Autoimmune Disease Diagnostics Market Size, Share, Growth and Forecast (2025 - 2035)
Autoimmune Disease Diagnostics Market Size and Share Forecast Outlook 2025 to 2035
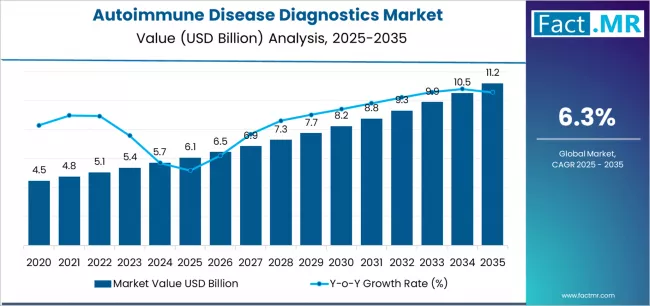
Autoimmune disease diagnostics market is projected to grow from USD 6.1 billion in 2025 to USD 11.2 billion by 2035, at a CAGR of 6.3%. Localized Autoimmune Disease Diagnostics will dominate with a 64.3% market share, while consumables and assays will lead the product segment with a 70.4% share.
Autoimmune Disease Diagnostics Market Forecast and Outlook 2025 to 2035
The global autoimmune disease diagnostics market is projected to reach USD 11.18 billion by 2035, recording an absolute increase of USD 5.11 billion over the forecast period. The market is valued at USD 6.07 billion in 2025 and is set to rise at a CAGR of 6.3% during the assessment period.
The market is expected to grow by 1.8 times during the same period, supported by increasing demand from clinical laboratory operations and expanding applications in early disease detection across both developed and emerging healthcare markets. Additionally, growing application diversity in autoantibody testing, biomarker identification, and personalized treatment monitoring creates expanding opportunities for diagnostic innovation and specialized assay development.
Quick Stats for Autoimmune Disease Diagnostics Market
- Autoimmune Disease Diagnostics Market Value (2025): USD 6.07 billion
- Autoimmune Disease Diagnostics Market Forecast Value (2035): USD 11.18 billion
- Autoimmune Disease Diagnostics Market Forecast CAGR: 6.3%
- Leading Type in Autoimmune Disease Diagnostics Market: Localized Autoimmune Disease Diagnostics (64.25%)
- Key Growth Regions in Autoimmune Disease Diagnostics Market: Asia Pacific, North America, and Europe
- Top Players in Autoimmune Disease Diagnostics Market: F. Hoffmann-La Roche Ltd, Siemens Healthcare Private Limited, Abbott, Beckman Coulter, Inc., Danaher Corporation, Quest Diagnostics, EUROIMMUN Medizinische Labordiagnostika AG, Nova Diagnostics Pte Ltd, BIOMÉRIEUX, Hemagen Diagnostics, Inc.
Rising disease prevalence in developed nations, coupled with expanding diagnostic infrastructure through advanced laboratory facilities and modern testing technologies, further accelerate market penetration across diverse healthcare segments. The growing global demand for accurate disease diagnosis, particularly in rheumatology and endocrinology applications, generates sustained requirements for high-sensitivity diagnostic assays and precision testing materials.
Technical advancements in immunoassay technologies demonstrating measurable improvements in detection accuracy, reduced turnaround times, and multiplexing capability optimization reinforce laboratory confidence in autoimmune diagnostics economics, while personalized medicine sector trends toward targeted therapy deployment expand addressable market opportunities beyond traditional screening applications into treatment monitoring supply chains.
Healthcare providers and specialty clinics increasingly incorporate comprehensive autoimmune testing into diagnostic protocols, patient management programs, and therapeutic decision-making processes, creating mainstream consumption channels that extend beyond specialty laboratory applications into conventional healthcare environments.
Fluctuating reimbursement policies for diagnostic testing and complex regulatory requirements for assay validation may pose challenges to market expansion. Test standardization constraints for autoantibody detection and result interpretation variability in certain regions also influence adoption capacity, requiring manufacturers to develop diversified product strategies catering to specific clinical requirements across different geographical markets.
Supply chain complexity during reagent procurement and the technical requirements for quality control protocols and laboratory accreditation may limit accessibility among smaller diagnostic facilities in developing regions with limited infrastructure for advanced autoimmune disease testing and reference laboratory capabilities.
Autoimmune Disease Diagnostics Market Year-over-Year Forecast 2025 to 2035
Between 2025 and 2030, the autoimmune disease diagnostics market is projected to expand from USD 6.07 billion to USD 8.23 billion, resulting in a value increase of USD 2.16 billion, which represents 42.3% of the total forecast growth for the decade. This phase of development will be shaped by rising demand for early disease detection systems and precision diagnostic assays, product innovation in multiplex immunoassay platforms and point-of-care testing configurations, as well as expanding integration with electronic health record initiatives and laboratory automation platforms. Companies are establishing competitive positions through investment in biomarker discovery technology development, high-throughput testing capabilities, and strategic market expansion across reference laboratory facilities, hospital-based testing segments, and specialty clinic applications.
From 2030 to 2035, the market is forecast to grow from USD 8.23 billion to USD 11.18 billion, adding another USD 2.95 billion, which constitutes 57.7% of the overall ten-year expansion. This period is expected to be characterized by the expansion of specialized diagnostic derivatives, including targeted autoantibody panel formulations and customized testing algorithms tailored for specific disease populations, strategic collaborations between diagnostic manufacturers and pharmaceutical companies, and an enhanced focus on companion diagnostic development and treatment monitoring certifications. The growing emphasis on precision medicine principles and therapeutic decision support technologies will drive demand for high-performance autoimmune diagnostic solutions across diverse clinical applications.
Autoimmune Disease Diagnostics Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 6.07 billion |
| Market Forecast Value (2035) | USD 11.18 billion |
| Forecast CAGR (2025-2035) | 6.3% |
Why is the Autoimmune Disease Diagnostics Market Experiencing Steady Growth?
The autoimmune disease diagnostics market grows by enabling healthcare providers, clinical laboratories, and specialty physicians to access reliable testing solutions that support accurate diagnosis while meeting clinical demand for consistent performance specifications.
Rheumatologists and laboratory directors face mounting pressure to develop comprehensive diagnostic protocols with proven sensitivity and specificity characteristics, with autoimmune diagnostic assays typically providing 85-95% accuracy levels and multiplex testing capabilities essential for differential diagnosis, disease monitoring, and therapeutic response assessment, making these tests essential for competitive positioning in rheumatology, endocrinology, and immunology categories.
The healthcare industry's need for dependable diagnostic infrastructure and consistent result quality creates demand for diverse testing platforms that can provide superior disease detection, maintain predictable performance across different patient populations, and ensure quality compliance without compromising clinical utility or laboratory economics.
Government initiatives promoting early disease intervention and preventive healthcare drive adoption in hospital laboratories, reference testing facilities, and specialty diagnostic centers, where test selection has a direct impact on patient outcomes and treatment pathway decisions.
The healthcare industry's growing focus on personalized medicine and targeted therapy development further expands market opportunities, with clinical research demonstrating measurable benefits from early autoimmune disease detection, including improved treatment outcomes and reduced disease progression.
Supply chain complexity during reagent sourcing and the technical requirements for assay validation and laboratory quality management processes may limit accessibility among smaller diagnostic facilities and developing regions with limited infrastructure for advanced autoimmune testing operations and specialist interpretation systems.
Segmental Analysis
The market is segmented by type, product, and region. By type, the market is divided into localized autoimmune disease diagnostics, systemic autoimmune disease diagnostics, rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus (SLE), multiple sclerosis, type 1 diabetes, Hashimoto's thyroiditis, idiopathic thrombocytopenic purpura, and others.
By product, the market is divided into consumables and assays, and instruments. Regionally, the market is divided into Asia Pacific, North America, Europe, Latin America, and Middle East & Africa.
Which Autoimmune Disease Diagnostics Type is the Dominant Category?
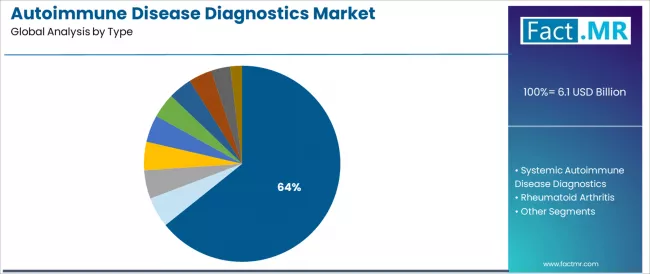
The localized autoimmune disease diagnostics segment represents the dominant force in the autoimmune disease diagnostics market, capturing 64.25% of the total market share in 2025. This established diagnostic category encompasses solutions featuring critical organ-specific testing functionality for thyroid disorders, type 1 diabetes, and gastrointestinal conditions, including thyroid peroxidase antibody tests, islet cell antibody assays, and tissue transglutaminase antibody detection that enable targeted disease identification, monitoring capabilities, and enhanced diagnostic precision across all endocrine and gastroenterology applications.
The segment's market leadership stems from its essential role in common autoimmune condition detection, with organ-specific tests capable of meeting diverse clinical requirements while maintaining cost-effective testing protocols and operational reliability across all laboratory environments. Within the localized autoimmune disease diagnostics segment, thyroid disorder testing accounts for the largest test volume share, driven by high disease prevalence and continuous screening initiatives across global healthcare systems.
The systemic autoimmune disease diagnostics segment maintains a substantial 35.75% market share, serving rheumatologists and immunologists who require comprehensive testing panels for multi-organ autoimmune conditions, systemic lupus erythematosus, and rheumatoid arthritis applications.
These diagnostic solutions offer broad autoantibody screening capabilities for complex disease presentations and differential diagnosis challenges while providing sufficient clinical sensitivity to meet diagnostic demands and disease classification requirements.
The rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus (SLE), multiple sclerosis, type 1 diabetes, Hashimoto's thyroiditis, idiopathic thrombocytopenic purpura, and others segments account for the remaining market share, serving specific disease categories and specialized testing applications requiring targeted biomarker detection and disease-specific diagnostic algorithms.
Key advantages driving the localized autoimmune disease diagnostics segment include:
- Established testing infrastructure with integrated laboratory platforms that reduce diagnostic complexity and ensure consistent result availability
- Critical organ-specific functionality allowing targeted disease detection across different autoimmune presentations without significant cost penalties
- Proven clinical utility, delivering reliable diagnostic information while maintaining competitiveness against broad screening approaches
- Broad healthcare acceptance enabling straightforward test ordering protocols and interpretation standards across multiple medical specialties
Which Product Category Leads the Autoimmune Disease Diagnostics Market?
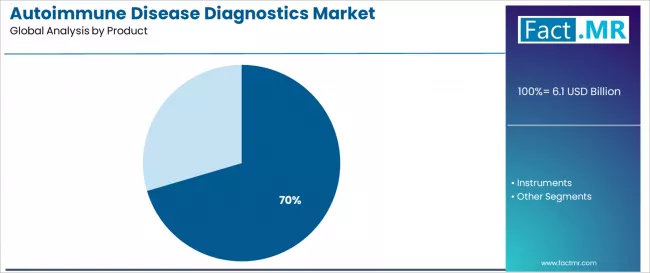
The consumables and assays segment captures 70.43% of the autoimmune disease diagnostics market in 2025, representing the most widely adopted product configuration across clinical laboratory operations. This product category offers recurring revenue characteristics, essential reagent functionality, and comprehensive test menu coverage that align with laboratory operational requirements.
The consumables and assays segment's dominance reflects its widespread adoption in immunoassay testing, ELISA procedures, and automated platform applications where reagent kits provide standardized testing protocols and quality-controlled diagnostic performance.
The instruments segment maintains a 29.57% market share, serving laboratory directors and healthcare systems requiring automated testing platforms, high-throughput analyzers, and integrated diagnostic systems across centralized testing operations.
These product types provide comprehensive testing automation across immunoassay workflows, enabling enhanced laboratory efficiency and standardized result generation in high-volume testing environments. The instruments category supports capital equipment investments for laboratory modernization and capacity expansion initiatives.
What are the Drivers, Restraints, and Key Trends of the Autoimmune Disease Diagnostics Market?
The market is driven by three concrete demand factors tied to disease prevalence and diagnostic advancement. First, increasing autoimmune disease incidence and improved disease awareness creates growing demand for diagnostic testing, with global autoimmune disorder cases expanding by 4-6% annually in major healthcare markets worldwide, requiring comprehensive diagnostic testing infrastructure. Second, expanding precision medicine initiatives for targeted therapy selection and treatment monitoring drives increased adoption of autoimmune biomarker testing, with many healthcare systems implementing comprehensive diagnostic protocols for autoimmune evaluation by 2030. Third, technological advancements in immunoassay platforms and multiplex testing technologies enable more effective and efficient disease detection that reduces diagnostic delays while improving sensitivity and specificity capabilities.
Market restraints include fluctuating reimbursement rates for autoimmune testing that can deter laboratories from maintaining comprehensive test menus, particularly in regions where payer policies and coverage limitations face budget pressures or utilization restrictions. Test result interpretation complexity and clinical guideline variability pose another significant challenge, as autoimmune diagnostics require specialist expertise and integrated clinical correlation, potentially causing diagnostic uncertainty and ordering hesitation. Assay standardization limitations and inter-laboratory variability concerns create additional market challenges for result comparability, demanding ongoing investment in reference material development and quality assurance improvements.
Key trends indicate accelerated diagnostic adoption in Asia Pacific markets, particularly China and India, where healthcare infrastructure development and disease awareness improvement drive comprehensive autoimmune testing implementation. Technology integration trends toward automated immunoassay analyzers with random access capabilities, multiplex bead-based platforms with simultaneous biomarker detection, and artificial intelligence-enabled result interpretation enable optimized laboratory workflows that reduce turnaround times and enhance diagnostic accuracy. However, the market thesis could face disruption if significant advances in point-of-care testing technologies or major shifts in diagnostic algorithm strategies reduce reliance on traditional laboratory-based autoimmune testing configurations.
Analysis of the Autoimmune Disease Diagnostics Market by Key Countries
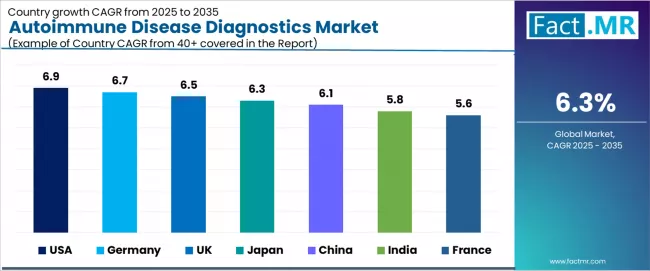
| Country | CAGR (2025-2035) |
|---|---|
| USA | 6.9% |
| Germany | 6.7% |
| UK | 6.5% |
| Japan | 6.3% |
| China | 6.1% |
| India | 5.8% |
| France | 5.6% |
The autoimmune disease diagnostics market is expanding steadily, with USA leading at a 6.9% CAGR through 2035, driven by substantial research funding, advanced diagnostic infrastructure, and comprehensive early detection adoption programs. Germany follows at 6.7%, supported by government-backed innovation initiatives, expanding diagnostic capabilities, and accelerated healthcare technology investments.
The UK records 6.5%, reflecting high autoimmune patient populations and increasing NHS diagnostic programs. Japan posts 6.3%, anchored by aging demographics and growing diagnostic demand operations. China grows at 6.1%, with rising disease prevalence and healthcare infrastructure expansion. India advances at 5.8%, emphasizing increasing diagnostic accessibility and disease awareness, while France grows steadily at 5.6%, focusing on personalized medicine adoption through established healthcare systems and diagnostic innovation.
USA Leads Global Market Expansion
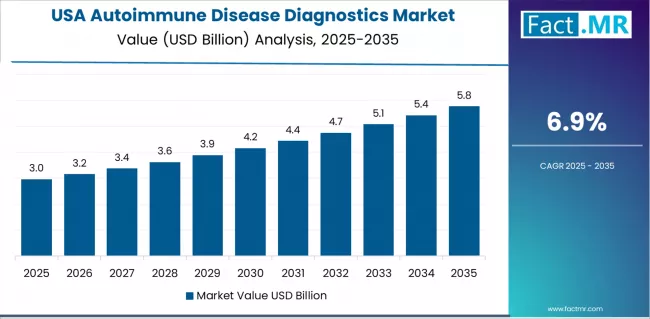
USA demonstrates the strongest growth potential in the autoimmune disease diagnostics market with a CAGR of 6.9% through 2035. The country's leadership position stems from substantial healthcare research investment, advanced laboratory infrastructure, and comprehensive diagnostic adoption enabling mainstream autoimmune testing utilization.
Growth is concentrated in major healthcare regions, including Northeast, Midwest, California, and Texas, where reference laboratories and expanding specialty clinic infrastructure are implementing advanced autoimmune testing protocols for patient diagnosis and treatment monitoring applications.
Distribution channels through integrated laboratory networks, hospital-based testing facilities, and specialty diagnostic centers expand deployment across healthcare providers and rheumatology practices. The country's growing autoimmune disease burden provides clinical momentum for diagnostic development, including research funding for biomarker discovery and assay validation.
Key market factors:
- Healthcare demand concentrated in rheumatology centers and reference laboratories with comprehensive testing networks
- Infrastructure development through laboratory automation expansion and specialty clinic improvements
- Comprehensive diagnostic ecosystem, including established quality standards with proven regulatory frameworks
- Technology integration featuring automated immunoassay platforms, multiplex testing systems, and laboratory information management solutions
Germany Emerges as Innovation Leader
In major healthcare regions including North Rhine-Westphalia, Bavaria, Baden-Württemberg, and Hesse, the adoption and utilization of autoimmune diagnostics is accelerating across hospital laboratories and specialized testing operations, driven by government innovation support and comprehensive diagnostic quality initiatives. The market demonstrates strong growth momentum with a CAGR of 6.7% through 2035, linked to federal healthcare innovation policies, expanding diagnostic infrastructure, and sustained medical technology investment.
German laboratory operators are implementing advanced autoimmune testing technologies and multiplex platforms to enhance diagnostic accuracy while meeting growing demand in expanding rheumatology sectors and immunology specialty areas. The country's healthcare quality emphasis creates persistent demand for diagnostic precision, while increasing focus on early intervention drives adoption of comprehensive autoimmune testing programs.
Key development areas:
- University hospitals and reference laboratories leading autoimmune diagnostic adoption with comprehensive research programs
- Integrated healthcare networks providing efficient diagnostic pathways with extensive specialist collaboration
- Technology partnerships between diagnostic manufacturers and academic medical centers are expanding innovation reach
- Integration of digital pathology systems and comprehensive quality management protocols
UK Shows Strong Clinical Adoption
The UK’s market expansion is driven by NHS diagnostic initiatives, including autoimmune testing programs in England and Scotland, and expanding specialty clinic implementations across multiple healthcare regions. The country demonstrates solid growth potential with a CAGR of 6.5% through 2035, supported by established healthcare infrastructure, high autoimmune disease prevalence, and growing integration with clinical pathway optimization across patient populations.
Healthcare providers face implementation priorities related to diagnostic access improvement and testing standardization, requiring investment in laboratory capacity enhancement and support from specialty diagnostic services. Growing disease recognition and established healthcare networks create compelling business cases for autoimmune diagnostic adoption, particularly in urban regions where specialty clinic operations have a direct impact on patient management quality.
Key market characteristics:
- NHS programs and specialty rheumatology services showing fastest growth with substantial increase in autoimmune testing requirements
- Regional expansion trends focused on diagnostic hubs in major metropolitan healthcare systems
- Future projections indicate the need for enhanced laboratory capacity and advanced testing technology development programs
- Growing emphasis on early diagnosis protocols and clinical pathway optimization in UK healthcare markets
Japan Demonstrates Aging Population Focus
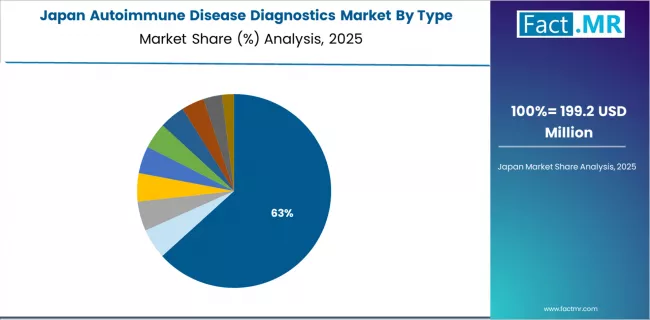
The Japanese market leads in comprehensive diagnostic applications based on integration with aging population healthcare and chronic disease management for enhanced patient outcomes. The country shows steady potential with a CAGR of 6.3% through 2035, driven by demographic aging trends, strong healthcare infrastructure, and the expansion of specialized diagnostic facilities in major healthcare centers, including Tokyo, Osaka, Nagoya, and Fukuoka.
Japanese healthcare providers are adopting advanced autoimmune diagnostic configurations for disease detection optimization and treatment monitoring applications, particularly in regions with sophisticated healthcare requirements and specialized testing demands requiring comprehensive quality standards. Technology deployment channels through established hospital laboratories and commercial diagnostic centers expand coverage across internal medicine practitioners and specialty physicians.
Leading market segments:
- University hospitals and commercial laboratories in major healthcare centers implementing comprehensive diagnostic strategies
- Strategic partnerships with diagnostic technology providers, achieving stringent quality specifications and performance standards
- Collaborative research between academic institutions and diagnostic companies are expanding innovation capabilities
- Focus on precision medicine initiatives and integrated care pathway methodologies
China Shows Infrastructure Development
In major healthcare regions including Beijing, Shanghai, Guangdong, and Zhejiang, hospital laboratories and diagnostic centers are implementing comprehensive autoimmune testing strategies to optimize disease detection and improve clinical outcomes, with documented case studies showing significant diagnostic capability improvements through advanced platform adoption and specialist training enhancement techniques.
The market shows moderate growth potential with a CAGR of 6.1% through 2035, linked to expanding healthcare infrastructure, growing disease awareness, and ongoing diagnostic capacity development requirements in major urban regions. Chinese healthcare providers are adopting proven autoimmune diagnostic technologies and quality protocols to enhance clinical laboratory services while maintaining performance standards demanded by specialty physician requirements and patient care expectations. The country's healthcare modernization initiatives create persistent demand for advanced diagnostic supply and technology solutions that integrate with expanding hospital networks.
Market development factors:
- Tertiary hospitals and specialty diagnostic centers leading autoimmune testing adoption across China
- Healthcare infrastructure investment and capacity expansion providing support for diagnostic development
- Strategic relationships between international diagnostic companies and Chinese healthcare institutions are expanding technology transfer
- Emphasis on laboratory quality improvement and clinical service enhancement across healthcare segments
India Shows Accessibility Expansion
India's autoimmune disease diagnostics market demonstrates growing positioning focused on diagnostic accessibility improvement and disease awareness integration, with documented adoption of immunoassay platforms, achieving enhanced testing availability through laboratory network expansion in Delhi, Mumbai, Bangalore, and Chennai healthcare regions.
The country maintains moderate growth momentum with a CAGR of 5.8% through 2035, driven by expanding healthcare access, growing middle class, and strategic emphasis on diagnostic service development. Major diagnostic chains, including operations in metropolitan healthcare centers and emerging city networks, showcase expanding autoimmune testing availability where increased awareness supports diagnostic utilization and comprehensive disease management programs.
Key market characteristics:
- Corporate hospitals and diagnostic chain laboratories driving autoimmune testing demand with emphasis on accessibility enhancement and quality standardization
- Healthcare awareness partnerships enabling diagnostic utilization growth with comprehensive education programs
- Technology collaboration between international manufacturers and Indian diagnostic providers is expanding market capabilities
- Emphasis on affordable diagnostics and healthcare accessibility optimization methodologies
France Demonstrates Personalized Medicine Focus
France's autoimmune disease diagnostics market demonstrates established and quality-focused landscape, characterized by integration of diagnostic testing with personalized medicine approaches across university hospitals, specialized clinics, and integrated healthcare applications. The country shows steady growth momentum with a CAGR of 5.6% through 2035, driven by established healthcare infrastructure and personalized medicine investments in diagnostic facilities concentrated in healthcare regions.
France's emphasis on therapeutic precision and diagnostic quality creates requirements for advanced autoimmune testing configurations that support comprehensive treatment selection initiatives and stringent clinical requirements in healthcare operations. The market benefits from strong partnerships between diagnostic manufacturers and healthcare institutions, creating comprehensive testing ecosystems that prioritize result accuracy and clinical utility. Hospital laboratories in major healthcare regions showcase established diagnostic implementations where testing quality achieves demanding clinical standards through integrated quality programs.
Key market characteristics:
- University hospitals and specialty clinics driving autoimmune diagnostic requirements with emphasis on treatment personalization and clinical precision
- Clinical utility partnerships enabling proven testing protocols with comprehensive validation programs
- Technology collaboration between French healthcare institutions and diagnostic innovators is expanding clinical capabilities
- Emphasis on therapeutic decision support and precision medicine optimization methodologies
Europe Market Split by Country
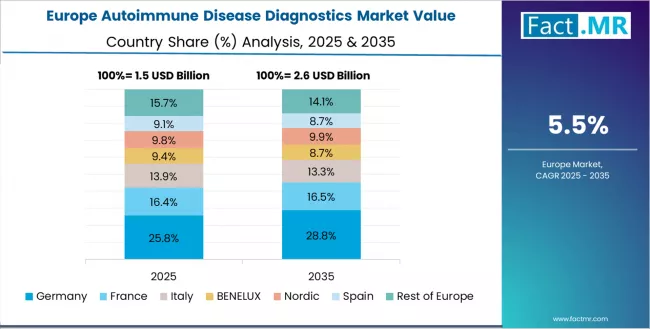
The autoimmune disease diagnostics market in Europe is projected to grow from USD 1.82 billion in 2025 to USD 3.27 billion by 2035, registering a CAGR of 6.0% over the forecast period. Germany is expected to maintain its leadership position with a 28.6% market share in 2025, supported by its advanced diagnostic infrastructure, comprehensive healthcare quality standards, and strong medical technology industry across major healthcare centers.
The UK follows with a 24.3% share in 2025, driven by established NHS diagnostic programs and integrated specialty clinic networks. France holds a 19.7% share in 2025 through its personalized medicine emphasis and university hospital diagnostic capabilities. Italy commands a 14.8% share, while the rest of Europe accounts for 12.6% in 2025.
By 2035, Germany is projected to hold 28.9% share, UK 24.1%, France 19.5%, Italy 14.6%, and Rest of Europe 12.9%, reflecting sustained growth momentum across all major European markets and increasing autoimmune diagnostic adoption in diversified healthcare applications implementing comprehensive early detection programs.
Competitive Landscape of the Autoimmune Disease Diagnostics Market
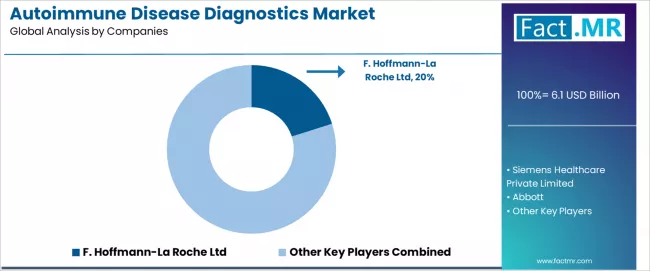
The autoimmune disease diagnostics market features approximately 30-40 meaningful players with moderate concentration, where the top three companies control roughly 45-55% of global market presence through established product portfolios and extensive customer relationships across laboratory and healthcare sectors.
Market leaders include F. Hoffmann-La Roche Ltd, Abbott, and Siemens Healthcare Private Limited, which maintain competitive advantages through comprehensive diagnostic test portfolios, global distribution networks, and deep expertise in the immunoassay technology and laboratory diagnostics sectors, creating strong customer relationships among hospital laboratories and reference testing facilities.
These companies leverage established manufacturing capabilities and ongoing product innovation initiatives to defend market positions while expanding into adjacent molecular diagnostics applications and companion diagnostic markets. F. Hoffmann-La Roche Ltd commands a 20.1% market share through integrated diagnostic solutions and strategic positioning in global healthcare markets.
Challengers encompass Beckman Coulter, Inc., Danaher Corporation, and Quest Diagnostics, which compete through diversified product portfolios and strong regional presence in key diagnostic markets. Specialty providers, including EUROIMMUN Medizinische Labordiagnostika AG, Nova Diagnostics Pte Ltd, Biomérieux, and Hemagen Diagnostics, Inc., focus on specific disease categories or testing platforms, offering differentiated capabilities in autoantibody detection, specialized assays, and technical laboratory services.
Regional players and emerging diagnostic companies create competitive pressure through innovative testing methodologies and strategic market positioning, particularly in high-growth markets including China and India, where local presence provides advantages in regulatory navigation and healthcare system integration.
Market dynamics favor companies that combine analytical performance excellence with comprehensive clinical validation that addresses the complete value chain from assay development through regulatory approval and market commercialization. Strategic investments in biomarker discovery and companion diagnostic development enable product differentiation and market leadership across healthcare applications.
Global Autoimmune Disease Diagnostics Market - Stakeholder Contribution Framework
Autoimmune disease diagnostic products represent a critical healthcare technology that enables physicians, clinical laboratories, and healthcare systems to achieve accurate disease detection and patient management optimization without substantial workflow modifications, typically providing essential diagnostic information and treatment guidance capabilities while supporting diverse clinical applications.
With the market projected to grow from USD 6.07 billion in 2025 to USD 11.18 billion by 2035 at a 6.3% CAGR, these diagnostics offer compelling advantages - high clinical sensitivity, specific disease identification, and therapeutic decision support - making them essential for localized disease testing (largest segment), comprehensive laboratory operations (dominant channel), and diverse healthcare applications seeking reliable diagnostic technologies. Scaling market adoption and clinical utilization requires coordinated action across healthcare policy, diagnostic standards, test manufacturers, laboratory operators, and medical education institutions.
How Governments Could Spur Local Production and Adoption?
- Healthcare Diagnostic Programs: Include autoimmune testing capabilities in national healthcare strategies, providing targeted funding for laboratory infrastructure in regions with established healthcare systems and supporting local diagnostic manufacturers through regulatory streamlining and development incentives.
- Reimbursement Policy & Access Support: Implement favorable coverage frameworks for autoimmune diagnostic testing, provide adequate reimbursement for comprehensive test panels, and establish clinical guidelines that support appropriate test utilization.
- Trade Policy & Market Access: Create balanced import-export frameworks across diagnostic and healthcare applications, establish fair regulatory practices for diagnostic products, and develop international cooperation protocols that facilitate test validation harmonization and quality standard alignment.
- Skills Development & Training: Fund medical laboratory scientist education programs, specialist physician training, and diagnostic interpretation courses. Invest in knowledge transfer initiatives that bridge diagnostic manufacturers with clinical laboratories and healthcare providers.
How Industry Bodies Could Support Market Development?
- Quality Standards & Certification: Define standardized performance specifications for autoimmune diagnostics across hospital laboratories, reference testing facilities, and specialty clinic applications, establish universal validation protocols and analytical metrics, and create certification programs for assay quality that laboratories can implement.
- Market Education & Best Practices: Lead communication initiatives that demonstrate autoimmune diagnostic value, emphasizing proven clinical utility, disease management benefits, and patient outcome improvements compared to empirical treatment approaches and delayed diagnosis.
- Clinical Utility Standards: Develop clinical practice guideline recommendations for autoimmune testing, comprehensive test utilization algorithms, and result interpretation best practices, ensuring appropriate diagnostic use across different clinical settings and patient populations.
How Manufacturers and Technology Players Could Strengthen the Ecosystem?
- Diagnostic Technology Development: Develop next-generation immunoassay platforms with enhanced sensitivity capabilities, automated testing systems with improved throughput performance, and integration-specific features that enhance laboratory efficiency while improving diagnostic accuracy and clinical utility.
- Clinical Support Platforms: Provide comprehensive medical education that integrates test interpretation guidance, clinical algorithm support, disease management information, and evidence generation, enabling healthcare providers to maximize diagnostic value and patient care quality.
- Supply Chain Integration: Offer flexible supply arrangements for hospital laboratories and diagnostic centers, including reagent rental agreements, instrument placement programs, and technical support services that keep diagnostic capabilities aligned with clinical demand and quality requirements.
Key Players in the Autoimmune Disease Diagnostics Market
- F. Hoffmann-La Roche Ltd
- Siemens Healthcare Private Limited
- Abbott
- Beckman Coulter, Inc.
- Danaher Corporation
- Quest Diagnostics
- EUROIMMUN Medizinische Labordiagnostika AG
- Nova Diagnostics Pte Ltd
- Biomérieux
- Hemagen Diagnostics, Inc.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD 6.07 Billion |
| Type | Localized Autoimmune Disease Diagnostics, Systemic Autoimmune Disease Diagnostics, Rheumatoid Arthritis, Ankylosing Spondylitis, Systemic Lupus Erythematosus (SLE), Multiple Sclerosis, Type 1 Diabetes, Hashimoto's Thyroiditis, Idiopathic Thrombocytopenic Purpura, Others |
| Product | Consumables and Assays, Instruments |
| Regions Covered | Asia Pacific, North America, Europe, Latin America, Middle East & Africa |
| Country Covered | USA, Germany, UK, Japan, China, India, France, and 40+ countries |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Siemens Healthcare Private Limited, Abbott, Beckman Coulter, Inc., Danaher Corporation, Quest Diagnostics, EUROIMMUN Medizinische Labordiagnostika AG, Nova Diagnostics Pte Ltd, BIOMÉRIEUX, Hemagen Diagnostics, Inc. |
| Additional Attributes | Dollar sales by disease type categories, regional adoption trends across Asia Pacific, North America, and Europe, competitive landscape with diagnostic manufacturers and laboratory service providers, test specifications and clinical performance requirements, integration with hospital laboratories and reference testing operations. |
Autoimmune Disease Diagnostics Market by Segments
-
Type :
- Localized Autoimmune Disease Diagnostics
- Systemic Autoimmune Disease Diagnostics
- Rheumatoid Arthritis
- Ankylosing Spondylitis
- Systemic Lupus Erythematosus (SLE)
- Multiple Sclerosis
- Type 1 Diabetes
- Hashimoto's Thyroiditis
- Idiopathic Thrombocytopenic Purpura
- Others
-
Product :
- Consumables and Assays
- Instruments
-
Region :
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- North America
- USA
- Canada
- Mexico
- Europe
- Russia
- Germany
- Norway
- France
- Rest of Europe
- Latin America
- Brazil
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Rest of Middle East & Africa
- Asia Pacific
- Frequently Asked Questions -
How big is the autoimmune disease diagnostics market in 2025?
The global autoimmune disease diagnostics market is estimated to be valued at USD 6.1 billion in 2025.
What will be the size of autoimmune disease diagnostics market in 2035?
The market size for the autoimmune disease diagnostics market is projected to reach USD 11.2 billion by 2035.
How much will be the autoimmune disease diagnostics market growth between 2025 and 2035?
The autoimmune disease diagnostics market is expected to grow at a 6.3% CAGR between 2025 and 2035.
What are the key product types in the autoimmune disease diagnostics market?
The key product types in autoimmune disease diagnostics market are localized autoimmune disease diagnostics, systemic autoimmune disease diagnostics, rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus (sle), multiple sclerosis, type 1 diabetes, hashimoto's thyroiditis, idiopathic thrombocytopenic purpura and others.
Which product segment to contribute significant share in the autoimmune disease diagnostics market in 2025?
In terms of product, consumables and assays segment to command 70.4% share in the autoimmune disease diagnostics market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type, 2025 to 2035
- Localized Autoimmune Disease Diagnostics
- Systemic Autoimmune Disease Diagnostics
- Rheumatoid Arthritis
- Ankylosing Spondylitis
- Systemic Lupus Erythematosus (SLE)
- Multiple Sclerosis
- Type 1 Diabetes
- Hashimoto's Thyroiditis
- Idiopathic Thrombocytopenic Purpura
- Others
- Y to o to Y Growth Trend Analysis By Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Type, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2025 to 2035
- Consumables and Assays
- Instruments
- Y to o to Y Growth Trend Analysis By Product, 2020 to 2024
- Absolute $ Opportunity Analysis By Product, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Product
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Product
- Competition Analysis
- Competition Deep Dive
- F. Hoffmann-La Roche Ltd
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Siemens Healthcare Private Limited
- Abbott
- Beckman Coulter, Inc.
- Danaher Corporation
- Quest Diagnostics
- EUROIMMUN Medizinische Labordiagnostika AG
- Nova Diagnostics Pte Ltd
- Biomérieux
- Hemagen Diagnostics, Inc.
- F. Hoffmann-La Roche Ltd
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Global Market Value (USD Million) Forecast by Type, 2020 to 2035
- Table 3: Global Market Value (USD Million) Forecast by Product, 2020 to 2035
- Table 4: North America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: North America Market Value (USD Million) Forecast by Type, 2020 to 2035
- Table 6: North America Market Value (USD Million) Forecast by Product, 2020 to 2035
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 8: Latin America Market Value (USD Million) Forecast by Type, 2020 to 2035
- Table 9: Latin America Market Value (USD Million) Forecast by Product, 2020 to 2035
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 11: Western Europe Market Value (USD Million) Forecast by Type, 2020 to 2035
- Table 12: Western Europe Market Value (USD Million) Forecast by Product, 2020 to 2035
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type, 2020 to 2035
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Product, 2020 to 2035
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 17: East Asia Market Value (USD Million) Forecast by Type, 2020 to 2035
- Table 18: East Asia Market Value (USD Million) Forecast by Product, 2020 to 2035
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type, 2020 to 2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2020 to 2035
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type, 2020 to 2035
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Product, 2020 to 2035
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020 to 2035
- Figure 3: Global Market Value Share and BPS Analysis by Type, 2025 and 2035
- Figure 4: Global Market Y to o to Y Growth Comparison by Type, 2025 to 2035
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 7: Global Market Y to o to Y Growth Comparison by Product, 2025 to 2035
- Figure 8: Global Market Attractiveness Analysis by Product
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2025 to 2035
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: North America Market Value Share and BPS Analysis by Type, 2025 and 2035
- Figure 21: North America Market Y to o to Y Growth Comparison by Type, 2025 to 2035
- Figure 22: North America Market Attractiveness Analysis by Type
- Figure 23: North America Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 24: North America Market Y to o to Y Growth Comparison by Product, 2025 to 2035
- Figure 25: North America Market Attractiveness Analysis by Product
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 27: Latin America Market Value Share and BPS Analysis by Type, 2025 and 2035
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Type, 2025 to 2035
- Figure 29: Latin America Market Attractiveness Analysis by Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Product, 2025 to 2035
- Figure 32: Latin America Market Attractiveness Analysis by Product
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type, 2025 and 2035
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Type, 2025 to 2035
- Figure 36: Western Europe Market Attractiveness Analysis by Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Product, 2025 to 2035
- Figure 39: Western Europe Market Attractiveness Analysis by Product
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type, 2025 and 2035
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Type, 2025 to 2035
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Product, 2025 to 2035
- Figure 46: Eastern Europe Market Attractiveness Analysis by Product
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 48: East Asia Market Value Share and BPS Analysis by Type, 2025 and 2035
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Type, 2025 to 2035
- Figure 50: East Asia Market Attractiveness Analysis by Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Product, 2025 to 2035
- Figure 53: East Asia Market Attractiveness Analysis by Product
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type, 2025 and 2035
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Type, 2025 to 2035
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Product, 2025 to 2035
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type, 2025 and 2035
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Type, 2025 to 2035
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Product, 2025 to 2035
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis