Alimentary Tract Implant Market Size, Share, Growth and Forecast (2026 - 2036)
Alimentary Tract Implant Market is segmented by Implant Type (Bariatric Restriction and Balloons, Gastric Electrical Stimulation or Neuromodulation, GI Stents or Endoluminal Scaffolds, and Continence or Anal Sphincter Implants), Indication (Obesity or Metabolic Control, Motility Disorders, GI Stricture Obstruction or Leak Management, and Fecal Incontinence or Continence Restoration), Procedure Approach (Endoscopic or Transoral, Laparoscopic Bariatric, Open or Revision Surgery, and Colorectal or Perineal Implantation), Care Setting (Bariatric and Metabolic Centers, GI and Advanced Endoscopy Suites, Colorectal or Pelvic Floor Centers, and Hospital OR or Other Acute Care), and Region. Forecast for 2026 to 2036.
Fact.MR is of the opinion that the alimentary tract implant market is structurally driven by procedure eligibility expansion and long term disease management pathways, where clinical outcomes, minimally invasive access, and reimbursement alignment influence adoption more than short term device development cycles.
Segmental Analysis
Alimentary Tract Implant Market Analysis by Implant Type
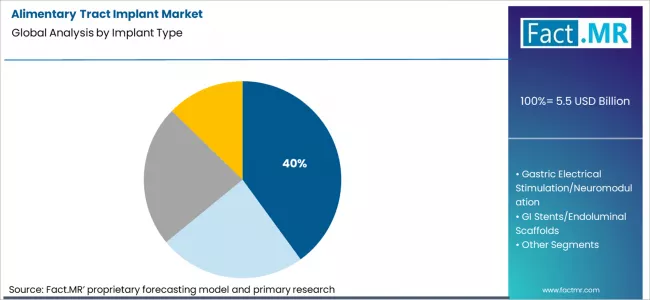
Based on Fact.MR analysis, consumption of bariatric restriction devices and gastric balloons is estimated to hold 40% share of the global alimentary tract implant market. Fact.MR analysts note that this implant type leads due to minimally invasive placement, reversibility, and shorter recovery periods compared with surgical bypass procedures. As per Fact.MR, these implants address clinical needs for temporary volume restriction and appetite control, particularly for patients requiring non-surgical or bridge-to-surgery obesity interventions under monitored treatment pathways across hospital and specialty clinic settings worldwide.
- Regulatory oversight: Implantable bariatric devices are regulated as medical devices requiring premarket authorization and post-market surveillance.[4]
- Clinical preference: Fact.MR analysts note gastric balloons are often selected for patients not eligible for permanent surgical implants.
- Procedure setting: These implants are commonly placed endoscopically in outpatient or short-stay hospital environments.
Alimentary Tract Implant Market Analysis by Indication
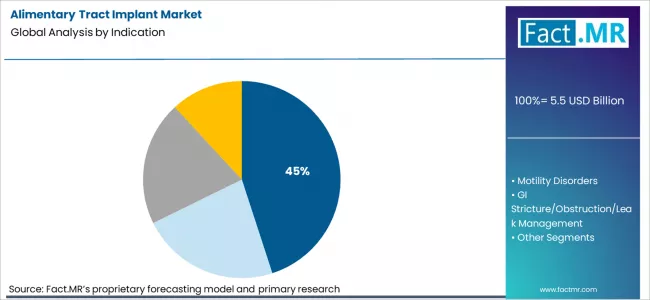
Based on Fact.MR analysis, consumption of alimentary tract implants for obesity and metabolic control is estimated to hold 45% share of the global market. Fact.MR opines that this indication leads due to rising prevalence of obesity-related conditions requiring interventional management beyond pharmacological therapy. As per Fact.MR, implants used for metabolic control address clinical goals related to weight reduction, glycemic improvement, and appetite regulation, supporting structured treatment programs delivered through bariatric centers and multidisciplinary care models globally.
- Public health context: Government health agencies recognize obesity as a chronic disease requiring medical and interventional treatment options. [5]
- Treatment pathways: Fact.MR analysts note implants are used alongside dietary and behavioral management programs.
- Care delivery: Obesity-focused implant procedures are concentrated in specialized bariatric and metabolic clinics.
Alimentary Tract Implant Market Drivers, Restraints, And Opportunities
Fact.MR analysis indicates that the alimentary tract implant market exists as a specialised medical device segment serving surgical restoration and support of the gastrointestinal tract following trauma, disease, or congenital anomalies. As per Fact.MR assessment, alimentary tract implants include mesh, stents, and prosthetic structures used to reinforce tissue, maintain lumen patency, or replace damaged anatomy; these devices are regulated through established medical device frameworks such as the U.S. Food and Drug Administration (FDA) Class II/III premarket pathways that govern design controls, clinical evidence, and post-marketing surveillance for implants. Fact.MR analysts observe that the current market size is driven by rising surgical volumes, adoption of minimally invasive techniques, and clinical guidelines that recommend device-assisted repair for conditions such as hernias and strictures.
Fact.MR is of the opinion that current market dynamics reflect a transition toward advanced implant materials and delivery mechanisms that improve biocompatibility and procedural efficiency. Based on Fact.MR assessment, conventional sutured repairs without implant support continue to be used in select cases because of cost and surgeon preference, but growth is occurring in segments where implantable meshes and stents provide measurable reductions in recurrence rates and procedural morbidity. Advanced alimentary tract implant products that incorporate novel polymers, anti-adhesive coatings, or resorbable elements typically carry higher per-unit pricing because of material complexity and regulatory testing requirements, meaning realised market value can increase even where shipment volumes grow at a measured pace. Fact.MR analysis suggests this balance between moderate volume growth and premium device placement defines near-term market trends.
- Regulatory medical device frameworks: Fact.MR analysts note that medical device regulation under the U.S. Food and Drug Administration (FDA) influences development, testing, and approval requirements for alimentary tract implants, affecting market entry timelines and product positioning.
- Shift to advanced materials: Based on Fact.MR assessment, demand is increasing for implantable meshes and stents that offer improved biocompatibility, reduced adhesion risks, and enhanced structural performance compared with conventional options.
- Asia Pacific surgical infrastructure growth: Fact.MR opines that expansion of surgical facilities and minimally invasive procedure adoption in China, India, and Southeast Asia supports regional uptake of alimentary tract implants as part of gastrointestinal surgical care.
Regional Analysis
Based on regional assessment, the alimentary tract implant sector is analyzed across North America, Europe, East Asia, Latin America, and other major healthcare markets, spanning more than 40 countries. Regional performance varies according to surgical procedure volumes, access to advanced gastrointestinal treatments, healthcare infrastructure quality, and adoption of implant-based therapeutic interventions. The analysis also presents a comparative market attractiveness evaluation grounded in region-specific demand trends.
Alimentary Tract Implant Market CAGR Analysis by Country (2026-2036)
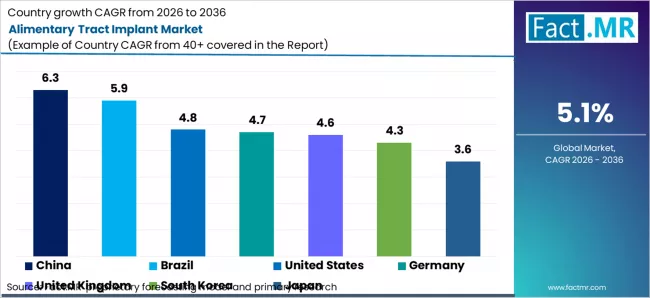
| Country | CAGR |
|---|---|
| China | 6.3% |
| Brazil | 5.9% |
| United States | 4.8% |
| Germany | 4.7% |
| United Kingdom | 4.6% |
| South Korea | 4.3% |
| Japan | 3.6% |
Source: Fact.MR analysis, based on proprietary forecasting models and primary research.
Asia Pacific Alimentary Tract Implant Market Analysis
Asia Pacific operates as a procedure volume driven alimentary tract implant region supported by expanding hospital capacity rising gastrointestinal disease burden and improving access to minimally invasive surgery. Demand is shaped by national medical device regulations reimbursement coverage and public hospital procurement systems. Adoption is concentrated in tertiary hospitals where endoscopic and surgical implants are integrated into colorectal bariatric and esophageal procedures. Regional growth reflects investment in surgical infrastructure clinician training programs and gradual expansion of insurance coverage across systems.
- China: Demand for alimentary tract implants in China is projected to rise at 6.3% CAGR through 2036. Usage is supported by high procedural volumes in public hospitals and adoption of minimally invasive gastrointestinal surgeries, as per Fact.MR.
- South Korea: Demand for alimentary tract implants in South Korea is projected to rise at 4.3% CAGR through 2036. Adoption is driven by advanced hospital infrastructure and widespread use of endoscopic and surgical gastrointestinal procedures, according to Fact.MR.
- Japan: Demand for alimentary tract implants in Japan is projected to rise at 3.6% CAGR through 2036. Consumption reflects stable surgical volumes and established reimbursement for gastrointestinal procedures, as per Fact.MR.
Fact.MR’s analysis of the Alimentary Tract Implant Market in Asia Pacific consists of country-wise assessment that includes China, South Korea, and Japan. Readers can find regulatory frameworks, hospital adoption trends, and procedure-driven demand references.
North America Alimentary Tract Implant Market Analysis
North America functions as a reimbursement and regulation led alimentary tract implant region characterized by established surgical standards and high procedure penetration. Demand is governed by FDA device approvals coding structures and hospital purchasing protocols. Adoption is concentrated within large hospital networks and ambulatory surgical centers where implants are used in bariatric colorectal and gastrointestinal oncology procedures. Market activity reflects replacement cycles clinical guideline adherence and stable funding through public and private payer systems supporting predictable procurement volumes nationwide annually.
- United States: Demand for alimentary tract implants in the United States is projected to rise at 4.8% CAGR through 2036. Usage is supported by high adoption of bariatric and colorectal procedures and stable reimbursement coverage across hospital systems, as per Fact.MR.
Fact.MR’s analysis of the Alimentary Tract Implant Market in North America consists of country-wise assessment that includes the United States. Readers can find reimbursement structures, hospital procurement practices, and procedure-based demand insights.
Europe Alimentary Tract Implant Market Analysis
Europe operates as a regulation led alimentary tract implant region shaped by harmonized medical device rules and public healthcare funding. Demand is guided by MDR compliance national reimbursement policies and hospital tendering systems. Adoption is centered in publicly funded hospitals where gastrointestinal implants are used in colorectal bariatric and upper digestive surgeries. Market growth reflects procedural demand demographic aging and standardized clinical pathways across Western and Northern European healthcare systems supported by consistent regulatory oversight and cross border procurement frameworks regionwide.
- Germany: Demand for alimentary tract implants in Germany is projected to rise at 4.7% CAGR through 2036. Adoption is driven by high procedural volumes in public hospitals and reimbursement under statutory health insurance systems, according to Fact.MR.
- United Kingdom: Demand for alimentary tract implants in the United Kingdom is projected to rise at 4.6% CAGR through 2036. Usage reflects public hospital procurement under NHS funding and standardized gastrointestinal care pathways, as per Fact.MR.
Fact.MR’s analysis of the Alimentary Tract Implant Market in Europe consists of country-wise assessment that includes Germany and the United Kingdom. Readers can find regulatory compliance requirements, procurement models, and procedure volume trends.
Latin America Alimentary Tract Implant Market Analysis
Latin America functions as an expanding alimentary tract implant region supported by growing surgical access and hospital investment. Demand is influenced by national regulatory approvals reimbursement expansion and public private healthcare procurement. Adoption remains concentrated in urban tertiary hospitals performing gastrointestinal and bariatric procedures. Market development reflects gradual insurance coverage growth increasing procedural volumes and ongoing modernization of surgical infrastructure across major healthcare markets driven by public hospital expansion programs clinician training initiatives and device import approvals regionwide today broadly.
- Brazil: Demand for alimentary tract implants in Brazil is projected to rise at 5.9% CAGR through 2036. Usage is supported by expanding access to bariatric and gastrointestinal surgeries within public and private hospital networks, as per Fact.MR.
Fact.MR’s analysis of the Alimentary Tract Implant Market in Latin America consists of country-wise assessment that includes Brazil. Readers can find healthcare investment trends, regulatory approvals, and procedure-based demand insights.
Competitive Landscape of Alimentary Tract Implant Market
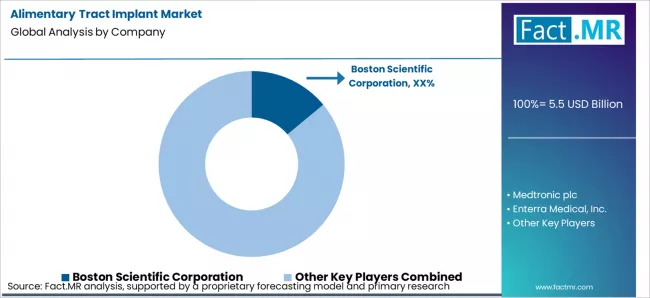
As per Fact.MR analysis, the 2026 alimentary tract implant market is defined by validated clinical efficacy, regulatory compliance, and hospital network adoption, which determine competitive advantage. Boston Scientific Corporation and Medtronic plc lead with fully tested implantable systems and long-term clinical outcomes, which strengthens adoption in gastroenterology and bariatric care programs. Enterra Medical, Inc. and EndoStim, Inc. focus on neuromodulation and gastric electrical stimulation devices with documented procedural efficacy, which improves patient outcomes and repeat utilization. Johnson & Johnson (Ethicon) and Cook Group provide precision-engineered implants with proven biocompatibility and multi-region regulatory approvals, which reduces procedural risk and enhances hospital trust. Coloplast A/S and B. Braun SE offer validated delivery systems and instrumentation, which increases reliability and simplifies surgeon workflow. Micro-Tech Endoscopy and Torax Medical provide innovative minimally invasive solutions with validated deployment mechanisms, which expands adoption in niche and emerging markets. Across the market, clinical validation, regulatory adherence, and integrated hospital support create enduring competitive moats, reducing reliance on price or isolated product claims.
Recent Industry Developments
- Johnson & Johnson (Ethicon): OTTAVA™ Robotic Filing: In January 2026, Johnson & Johnson officially submitted its OTTAVA™ Robotic Surgical System to the FDA for De Novo classification. Specifically designed for "multi-quadrant" surgery, the system is being targeted for complex alimentary tract procedures, including gastric bypass, sleeve gastrectomy, and hiatal hernia repairs. [6]
- Medtronic: Hugo™ Robotics & AI Integration: In February 2026, Medtronic announced the first U.S. surgery using its Hugo™ robotic-assisted surgery system at the Cleveland Clinic. This follows a 2025 study showing the Hugo system's success in hernia repair and GI tract interventions, marking Medtronic's aggressive push to challenge the monopoly in soft-tissue robotic implants. [7]
Key Players in Alimentary Tract Implant Market
- Boston Scientific Corporation
- Medtronic plc
- Enterra Medical, Inc.
- Johnson & Johnson Services, Inc. (Ethicon)
- Coloplast A/S
- Cook Group Incorporated (Cook Medical)
- B. Braun SE
- Micro-Tech Endoscopy (Hangzhou) Co., Ltd.
- EndoStim, Inc.
- Torax Medical, Inc.
Bibliography
- [1] U.S. Food and Drug Administration. (2024). Medical devices: Overview, regulation, and safety. U.S. Department of Health and Human Services; World Health Organization. (2024). Medical devices. World Health Organization.
- [2] Centers for Disease Control and Prevention. (2024). Obesity basics: Causes, health effects, and treatment approaches. U.S. Department of Health and Human Services.
- [3] U.S. Food and Drug Administration. (2024). Medical devices regulatory framework and device classification. U.S. Department of Health and Human Services.
- [4] U.S. Food and Drug Administration. (2024). Weight management medical devices: Gastric balloons and bariatric implants. U.S. Department of Health and Human Services.
- [5] Centers for Disease Control and Prevention. (2024). Adult obesity facts and chronic disease classification. U.S. Department of Health and Human Services.
- [6] Johnson & Johnson Services, Inc. (2026). Johnson & Johnson submits OTTAVA™ robotic surgical system for FDA review. Johnson & Johnson - Investor & Media Relations.
- [7] Medtronic plc. (2026). Medtronic announces first U.S. clinical use of the Hugo™ robotic-assisted surgery system. Medtronic Newsroom.
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 5.50 billion (2026) to USD 9.10 billion (2036), at a CAGR of 5.1% |
| Market Definition | The alimentary tract implant market covers medical implants designed to support, repair, or restore function within the gastrointestinal tract. These implants include stents, meshes, gastric balloons, neuromodulation devices, and continence implants, and are used in surgical or endoscopic procedures to treat obesity, motility disorders, obstruction, and fecal incontinence. |
| Implant Type Segmentation | Bariatric Restriction & Balloons, Gastric Electrical Stimulation/Neuromodulation, GI Stents/Endoluminal Scaffolds, Continence/Anal Sphincter Implants |
| Indication Segmentation | Obesity/Metabolic Control, Motility Disorders, GI Stricture/Obstruction/Leak Management, Fecal Incontinence/Continence Restoration |
| Procedure Approach Segmentation | Endoscopic/Transoral, Laparoscopic Bariatric, Open/Revision Surgery, Colorectal/Perineal Implantation |
| Care Setting Segmentation | Bariatric & Metabolic Centers, GI & Advanced Endoscopy Suites, Colorectal/Pelvic Floor Centers, Hospital OR/Other Acute Care |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Japan, South Korea, Indonesia, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, Italy, France, United Kingdom, Spain, Benelux, Nordics, Central & Eastern Europe, Rest of Europe, United States, Canada, Mexico, Brazil, Argentina, Chile, Rest of Latin America, Kingdom of Saudi Arabia, United Arab Emirates, South Africa, Turkey, Rest of Middle East & Africa |
| Key Companies Profiled | Boston Scientific Corporation, Medtronic plc, Enterra Medical, Inc., Johnson & Johnson Services, Inc. (Ethicon), Coloplast A/S, Cook Group Incorporated (Cook Medical), B. Braun SE, Micro-Tech Endoscopy (Hangzhou) Co., Ltd., EndoStim, Inc., Torax Medical, Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up modeling validated through primary interviews with implant manufacturers, gastrointestinal surgeons, hospital procurement managers, and clinical engineering staff. Market sizing supported by procedure volumes, implant utilization rates by indication, and revision/replacement frequency, aligned with FDA and WHO medical device frameworks. |
Alimentary Tract Implant Market Analysis by Segments
-
Implant Type :
- Bariatric Restriction & Balloons
- Gastric Electrical Stimulation/Neuromodulation
- GI Stents/Endoluminal Scaffolds
- Continence/Anal Sphincter Implants
-
Indication :
- Obesity/Metabolic Control
- Motility Disorders
- GI Stricture/Obstruction/Leak Management
- Fecal Incontinence/Continence Restoration
-
Procedure Approach :
- Endoscopic/Transoral
- Laparoscopic Bariatric
- Open/Revision Surgery
- Colorectal/Perineal Implantation
-
Care Setting :
- Bariatric & Metabolic Centers
- GI & Advanced Endoscopy Suites
- Colorectal/Pelvic Floor Centers
- Hospital OR/Other Acute Care
-
Region :
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
- Asia Pacific
- Frequently Asked Questions -
How large is the alimentary tract implant market in 2026?
The market is estimated to reach USD 5.50 billion in 2026 based on Fact.MR analysis.
What will the alimentary tract implant market size be by 2036?
By 2036, the market is projected to reach nearly USD 9.10 billion.
What CAGR is expected for the alimentary tract implant market during 2026–2036?
Fact.MR projects a 5.1% CAGR over the forecast period.
What was the market value of alimentary tract implants in 2025?
In 2025, the market was valued at approximately USD 5.25 billion.
How much absolute value is the market expected to add over the forecast period?
The market is expected to add close to USD 3.9 billion in value between 2026 and 2036.
Which implant type currently leads the alimentary tract implant market?
Bariatric restriction devices and gastric balloons lead the market with about 40% share.
Why do bariatric restriction devices and gastric balloons dominate demand?
They offer minimally invasive, reversible treatment options with shorter recovery periods, supporting wider clinical adoption.
Which indication accounts for the largest share of alimentary tract implant demand?
Obesity and metabolic control account for roughly 45% share of the global market.
What factors are driving growth in obesity-related alimentary tract implants?
Rising obesity prevalence and expanded eligibility for interventional treatment beyond pharmacotherapy are supporting demand growth.
Which procedure approach is most influential in market expansion?
Growth is strongly supported by endoscopic and laparoscopic procedures, which reduce hospital stay and recovery time.
Which country is expected to grow the fastest in the alimentary tract implant market?
China leads growth with a projected 6.3% CAGR through 2036.
How fast is the Brazilian alimentary tract implant market expected to grow?
Brazil is projected to grow at a 5.9% CAGR, driven by obesity treatment uptake and private hospital investment.
What is the growth outlook for the United States market?
The United States is expected to grow at a 4.8% CAGR, supported by steady procedural volumes and established reimbursement.
How does the United Kingdom compare in terms of growth rate?
The United Kingdom is projected to post a 4.6% CAGR through 2036.
What is Germany’s expected CAGR in the alimentary tract implant market?
Germany is forecast to grow at a 4.7% CAGR, reflecting adoption within regulated clinical pathways.
How does South Korea’s market growth compare globally?
South Korea is expected to grow at a 4.3% CAGR, supported by advanced hospital infrastructure and endoscopic capacity.
Why is growth more moderate in Japan compared with other countries?
Japan’s market grows at 3.6% CAGR, constrained by conservative surgical eligibility criteria and longer clinical evaluation cycles.
What supports early-period market growth during the forecast window?
Early growth is supported by increasing endoscopic and laparoscopic bariatric procedures, particularly balloon-based and neuromodulation implants.
What drives mid-period demand in the alimentary tract implant market?
Mid-period performance aligns with wider clinical use of GI stents and endoluminal scaffolds for obstruction and leak management.
What factors shape value growth in the later years of the forecast?
Later growth is driven by continence restoration implants and repeat interventions, where therapy durability supports recurring device demand.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Implant Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Implant Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Implant Type , 2026 to 2036
- Bariatric Restriction & Balloons
- Gastric Electrical Stimulation/Neuromodulation
- GI Stents/Endoluminal Scaffolds
- Continence/Anal Sphincter Implants
- Bariatric Restriction & Balloons
- Y to o to Y Growth Trend Analysis By Implant Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Implant Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Obesity/Metabolic Control
- Motility Disorders
- GI Stricture/Obstruction/Leak Management
- Fecal Incontinence/Continence Restoration
- Obesity/Metabolic Control
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Implant Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Indication
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Implant Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Indication
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Implant Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Indication
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Implant Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Indication
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Implant Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Indication
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Implant Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Indication
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Implant Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Indication
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Indication
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Implant Type
- By Indication
- Competition Analysis
- Competition Deep Dive
- Boston Scientific Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Medtronic plc
- Enterra Medical, Inc.
- Johnson & Johnson Services, Inc. (Ethicon)
- Coloplast A/S
- Cook Group Incorporated (Cook Medical)
- B. Braun SE
- Micro-Tech Endoscopy (Hangzhou) Co., Ltd.
- EndoStim, Inc.
- Torax Medical, Inc.
- Boston Scientific Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Implant Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Implant Type,2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Implant Type,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Implant Type
- Figure 6: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Implant Type,2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Implant Type,2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Implant Type
- Figure 23: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Indication
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Implant Type,2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Implant Type,2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Implant Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Indication
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Implant Type,2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Implant Type,2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Implant Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Indication
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Implant Type,2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Implant Type,2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Implant Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Implant Type,2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Implant Type,2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Implant Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Indication
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Implant Type,2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Implant Type,2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Implant Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Implant Type,2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Implant Type,2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Implant Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis