specialty diagnostic tests remain uneven across regions. Regulatory validation timelines for companion diagnostics are long, and the expertise required to design and interpret biomarker panels for rare conditions is concentrated in a small number of specialized centers. Smaller biotech sponsors and labs in emerging markets face real access barriers, which slows the pace of geographic diffusion. Fragmented data standards across genomics, proteomics, and clinical datasets also create interoperability friction that raises the effective cost of biomarker adoption.
Opportunities
The largest opportunity is in integrated biomarker service offerings that combine discovery, validation, regulatory support, and commercial companion diagnostic rollout under one commercial contract. AI and multi-omics integration represents a second opportunity pool, with pharmaceutical sponsors willing to pay premium rates for platforms that improve target identification and patient stratification accuracy. A third opportunity sits in emerging Asia Pacific markets, where rare disease registries, local NGS capacity, and domestic orphan drug pipelines are all scaling together, creating a structural demand pull that has not yet been fully priced into global forecasts.
Regional Outlook Across Key Markets
.webp)
- North America leads global revenue, with the United States at 12.8% CAGR supported by FDA orphan drug designation programs, deep biotech venture capital, and concentrated pharmaceutical research infrastructure.
- Europe is anchored by Germany (12.2%) and the United Kingdom (11.8%), both supported by EMA orphan drug frameworks and national rare disease strategies that direct public funding into biomarker-enabled clinical research.
- Asia Pacific growth is led by Japan (11.4%) and South Korea (11.0%), driven by precision medicine programs, growing rare disease registries, and domestic investment in genomics and proteomics platforms.
- France (10.6%) represents a steady performer, supported by public research funding, an established pharmaceutical sector, and advancing national genome programs.
| Country | CAGR (2026-2036) |
|---|---|
| United States | 12.8% |
| Germany | 12.2% |
| United Kingdom | 11.8% |
| Japan | 11.4% |
| South Korea | 11.0% |
| France | 10.6% |
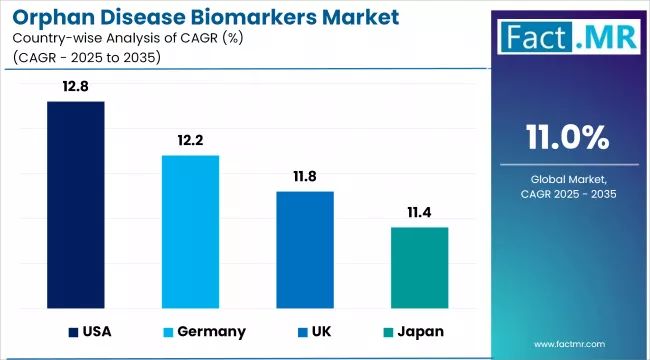
Regional performance tracks the distribution of orphan drug development activity and precision medicine infrastructure. North America leads both in absolute revenue and in growth pace because it combines regulatory incentives, private capital flowing into rare disease biotech, and the highest concentration of specialized diagnostic laboratories. European growth is more regulation-driven, with orphan drug frameworks and national rare disease plans translating into consistent demand. Asia Pacific is still catching up in absolute size but is scaling fastest relative to its base through precision medicine investment.
United States
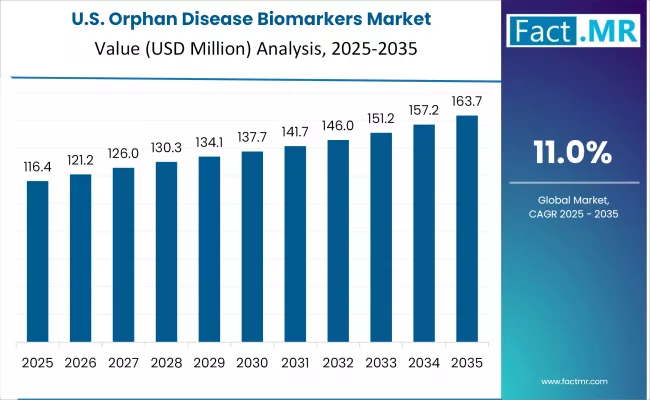
The United States is the largest single-country market, with 2026 revenue of approximately USD 199.9 million and a 12.8% CAGR through 2036. FDA orphan drug designation programs, the Rare Disease Act, and strong venture capital funding for rare disease biotech create a structural pull that is deeper than any other national market. Demand is concentrated in Boston, San Francisco, San Diego, and Research Triangle Park, where pharmaceutical and biotech sponsors run structured biomarker discovery and validation programs, and specialty labs handle companion diagnostic and clinical confirmatory testing.
- FDA orphan drug designations and breakthrough therapy pathways drive biomarker-anchored clinical trial design.
- Dense academic medical center and biotech ecosystems support high-volume discovery and validation work.
- Commercial payer engagement on companion diagnostics creates recurring post-launch revenue pools.
Germany
Germany anchors European demand with a 12.2% CAGR through 2036. The country combines an established pharmaceutical sector, strong public research infrastructure through Max Planck and Helmholtz networks, and active orphan drug development programs. Biomarker demand is being pulled into clinical research through German rare disease centers and expanding collaborations between pharma and academic hospitals in Munich, Heidelberg, and Berlin.
- National rare disease action plans and EMA orphan drug frameworks shape structured biomarker demand.
- Strong academic-industry links in Munich, Heidelberg, and Berlin accelerate discovery-to-clinic workflows.
- Public reimbursement for specialty diagnostic tests supports commercial diagnostic rollout.
United Kingdom
The United Kingdom posts an 11.8% CAGR through 2036. NHS-linked genomics programs, particularly the 100,000 Genomes Project and its successor NHS Genomic Medicine Service, have built a national infrastructure for sequencing-based biomarker workflows in rare diseases. That infrastructure is now pulling in commercial demand for validated kits and services, and it supports ongoing collaboration between biotech sponsors and academic medical centers.
- NHS Genomic Medicine Service anchors high-volume sequencing and biomarker testing in clinical settings.
- Strong biotech clustering around Cambridge, Oxford, and London supports discovery-side demand.
- MHRA orphan drug pathways and MIA regulatory frameworks shape predictable companion diagnostic approval routes.
Japan
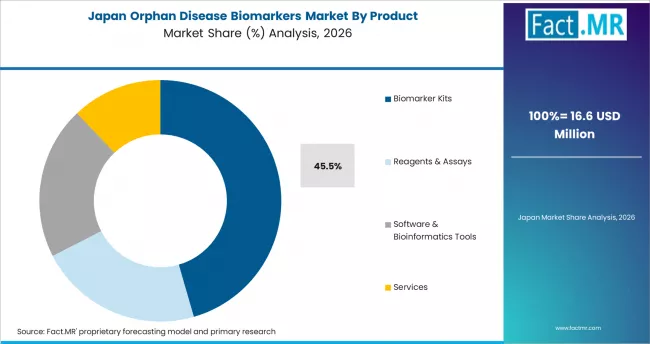
Japan is the leading Asian market for orphan disease biomarkers, with an 11.4% CAGR through 2036. PMDA orphan drug designations, an aging population with high prevalence of specific genetic disorders, and national precision medicine programs are driving structured biomarker demand. Domestic companies are co-developing companion diagnostics with global pharma, and universities in Tokyo, Osaka, and Kyoto are running large rare disease registries that pull biomarker testing volume.
- PMDA orphan drug framework and national precision medicine programs drive biomarker-anchored trial design.
- Strong domestic diagnostic firms are co-developing companion diagnostics with global pharma.
- Academic rare disease registries in Tokyo, Osaka, and Kyoto support biomarker validation at scale.
South Korea
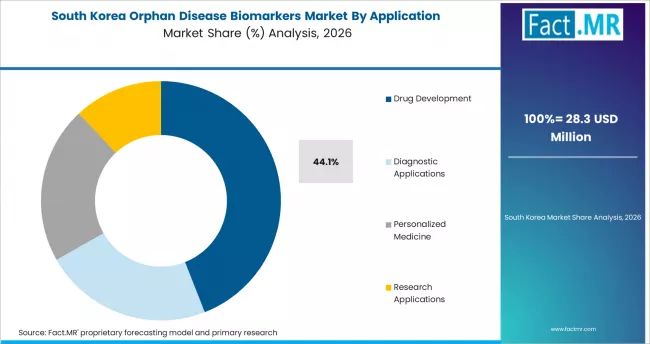
South Korea posts an 11.0% CAGR through 2036. The country combines rising domestic pharmaceutical R&D, a strong hospital-based clinical research base, and government funding for precision medicine through the Korea National Institute of Health. Seoul and Daejeon are emerging as biomarker discovery hubs, and Korean biotech firms are partnering with global diagnostic players to localize companion diagnostic offerings.
- Government-funded precision medicine programs direct public spending into rare disease biomarker research.
- Hospital-based clinical research networks support structured biomarker validation workflows.
- Growing domestic biotech pipeline is creating local companion diagnostic demand alongside global partnerships.
France
France posts a 10.6% CAGR through 2036. The country has one of Europe's most developed national rare disease plans, anchored by the Plan National Maladies Rares, and a dense network of rare disease reference centers that pull biomarker testing into routine specialty care. INSERM and CEA networks support public biomarker discovery research, and Paris and Lyon are emerging commercial hubs for diagnostic firms.
- National rare disease plans and INSERM-backed research networks support sustained public-sector demand.
- Rare disease reference centers pull biomarker testing into routine specialty clinical workflows.
- Paris and Lyon host expanding commercial operations of global diagnostic and biotech firms.
Competitive Benchmarking and Company Positioning
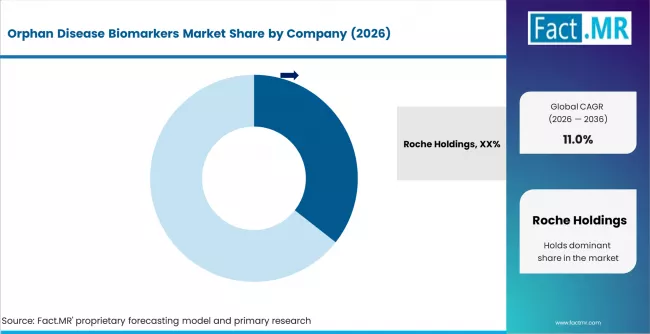
- The top three players, Roche Holdings, Thermo Fisher Scientific, and Abbott Laboratories, together control an estimated 40 to 50% of global market share in 2026, supported by broad biomarker portfolios and deep pharmaceutical customer relationships.
- Challengers including Qiagen, Bio-Rad, Agilent, and Illumina compete through technology-specific depth in genomics, proteomics, or application-specific panels rather than portfolio breadth.
- Competitive advantage is shifting toward integrated offerings that combine biomarker discovery, validation, and regulatory-grade commercial rollout rather than standalone reagent or instrument sales.
The orphan disease biomarkers market is moderately concentrated. Competition is less about unit pricing and more about scientific depth, regulatory validation capability, and the ability to co-develop companion diagnostics alongside orphan drug approvals. Switching costs are high because validated biomarker panels get embedded in pharmaceutical clinical trial designs and in regulatory submissions, which locks in supplier relationships for the life of a drug program.
Roche Holdings leads the market with an estimated 35.6% share in 2026, built on a deep diagnostic portfolio, strong pharmaceutical in-house customer base, and long-standing companion diagnostic co-development relationships. Thermo Fisher Scientific competes on platform breadth, combining mass spectrometry, NGS, and immunoassay capabilities with a strong services arm. Abbott Laboratories is anchored in immunoassay and molecular diagnostic strengths that translate well into rare disease commercial diagnostic rollout.
Challengers compete through focused depth. Qiagen brings sample preparation and molecular diagnostic strength. Bio-Rad Laboratories serves immunoassay and PCR-based rare disease applications. Agilent Technologies differentiates through mass spectrometry and bioinformatics, and Illumina is central to any NGS-based rare disease biomarker workflow. PerkinElmer, Danaher, and Merck KGaA round out the second tier with specialized strengths across reagents, instruments, and laboratory services.
Regional and specialized players create pricing pressure in emerging markets and in specific applications such as proteomics discovery or bioinformatics-only platforms. The strategic direction across the market is toward integrated offerings, where suppliers combine biomarker discovery, regulatory validation, and post-approval commercial diagnostic rollout into a single customer relationship that sits behind an orphan drug for its full commercial lifecycle.
Competitive Analysis
Competitive dynamics favor firms that can pair laboratory services with kit-based commercial diagnostics. Roche, Thermo Fisher, and Abbott all operate integrated businesses that sell kits, instruments, reagents, and services to the same pharmaceutical customers, which creates revenue stickiness and supports cross-selling into new orphan drug programs as they emerge. Specialized challengers are building targeted strengths, but the long-term direction of the market is consolidation around fewer, deeper platform relationships rather than fragmentation across specialist vendors.
Benchmarking Table
| Company | Portfolio Breadth | Regulatory & Companion Dx Track Record | Regional Reach | Pharma Customer Depth |
|---|---|---|---|---|
| Roche Holdings | High | High | Global | High |
| Thermo Fisher Scientific | High | Medium-High | Global | High |
| Abbott Laboratories | Medium-High | High | Global | Medium-High |
| Qiagen N.V. | Medium | Medium | Global | Medium |
| Illumina Inc. | Medium (NGS-focused) | Medium-High | Global | High |
Recent Developments
- Roche expanded its companion diagnostic portfolio in 2025 with new rare disease panel approvals tied to orphan oncology and hereditary disease indications, reinforcing its lead position in the market.
- Illumina launched updated rare disease NGS workflows in 2025 that integrate directly with clinical interpretation platforms, targeting specialty diagnostic labs and orphan drug sponsors.
- Thermo Fisher Scientific announced expanded CDMO and biomarker service capacity in 2026 to support orphan drug clinical trial work, reflecting rising pharmaceutical sponsor demand.
Leading Companies Shaping Orphan Disease Biomarkers Market
-
Major Players
- Roche Holdings
- Thermo Fisher Scientific
- Abbott Laboratories
- Qiagen N.V.
- Bio-Rad Laboratories
- Agilent Technologies
- Illumina Inc.
-
Emerging Players
- PerkinElmer
- Danaher Corporation
- Merck KGaA
- SOPHiA GENETICS
- Centogene
- Invitae Corporation
- Blueprint Genetics
- Fulgent Genetics
- 10x Genomics
- Genedata AG
Sources and Research References
- World Health Organization (WHO) rare disease data and genomics program publications
- National Institutes of Health (NIH) Genetic and Rare Diseases Information Center (GARD) and Rare Diseases Clinical Research Network
- European Medicines Agency (EMA) Committee for Orphan Medicinal Products (COMP) designation lists and orphan drug approval records
- U.S. Food and Drug Administration (FDA) Office of Orphan Products Development designation and approval databases
- Roche Holdings, Thermo Fisher Scientific, Abbott Laboratories, Illumina Inc., and Qiagen N.V. annual reports and investor presentations
- Orphanet rare disease registry and epidemiology publications
- Primary interviews with biomarker product managers, rare disease clinical researchers, diagnostic lab directors, and pharmaceutical procurement leads
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Key Questions This Report Addresses
- What is the size of the orphan disease biomarkers market in 2026 and 2036?
- What CAGR is the market expected to record between 2026 and 2036?
- Which product segment is projected to lead the market in 2026?
- Which application segment accounts for the largest share of market demand?
- How is the shift from discovery to commercial companion diagnostics changing biomarker revenue profiles?
- Which countries are the fastest-growing markets through 2036 and why?
- Who are the leading companies in the orphan disease biomarkers market, and how are they differentiated?
- How does Fact.MR estimate and validate the market forecast?
Orphan Disease Biomarkers Market Definition
The orphan disease biomarkers market covers biological markers and associated testing products, technologies, and services used to detect, diagnose, stratify, and monitor rare diseases that affect small patient populations. The scope includes biomarker kits, reagents and assays, discovery and validation services, and bioinformatics platforms applied across orphan drug development, rare disease diagnostics, personalized medicine, and academic research.
Orphan Disease Biomarkers Market Inclusions
The market includes protein, genetic, and metabolic biomarker products used in rare disease applications. It covers biomarker discovery reagents, validated clinical kits, companion diagnostic panels, NGS and PCR-based detection kits, immunoassay reagents, mass spectrometry-based proteomics workflows, and bioinformatics and AI tools used to identify and validate rare disease biomarkers. The scope also includes biomarker discovery services, validation and testing services, and outsourced biomarker analysis provided by contract research organizations.
Orphan Disease Biomarkers Market Exclusions
The market excludes general-purpose diagnostic tests not specifically developed or marketed for rare disease applications, routine clinical chemistry panels, non-orphan oncology diagnostic platforms, and over-the-counter consumer genetic testing services. Standalone laboratory instruments without linked rare disease assay content, generic research reagents used outside biomarker workflows, and pure therapeutic products such as orphan drugs themselves fall outside the scope. Rental or leasing of diagnostic instruments without associated biomarker assay revenue is also excluded.
Orphan Disease Biomarkers Market Research Methodology
- Primary Research: Interviews with biomarker product managers, rare disease clinical researchers, diagnostic lab directors, orphan drug developers, and procurement leads at pharmaceutical and biotechnology companies across all covered regions.
- Desk Research: Regulatory filings, orphan drug designation databases, published clinical trial data, annual reports and investor presentations of listed diagnostic and reagent companies, rare disease registry publications, and peer-reviewed biomarker literature.
- Market Sizing: Demand-side model built from assay volumes, kit pricing, service contract values, and installed base of NGS, mass spectrometry, and immunoassay platforms, cross-validated through top-down reconciliation with reported segment revenues of listed players.
- Data Validation: Triangulation across primary interviews, reported company financials, regulatory approval data, and shipment benchmarks. Sensitivity testing on pricing, kit adoption rates, and pipeline-to-commercial conversion ratios.
Scope of Analysis
| Parameter | Details |
|---|---|
| Quantitative Units | USD 538.35 million in 2026 to USD 1,528.6 million by 2036, at a CAGR of 11.0% |
| Market Definition | Biological markers, associated kits, reagents, platforms, and services used for detection, diagnosis, stratification, and monitoring of rare diseases across pharmaceutical R&D, clinical diagnostics, and research applications. |
| Regions | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries | United States, Germany, United Kingdom, Japan, South Korea, France, and 40+ additional countries |
| Companies | Roche Holdings, Thermo Fisher Scientific, Abbott Laboratories, Qiagen N.V., Bio-Rad Laboratories, Agilent Technologies, PerkinElmer, Illumina Inc., Danaher Corporation, Merck KGaA |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid demand-side and top-down methodology built on assay volumes, kit pricing, service contract values, installed base benchmarking, and primary interviews across manufacturers, pharmaceutical sponsors, specialty diagnostic labs, and regulators. |
Analysis by Product, Application, End Use, Technology, Distribution Channel, Packaging, and Region
-
Orphan Disease Biomarkers Market Market Segmented by Product:
- Biomarker Kits
- Genetic Testing Kits
- PCR-Based Detection Kits
- Next-Generation Sequencing (NGS) Kits
- Genetic Testing Kits
- Reagents & Assays
- Immunoassay Reagents
- ELISA Kits
- Multiplex Assay Kits
- Immunoassay Reagents
- Software & Bioinformatics Tools
- Data Analysis Platforms
- Biomarker Discovery Software
- Data Analysis Platforms
- Services
- Biomarker Discovery Services
- Validation & Testing Services
- Biomarker Discovery Services
- Biomarker Kits
-
Orphan Disease Biomarkers Market Market Segmented by Application:
- Drug Development
- Target Identification
- Clinical Trial Stratification
- Target Identification
- Diagnostic Applications
- Rare Disease Diagnosis
- Early Detection Tools
- Rare Disease Diagnosis
- Personalized Medicine
- Patient Stratification
- Therapy Response Monitoring
- Patient Stratification
- Research Applications
- Genomic & Proteomic Studies
- Biomarker Discovery Research
- Genomic & Proteomic Studies
- Drug Development
-
Orphan Disease Biomarkers Market Market Segmented by End Use:
- Pharmaceutical & Biotechnology Companies
- Orphan Drug Developers
- Biotech Research Firms
- Orphan Drug Developers
- Academic & Research Institutes
- University Research Labs
- Government Research Centers
- University Research Labs
- Diagnostic Laboratories
- Clinical Testing Labs
- Specialty Rare Disease Labs
- Clinical Testing Labs
- Contract Research Organizations (CROs)
- Clinical Trial Services
- Biomarker Validation Providers
- Clinical Trial Services
- Pharmaceutical & Biotechnology Companies
-
Orphan Disease Biomarkers Market Market Segmented by Technology:
- Genomics-Based Technology
- Next-Generation Sequencing (NGS)
- Whole Genome & Exome Sequencing
- Next-Generation Sequencing (NGS)
- Proteomics Technology
- Mass Spectrometry-Based Proteomics
- Protein Biomarker Identification
- Mass Spectrometry-Based Proteomics
- Immunoassay Technology
- ELISA & Multiplex Assays
- High-Sensitivity Detection Systems
- ELISA & Multiplex Assays
- Bioinformatics & AI
- Data Mining & Pattern Recognition
- Predictive Biomarker Modeling
- Data Mining & Pattern Recognition
- Genomics-Based Technology
-
Orphan Disease Biomarkers Market Market Segmented by Distribution Channel:
- Direct Sales
- Pharma & Biotech Contracts
- Research Institution Supply
- Pharma & Biotech Contracts
- Distributors
- Laboratory Equipment Suppliers
- Diagnostic Product Vendors
- Laboratory Equipment Suppliers
- Service Providers
- CRO & Testing Services
- Outsourced Biomarker Analysis
- CRO & Testing Services
- Direct Sales
-
Orphan Disease Biomarkers Market Market Segmented by Packaging:
- Kit-Based Packaging
- Diagnostic Test Kits
- Pre-Configured Assay Kits
- Diagnostic Test Kits
- Reagent Packaging
- Bottles & Vials
- Stability-Controlled Containers
- Bottles & Vials
- Cold Chain Packaging
- Temperature-Controlled Shipping
- Biological Sample Transport Kits
- Temperature-Controlled Shipping
- Kit-Based Packaging
-
Orphan Disease Biomarkers Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
How big is the orphan disease biomarkers market in 2026?
The global orphan disease biomarkers market is valued at USD 538.35 million in 2026.
What will be the size of the orphan disease biomarkers market in 2036?
The market is projected to reach USD 1,528.6 million by 2036.
How fast is the orphan disease biomarkers market expected to grow between 2026 and 2036?
The market is expected to expand at a CAGR of 11.0% between 2026 and 2036.
Which product segment leads the orphan disease biomarkers market in 2026?
Biomarker Kits lead the product segment with 46% share in 2026.
Which country is the fastest-growing market for orphan disease biomarkers?
The United States is the fastest-growing major market with a 12.8% CAGR through 2036.
Which is the second-fastest-growing market?
Germany is the second-fastest-growing market, expanding at a 12.2% CAGR through 2036.
What does the orphan disease biomarkers market include?
The market covers biomarker kits, reagents and assays, bioinformatics software, and services used for rare disease detection, diagnosis, stratification, and monitoring across pharmaceutical R&D, clinical diagnostics, and research applications.
How does Fact.MR estimate and validate the market forecast?
The forecast is built on a hybrid demand-side and top-down methodology using assay volumes, kit pricing, service contract values, installed base benchmarking, and primary interviews with manufacturers, pharmaceutical sponsors, specialty diagnostic labs, and regulators.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Biomarker Kits
- Genetic Testing Kits
- _PCR-Based Detection Kits
- _Next-Generation Sequencing (NGS) Kits
- Reagents & Assays
- Immunoassay Reagents
- _ELISA Kits
- _Multiplex Assay Kits
- Software & Bioinformatics Tools
- Data Analysis Platforms
- _Biomarker Discovery Software
- Services
- Biomarker Discovery Services
- _Validation & Testing Services
- Biomarker Kits
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Drug Development
- Target Identification
- _Clinical Trial Stratification
- Diagnostic Applications
- Rare Disease Diagnosis
- _Early Detection Tools
- Personalized Medicine
- Patient Stratification
- _Therapy Response Monitoring
- Research Applications
- Genomic & Proteomic Studies
- _Biomarker Discovery Research
- Drug Development
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical & Biotechnology Companies
- Orphan Drug Developers
- _Biotech Research Firms
- Academic & Research Institutes
- University Research Labs
- _Government Research Centers
- Diagnostic Laboratories
- Clinical Testing Labs
- _Specialty Rare Disease Labs
- Contract Research Organizations (CROs)
- Clinical Trial Services
- _Biomarker Validation Providers
- Pharmaceutical & Biotechnology Companies
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Genomics-Based Technology
- Next-Generation Sequencing (NGS)
- _Whole Genome & Exome Sequencing
- Proteomics Technology
- Mass Spectrometry-Based Proteomics
- _Protein Biomarker Identification
- Immunoassay Technology
- ELISA & Multiplex Assays
- _High-Sensitivity Detection Systems
- Bioinformatics & AI
- Data Mining & Pattern Recognition
- _Predictive Biomarker Modeling
- Genomics-Based Technology
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct Sales
- Pharma & Biotech Contracts
- _Research Institution Supply
- Distributors
- Laboratory Equipment Suppliers
- _Diagnostic Product Vendors
- Service Providers
- CRO & Testing Services
- _Outsourced Biomarker Analysis
- Direct Sales
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging, 2026 to 2036
- Kit-Based Packaging
- Diagnostic Test Kits
- _Pre-Configured Assay Kits
- Reagent Packaging
- Bottles & Vials
- _Stability-Controlled Containers
- Cold Chain Packaging
- Temperature-Controlled Shipping
- _Biological Sample Transport Kits
- Kit-Based Packaging
- Y to o to Y Growth Trend Analysis By Packaging, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Competition Analysis
- Competition Deep Dive
- Roche Holdings
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Thermo Fisher Scientific
- Abbott Laboratories
- Qiagen N.V.
- Bio-Rad Laboratories
- Agilent Technologies
- PerkinElmer
- Illumina Inc.
- Danaher Corporation
- Merck KGaA
- Roche Holdings
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Packaging, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Distribution Channel
- Figure 18: Global Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Packaging
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 28: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 46: North America Market Attractiveness Analysis by Distribution Channel
- Figure 47: North America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 49: North America Market Attractiveness Analysis by Packaging
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Application
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 62: Latin America Market Attractiveness Analysis by Technology
- Figure 63: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 65: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 66: Latin America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 68: Latin America Market Attractiveness Analysis by Packaging
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Application
- Figure 76: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 78: Western Europe Market Attractiveness Analysis by End Use
- Figure 79: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 81: Western Europe Market Attractiveness Analysis by Technology
- Figure 82: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 84: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 85: Western Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 87: Western Europe Market Attractiveness Analysis by Packaging
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Application
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Packaging
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 113: East Asia Market Attractiveness Analysis by Application
- Figure 114: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 116: East Asia Market Attractiveness Analysis by End Use
- Figure 117: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 119: East Asia Market Attractiveness Analysis by Technology
- Figure 120: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 122: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 123: East Asia Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 125: East Asia Market Attractiveness Analysis by Packaging
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Packaging
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Packaging
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis