Structured Product Label Management Market Size, Share, Growth and Forecast (2026 - 2036)
Structured Product Label Management Market is segmented by Solutions (Software and Services), Deployment (Cloud-based and On-Premise), End User (Pharmaceutical Companies, Biotechnology Companies, Medical Device Companies, Regulatory Authorities, and Contract Research Organizations (CROs)), and Region. Forecast for 2026 to 2036.
Fact MR opines that the structured product label management market was valued at USD 65.0 billion in 2025. Demand is forecast to grow to USD 73.1 billion in 2026 and reach USD 237.0 billion by 2036, registering a 12.5% CAGR during the forecast period. Cloud based deployment is expected to dominate the deployment segment with 65%, while solutions are projected to lead the component segment with 50%.
Summary of the Structured Product Label Management Market
- Market Definition
- The market comprises software platforms and services used to create, manage, and standardize regulated product labeling content in structured digital formats. These systems organize product information, safety data, and regulatory content to ensure consistency and compliance across global markets. They support version control, content reuse, and automated workflows for regulatory submissions. Structured labeling platforms are widely used in pharmaceutical, biotechnology, and medical device industries to streamline documentation and compliance processes.
- Demand Drivers
- Increasing regulatory complexity across global markets requiring standardized and compliant product labeling systems.
- Rising adoption of digital platforms for managing regulatory submissions, multilingual labeling, and documentation workflows.
- Growth in pharmaceutical and biotechnology production driving demand for centralized labeling management systems.
- Need for accurate, consistent, and traceable product information across multiple regulatory environments.
- Key Segments Analyzed
- Deployment: Cloud based systems account for about 65% share due to scalability and centralized access across global operations.
- Component: Solutions represent roughly 50% share supported by core functionality for labeling content management and workflow automation.
- End User: Pharmaceutical companies remain the primary users due to regulatory requirements and global product distribution.
- Geography: India records the fastest expansion supported by growth in pharmaceutical manufacturing and adoption of digital compliance systems.
- Analyst Opinion at Fact MR
- Shambhu Nath Jha, Principal Consultant, Fact MR, opines, "In this updated edition of the Structured Product Label Management Market report, industry participants will observe that regulatory data management systems are increasingly transitioning toward structured digital platforms. These systems enable efficient management of labeling content, compliance workflows, and regulatory submissions. Companies offering integrated and scalable labeling solutions will maintain competitive positions in life sciences software markets."
- Strategic Implications or Executive Takeaways
- Invest in development of structured labeling platforms supporting regulatory compliance and automated documentation workflows.
- Strengthen integration with regulatory information management, quality management, and enterprise content systems.
- Expand cloud based deployment capabilities to support global collaboration and real time data access.
- Enhance system validation and compliance features aligned with evolving regulatory standards across regions.
- Methodology
- Primary interviews conducted with regulatory affairs professionals, life sciences software providers, compliance specialists, and contract research organizations.
- Desk research based on pharmaceutical industry publications, regulatory agency guidelines, company reports, and clinical research databases.
- Market sizing developed through a hybrid approach combining bottom up software revenue analysis and top down demand assessment across life sciences and regulatory compliance sectors.
- Findings validated through multiple industry data sources and expert consultations following Fact MR internal modeling standards.
Segmental Analysis
Structured Product Label Management Market Analysis by Deployment
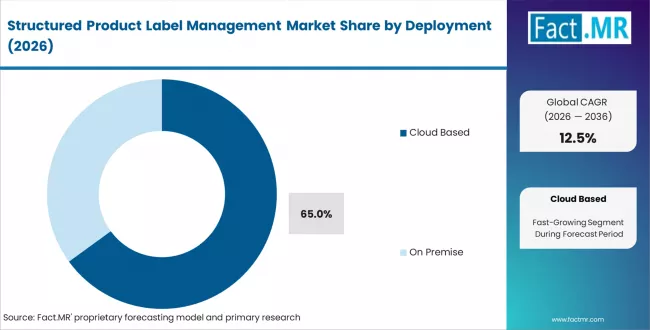
- Market Overview: Cloud based deployment is projected to account for around 65% share of the structured product label management market by 2026. Cloud platforms provide scalable infrastructure for managing product labeling data, regulatory content, and document workflows across distributed teams. Organizations deploy cloud based label management systems to centralize labeling processes, enable real time updates, and maintain compliance with regional regulatory requirements. Pharmaceutical, biotechnology, and medical device companies rely on cloud environments to manage multilingual labeling, version control, and approval workflows. The flexibility of cloud deployment supports collaboration across global operations and reduces dependency on on premise infrastructure.
- Demand Drivers:
- Centralized Label Management: Cloud platforms allow organizations to manage product labeling content, updates, and approvals within a unified system.
- Regulatory Compliance Support: Label management systems help organizations maintain compliance with regional regulatory standards through controlled documentation workflows.
- Global Collaboration: Cloud based systems enable teams across multiple locations to access and update labeling information in real time.
Structured Product Label Management Market Analysis by Component
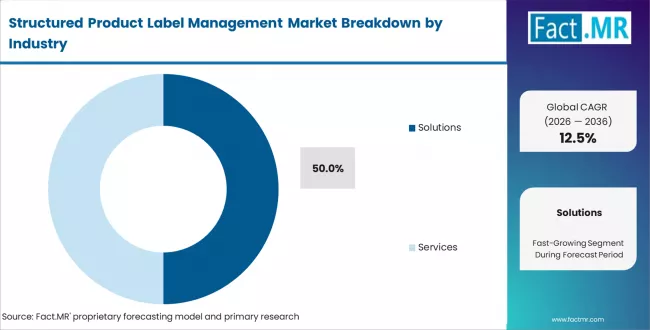
- Market Overview: Solutions are expected to represent approximately 50% share of the structured product label management market in 2026. Solution platforms provide the core functionality required to create, manage, and distribute structured labeling content across regulatory and commercial channels. These platforms integrate with enterprise systems such as regulatory information management systems, document management systems, and quality management platforms. Organizations deploy label management solutions to standardize labeling formats, automate document generation, and ensure accuracy in product information across markets.
- Demand Drivers:
- Labeling Workflow Automation: Solution platforms automate the creation, review, and approval of product labeling documents.
- Integration with Enterprise Systems: Label management solutions connect with regulatory, quality, and document management systems to support data consistency.
- Standardization of Label Content: Organizations use structured labeling solutions to maintain consistent product information across different regions and regulatory environments.
Structured Product Label Management Market Drivers, Restraints, and Opportunities
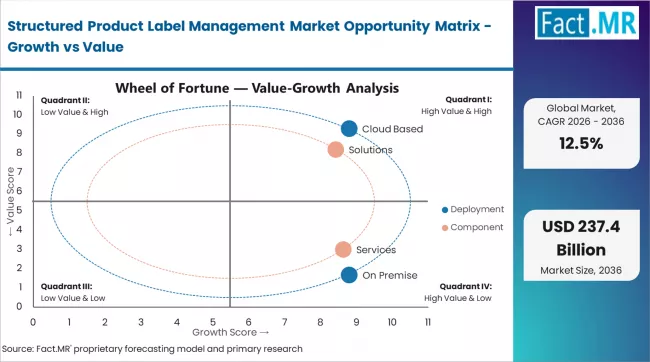
Fact MR analysis indicates that the structured product label management market developed from regulatory documentation systems used by pharmaceutical and life sciences companies to manage product labeling across multiple markets. Early labeling processes relied on manual documentation and fragmented content management systems that created inefficiencies in regulatory submissions. The current market valuation reflects the adoption of centralized labeling platforms that manage product information, regulatory content, and multilingual documentation within unified systems. Demand persists because pharmaceutical companies must maintain accurate, compliant labeling across global markets with varying regulatory requirements.
A structural shift is occurring as traditional document-based labeling workflows give way to structured content management systems that enable modular content reuse and automated updates. Conventional labeling processes remain present in smaller organizations where manual workflows continue due to cost constraints. Advanced label management platforms integrate regulatory databases, version control systems, and submission management tools that support faster approval cycles and compliance tracking. These platforms require higher implementation and licensing costs due to system integration and validation requirements. Even with gradual adoption across mid-sized companies, higher pricing associated with enterprise labeling systems supports steady market value expansion.
- Regulatory Content Automation: Pharmaceutical companies deploy structured labeling systems to manage product information, automate document updates, and support global regulatory submissions.
- Pharmaceutical Labeling Regulation: Frameworks such as the U.S. Food and Drug Administration Structured Product Labeling (SPL) standard and European Medicines Agency electronic product information (ePI) initiatives govern labeling formats and submission processes.
- Europe Life Sciences Compliance: Germany, Switzerland, and the United Kingdom maintain strong pharmaceutical industries that adopt structured label management platforms to meet regulatory documentation requirements.
Regional Analysis
The structured product label management market is assessed across North America, Europe, Asia Pacific, Latin America, and Middle East & Africa, segmented by country-level regulatory compliance, pharmaceutical labeling standards, and digital documentation systems. Regional demand reflects regulatory mandates and lifecycle management requirements. The full report offers market attractiveness analysis.
.webp)
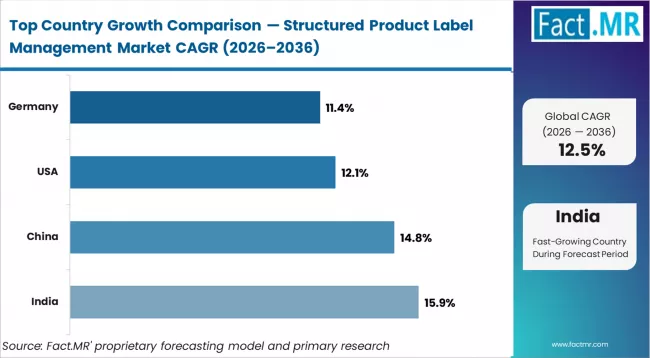
| Country | CAGR (2026-2036) |
|---|---|
| India | 15.9% |
| China | 14.8% |
| Germany | 11.4% |
| USA | 12.1% |
Source: Fact MR analysis, based on proprietary forecasting model and primary research
North America
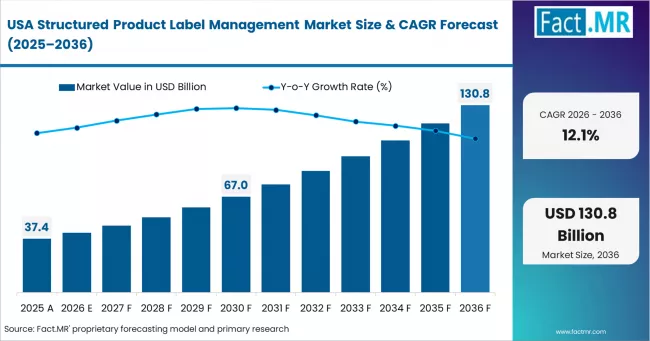
North America functions as the global regulatory compliance and digital health documentation hub, supported by structured submission requirements and advanced enterprise software adoption. Veeva Systems Inc. maintains leadership in regulatory information management platforms. Oracle Corporation strengthens presence through health sciences solutions. DXC Technology Company supports large-scale enterprise implementations.
- USA: Demand for structured product label management in USA is projected to rise at 12.1% CAGR through 2036. Growth is supported by Structured Product Labeling requirements (U.S. Food and Drug Administration, updated 2020), mandating standardized submissions. Veeva Systems Inc. expanded Vault RIM capabilities for labeling management (March 2023), supporting compliance workflows.
Europe
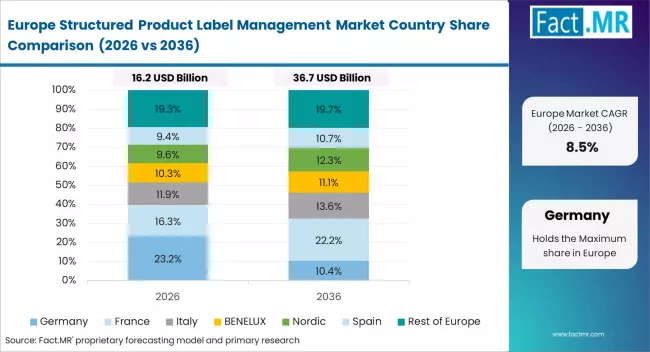
Europe operates as the regulatory harmonization and pharmacovigilance base, shaped by centralized drug approval processes and documentation standards. ArisGlobal LLC maintains strong positioning in regulatory software. EXTEDO GmbH specializes in submission and labeling solutions. PharmaLex GmbH supports compliance and regulatory services.
- Germany: Demand for structured product label management in Germany is projected to rise at 11.4% CAGR through 2036. The EU Clinical Trials Regulation (European Commission, January 2022) strengthens data transparency requirements. EXTEDO GmbH expanded regulatory software solutions (February 2023), supporting compliance adoption.
Asia Pacific
Asia Pacific serves as the high-growth regulatory digitization market, supported by expanding pharmaceutical manufacturing and evolving compliance frameworks. Parexel International Corporation supports regulatory outsourcing services. Ennov SAS expands labeling and compliance solutions. Acolad Group strengthens structured content management capabilities.
- India: Demand for structured product label management in India is projected to rise at 15.9% CAGR through 2036. The New Drugs and Clinical Trials Rules (CDSCO, March 2019) strengthened regulatory submission requirements. Parexel International Corporation expanded regulatory service capabilities in India (April 2023), supporting compliance demand.
- China: Demand for structured product label management in China is projected to rise at 14.8% CAGR through 2036. The Drug Administration Law amendment (National Medical Products Administration, December 2019) enhanced labeling and traceability requirements. Ennov SAS expanded regulatory software deployment in China (May 2023), supporting adoption.
Fact MR's analysis of structured product label management market in global regions consists of country-wise assessment that includes India, China, Germany, and USA. Readers can find regulatory trends, compliance requirements, enterprise adoption patterns, and competitive positioning across key markets.
Competitive Structure and Buyer Dynamics in the Structured Product Label Management Market
The competitive structure of the Structured Product Label Management Market is moderately concentrated, with a group of specialized regulatory software providers and enterprise technology firms controlling a significant share of deployments. Companies such as Veeva Systems Inc., ArisGlobal LLC, Oracle Corporation (Oracle Health Sciences division), and MasterControl, Inc. maintain strong positions through dedicated life sciences software platforms designed for regulatory compliance and product labeling management. These firms provide solutions used by pharmaceutical, biotechnology, and medical device companies to manage labeling content, submission workflows, and regulatory documentation. Additional participants including DXC Technology Company, Acolad Group, Parexel International Corporation, Ennov SAS, PharmaLex GmbH, and EXTEDO GmbH contribute through regulatory consulting, labeling services, and specialized compliance software. Competition in the market is primarily driven by regulatory compliance capability, system integration with clinical and regulatory platforms, and the ability to manage complex global labeling requirements.
Several companies maintain structural advantages through deep domain expertise in life sciences regulation and long term relationships with pharmaceutical companies. Firms such as Veeva Systems Inc. and ArisGlobal LLC benefit from established platforms widely adopted across global pharmaceutical organizations. Oracle Corporation leverages its broader enterprise software ecosystem to support integrated regulatory and clinical data management solutions. Service providers such as Parexel International Corporation and PharmaLex GmbH maintain advantages through regulatory consulting expertise and long term client engagements. Pharmaceutical companies typically adopt multi vendor strategies to reduce dependency on a single software provider and ensure compliance flexibility across regions. Procurement decisions evaluate vendors based on regulatory expertise, system reliability, and validation support. This purchasing behavior moderates supplier pricing leverage, though vendors with validated platforms and strong compliance track records retain stronger negotiating influence.
Key Players of the Structured Product Label Management Market
- Veeva Systems Inc.
- ArisGlobal LLC
- DXC Technology Company
- MasterControl, Inc.
- Acolad Group (parent company of Amplexor)
- Oracle Corporation (Oracle Health Sciences division)
- Parexel International Corporation
- Ennov SAS
- PharmaLex GmbH
- EXTEDO GmbH
Report Scope

| Metric | Value |
|---|---|
| Quantitative Units | USD 73.1 billion (2026) to USD 237 billion (2036), at a CAGR of 12.5% |
| Market Definition | The structured product label management market includes software platforms and related services used to create, manage, standardize, and distribute regulated product labeling information across life sciences organizations, ensuring compliance with regional regulatory requirements and consistency across pharmaceutical, biotechnology, and medical device documentation. |
| Solution Segmentation | Software, Services |
| Deployment Segmentation | Cloud-based, On-Premise |
| End User Segmentation | Pharmaceutical Companies, Biotechnology Companies, Medical Device Companies, Regulatory Authorities, Contract Research Organizations (CROs) |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, Australia, and 40+ countries |
| Key Companies Profiled | Veeva Systems Inc., ArisGlobal LLC, DXC Technology Company, MasterControl, Inc., Acolad Group (parent company of Amplexor), Oracle Corporation (Oracle Health Sciences division), Parexel International Corporation, Ennov SAS, PharmaLex GmbH, EXTEDO GmbH |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling supported by regulatory data management adoption analysis, life sciences IT spending assessment, product labeling workflow evaluation, and validation through interviews with pharmaceutical companies, regulatory specialists, and software providers. |
Bibliographies
- [1] U.S. Food and Drug Administration. (2020). Structured Product Labeling (SPL) requirements.
- [2] European Commission. (2022). EU Clinical Trials Regulation.
- [3] Central Drugs Standard Control Organization, India. (2019). New Drugs and Clinical Trials Rules.
- [4] National Medical Products Administration, China. (2019). Drug Administration Law amendment.
- [5] Veeva Systems Inc. (2023). Vault RIM labeling management expansion.
- [6] EXTEDO GmbH. (2023). Regulatory software solutions expansion.
- [7] Parexel International Corporation. (2023). Regulatory services expansion in India.
- [8] Ennov SAS. (2023). Regulatory software deployment in China.
- [9] European Medicines Agency. (ongoing). Electronic product information (ePI) initiatives.
This Report Addresses
- Market size estimation and revenue forecasts for the Structured Product Label Management Market from 2026 to 2036, supported by life sciences software adoption benchmarks and regulatory data management demand indicators across pharmaceutical and biotechnology sectors.
- Growth opportunity analysis across software solutions and associated services, along with cloud based and on premise deployment models used in structured labeling and regulatory content management systems.
- Segment and regional revenue forecasts covering pharmaceutical companies, biotechnology firms, medical device manufacturers, regulatory authorities, and contract research organizations managing product labeling and compliance documentation.
- Competitive strategy assessment including regulatory information management integration capability, labeling workflow automation, and enterprise compliance platform benchmarking among life sciences software providers.
- Regulatory and compliance evaluation covering frameworks such as United States Food and Drug Administration Structured Product Labeling standards and European Medicines Agency electronic product information initiatives governing digital labeling formats and submission processes.
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats designed for regulatory compliance planning, life sciences IT strategy evaluation, and product labeling workflow optimization.
- Supply chain and operational risk analysis identifying integration complexity with legacy regulatory systems, high implementation and validation costs, and evolving global compliance requirements affecting adoption of structured product label management platforms.
Structured Product Label Management Market Key Segments
-
Solutions
- Software
- Services
-
Deployment
- Cloud Based
- On Premise
-
End User
- Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Companies
- Regulatory Authorities
- Contract Research Organizations (CROs)
-
Region
- North America
- USA
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East and Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Rest of Middle East and Africa
- Other Regions
- Oceania
- Central Asia
- Other Markets
- North America
- Frequently Asked Questions -
How large is the demand for Structured Product Label Management in the global market in 2026?
Demand for Structured Product Label Management in the global market is estimated to be valued at USD 73.1 billion in 2026.
What will be the market size of Structured Product Label Management in the global market by 2036?
Market size for Structured Product Label Management is projected to reach USD 237 billion by 2036.
What is the expected demand growth for Structured Product Label Management in the global market between 2026 and 2036?
Demand for Structured Product Label Management in the global market is expected to grow at a CAGR of 12.5% during 2026 to 2036.
Which Deployment Segment is poised to lead global demand by 2026?
Cloud based deployment is expected to remain the dominant segment, accounting for approximately 65% of the global market share in 2026.
Which Company is Identified as a Leading Participant in the Structured Product Label Management Market?
Veeva Systems Inc. is identified as a leading participant in the structured product label management market due to its strong presence in cloud based life sciences software solutions.
What is Driving Structured Product Label Management Demand in the United States?
Demand growth in the United States is supported by increasing regulatory requirements and adoption of digital labeling and compliance management systems.
What is the United States Growth Outlook in this Report?
The United States structured product label management market is projected to expand at a CAGR of 12.1% during 2026 to 2036.
Why is China an Important Market for Structured Product Label Management?
China represents a significant market due to expanding pharmaceutical manufacturing and increasing adoption of regulatory compliance software platforms.
What is the China Growth Outlook in this Report?
The China structured product label management market is projected to expand at a CAGR of 14.8% during 2026 to 2036.
What Factors Support Structured Product Label Management Demand in India?
India demand growth is associated with growing pharmaceutical production and increasing need for standardized product labeling and regulatory documentation systems.
What is the India Growth Outlook in this Report?
The India structured product label management market is projected to expand at a CAGR of 15.9% during 2026 to 2036.
Why is Germany Considered an Important Market for Structured Product Label Management in Europe?
Germany represents a key European market due to strong pharmaceutical and chemical manufacturing industries and strict regulatory compliance requirements.
What is the Germany Growth Outlook in this Report?
The Germany structured product label management market is projected to expand at a CAGR of 11.4% during 2026 to 2036.
What is Structured Product Label Management and What is It Mainly Used For?
Structured product label management refers to software systems used to create, manage, and standardize product labeling information across regulated industries.
What does Structured Product Label Management Market Mean in this Report?
Structured product label management market refers to global development, deployment, and adoption of software platforms used for product labeling and regulatory compliance.
What is Included in the Scope of this Structured Product Label Management Market Report?
The scope includes structured product label management solutions categorized by deployment and their use across pharmaceutical, chemical, and regulated product industries.
What is Excluded from the Scope of this Report?
General document management systems and unrelated enterprise software are excluded unless integrated with structured labeling platforms.
What does Market Forecast Mean on this Page?
Market forecast represents a projection of future demand based on regulatory compliance requirements and adoption of digital labeling systems.
How is the Structured Product Label Management Market Forecast Developed in this Report?
Forecast analysis is developed using enterprise software adoption data, regulatory compliance trends, and pharmaceutical industry IT investment statistics.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- Cloud Based
- On Premise
- Cloud Based
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Component
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Component, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Component, 2026 to 2036
- Solutions
- Services
- Solutions
- Y to o to Y Growth Trend Analysis By Component, 2021 to 2025
- Absolute $ Opportunity Analysis By Component, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Deployment
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Component
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Deployment
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Component
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Deployment
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Component
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Deployment
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Component
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Deployment
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Component
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Deployment
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Component
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Deployment
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Deployment
- By Component
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Deployment
- By Component
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Deployment
- By Component
- Competition Analysis
- Competition Deep Dive
- Veeva Systems Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- ArisGlobal LLC
- DXC Technology Company
- MasterControl, Inc.
- Acolad Group (parent company of Amplexor)
- Oracle Corporation (Oracle Health Sciences division)
- Parexel International Corporation
- Ennov SAS
- PharmaLex GmbH
- EXTEDO GmbH
- Veeva Systems Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Component, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Deployment
- Figure 6: Global Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Component
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Deployment
- Figure 23: North America Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Component
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Deployment
- Figure 30: Latin America Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Component
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Deployment
- Figure 37: Western Europe Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Component
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Component
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Deployment
- Figure 51: East Asia Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Component
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Component
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Component
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis