Disease-on-Chip Platforms Market Size, Share, Growth and Forecast (2026 - 2036)
The global Disease-on-Chip Platforms Market is segmented by Product, Platform Type, Multi-Disease Integrated Platforms, Zechnology (Microfluidic Platforms, 3D Cell Culture Platforms, Biosensor-Integrated Platforms, AI/Automation-Integrated Platforms, Stem Cell Technologie), with forecasts covering the period from 2026 to 2036.
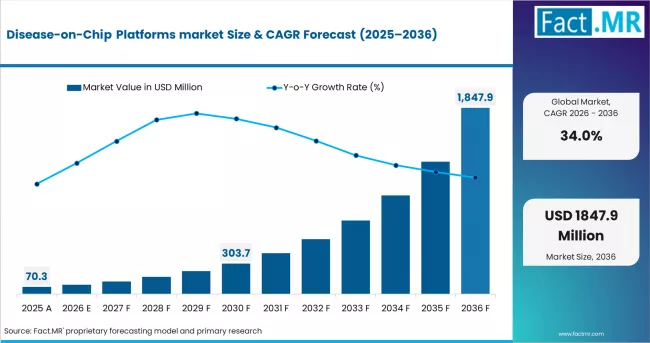
According to Fact.MR analysis, the global Disease-on-Chip Platforms market was valued at USD 70.3 million in 2025 and is projected to reach USD 99.0 million in 2026, expanding to US$ 1,847.9 million by 2036 at a CAGR of 34.0%. Drug discovery dominates with 47% share, while pharmaceutical & biotechnology companies lead the end-user segment with 59% share, driven by increasing adoption of predictive preclinical models and precision medicine.
Disease-On-Chip Platforms Market Forecast and Outlook By Fact.MR
In 2025, the Disease-On-Chip Platforms Market was valued at approximately USD 70.3 million. In 2026, the global market is estimated to reach USD 99.0 million and is projected to expand significantly to around USD 1,847.9 million (USD 1.85 billion) by 2036, exhibiting a CAGR of approximately 34.0% during the forecast period. The absolute dollar growth over the period is expected to be approximately USD 1.75 billion.
Growth in the market is primarily driven by the increasing demand for advanced in vitro disease models as alternatives to animal testing, along with the rising adoption of precision and personalized medicine approaches. The growing focus on disease-specific drug discovery, particularly in oncology and neurological conditions such as Alzheimer’s disease, is further accelerating market adoption. Additionally, rapid technological advancements in microfluidics, stem cell engineering, and 3D cell culture systems, coupled with the integration of biosensors and AI-enabled platforms, are enhancing the predictive accuracy of preclinical models.
Moreover, the growing number of partnerships between pharmaceutical and biotechnology firms, academic centers, and contract research organizations (CROs), as well as the necessity to decrease the cost and time of drug development, are likely to contribute to the further market growth. The increasing regulatory trend to reduce animal testing and the development of research facilities in the emerging economies are also expected to play a significant role in the market growth during the forecast period.
Summary of Disease-on-Chip Platforms Market
- Market Snapshot
- Global Disease-On-Chip Platforms market revenue stood at USD 70.3 million in 2025 and is estimated to reach USD 99.0 million in 2026, further projected to expand to USD 1,847.9 million by 2036.
- At a 34.0% CAGR from 2026 to 2036, the market is set to expand 18.7x in value, adding USD 1,748.9 million in absolute opportunity.
- Market growth is driven by the increasing shift toward human-relevant preclinical models, reducing reliance on animal testing.
- Advancements in microfluidics, 3D cell culture, stem cell engineering, and AI-integrated platforms are significantly improving predictive accuracy in drug development.
- Demand and Growth Drivers
- Rising demand for precision medicine and patient-specific therapies is accelerating adoption of disease-on-chip platforms.
- Increasing pharmaceutical R&D investments and need to reduce drug development costs and timelines are supporting market expansion.
- Growing regulatory pressure to minimize animal testing is further strengthening adoption.
- Expanding use in oncology and neurological disease modeling, particularly Alzheimer’s and cancer research, is driving demand.
- Strategic collaborations between pharmaceutical companies, CROs, and academic institutions are enhancing innovation and commercialization.
- Product and Segment View
- Devices (chips) and services collectively form the core market, with increasing demand for integrated platform-based solutions.
- Drug discovery & development accounts for 47.1% application share, making it the leading segment due to its role in early-stage validation and screening.
- Pharmaceutical & biotechnology companies hold 59.4% end-user share, reflecting strong adoption driven by high R&D spending.
- Single disease model chips dominate current adoption, while multi-organ integrated platforms are emerging for complex disease simulation.
- Technologies such as microfluidic platforms and biosensor-integrated systems are central to market advancement.
- Geography and Competitive Outlook
- North America remains the leading market, supported by strong R&D infrastructure and early regulatory adoption.
- Europe follows with policy-driven growth emphasizing reduction of animal testing under regulatory frameworks.
- East Asia and South Asia & Pacific are emerging as high-growth regions, driven by expanding biotech capabilities and cost-efficient research environments.
- Key companies operating in this market include Emulate Inc., Mimetas, CN Bio Innovations, TissUse GmbH, and InSphero, focusing on platform innovation, partnerships, and commercialization strategies.
Disease-on-Chip Platforms Market — At a Glance
| Attribute | Details |
|---|---|
| Market Value 2025 | USD 70.3 million |
| Market Value 2026 | USD 99.0 million |
| Market Value 2036 | USD 1,847.9 million |
| Absolute Dollar Opportunity | USD 1,748.9 million |
| Total Growth | 2,530% |
| CAGR | 34.00% |
| Growth Multiple | 18.7x |
| Key Demand Theme | Rising need for predictive, human-relevant preclinical models and alternatives to animal testing |
| Leading Application | Drug Discovery & Development |
| Application Share | 47.10% |
| Leading End User | Pharmaceutical & Biotechnology Companies |
| End-user Share | 59.40% |
| Key Growth Regions | North America, Europe, East Asia, South Asia & Pacific |
| Key Companies | Emulate, Mimetas, CN Bio, TissUse, InSphero |
| Segmentation by Technology | Microfluidic Platforms, 3D Cell Culture Platforms, Biosensor-Integrated Platforms, AI/Automation-Integrated Platforms |
| Segmentation by Platform | Single Disease Models, Multi-Disease Integrated Platforms |
| Segmentation by Material Type | PDMS-Based Chips, Glass-Based Chips, Polymer-Based Chips, Silicon-Based Chips, Hydrogel-Based Disease Platforms |
| Segmentation by Application | Drug Discovery, Toxicology, Personalized Medicine |
| Segmentation by End User | Pharma & Biotech, Academic Institutes, CROs, Regulatory Bodies |
| Segmentation by Region | North America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Latin America, MEA |
Where is the Real Growth Coming From?
Precision Medicine, Disease Modeling Demand, and Advanced Microfluidic Technologies Driving Market Expansion
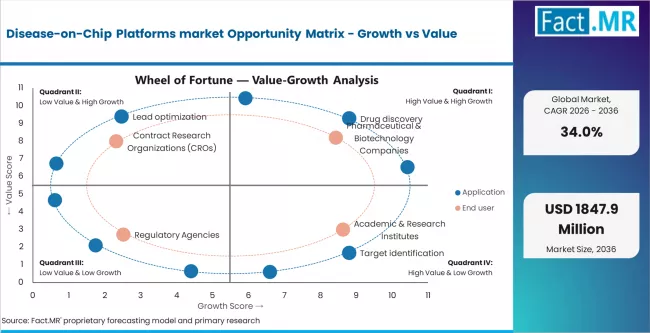
Growth in the Disease-on-Chip Platforms market is not driven purely by increasing adoption volumes, but rather by the rising need for more predictive, human-relevant preclinical models and a structural shift away from traditional animal testing. The market is witnessing accelerated expansion due to increasing demand for disease-specific modeling, particularly in high-burden therapeutic areas such as oncology and neurological disorders including Alzheimer’s disease.
The growing prevalence of chronic and complex diseases, coupled with rising R&D investments by pharmaceutical and biotechnology companies, is significantly driving demand for advanced in vitro platforms. Additionally, increasing pressure to reduce drug development costs and improve clinical success rates is encouraging the adoption of disease-on-chip systems for early-stage drug screening and validation. Expanding collaboration between industry players, academic institutions, and contract research organizations (CROs) is further strengthening the innovation ecosystem and accelerating market penetration.
According to industry analysis, technological advancements such as microfluidics, stem cell engineering, 3D cell culture systems, and biosensor integration are emerging as key growth enablers. These advanced platforms offer improved simulation of human disease physiology, enabling better prediction of drug efficacy and toxicity. Furthermore, the integration of AI and automation in disease modeling workflows is enhancing throughput and reproducibility, creating new opportunities for scalable adoption.
As the industry shifts toward precision medicine and patient-specific drug development, high-value applications such as personalized disease modeling and biomarker validation are gaining traction. These capabilities not only improve therapeutic outcomes but also support lifecycle management and targeted therapy development, positioning Disease-on-Chip platforms as a critical tool in next-generation drug discovery and development.
Which Application Segment Dominates the Disease-on-Chip Platforms Market?
Drug Discovery Leading Adoption Due to High Demand for Predictive Preclinical Models
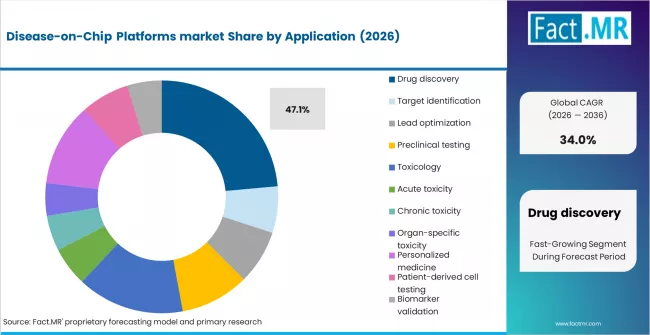
Drug discovery represents the largest application segment within the Disease-on-Chip Platforms market, accounting for approximately 47.1% of total market value in 2026. This dominance is primarily driven by the increasing need for more predictive, human-relevant preclinical testing models, which can significantly improve drug success rates and reduce late-stage failures.
Within drug discovery, sub-segments such as target identification (12.9%), lead optimization (15.1%), and preclinical testing (19.1%) are witnessing strong adoption as pharmaceutical companies increasingly integrate disease-on-chip platforms into early-stage R&D workflows. These platforms enable better simulation of human disease conditions, allowing for more accurate evaluation of drug efficacy and safety before clinical trials.
The growing complexity of diseases, particularly in oncology and neurological disorders, is further accelerating demand for advanced screening tools. Additionally, the need to reduce drug development costs and timelines, along with regulatory pressure to minimize animal testing, is reinforcing the role of disease-on-chip systems in drug discovery pipelines.
As pharmaceutical companies continue to prioritize efficiency and precision in R&D, drug discovery is expected to maintain its leadership position, serving as the primary revenue-generating application segment throughout the forecast period.
Which End-User Segment Dominates the Disease-on-Chip Platforms Market?
Pharmaceutical & Biotechnology Companies Leading Due to High R&D Investments and Early Technology Adoption
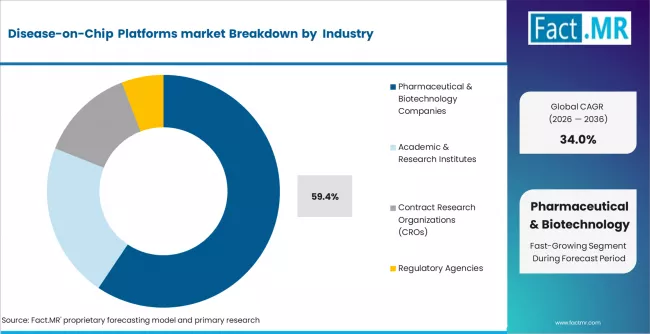
Pharmaceutical & biotechnology companies represent the largest end-user segment in the Disease-on-Chip Platforms market, accounting for approximately 59.4% of total market value in 2026. Their dominance is driven by substantial investments in drug discovery and development, along with the increasing adoption of advanced in vitro models to enhance preclinical research outcomes.
These companies are at the forefront of integrating disease-on-chip platforms into their R&D pipelines to improve predictive accuracy, reduce dependency on animal models, and accelerate time-to-market for new therapeutics. The growing focus on complex and chronic diseases has further intensified the need for more sophisticated disease modeling systems Academic & research institutes, accounting for 21.6% share, play a critical role in early-stage innovation and technology development, often collaborating with industry players. Meanwhile, contract research organizations (CROs), with a 13.2% share, are benefiting from increased outsourcing trends, as pharmaceutical companies seek cost-efficient and scalable research solutions.
Regulatory agencies, although holding a smaller share (5.8%), are gradually increasing their involvement in validating and standardizing these platforms for broader adoption.
With continued growth in pharmaceutical R&D spending and increasing reliance on advanced preclinical technologies, pharmaceutical & biotechnology companies are expected to remain the dominant end-user segment over the forecast period.
Cross-Sectional Analysis – Disease-on-Chip Platforms Market
Fact.MR analysis indicates that the Disease-On-Chip Platforms market is shaped by a strong interplay of demand-side drivers, supply-side innovation, and ecosystem-level collaboration. On the demand side, the rising prevalence of complex and chronic diseases, particularly in oncology and neurological conditions such as Alzheimer’s disease, is accelerating the need for more predictive and human-relevant preclinical models. Pharmaceutical and biotechnology companies are increasingly adopting disease-on-chip platforms to improve drug development efficiency, reduce late-stage failures, and minimize reliance on animal testing, thereby creating sustained demand across global markets.
From a supply-side perspective, innovation is highly concentrated in North America and Europe, supported by advanced research infrastructure, strong funding environments, and early technology adoption. These regions lead in the development of high-value microfluidic, stem cell-based, and biosensor-integrated platforms. Meanwhile, Asia-Pacific is emerging as a key growth region, driven by expanding biotechnology capabilities, cost-efficient research environments, and increasing participation in global R&D activities.
At the ecosystem level, the market operates through a collaborative network involving pharmaceutical companies, academic institutions, and contract research organizations (CROs). Pharmaceutical firms drive commercialization and demand, while academic institutions contribute to early-stage innovation, and CROs enable scalability through outsourced research services. Strategic partnerships, co-development agreements, and technology licensing are increasingly shaping market expansion and competitive positioning.
Fact.MR analysis further highlights that the integration of advanced technologies, including microfluidics, 3D cell culture systems, stem cell engineering, and AI-enabled analytics, is enhancing platform accuracy and scalability. However, challenges related to standardization, regulatory validation, and large-scale adoption remain key considerations.
Overall, the Disease-on-Chip Platforms market reflects a highly interconnected and innovation-driven structure, where demand growth, technological advancement, and collaborative ecosystems must align to ensure long-term scalability and market maturity.
ROI Analysis – Disease-on-Chip Platforms vs Traditional Animal Testing
Cost Efficiency, Accelerated Timelines, and Improved Clinical Predictability Driving Superior R&D Returns
Fact.MR analysis indicates that the adoption of Disease-On-Chip platforms is emerging as a high-impact value driver in pharmaceutical R&D, offering measurable advantages over traditional animal-based preclinical models across cost, time, and clinical success parameters.
From a cost perspective, traditional animal-based preclinical studies typically require investments in the range of US$ 3–6 million per program, driven by animal procurement, maintenance, and extended study durations. In comparison, Disease-On-Chip platforms reduce preclinical costs to approximately US$ 1–2.5 million per program, resulting in cost savings of nearly 40–60%. These savings are further amplified by higher throughput capabilities and reduced experimental redundancy.
In terms of development timelines, animal-based preclinical testing generally spans 18–24 months, whereas chip-based platforms can compress this timeline to 9–15 months, enabling a time advantage of approximately 6–9 months. This acceleration directly translates into earlier clinical trial initiation and faster market entry, significantly enhancing the net present value (NPV) of drug assets, particularly in high-value therapeutic areas.
Fact.MR analysis further highlights that Disease-On-Chip platforms improve the predictive accuracy of preclinical testing, thereby reducing downstream clinical failure rates. Traditional models are associated with Phase II failure rates of approximately 60–70% and Phase III failure rates of 30–40%, largely due to limited human relevance. In contrast, Disease-on-Chip systems are estimated to reduce failure rates by 15–25%, driven by improved simulation of human disease biology and more accurate toxicity profiling. Strategic Implications
Fact.MR analysis suggests that Disease-on-Chip platforms are transitioning from a niche research tool to a strategic R&D enabler for pharmaceutical companies. Their ability to improve decision-making quality in early-stage development significantly enhances overall pipeline productivity.
At a portfolio level, even a modest improvement of 5–10% in clinical success rates can translate into substantial financial upside, particularly for companies managing large and diversified drug pipelines. Additionally, the ability to accelerate development timelines provides a critical competitive advantage in high-value therapeutic areas, where early market entry can yield significant revenue gains.
Furthermore, the integration of Disease-on-Chip platforms supports the advancement of precision medicine strategies, enabling patient-specific testing and more targeted clinical trial designs. Combined with increasing regulatory support for alternatives to animal testing, these platforms are expected to play a central role in the future of drug development.
Global Regulatory Landscape – Disease-on-Chip Platforms
| Region / Authority | Acceptance Level | Approved / Accepted Use Cases | Key Limitations | Strategic Impact for Market Entry |
|---|---|---|---|---|
| US (FDA) | Moderate → High (Fastest evolving) | Toxicology screening, disease modeling, mechanistic studies, supportive preclinical data | Not yet accepted as standalone replacement for animal testing in approvals | Best entry market – early adoption, pilot validation, and regulatory engagement (e.g., FDA alternative methods programs) |
| Europe (EMA) | Moderate (Policy-driven) | Supplementary data under 3Rs framework, exploratory research support | Limited use as primary decision-making data; slower validation processes | Compliance-driven entry – strong ESG/animal reduction positioning but slower commercialization |
| OECD (Global Framework) | Emerging (Standardization role) | Validation of New Approach Methodologies (NAMs), data harmonization frameworks | No direct approval authority; requires multi-country validation | Critical for scaling – enables global acceptance via standardization (Mutual Acceptance of Data) |
| China | Low → Emerging (Rapidly developing) | Early-stage drug discovery, pilot research programs | Lack of formal regulatory pathways, limited harmonization with global standards | High growth opportunity – invest early, but expect regulatory uncertainty |
| Japan | Emerging (Innovation-focused) | Preclinical research, translational studies, limited regulatory pilots | Fragmented guidelines, slow integration into approval frameworks | Strategic partnership market – strong R&D ecosystem but gradual regulatory adoption |
Comparative Analysis – Disease-on-Chip vs Organ-on-a-Chip vs Organoid Models
Differentiation Across Predictive Accuracy, Cost Efficiency, and Development Stage Relevance
Fact.MR analysis indicates that Disease-on-Chip platforms, Organ-on-Chip systems, and organoid models serve distinct but complementary roles across the drug development value chain, differentiated primarily by predictive accuracy, scalability, and application depth.
| Parameter | Disease-on-Chip Platforms | Organ-on-Chip | Organoid Models |
|---|---|---|---|
| Accuracy (Human Relevance) | Very high - replicates disease-specific microenvironments and pathological responses | High - models healthy organ physiology with controlled dynamics | Moderate to high - strong cellular architecture but limited systemic interaction |
| Cost Structure | Moderate - higher than organoids due to system complexity, lower than full animal studies | Moderate to high - requires specialized devices and integration systems | Low to moderate - cost-efficient cell culture systems |
| Scalability | Moderate - improving with automation but constrained by complexity | Moderate - scalable with standardized platforms but infrastructure-dependent | High - suitable for large-scale screening and parallel testing |
| System Complexity | High - integrates disease pathways, immune responses, and multi-factor interactions | High - simulates organ-level function with microfluidic flow | Low to moderate - lacks vascularization and dynamic flow systems |
| Primary Use Cases | Disease modeling, drug efficacy validation, personalized medicine | Toxicology testing, pharmacokinetics (ADME), organ interaction studies | Target identification, early-stage screening, developmental biology |
Strategic Technology Positioning
Fact.MR analysis suggests that Disease-on-Chip platforms offer the highest predictive accuracy for clinical outcomes, as they replicate disease-specific conditions, including tumor microenvironments and neurodegenerative pathways such as Alzheimer’s disease. These platforms are increasingly used in late-stage preclinical validation and precision medicine applications, where decision accuracy is critical.
Organ-On-Chip systems, in contrast, are primarily optimized for physiological modeling and safety assessment, making them highly relevant for toxicology and pharmacokinetic studies. Their ability to simulate dynamic biological processes positions them as a translational bridge between traditional in vitro and in vivo models, with relatively higher regulatory familiarity.
Organoid models provide a cost-efficient and scalable solution for early-stage research, particularly in target identification and high-throughput screening. However, their lack of systemic integration and limited ability to simulate dynamic biological environments restrict their standalone predictive value in later stages of drug development.
Real-World Use Cases – Disease-on-Chip Platforms
Driving Predictive Accuracy, De-Risking Clinical Development, and Enabling Precision Therapeutics
Fact.MR analysis indicates that Disease-on-Chip platforms are increasingly being integrated into pharmaceutical R&D workflows where traditional preclinical models fail to provide reliable human-relevant insights. Their adoption is primarily concentrated in areas where decision accuracy directly influences downstream clinical outcomes and overall portfolio performance.
In oncology drug development, pharmaceutical companies face significant challenges in translating preclinical success into clinical efficacy due to the inability of conventional models to capture tumor heterogeneity and microenvironment complexity. Disease-on-Chip platforms address this limitation by enabling realistic simulation of tumor–immune interactions and disease progression under controlled conditions. This allows researchers to identify efficacy gaps, resistance mechanisms, and drug penetration limitations at an early stage. As a result, companies can refine or deprioritize candidates before entering costly clinical trials, thereby reducing late-stage attrition and improving pipeline efficiency. The financial implications are substantial, as avoiding a single failed oncology program can result in savings exceeding tens of millions of dollars while also accelerating time-to-market for viable candidates.
In neurodegenerative disease research, particularly in conditions such as Alzheimer’s disease, the lack of predictive animal models has historically led to high failure rates in clinical trials. Disease-on-Chip systems enable the replication of human neuronal networks, disease progression pathways, and blood–brain barrier interactions, offering a more physiologically relevant testing environment. This allows for early evaluation of drug delivery challenges and therapeutic efficacy, leading to more informed go/no-go decisions. By improving translational accuracy, these platforms help reduce mid-stage clinical failures and enable more efficient allocation of R&D resources, which is critical in high-risk therapeutic areas.
In the context of personalized medicine, Disease-on-Chip platforms are transforming the way therapies are developed and administered by enabling patient-specific disease modeling. Using cells derived from individual patients, these platforms allow for rapid testing of multiple treatment options under conditions that closely mimic the patient’s biological environment. This facilitates the identification of the most effective therapy for each individual, supporting biomarker-driven treatment strategies and improving clinical outcomes. From a commercial perspective, this capability enables the development of high-value, personalized therapeutic offerings, creating new revenue streams and strengthening competitive positioning in precision medicine markets.
Overall, Disease-on-Chip platforms are emerging as a critical enabler of next-generation drug development by improving early-stage decision-making, reducing clinical risk, and supporting the transition toward more targeted and efficient therapeutic strategies.
Regional Analysis
The report on the global Disease-on-Chip Platforms Market categorizes key regions, including South Asia & Pacific, Middle East & Africa, North America, Latin America, Europe, and East Asia. It provides geographic segmentation where market dynamics vary based on factors such as research and development intensity, regulatory acceptance of alternative testing models, biotechnology infrastructure, funding availability, and adoption of advanced in vitro technologies.
The analysis further offers a comprehensive assessment of regional market attractiveness over the forecast period using parameters such as innovation capacity, pharmaceutical R&D expenditure, regulatory support for non-animal testing, collaboration ecosystems, and technological adoption rates. Regional demand is influenced by differences in disease burden, healthcare investment, availability of skilled research talent, and integration of precision medicine approaches, which collectively shape the growth trajectory of the global Disease-on-Chip Platforms market.
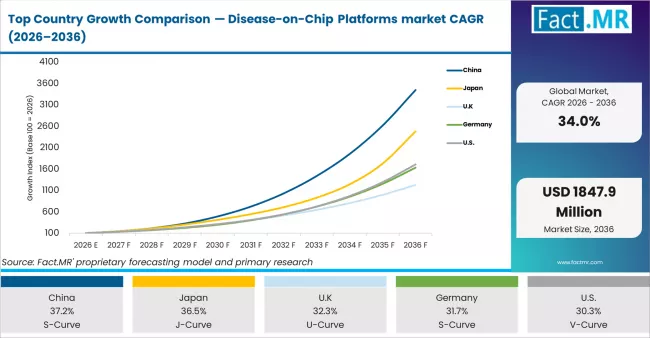
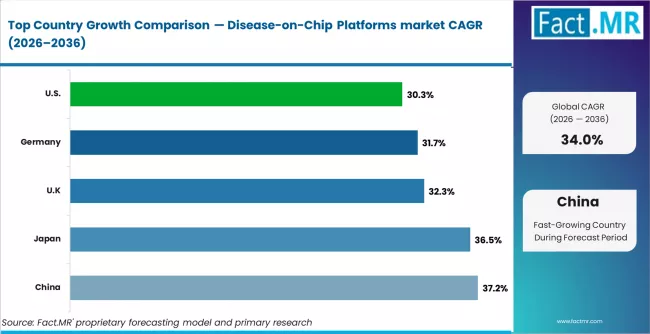
CAGR Table of Disease-on-Chip Platforms Market
| Country | CAGR (%) |
|---|---|
| China | 37.2% |
| Japan | 36.5% |
| UK | 32.3% |
| Germany | 31.7% |
| U.S. | 30.3% |
Source: Fact MR (FMR) analysis, based on proprietary forecasting model and primary research.
South Asia & Pacific
South Asia & Pacific is emerging as a high-growth region, driven by expanding biotechnology capabilities, increasing research outsourcing, and cost-efficient innovation environments.
- India: Growth is supported by rising pharmaceutical R&D activities, expanding CRO presence, and increasing adoption of advanced preclinical technologies.
- Southeast Asia & Australia: Demand is driven by improving research infrastructure, growing investment in life sciences, and increasing focus on precision medicine.
Middle East & Africa
The Middle East & Africa represents a developing, high-potential market, supported by gradual improvements in healthcare and research infrastructure.
- Growth is driven by increasing government investment in healthcare and biotechnology, along with rising interest in advanced research methodologies.
- Adoption remains at an early stage but is expected to increase with expanding academic and clinical research capabilities.
North America
North America remains the largest and most innovation-driven market, characterized by strong regulatory support and advanced research ecosystems.
- United States: Growth is driven by high pharmaceutical R&D spending, early adoption of Disease-on-Chip technologies, and supportive regulatory initiatives promoting alternatives to animal testing.
- Canada: Demand is supported by strong academic research and increasing collaboration with biotechnology firms.
Latin America
Latin America represents a moderate-growth market, supported by improving healthcare access and increasing participation in clinical research.
- Brazil & Mexico: Growth is driven by expanding pharmaceutical sectors, rising research collaborations, and gradual adoption of advanced preclinical technologies.
- Increasing focus on innovation and regulatory alignment is expected to support future market expansion.
Europe
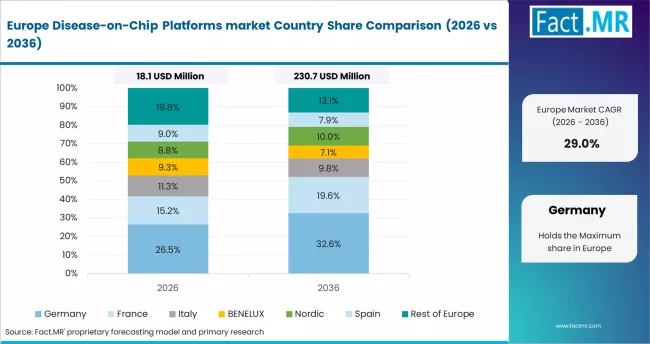
Europe is a mature and policy-driven market, supported by strong regulatory frameworks and emphasis on reducing animal testing.
- Germany, UK, France: Growth is driven by advanced research infrastructure, strong academic-industry collaboration, and regulatory support under the 3Rs framework.
- Adoption is supported by innovation funding and increasing focus on precision medicine and translational research.
East Asia
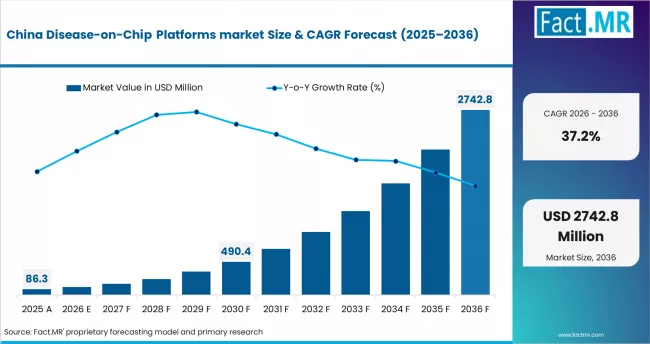
East Asia is a fast-growing and high-potential region, supported by strong manufacturing capabilities and increasing biotechnology investment.
- China: Growth is driven by expanding biotech industry, government support, and increasing R&D investment in advanced platforms.
- Japan: Demand is supported by strong research capabilities, aging population-driven healthcare needs, and focus on innovative drug development approaches.
Fact.MR’s analysis of the Disease-on-Chip Platforms Market across South Asia & Pacific, Middle East & Africa, North America, Latin America, Europe, and East Asia includes country-level assessments covering India, China, Japan, the United States, Germany, and other key markets. The report evaluates critical factors such as regulatory adoption, R&D intensity, technology infrastructure, and collaboration ecosystems across regions.
It further examines evolving industry trends, including the increasing shift toward non-animal testing methodologies, integration of AI and advanced cell-based technologies, and growing collaboration between pharmaceutical companies, academic institutions, and CROs. Additionally, the study analyzes cost structures, funding patterns, innovation pipelines, and global research networks, which collectively shape the long-term growth trajectory of the Disease-on-Chip Platforms market across key regions.
Competitive Landscape
How Are Key Players Competing in the Disease-on-Chip Platforms Market?
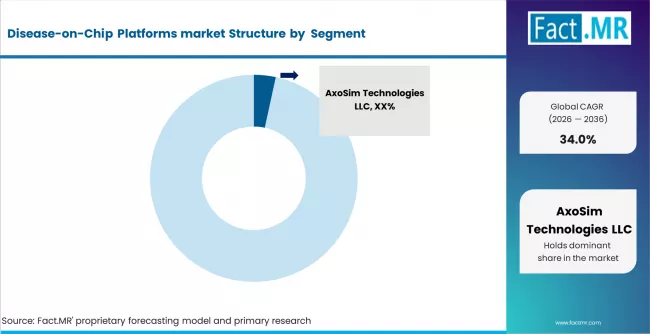
The Disease-on-Chip platforms market is led by specialized biotechnology and micro physiological systems companies such as Emulate, Inc., MIMETAS B.V., InSphero AG, CN Bio Innovations Ltd., TissUse GmbH, and Hesperos, Inc., alongside a growing base of emerging startups and niche technology providers.
These players are increasingly focused on platform innovation, strategic collaborations, and commercialization partnerships with pharmaceutical and biotechnology companies, strengthening their presence across drug discovery and preclinical research workflows. The market remains highly fragmented, with competition driven more by technological differentiation and scientific validation than scale alone.
A key competitive trend is the expansion of end-to-end solutions, where companies offer integrated capabilities spanning disease modeling, assay development, validation, and data analytics, often in collaboration with CROs and pharma clients. Strategic partnerships and co-development agreements are critical, enabling companies to embed their platforms into drug development pipelines.
Product innovation is centered around advanced disease modeling capabilities, including multi-organ systems, patient-derived models, and AI-integrated platforms, enabling more accurate simulation of human pathophysiology. Companies are investing heavily in organ-specific chips (liver, lung, brain) and disease-specific models (oncology, neurodegenerative disorders) to improve predictive accuracy and translational outcomes.
Market participants are also expanding their technology capabilities and application scope, focusing on areas such as toxicology testing, personalized medicine, and biomarker validation, which are emerging as high-growth segments. Additionally, increasing emphasis is placed on standardization, reproducibility, and regulatory acceptance, particularly for use in safety testing and potential replacement of animal models.
Key platform attributes include physiological relevance, scalability, reproducibility, compatibility with human cells, and integration with analytical systems, along with ease of use and adaptability across multiple therapeutic areas.
Competitive differentiation is driven by technological sophistication, validation through pharma partnerships, breadth of application, and ability to deliver reliable, human-relevant data, rather than pricing alone. Companies that successfully combine robust platform technology, strong industry collaborations, and scalable commercial models are best positioned to capture market share and drive long-term growth in the Disease-on-Chip platform market.
Recent Industry Developments
- Quantum Brilliance – Consortium / Technology Development (2025)
- Quantum Brilliance joined a consortium to develop a quantum-enabled brain-on-chip platform aimed at advancing neurological drug discovery and screening. The initiative integrates quantum computing with organ-on-chip systems to enhance complex brain disease modeling and predictive analytics. (Quantum Brilliance)
- CN Bio Innovations Ltd. – Strategic Partnership / Technology Expansion (April 2025)
- CN Bio partnered with Pharmaron to advance organ-on-chip (OOC) technologies globally, focusing on integrating PhysioMimix platforms into drug discovery and development workflows. The collaboration strengthens global R&D capabilities and expands adoption of human-relevant disease models. (CN Bio Innovations Ltd.)
- CN Bio Innovations Ltd. – Product Launch / Platform Innovation (October 2025)
- CN Bio launched PhysioMimix® Core, an advanced organ-on-chip system designed to support single-organ and multi-organ disease modeling. The platform enhances scalability and usability for drug discovery, toxicology, and preclinical testing applications. (CN Bio Innovations Ltd.)
- Emulate, Inc. – Product Launch / Technology Innovation (September 2024)
- Emulate introduced the Chip-R1™ Rigid Chip, a next-generation organ-on-chip platform with a minimally drug-absorbing profile. The innovation improves accuracy in ADME and toxicology studies, enabling more predictive biological modeling. (Emulate, Inc.)
- Emulate, Inc. – Product Launch & Strategic Partnership (November 2025)
- Emulate launched Brain-Chip R1 in partnership with FUJIFILM Cellular Dynamics to advance neurological disease modeling and drug development. The platform leverages human iPSC-derived cells to improve predictive accuracy in neurodegenerative research. (Emulate, Inc.)
- MIMETAS – Drug Development Collaboration / Regulatory Support (July 2024)
- MIMETAS contributed human organ-on-chip data to an Investigational New Drug (IND) application by argenx, demonstrating the use of its OrganoPlate platform in regulatory drug development. This highlights growing acceptance of organ-on-chip systems in preclinical and regulatory workflows. (MIMETAS B.V.)
Key Players of the Disease-on-Chip Platforms market
- Emulate Inc.
- Mimetas
- CN Bio Innovations
- TissUse GmbH
- InSphero
- Hesperos
- AxoSim Technologies
- Altis Biosystems
- Nortis
- BiomimX
- NETRI
- AlveoliX AG
- Tara Biosystems
- Organovo Holdings
Research Methodology – Disease-On-Chip Platforms Market
Fact.MR analysis indicates that the study of the Disease-On-Chip Platforms market is built on a combination of secondary research, primary validation, and analytical modeling to ensure high accuracy and strategic relevance. Secondary research forms the foundation of the study, leveraging sources such as company annual reports, investor presentations, scientific literature, regulatory publications, industry white papers, and proprietary databases. Key parameters including market size, technology landscape (microfluidics, stem cell technologies, 3D cell culture, biosensor-integrated systems), application areas, and regional adoption trends are cross-verified across multiple sources to maintain data consistency.
Market sizing is conducted using a demand-led approach, wherein data on active pharmaceutical R&D pipelines, number of preclinical programs, and adoption rates of advanced in vitro models are aggregated at global and regional levels. This is further validated through a top-down approach, incorporating macro indicators such as pharmaceutical R&D expenditure, biotechnology funding trends, regulatory acceptance of non-animal testing methodologies, and the expansion of precision medicine. Historical trends are analyzed to project future growth, with forecasting models integrating factors such as increasing demand for alternatives to animal testing, technological advancements, and growing application of disease-specific models in areas such as oncology and neurological disorders including Alzheimer’s disease.
To ensure robustness, data triangulation is applied by integrating supply-side insights—such as platform development capabilities, innovation pipelines, and commercialization strategies—with demand-side dynamics including research intensity, disease burden, and adoption patterns across pharmaceutical companies, academic institutions, and CROs. Statistical tools and forecasting techniques are used to refine estimates and align projections with market realities.
Primary research forms a critical component of the methodology. Between 2025 and 2026, extensive interviews were conducted across the value chain to validate assumptions and address data gaps. Respondents included platform developers and technology providers, pharmaceutical and biotechnology companies, academic and research institutions, contract research organizations (CROs), and industry experts across North America, Europe, East Asia, South Asia & Pacific, and the Middle East & Africa. Discussions focused on pricing dynamics, adoption trends, platform capabilities, regulatory acceptance, and emerging use cases. Insights gathered from these interactions were used to validate market estimates and strengthen forecast accuracy.
The final dataset is validated through a multi-layered triangulation process combining expert inputs, supply chain insights, and recalibration of key variables such as adoption rates, pricing structures, utilization levels, and regional dynamics. Fact.MR analysis concludes that this integrated methodology ensures a high degree of precision, reliability, and decision-making value, enabling stakeholders to effectively assess market opportunities, optimize investment strategies, and plan long-term growth in the Disease-on-Chip Platforms market.
Strategic Decision Analysis – Disease-on-Chip Platforms Market
Fact.MR analysis indicates that a leading pharmaceutical company undertook a strategic evaluation to integrate Disease-on-Chip platforms into its R&D framework with the objective of improving clinical success rates, reducing development timelines, and optimizing preclinical costs. The company was facing challenges related to low translational accuracy of traditional animal models, high attrition in mid- and late-stage clinical trials, and increasing pressure to adopt more predictive and human-relevant testing approaches.
The study conducted by Fact.MR began with a comprehensive assessment of the company’s global drug development pipeline, with particular focus on high-risk therapeutic areas such as oncology and neurological disorders including Alzheimer’s disease. Analysis of historical clinical performance revealed that a significant proportion of failures were linked to inadequate preclinical models, highlighting the need for more advanced disease-specific platforms. Using a bottom-up approach, the potential impact of Disease-on-Chip adoption was quantified across different stages of the development cycle, particularly in target validation, lead optimization, and preclinical testing.
On the supply side, Fact.MR evaluated leading platform providers based on technological capabilities, scalability, integration potential, and regulatory readiness. This enabled the identification of suitable partners and benchmarking of available solutions against the company’s R&D requirements. A detailed techno-commercial model was developed to assess cost savings from reduced reliance on animal studies, improvements in time-to-market, and the financial impact of lowering late-stage clinical failures.
The analysis demonstrated that integrating Disease-on-Chip platforms could significantly enhance decision accuracy in early-stage development, thereby reducing the likelihood of costly failures in later phases. Scenario modeling further indicated that even modest improvements in clinical success rates could translate into substantial financial gains at the portfolio level, while accelerated development timelines would improve asset value and competitive positioning.
Based on these findings, Fact.MR recommended a phased adoption strategy, prioritizing the use of Disease-on-Chip platforms in high-risk and high-value programs while leveraging external partnerships to ensure flexibility and cost efficiency. Sensitivity analysis across key variables, including adoption rates, platform costs, and regulatory acceptance, confirmed the robustness of this approach under varying market conditions.
Fact.MR concludes that the implementation of a structured, hybrid adoption model enabled the client to align technological innovation with strategic R&D objectives, resulting in improved pipeline productivity, reduced development risk, and enhanced long-term value creation in the evolving Disease-on-Chip Platforms market.
Bibliographies
- Quantum Brilliance joins consortium to develop a quantum-enabled brain-on-chip platform for neurological drug discovery and screening [1]]
- CN Bio Innovations Ltd. establishes strategic partnership with Pharmaron to expand global organ-on-chip R&D capabilities [2]
- CN Bio Innovations Ltd. launches PhysioMimix® Core, an advanced organ-on-chip system for disease modeling and preclinical applications [3]]
- Emulate Inc. unveils Chip-R1™ Rigid Chip to enhance predictive accuracy in ADME and toxicology studies [4]
- Emulate Inc. launches Brain-Chip R1 in partnership with FUJIFILM Cellular Dynamics to advance neurological drug development [5]
- MIMETAS contributes organ-on-chip data to an IND application by argenx, supporting regulatory adoption of advanced in vitro models [6]
Disease-on-Chip Platforms Market Definition
Disease-on-Chip platforms are advanced microfluidic in vitro systems that replicate human disease conditions using living cells within biomimetic environments. These platforms simulate disease-specific physiological and pathological processes, enabling more predictive and human-relevant insights than traditional cell culture and animal models.
They integrate human primary cells, stem cells (iPSCs), and organoids with engineered microenvironments that mimic tissue interactions, fluid dynamics, and mechanical cues. The market includes various configurations such as single-organ disease models, multi-organ systems, and patient-specific platforms, along with technologies like microfluidic chips, 3D culture systems, and analytical tools.
These platforms are widely used in drug discovery, preclinical testing, toxicity assessment, and personalized medicine, including patient-derived cell testing and biomarker validation. The market also encompasses devices and services, such as assay development and contract research, ensuring scalability, reproducibility, and regulatory compliance.
Disease-on-Chip platforms are adopted across pharmaceutical & biotechnology companies, academic institutes, CROs, and regulatory bodies, driven by the need for accurate disease modeling, reduced animal testing, and improved drug development efficiency.
Disease-on-Chip Platforms Market Inclusions
The report covers global and regional market size estimates and forecasts for the Disease-on-Chip Platforms Market from 2026 to 2036, including:
- Segment Analysis: Based on Product Type (Devices (Chips), Services), Platform Type (Single Disease Models, Multi-Disease Integrated Platforms), Organ Type (Liver, Lung, Heart, Brain, Kidney, Others), Technology (Microfluidics, Stem Cell Technologies, 3D Cell Culture, Biosensor-Integrated Systems), Therapeutic Area (Oncology, Neurology including Alzheimer’s disease, Infectious Diseases, Cardiovascular Diseases, Respiratory Diseases, Metabolic Disorders), Application (Drug Discovery & Development, Toxicology Testing, Disease Modeling, Personalized Medicine), End User (Pharmaceutical & Biotechnology Companies, Academic & Research Institutes, Contract Research Organizations (CROs), Regulatory Agencies, Hospitals & Clinical Laboratories), and Region (North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa).
- Regional Play: Analysis of demand variations across North America, Europe, East Asia, South Asia & Pacific, Latin America, and Middle East & Africa, considering factors such as R&D investment intensity, regulatory adoption of alternative testing models, biotechnology infrastructure, and availability of skilled research capabilities.
- Pricing and Cost Analysis: Assessment of platform pricing trends, cost components including chip fabrication, microfluidic system integration, cell sourcing (including stem cells), assay development, and operational costs, along with evaluation of service-based revenue models and regional cost differentials.
- Technology and Innovation Landscape: Insights into advancements in microfluidics, induced pluripotent stem cells (iPSCs), 3D cell culture systems, biosensor integration, and AI-enabled disease modeling platforms, supporting improved predictive accuracy, scalability, and automation in preclinical research.
- Market Trends and Dynamics: Analysis of key growth drivers and opportunities, including increasing demand for alternatives to animal testing, rising adoption of precision medicine, growing pharmaceutical R&D investments, and expanding use of disease-specific models in oncology and neurological research, along with challenges related to standardization and regulatory validation.
- Competitive Landscape: Coverage of key market participants such as Emulate Inc., Mimetas, TissUse GmbH, CN Bio Innovations, and InSphero, with insights into technological capabilities, strategic collaborations, product innovation, and commercialization strategies.
Disease-on-Chip Platforms Market Exclusions
The scope excludes:
- Traditional in vivo animal testing models: Conventional animal-based preclinical studies and related services are excluded, except where direct comparisons with Disease-on-Chip platforms are relevant for benchmarking or validation purposes.
- Standalone Organ-on-a-Chip platforms (non-disease-specific): General organ-on-chip systems used solely for physiological or toxicity studies without disease modeling applications are excluded unless integrated into disease-specific platforms.
- Basic 2D and non-advanced 3D cell culture systems: Standard cell culture models and simple in vitro assays that do not replicate disease-specific microenvironments or dynamic biological interactions are excluded.
- Standalone microfluidic components and materials: Individual components such as microfluidic chips, polymers, or fabrication materials are excluded unless part of a complete Disease-on-Chip platform solution.
- Drug discovery services (standalone CRO activities): Contract research services related to general drug discovery, clinical trials, and preclinical testing are excluded unless directly linked to Disease-on-Chip platform utilization.
- Medical devices and diagnostic platforms: Devices used for diagnostics, imaging, or therapeutic delivery (e.g., implantable devices, biosensors not integrated into disease-on-chip systems) are excluded.
- Regenerative medicine and tissue engineering (standalone): Applications focused purely on tissue regeneration or transplantation without disease modeling functionality are excluded.
- Unregulated or experimental research applications: Informal research setups, academic prototypes without standardization, and non-commercial or unvalidated systems are excluded where data reliability and reproducibility cannot be ensured.
- Veterinary and animal health applications: Disease-on-Chip platforms developed exclusively for animal health research are excluded, with the scope limited to human disease modeling and pharmaceutical applications.
Disease-On-Chip Platforms Market Key Segments
-
By Platform Type:
- Single Disease Model Chips
- Cancer-on-Chip
- Neurodegenerative Disease-on-Chip
- Cardiovascular Disease-on-Chip
- Respiratory Disease-on-Chip
- Infectious Disease-on-Chip
- Metabolic Disorder-on-Chip
- Autoimmune Disease-on-Chip
- Multi-Disease Integrated Platforms
- Multi-Organ Disease Simulation Chips
- Systemic Disease Interaction Platforms
- Metastasis/Progression Modeling Platforms
- Single Disease Model Chips
-
By Technology:
- Microfluidic Platforms
- 3D Cell Culture Platforms
- Biosensor-Integrated Platforms
- AI/Automation-Integrated Platforms
-
By Material Type:
- PDMS-Based Chips
- Glass-Based Chips
- Polymer-Based Chips
- MMA
- COC/COP
- Silicon-Based Chips
- Hydrogel-Based Disease Platforms
-
By Application:
- Drug Discovery
- Target Identification
- Lead Optimization
- Preclinical Testing
- Toxicology
- Acute Toxicity
- Chronic Toxicity
- Organ-Specific Toxicity
- Personalized Medicine
- Patient-Derived Cell Testing
- Biomarker Validation
- Drug Discovery
-
By End-User:
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
- Contract Research Organizations (CROs)
- Regulatory Agencies
-
By Region:
- North America
- U.S.
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- Italy
- France
- U.K.
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- Eastern Europe
- Russia
- Hungary
- Poland
- Balkan & Baltic Countries
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Türkiye
- South Africa
- Rest of Africa
- Rest of Middle East & Africa
- North America
Table of Content
- Disease-On-Chip Platforms Market – Executive Summary
- Market Overview
- Market Definition & Introduction
- Market Taxonomy & Research Scope
- Global Disease-On-Chip Platforms Market Demand Analysis 2021-2025 and Forecast, 2026–2036
- Historical Market Volume (Units) Analysis, 2021-2025
- Current and Future Market Volume (Units) Projections, 2026–2036
- Y-o-Y Growth Trend Analysis
- Global Disease-On-Chip Platforms Market - Pricing Analysis
- Product Category and Country-Level Pricing Analysis
- Region Average Pricing Analysis Benchmark
- Price Trends and Forecasts
- Historical price trends
- Future price forecasts
- Factors Influencing Pricing
- Competitive Pricing Strategies
- Cost Structure Analysis: Elementary Deep Dive
- Global Disease-On-Chip Platforms Market Demand (in Value or Size in US$ Mn) Analysis 2021-2025 and Forecast, 2026–2036
- Historical Market Value (US$ Mn) Analysis, 2021-2025
- Current and Future Market Value (US$ Mn) Projections, 2026–2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Market Background and Foundational Data Points
- Global Adoption and Utilization Overview
- Total Platform Deployment (Units / Installed Base)
- Total Market Value (USD Million / Billion)
- Historical Growth of Advanced In Vitro Modeling Demand
- Regional Market Distribution
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
- Market by Product Type
- Single Disease Model Chips
- Multi-Disease Integrated Platforms
- Market by Technology Type
- Microfluidic Platforms
- 3D Cell Culture Platforms
- Biosensor-Integrated Platforms
- AI/Automation-Integrated Platforms
- Product Lifecycle and Replacement Dynamics
- Technology Validation and Regulatory Acceptance Trends
- Platform Upgradation and Integration Strategies
- Expansion of Disease-Specific and Multi-Organ Models
- Market Forecast (2025–2036)
- Net Market Growth
- Regional Expansion Trends
- Application-wise Growth Split (Drug Discovery vs Toxicology vs Personalized Medicine)
- Global Adoption and Utilization Overview
- Disease-On-Chip Platforms Market Analysis 2021-2025 and Forecast 2026–2036, By Product Type
- Introduction / Key Findings
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Product Type, 2021-2025
- Current and Future Market Size (US$ Mn) & Volume (Units) Analysis and Forecast by Product Type, 2026-2036
- Single Disease Model Chips
- Cancer-on-Chip
- Neurodegenerative Disease-on-Chip
- Cardiovascular Disease-on-Chip
- Respiratory Disease-on-Chip
- Infectious Disease-on-Chip
- Metabolic Disorder-on-Chip
- Autoimmune Disease-on-Chip
- Multi-Disease Integrated Platforms
- Multi-Organ Disease Simulation Chips
- Systemic Disease Interaction Platforms
- Metastasis/Progression Modeling Platforms
- Single Disease Model Chips
- Market Attractiveness Analysis by Product Type
- Disease-On-Chip Platforms Market Analysis 2021-2025 and Forecast 2026–2036, By Technology
- Introduction / Key Findings
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Technology, 2021-2025
- Current and Future Market Size (US$ Mn) & Volume (Units) Analysis and Forecast by Technology, 2026-2036
- Microfluidic Platforms
- 3D Cell Culture Platforms
- Biosensor-Integrated Platforms
- AI/Automation-Integrated Platforms
- Market Attractiveness Analysis by Technology
- Disease-On-Chip Platforms Market Analysis 2021-2025 and Forecast 2026–2036, By Material Type
- Introduction / Key Findings
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Material Type, 2021-2025
- Current and Future Market Size (US$ Mn) & Volume (Units) Analysis and Forecast by Material Type, 2026-2036
- PDMS-Based Chips
- Glass-Based Chips
- Polymer-Based Chips
- PMMA
- COC/COP
- Silicon-Based Chips
- Hydrogel-Based Disease Platforms
- Market Attractiveness Analysis by Material Type
- Disease-On-Chip Platforms Market Analysis 2021-2025 and Forecast 2026–2036, By Application
- Introduction / Key Findings
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Application, 2021-2025
- Current and Future Market Size (US$ Mn) & Volume (Units) Analysis and Forecast by Application, 2026-2036
- Drug discovery
- Target identification
- Lead optimization
- Preclinical testing
- Toxicology
- Acute toxicity
- Chronic toxicity
- Organ-specific toxicity
- Personalized medicine
- Patient-derived cell testing
- Biomarker validation
- Drug discovery
- Market Attractiveness Analysis by Application
- Disease-On-Chip Platforms Market Analysis 2021-2025 and Forecast 2026–2036, By End-User
- Introduction / Key Findings
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by End-User, 2021-2025
- Current and Future Market Size (US$ Mn) & Volume (Units) Analysis and Forecast by End-User, 2026-2036
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
- Contract Research Organizations (CROs)
- Regulatory Agencies
- Market Attractiveness Analysis by End-User
- Global Disease-On-Chip Platforms Market Analysis and Forecast, by Region
- Introduction / Key Findings
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Region, 2021-2025
- Current and Future Market Size (US$ Mn) & Volume (Units) Analysis and Forecast by Region, 2026-2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
- Market Attractiveness Analysis by Region
- North America Disease-On-Chip Platforms Market Analysis and Forecast
- Introduction / Key Findings
- Pricing Analysis
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Market Taxonomy, 2021-2025
- Current and Future Market Size (US$ Mn) and Volume (Units) Analysis & Forecast by Market Taxonomy, 2026-2036
- By Country
- United States
- Canada
- Mexico
- By Taxonomy
- By Platform Type
- By Technology
- By Material Type
- By Application
- By End-User
- Market Attractiveness Analysis by Country & Taxonomy
- Market Trends
- Key Market Participants – Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Latin America Disease-On-Chip Platforms Market Analysis and Forecast
- Introduction / Key Findings
- Pricing Analysis
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Market Taxonomy, 2021-2025
- Current and Future Market Size (US$ Mn) and Volume (Units) Analysis & Forecast by Market Taxonomy, 2026-2036
- By Country
- Brazil
- Chile
- Rest of LATAM
- By Taxonomy
- By Platform Type
- By Technology
- By Material Type
- By Application
- By End-User
- Market Attractiveness Analysis By Country & Taxonomy
- Market Trends
- Key Market Participants – Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Western Europe Disease-On-Chip Platforms Market Analysis and Forecast
- Introduction / Key Findings
- Pricing Analysis
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Market Taxonomy, 2021-2025
- Current and Future Market Size (US$ Mn) and Volume (Units) Analysis & Forecast by Market Taxonomy, 2026-2036
- By Country
- Germany
- Italy
- France
- United Kingdom
- Spain
- BENELUX
- Nordics
- Rest of Western Europe
- By Taxonomy
- By Platform Type
- By Technology
- By Material Type
- By Application
- By End-User
- Market Attractiveness Analysis By Country & Taxonomy
- Market Trends
- Key Market Participants – Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Eastern Europe Disease-On-Chip Platforms Market Analysis and Forecast
- Introduction / Key Findings
- Pricing Analysis
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Market Taxonomy, 2021-2025
- Current and Future Market Size (US$ Mn) and Volume (Units) Analysis & Forecast by Market Taxonomy, 2026-2036
- By Country
- Russia
- Hungary
- Poland
- Balkan & Baltics
- Rest of Eastern Europe
- By Taxonomy
- By Platform Type
- By Technology
- By Material Type
- By Application
- By End-User
- Market Attractiveness Analysis By Country & Taxonomy
- Market Trends
- Key Market Participants – Intensity Mapping
- Drivers and Restraints - Impact Analysis
- East Asia Disease-On-Chip Platforms Market Analysis and Forecast
- Introduction / Key Findings
- Pricing Analysis
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Market Taxonomy, 2021-2025
- Current and Future Market Size (US$ Mn) and Volume (Units) Analysis & Forecast by Market Taxonomy, 2026-2036
- By Country
- China
- Japan
- South Korea
- By Taxonomy
- By Platform Type
- By Technology
- By Material Type
- By Application
- By End-User
- Market Attractiveness Analysis by Country & Taxonomy
- Market Trends
- Key Market Participants – Intensity Mapping
- Drivers and Restraints - Impact Analysis
- South Asia & Pacific Disease-On-Chip Platforms Market Analysis and Forecast
- Introduction / Key Findings
- Pricing Analysis
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Market Taxonomy, 2021-2025
- Current and Future Market Size (US$ Mn) and Volume (Units) Analysis & Forecast by Market Taxonomy, 2026-2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia & Pacific
- By Taxonomy
- By Platform Type
- By Technology
- By Material Type
- By Application
- By End-User
- Market Attractiveness Analysis by Country & Taxonomy
- Market Trends
- Key Market Participants – Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Middle East & Africa Disease-On-Chip Platforms Market Analysis and Forecast
- Introduction / Key Findings
- Pricing Analysis
- Historical Market Size (US$ Mn) & Volume (Units) Trend Analysis by Market Taxonomy, 2021-2025
- Current and Future Market Size (US$ Mn) and Volume (Units) Analysis & Forecast by Market Taxonomy, 2026-2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- By Taxonomy
- By Platform Type
- By Technology
- By Material Type
- By Application
- By End-User
- Market Attractiveness Analysis By Country & Taxonomy
- Market Trends
- Key Market Participants – Intensity Mapping
- Drivers and Restraints - Impact Analysis
- Country-level Disease-On-Chip Platforms Market Analysis and Forecast
- Introduction
- Market Value Proportion Analysis, By Key Countries
- Global Vs. Country Growth Comparison
- United States Disease-On-Chip Platforms Market Analysis
- Market Size (US$ Mn) & Volume (Units) Analysis by Market Taxonomy, 2021–2036
- By Product Type
- By Technology
- By Material Type
- By Application
- By End-User
- United States Disease-On-Chip Platforms Market Analysis
- *Same Analysis will be providing for each of the countries mentioned below
-
- Canada Disease-On-Chip Platforms Market Analysis
- Mexico Disease-On-Chip Platforms Market Analysis
- Brazil Disease-On-Chip Platforms Market Analysis
- Chile Disease-On-Chip Platforms Market Analysis
- Germany Disease-On-Chip Platforms Market Analysis
- France Disease-On-Chip Platforms Market Analysis
- Italy Disease-On-Chip Platforms Market Analysis
- Spain Disease-On-Chip Platforms Market Analysis
- United Kingdom Disease-On-Chip Platforms Market Analysis
- BENELUX Disease-On-Chip Platforms Market Analysis
- Nordics Disease-On-Chip Platforms Market Analysis
- Poland Disease-On-Chip Platforms Market Analysis
- Russia Disease-On-Chip Platforms Market Analysis
- Hungary Disease-On-Chip Platforms Market Analysis
- Balkan & Baltics Disease-On-Chip Platforms Market Analysis
- China Disease-On-Chip Platforms Market Analysis
- Japan Disease-On-Chip Platforms Market Analysis
- South Korea Disease-On-Chip Platforms Market Analysis
- India Disease-On-Chip Platforms Market Analysis
- ASEAN Disease-On-Chip Platforms Market Analysis
- ANZ Disease-On-Chip Platforms Market Analysis
- KSA Disease-On-Chip Platforms Market Analysis
- Other GCC Countries Disease-On-Chip Platforms Market Analysis
- South Africa Disease-On-Chip Platforms Market Analysis
- Turkiye Disease-On-Chip Platforms Market Analysis
-
- Disease-On-Chip Platforms Market Structure Analysis
- Market Analysis by Tier of Companies
- Market Concentration
- Market Share Analysis of Top Players
- Market Space for New Brands and Dollar Opportunity
- Market Presence Analysis
- Regional Footprint of Players
- Product Footprint by Players
- Disease-On-Chip Platforms Competition Analysis
- Competition Dashboard
- Pricing Analysis by Competition
- Competition Benchmarking
- Competition Deep Dive
- Emulate Inc.
- Overview
- Product Portfolio
- Sales Footprint
- Key Developments
- SWOT Analysis
- Strategy Overview
- Key Financials
- Mimetas
- CN Bio Innovations
- TissUse GmbH
- InSphero
- Hesperos
- AxoSim Technologies
- Altis Biosystems
- Nortis
- BiomimX
- NETRI
- AlveoliX AG
- Tara Biosystems
- Organovo Holdings
- Other Prominent Players
- Emulate Inc.
- Primary insights
- Assumptions & Acronyms Used
- Research Methodology & Data Sources
List Of Table
- Table 1: Global Market Value (USD Mn) Forecast by Region, 2021 to 2036
- Table 2: Global Market Volume (Units) Forecast by Region, 2021 to 2036
- Table 3: Global Market Value (USD Mn) Forecast by Platform Type, 2021 to 2036
- Table 4: Global Market Volume (Units) Forecast by Platform Type, 2021 to 2036
- Table 5: Global Market Value (USD Mn) Forecast by Technology, 2021 to 2036
- Table 6: Global Market Volume (Units) Forecast by Technology, 2021 to 2036
- Table 7: Global Market Value (USD Mn) Forecast by Material Type, 2021 to 2036
- Table 8: Global Market Volume (Units) Forecast by Material Type, 2021 to 2036
- Table 9: Global Market Value (USD Mn) Forecast by Application, 2021 to 2036
- Table 10: Global Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 11: Global Market Value (USD Mn) Forecast by End-User, 2021 to 2036
- Table 12: Global Market Volume (Units) Forecast by End-User, 2021 to 2036
- Table 13: North America Market Value (USD Mn) Forecast by Country, 2021 to 2036
- Table 14: North America Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 15: North America Market Value (USD Mn) Forecast by Platform Type, 2021 to 2036
- Table 16: North America Market Volume (Units) Forecast by Platform Type, 2021 to 2036
- Table 17: North America Market Value (USD Mn) Forecast by Technology, 2021 to 2036
- Table 18: North America Market Volume (Units) Forecast by Technology, 2021 to 2036
- Table 19: North America Market Value (USD Mn) Forecast by Material Type, 2021 to 2036
- Table 20: North America Market Volume (Units) Forecast by Material Type, 2021 to 2036
- Table 21: North America Market Value (USD Mn) Forecast by Application, 2021 to 2036
- Table 22: North America Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 23: North America Market Value (USD Mn) Forecast by End-User, 2021 to 2036
- Table 24: North America Market Volume (Units) Forecast by End-User, 2021 to 2036
- Table 25: Latin America Market Value (USD Mn) Forecast by Country, 2021 to 2036
- Table 26: Latin America Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 27: Latin America Market Value (USD Mn) Forecast by Platform Type, 2021 to 2036
- Table 28: Latin America Market Volume (Units) Forecast by Platform Type, 2021 to 2036
- Table 29: Latin America Market Value (USD Mn) Forecast by Technology, 2021 to 2036
- Table 30: Latin America Market Volume (Units) Forecast by Technology, 2021 to 2036
- Table 31: Latin America Market Value (USD Mn) Forecast by Material Type, 2021 to 2036
- Table 32: Latin America Market Volume (Units) Forecast by Material Type, 2021 to 2036
- Table 33: Latin America Market Value (USD Mn) Forecast by Application, 2021 to 2036
- Table 34: Latin America Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 35: Latin America Market Value (USD Mn) Forecast by End-User, 2021 to 2036
- Table 36: Latin America Market Volume (Units) Forecast by End-User, 2021 to 2036
- Table 37: Western Europe Market Value (USD Mn) Forecast by Country, 2021 to 2036
- Table 38: Western Europe Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 39: Western Europe Market Value (USD Mn) Forecast by Platform Type, 2021 to 2036
- Table 40: Western Europe Market Volume (Units) Forecast by Platform Type, 2021 to 2036
- Table 41: Western Europe Market Value (USD Mn) Forecast by Technology, 2021 to 2036
- Table 42: Western Europe Market Volume (Units) Forecast by Technology, 2021 to 2036
- Table 43: Western Europe Market Value (USD Mn) Forecast by Material Type, 2021 to 2036
- Table 44: Western Europe Market Volume (Units) Forecast by Material Type, 2021 to 2036
- Table 45: Western Europe Market Value (USD Mn) Forecast by Application, 2021 to 2036
- Table 46: Western Europe Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 47: Western Europe Market Value (USD Mn) Forecast by End-User, 2021 to 2036
- Table 48: Western Europe Market Volume (Units) Forecast by End-User, 2021 to 2036
- Table 49: Eastern Europe Market Value (USD Mn) Forecast by Country, 2021 to 2036
- Table 50: Eastern Europe Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 51: Eastern Europe Market Value (USD Mn) Forecast by Platform Type, 2021 to 2036
- Table 52: Eastern Europe Market Volume (Units) Forecast by Platform Type, 2021 to 2036
- Table 53: Eastern Europe Market Value (USD Mn) Forecast by Technology, 2021 to 2036
- Table 54: Eastern Europe Market Volume (Units) Forecast by Technology, 2021 to 2036
- Table 55: Eastern Europe Market Value (USD Mn) Forecast by Material Type, 2021 to 2036
- Table 56: Eastern Europe Market Volume (Units) Forecast by Material Type, 2021 to 2036
- Table 57: Eastern Europe Market Value (USD Mn) Forecast by Application, 2021 to 2036
- Table 58: Eastern Europe Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 59: Eastern Europe Market Value (USD Mn) Forecast by End-User, 2021 to 2036
- Table 60: Eastern Europe Market Volume (Units) Forecast by End-User, 2021 to 2036
- Table 61: East Asia Market Value (USD Mn) Forecast by Country, 2021 to 2036
- Table 62: East Asia Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 63: East Asia Market Value (USD Mn) Forecast by Platform Type, 2021 to 2036
- Table 64: East Asia Market Volume (Units) Forecast by Platform Type, 2021 to 2036
- Table 65: East Asia Market Value (USD Mn) Forecast by Technology, 2021 to 2036
- Table 66: East Asia Market Volume (Units) Forecast by Technology, 2021 to 2036
- Table 67: East Asia Market Value (USD Mn) Forecast by Material Type, 2021 to 2036
- Table 68: East Asia Market Volume (Units) Forecast by Material Type, 2021 to 2036
- Table 69: East Asia Market Value (USD Mn) Forecast by Application, 2021 to 2036
- Table 70: East Asia Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 71: East Asia Market Value (USD Mn) Forecast by End-User, 2021 to 2036
- Table 72: East Asia Market Volume (Units) Forecast by End-User, 2021 to 2036
- Table 73: South Asia and Pacific Market Value (USD Mn) Forecast by Country, 2021 to 2036
- Table 74: South Asia and Pacific Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 75: South Asia and Pacific Market Value (USD Mn) Forecast by Platform Type, 2021 to 2036
- Table 76: South Asia and Pacific Market Volume (Units) Forecast by Platform Type, 2021 to 2036
- Table 77: South Asia and Pacific Market Value (USD Mn) Forecast by Technology, 2021 to 2036
- Table 78: South Asia and Pacific Market Volume (Units) Forecast by Technology, 2021 to 2036
- Table 79: South Asia and Pacific Market Value (USD Mn) Forecast by Material Type, 2021 to 2036
- Table 80: South Asia and Pacific Market Volume (Units) Forecast by Material Type, 2021 to 2036
- Table 81: South Asia and Pacific Market Value (USD Mn) Forecast by Application, 2021 to 2036
- Table 82: South Asia and Pacific Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 83: South Asia and Pacific Market Value (USD Mn) Forecast by End-User, 2021 to 2036
- Table 84: South Asia and Pacific Market Volume (Units) Forecast by End-User, 2021 to 2036
- Table 85: Middle East & Africa Market Value (USD Mn) Forecast by Country, 2021 to 2036
- Table 86: Middle East & Africa Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 87: Middle East & Africa Market Value (USD Mn) Forecast by Platform Type, 2021 to 2036
- Table 88: Middle East & Africa Market Volume (Units) Forecast by Platform Type, 2021 to 2036
- Table 89: Middle East & Africa Market Value (USD Mn) Forecast by Technology, 2021 to 2036
- Table 90: Middle East & Africa Market Volume (Units) Forecast by Technology, 2021 to 2036
- Table 91: Middle East & Africa Market Value (USD Mn) Forecast by Material Type, 2021 to 2036
- Table 92: Middle East & Africa Market Volume (Units) Forecast by Material Type, 2021 to 2036
- Table 93: Middle East & Africa Market Value (USD Mn) Forecast by Application, 2021 to 2036
- Table 94: Middle East & Africa Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 95: Middle East & Africa Market Value (USD Mn) Forecast by End-User, 2021 to 2036
- Table 96: Middle East & Africa Market Volume (Units) Forecast by End-User, 2021 to 2036
List Of Figures
- Figure 1: Global Market Volume (Units) Forecast 2021 to 2036
- Figure 2: Global Market Pricing Analysis
- Figure 3: Global Market Value (USD Mn) Forecast 2021 to 2036
- Figure 4: Global Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 5: Global Market Y-o-Y Growth Comparison by Platform Type, 2021 to 2036
- Figure 6: Global Market Attractiveness Analysis by Platform Type
- Figure 7: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 8: Global Market Y-o-Y Growth Comparison by Technology, 2021 to 2036
- Figure 9: Global Market Attractiveness Analysis by Technology
- Figure 10: Global Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 11: Global Market Y-o-Y Growth Comparison by Material Type, 2021 to 2036
- Figure 12: Global Market Attractiveness Analysis by Material Type
- Figure 13: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 14: Global Market Y-o-Y Growth Comparison by Application, 2021 to 2036
- Figure 15: Global Market Attractiveness Analysis by Application
- Figure 16: Global Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 17: Global Market Y-o-Y Growth Comparison by End-User, 2021 to 2036
- Figure 18: Global Market Attractiveness Analysis by End-User
- Figure 19: Global Market Value (USD Mn) Share and BPS Analysis by Region, 2026 and 2036
- Figure 20: Global Market Y-o-Y Growth Comparison by Region, 2021 to 2036
- Figure 21: Global Market Attractiveness Analysis by Region
- Figure 22: North America Market Incremental $ Opportunity, 2021 to 2036
- Figure 23: Latin America Market Incremental $ Opportunity, 2021 to 2036
- Figure 24: Western Europe Market Incremental $ Opportunity, 2021 to 2036
- Figure 25: Eastern Europe Market Incremental $ Opportunity, 2021 to 2036
- Figure 26: East Asia Market Incremental $ Opportunity, 2021 to 2036
- Figure 27: South Asia and Pacific Market Incremental $ Opportunity, 2021 to 2036
- Figure 28: Middle East & Africa Market Incremental $ Opportunity, 2021 to 2036
- Figure 29: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 30: North America Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 31: North America Market Y-o-Y Growth Comparison by Platform Type, 2021 to 2036
- Figure 32: North America Market Attractiveness Analysis by Platform Type
- Figure 33: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 34: North America Market Y-o-Y Growth Comparison by Technology, 2021 to 2036
- Figure 35: North America Market Attractiveness Analysis by Technology
- Figure 36: North America Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 37: North America Market Y-o-Y Growth Comparison by Material Type, 2021 to 2036
- Figure 38: North America Market Attractiveness Analysis by Material Type
- Figure 39: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 40: North America Market Y-o-Y Growth Comparison by Application, 2021 to 2036
- Figure 41: North America Market Attractiveness Analysis by Application
- Figure 42: North America Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 43: North America Market Y-o-Y Growth Comparison by End-User, 2021 to 2036
- Figure 44: North America Market Attractiveness Analysis by End-User
- Figure 45: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 46: Latin America Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 47: Latin America Market Y-o-Y Growth Comparison by Platform Type, 2021 to 2036
- Figure 48: Latin America Market Attractiveness Analysis by Platform Type
- Figure 49: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 50: Latin America Market Y-o-Y Growth Comparison by Technology, 2021 to 2036
- Figure 51: Latin America Market Attractiveness Analysis by Technology
- Figure 52: Latin America Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 53: Latin America Market Y-o-Y Growth Comparison by Material Type, 2021 to 2036
- Figure 54: Latin America Market Attractiveness Analysis by Material Type
- Figure 55: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 56: Latin America Market Y-o-Y Growth Comparison by Application, 2021 to 2036
- Figure 57: Latin America Market Attractiveness Analysis by Application
- Figure 58: Latin America Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 59: Latin America Market Y-o-Y Growth Comparison by End-User, 2021 to 2036
- Figure 60: Latin America Market Attractiveness Analysis by End-User
- Figure 61: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Western Europe Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 63: Western Europe Market Y-o-Y Growth Comparison by Platform Type, 2021 to 2036
- Figure 64: Western Europe Market Attractiveness Analysis by Platform Type
- Figure 65: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 66: Western Europe Market Y-o-Y Growth Comparison by Technology, 2021 to 2036
- Figure 67: Western Europe Market Attractiveness Analysis by Technology
- Figure 68: Western Europe Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 69: Western Europe Market Y-o-Y Growth Comparison by Material Type, 2021 to 2036
- Figure 70: Western Europe Market Attractiveness Analysis by Material Type
- Figure 71: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 72: Western Europe Market Y-o-Y Growth Comparison by Application, 2021 to 2036
- Figure 73: Western Europe Market Attractiveness Analysis by Application
- Figure 74: Western Europe Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 75: Western Europe Market Y-o-Y Growth Comparison by End-User, 2021 to 2036
- Figure 76: Western Europe Market Attractiveness Analysis by End-User
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: Eastern Europe Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 79: Eastern Europe Market Y-o-Y Growth Comparison by Platform Type, 2021 to 2036
- Figure 80: Eastern Europe Market Attractiveness Analysis by Platform Type
- Figure 81: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 82: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2021 to 2036
- Figure 83: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 84: Eastern Europe Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 85: Eastern Europe Market Y-o-Y Growth Comparison by Material Type, 2021 to 2036
- Figure 86: Eastern Europe Market Attractiveness Analysis by Material Type
- Figure 87: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 88: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2021 to 2036
- Figure 89: Eastern Europe Market Attractiveness Analysis by Application
- Figure 90: Eastern Europe Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 91: Eastern Europe Market Y-o-Y Growth Comparison by End-User, 2021 to 2036
- Figure 92: Eastern Europe Market Attractiveness Analysis by End-User
- Figure 93: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 94: East Asia Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 95: East Asia Market Y-o-Y Growth Comparison by Platform Type, 2021 to 2036
- Figure 96: East Asia Market Attractiveness Analysis by Platform Type
- Figure 97: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 98: East Asia Market Y-o-Y Growth Comparison by Technology, 2021 to 2036
- Figure 99: East Asia Market Attractiveness Analysis by Technology
- Figure 100: East Asia Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 101: East Asia Market Y-o-Y Growth Comparison by Material Type, 2021 to 2036
- Figure 102: East Asia Market Attractiveness Analysis by Material Type
- Figure 103: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 104: East Asia Market Y-o-Y Growth Comparison by Application, 2021 to 2036
- Figure 105: East Asia Market Attractiveness Analysis by Application
- Figure 106: East Asia Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 107: East Asia Market Y-o-Y Growth Comparison by End-User, 2021 to 2036
- Figure 108: East Asia Market Attractiveness Analysis by End-User
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 110: South Asia and Pacific Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 111: South Asia and Pacific Market Y-o-Y Growth Comparison by Platform Type, 2021 to 2036
- Figure 112: South Asia and Pacific Market Attractiveness Analysis by Platform Type
- Figure 113: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 114: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2021 to 2036
- Figure 115: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 116: South Asia and Pacific Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 117: South Asia and Pacific Market Y-o-Y Growth Comparison by Material Type, 2021 to 2036
- Figure 118: South Asia and Pacific Market Attractiveness Analysis by Material Type
- Figure 119: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 120: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2021 to 2036
- Figure 121: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 122: South Asia and Pacific Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 123: South Asia and Pacific Market Y-o-Y Growth Comparison by End-User, 2021 to 2036
- Figure 124: South Asia and Pacific Market Attractiveness Analysis by End-User
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 126: Middle East & Africa Market Value Share and BPS Analysis by Platform Type, 2026 and 2036
- Figure 127: Middle East & Africa Market Y-o-Y Growth Comparison by Platform Type, 2021 to 2036
- Figure 128: Middle East & Africa Market Attractiveness Analysis by Platform Type
- Figure 129: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 130: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2021 to 2036
- Figure 131: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 132: Middle East & Africa Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 133: Middle East & Africa Market Y-o-Y Growth Comparison by Material Type, 2021 to 2036
- Figure 134: Middle East & Africa Market Attractiveness Analysis by Material Type
- Figure 135: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 136: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2021 to 2036
- Figure 137: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 138: Middle East & Africa Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 139: Middle East & Africa Market Y-o-Y Growth Comparison by End-User, 2021 to 2036
- Figure 140: Middle East & Africa Market Attractiveness Analysis by End-User
- Figure 141: Global Market – Tier Structure Analysis
- Figure 142: Global Market – Company Share Analysis