Investigator Payment Management Solutions Market Size, Share, Growth and Forecast (2026 - 2036)
The Investigator Payment Management Solutions Market is segmented by Component (Software and Services), Functionality (Payment Tracking & Automation, Multi-Currency & Cross-Border Payments, and Others), End User (Pharmaceutical & Biotechnology Companies, Contract Research Organizations, and Academic & Research Institutes) and Region. Forecast for 2026 to 2036.
Fact.MR highlights that the investigator payment management solutions market is reaching a tipping point between compliance requirements and operational scalability, where traditional manual payment workflows are becoming unmanageable as clinical trial volumes grow and global site networks expand, driving pharma sponsors and CROs toward automated, audit-ready platforms despite higher upfront investments.
Investigator Payment Management Solutions Market Forecast and Outlook By Fact.MR
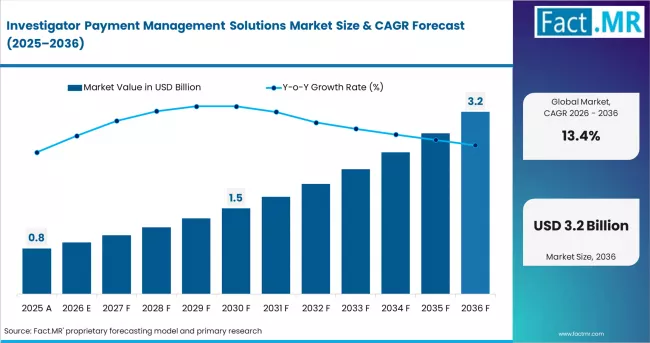
- In 2025, the investigator payment management solutions market was valued at USD 0.24 billion.
- Based on Fact.MR analysis, demand for investigator payment management solutions is estimated to grow to USD 0.7 billion in 2026 and USD 2.3 billion by 2036.
- FACT.MR projects a CAGR of 13.4% during the forecast period.
| Metric | Value |
|---|---|
| Estimated Value in 2026 | USD 0.7 billion |
| Forecast Value in 2036 | USD 2.3 billion |
| Forecast CAGR (2026 to 2036) | 13.4% |
Summary of Investigator Payment Management Solutions Market
- Market Definition
- Market covers software and services for automating investigator and site payments in clinical trials, including milestone tracking, multi-currency disbursement, tax compliance, and audit documentation.
- Demand Drivers
- Trial Site Network Expansion: Pharmaceutical sponsors are running trials across 50+ country site networks. Manual payment processing at this scale creates reconciliation errors and compliance risk, accelerating platform adoption.
- FDA Financial Disclosure Requirements: FDA 21 CFR Part 54 mandates financial disclosure from clinical investigators. Automated solutions reduce sponsor liability by maintaining auditable payment records throughout the trial lifecycle.
- CRO Outsourcing Growth: As sponsors outsource trial operations to CROs, payment management complexity increases. CROs are standardising on centralised platforms to serve multiple sponsor programmes simultaneously.
- Key Segments Analyzed
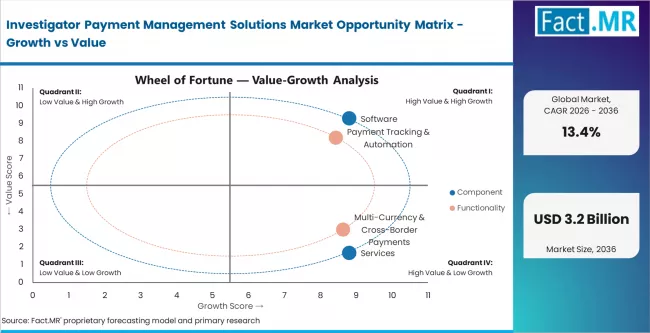
- By Component: Software leads with approximately 68% share in 2026, driven by recurring licence revenue and platform integration with CTMS and EDC systems across large pharma sponsors.
- By Functionality: Payment tracking and automation holds approximately 35% share in 2026. Multi-currency and cross-border payments represent the fastest-growing sub-segment at approximately 16% CAGR.
- By End User: Pharmaceutical and biotechnology companies account for approximately 60% share in 2026, driven by large-volume multi-site trial programmes and regulatory compliance requirements.
- Analyst Opinion at FACT.MR
- Shambhu Nath Jha, Senior Analyst at Fact.MR, opines, CXOs will observe that platform selection is moving away from cost-driven decisions. The focus is shifting toward compliance capability and operational control. Vendors offering built-in audit trails and multi-currency payment modules are gaining priority. These features are critical for managing global clinical trials and regulatory requirements.
- Strategic Implications
- Invest in EMA and FDA-aligned audit trail functionality. This is the primary procurement qualifier in Europe and North America for large pharma sponsors evaluating payment platforms.
- Develop embedded multi-currency and cross-border payment modules. This targets the fastest-growing functionality segment and addresses the needs of global trial networks in Asia Pacific.
- Partner with leading CROs for platform integration. Bundled CRO-software arrangements are expanding market access and locking in multi-year recurring revenue streams.
- Methodology
- Market sizing combines software licence revenues, professional services fees, and per-site payment management spend benchmarks across trial phases and therapeutic areas.
- Incorporates FDA clinical trial registry data, EMA GCP inspection outcomes, and CRO annual financial disclosures covering 2024 and 2025.
- Forecasts account for clinical trial volume growth, site network complexity trends, and regulatory tightening of financial transparency requirements across key markets.
The market is projected to generate USD 1.6 billion in absolute opportunity between 2026 and 2036. Growth is structurally driven by rising clinical trial site counts, multi-currency payment complexity, and regulatory audit pressure. Adoption is constrained by integration challenges with legacy clinical trial management systems and variable digital readiness among academic research sites.
Multi-site trial sponsors are increasingly standardising on centralised payment platforms to reduce reconciliation errors and meet FDA and EMA financial transparency requirements. Long-term software agreements bundled with CRO service contracts are becoming the norm, particularly in oncology and rare disease programmes across North America and Europe.
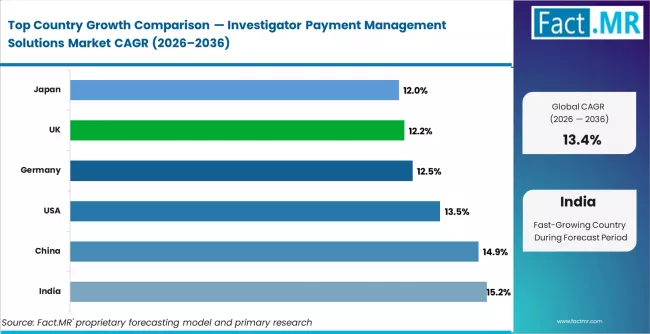
India leads with a projected CAGR of approximately 15.2% through 2036, driven by expanding CRO activity and regulatory harmonisation with ICH guidelines. China follows at 14.9%, supported by NMPA-driven clinical trial site expansion and rising sponsor outsourcing. The United States grows at 13.5%, anchored by large-volume trial networks and FDA financial disclosure mandates. Germany records 12.5%, led by precision trial infrastructure and EMA compliance demands. The United Kingdom grows at 12.2%, shaped by NHS-linked research site payment integration and MHRA oversight. Japan follows at 12.0%, driven by PMDA modernisation and growing site management organisation networks.
Segmental Analysis
Investigator Payment Management Solutions Market Analysis by Component
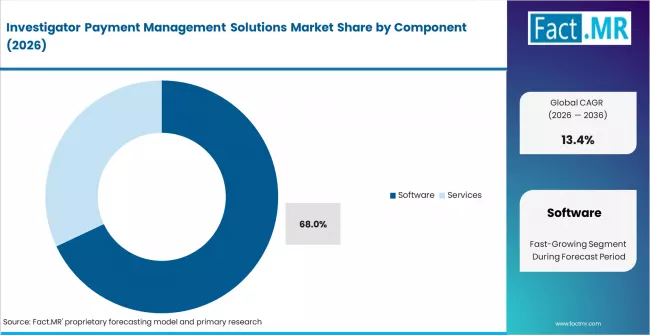
Based on FACT.MR analysis, software holds approximately 68% share in 2026. It leads due to recurring licence structures and deep integration with CTMS, EDC, and financial systems at large pharma sponsors. Services are the faster-growing segment as implementation complexity and training demand increase with platform adoption across mid-market and CRO clients.
- Veeva Payments Module: Veeva Systems launched its Vault Payments module as an extension of Vault CTMS in Q3 2024. The module automates investigator payment scheduling and milestone tracking. It integrates with existing Vault EDC and eTMF deployments across global pharma sponsor organisations [4].
- Oracle Health Sciences Update: Oracle Corporation released an enhanced investigator payment workflow within Oracle Clinical One in early 2025. The update includes multi-currency disbursement and real-time budget tracking. It targets enterprise pharma clients managing 500+ site trials across multiple geographies [5].
- Services Demand Trend: Parexel International disclosed in its 2024 annual results that professional services revenue related to site payment implementation and training grew 19% year-on-year. Growth reflects increasing demand from mid-size biotech sponsors transitioning from manual to automated payment workflows [3].
Investigator Payment Management Solutions Market Analysis by Functionality
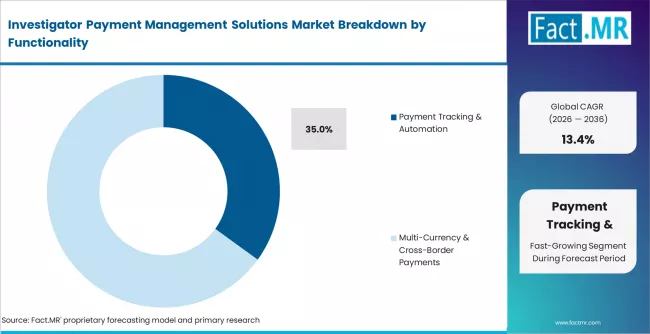
Based on FACT.MR analysis, payment tracking and automation holds approximately 35% share in 2026. It leads because milestone-based site payment reconciliation is the core operational pain point for sponsors and CROs. Multi-currency and cross-border payments represent the fastest-growing sub-segment, driven by increasing globalisation of clinical trial site networks into Asia Pacific and Latin America.
- Medidata Pay Launch: Medidata Solutions launched Medidata Pay in 2024, a purpose-built investigator payment platform integrated with Rave CTMS. It automates milestone-triggered disbursements and supports 130+ currency transactions. The platform was adopted by six of the top-20 global pharma companies by Q4 2024 [6].
- Multi-Currency Capability: IQVIA expanded its Site Payments platform in 2025 to include real-time foreign exchange rate locking and tax withholding automation for 40+ countries. This directly addresses cross-border payment reconciliation challenges at global trial sites in India, China, and Brazil [1].
- Audit Trail Trend: FDA's updated clinical investigator guidance in 2024 emphasised real-time payment audit trails as part of GCP compliance documentation. Platform vendors including Veeva and Oracle reported increased enterprise RFP activity for audit-capable payment modules in H2 2024 following this guidance update [4].
Drivers, Restraints, and Opportunities
FACT.MR analysts observe that the investigator payment management solutions market is in an early-to-growth phase, transitioning from manual and spreadsheet-based site payment workflows to integrated software platforms.
Legacy trial management systems lack native payment automation, creating integration friction. Platform vendors offering API-based CTMS connectivity are accelerating enterprise adoption.
- FDA 21 CFR Part 54 Compliance: FDA's financial disclosure regulations require sponsors to certify investigator payment independence. Automated audit trails generated by payment platforms directly support FDA submission requirements. IQVIA updated its site payment compliance module in Q2 2024 to align with FDA guidance revisions, supporting sponsor submission readiness [1].
- APAC Clinical Trial Expansion: China's NMPA approved 1,800+ clinical trial applications in 2024, generating significant new site payment volume. India's CDSCO harmonised ICH E6(R3) GCP guidelines in 2025, increasing sponsor confidence in Indian sites and driving demand for compliant payment management platforms [2].
- CRO Platform Standardisation: ICON plc and Parexel International both disclosed investments in payment automation platforms in 2024 annual reports. Standardised payment infrastructure across CRO networks reduces per-trial setup cost and supports multi-sponsor operations on shared technology stacks [3].
Regional Analysis
The investigator payment management solutions market is assessed across North America, Europe, Asia Pacific, Latin America, and Middle East and Africa, covering 40+ countries with demand shaped by clinical trial volume, regulatory transparency requirements, CRO ecosystem maturity, and digital adoption among research sites. The full report offers market attractiveness analysis by region and country.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 15.2% |
| China | 14.9% |
| United States | 13.5% |
| Germany | 12.5% |
| United Kingdom | 12.2% |
| Japan | 12.0% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
Asia Pacific Investigator Payment Management Solutions Market Analysis
Asia Pacific is the fastest-growing regional market. China and India are expanding their clinical trial site bases rapidly under government and regulatory reform initiatives. Platform vendors are entering through CRO partnerships rather than direct enterprise sales.
- China: Demand for investigator payment management solutions in China is projected to rise at 14.9% CAGR through 2036. China's NMPA approved more than 1,800 clinical trial applications in 2024, expanding the active site base significantly. International sponsors running China-inclusive global trials require RMB disbursement capability and NMPA-compliant financial documentation, driving platform demand.
- India: India is projected to rise at approximately 15.2% CAGR through 2036, the fastest among covered countries. India's CDSCO adopted ICH E6(R3) GCP guidelines in 2025, directly raising financial transparency and audit documentation requirements for trial sites. The Indian CRO sector, led by firms such as Fortrea and Syneos Health India operations, is scaling rapidly and standardising on platform-based payment management for multi-sponsor programmes. The number of CDSCO-approved clinical trial sites grew by 18% between 2023 and 2024.
- Japan: Investigator payment management solutions in Japan is projected to rise at 12.0% CAGR through 2036. Japan's PMDA released updated GCP compliance guidelines in 2024, reinforcing financial documentation requirements for investigator payments at domestic trial sites. AGC Biologics and Parexel's Japan operations expanded site management services in 2024, creating new demand for integrated payment platforms linked to Japanese clinical trial infrastructure.
FACT.MR's analysis of the investigator payment management solutions market in Asia Pacific consists of country-wise assessment that includes China, India, Japan, South Korea, and ASEAN. Readers can find NMPA and CDSCO regulatory impact data, CRO platform adoption trends, and multi-currency payment demand profiles.
North America Investigator Payment Management Solutions Market Analysis
North America is the revenue-dominant region for investigator payment management solutions. FDA regulatory requirements and the highest concentration of active Phase II and Phase III trials globally drive platform adoption. IQVIA and Medidata Solutions anchor the competitive landscape through integrated CTMS-payment platform ecosystems. Enterprise pharma procurement cycles are long and renewal rates are high.
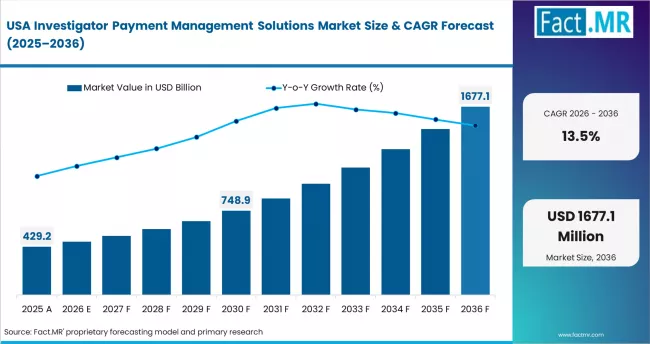
- United States: The market is set to expand at 13.5% CAGR through 2036. The US remains the global hub for clinical trials. ClinicalTrials.gov recorded over 490,000 studies by 2024. FDA 21 CFR Part 54 mandates strict audit trails. IQVIA enhanced compliance modules in 2024. Medidata Solutions gained traction among top pharma firms. Academic institutions are also upgrading systems for compliance.
FACT.MR's analysis of North America includes the United States, Canada, and Mexico. It highlights FDA-driven adoption, CRO standardisation, and enterprise procurement patterns.
Europe Investigator Payment Management Solutions Market Analysis
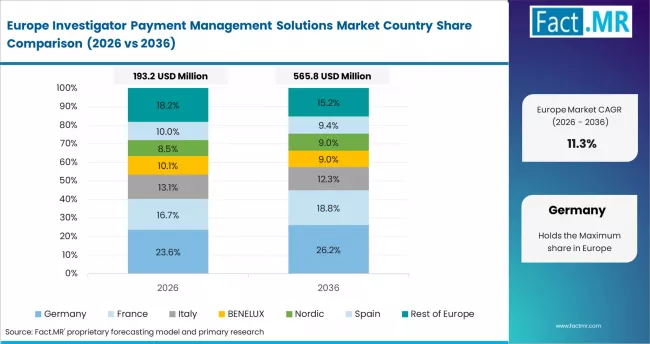
Europe is a compliance-driven market structured around EMA GCP inspection requirements and GDPR data governance obligations. Payment platforms must satisfy EMA financial transparency standards and secure cross-border data transfer rules. Veeva Systems and Oracle Health Sciences maintain strong positions in enterprise pharma accounts across Germany, the UK, and France.
- Germany: The market is on track to register 12.5% CAGR through 2036. Germany has one of the largest trial site networks in Europe. EMA inspections increased in 2024. Financial documentation gaps were frequently cited. Boehringer Ingelheim upgraded payment systems in 2024. BfArM issued updated transparency guidance in 2025, boosting software demand.
- United Kingdom: The market is positioned to expand at 12.2% CAGR through 2036. MHRA revised GCP guidelines in 2024. Financial disclosure requirements were strengthened. NHS sites are modernising payment systems under NIHR funding. AstraZeneca and GSK are transitioning to centralised platforms.
FACT.MR's analysis of the investigator payment management solutions market in Europe consists of country-wise assessment that includes Germany, the United Kingdom, France, Italy, Spain, and BENELUX. Readers can find EMA compliance trends, GDPR impact on payment data management, and enterprise pharma platform adoption profiles.
Competitive Aligners for Market Players
Competition in the investigator payment management solutions market is shifting toward compliance-led differentiation. Vendors must prioritise audit readiness and regulatory alignment over basic payment functionality. Platforms that support global compliance frameworks are gaining preference in enterprise procurement.
Integrated ecosystem capability is critical. Companies such as IQVIA and Medidata Solutions lead by embedding payment modules within CTMS and clinical data platforms. This creates workflow continuity and increases switching costs for sponsors and CROs.
Multi-currency and cross-border payment functionality is a key competitive lever. Vendors must support complex disbursement structures across regions such as the US, EU, China, and India. Platforms lacking automated currency handling and tax compliance features are losing relevance in global trials. Cloud-based deployment is becoming standard. SaaS models enable real-time tracking, scalability, and regulatory updates. Vendors must invest in secure, compliant cloud infrastructure to meet GDPR, FDA, and ICH requirements.
Partnerships with CROs are expanding market access. Vendors entering Asia Pacific are leveraging CRO relationships instead of direct enterprise sales. This approach accelerates adoption in fragmented trial ecosystems.
AI and automation are emerging differentiators. Automated payment scheduling, milestone tracking, and reconciliation reduce manual errors and improve operational efficiency. Vendors that embed intelligent workflows gain a competitive edge. Thus, enterprise sales capability is essential. Procurement cycles are long and documentation-intensive. Vendors must demonstrate compliance, scalability, and integration depth to secure large pharma contracts. Success depends on compliance capability, platform integration, and global scalability rather than standalone payment features.
Key Players
- IQVIA Holdings Inc.
- Medidata Solutions, Inc.
- Oracle Corporation (Oracle Health Sciences)
- Veeva Systems Inc.
- Parexel International Corporation
- ICON plc
Bibliography
- [1] IQVIA Holdings Inc. (Q2 2024). Site Payments Platform Update: Multi-Currency Foreign Exchange Rate Locking, FDA 21 CFR Part 54-Aligned Audit Trail Enhancement, and Enterprise Sponsor Compliance Module Release Notes. Q2 2024. iqvia.com/newsroom
- [2] Central Drugs Standard Control Organisation (CDSCO), Ministry of Health and Family Welfare, Government of India. (2025). ICH E6(R3) Good Clinical Practice Guidelines Adoption Notification and Updated Financial Transparency Requirements for Clinical Trial Sites in India. January 2025. cdsco.gov.in
- [3] ICON plc. (2024). Annual Report 2024: Site Payment Infrastructure Investment, Symphony CTMS Integration Update, and Professional Services Revenue Growth in Clinical Operations Technology. 2024. iconplc.com/investors
- [4] Veeva Systems Inc. (Q3 2024). Vault Payments Module Launch Press Release: Investigator Payment Scheduling Automation, Milestone Tracking Integration with Vault CTMS, and eTMF Audit Trail Documentation for Global Pharmaceutical Sponsor Trials. Q3 2024. veeva.com/news
- [5] Oracle Corporation. (Early 2025). Oracle Clinical One Release Notes: Enhanced Investigator Payment Workflow, Multi-Currency Disbursement Automation, and Real-Time Budget Tracking for Enterprise Pharma Trial Networks. January 2025. oracle.com/life-sciences/clinical-one
- [6] Medidata Solutions, Inc. (2024). Medidata Pay Product Launch: Purpose-Built Investigator Payment Platform Integrated with Rave CTMS Supporting 130+ Currency Transactions and Milestone-Based Automation for Top-20 Global Pharmaceutical Company Clients. 2024. medidata.com/news
- [7] National Institutes of Health (NIH). (2024). Clinical Trial Financial Reporting Requirements Update 2024: Enhanced Payment Documentation Standards for Federally Funded Research at Academic Medical Centres. 2024. grants.nih.gov/policy
- [8] National Medical Products Administration (NMPA), China. (2024). Clinical Trial Application Approvals Report 2024: Volume of Approved Applications, Site Network Expansion, and ICH E6 GCP Alignment Progress for International Sponsor Trials in China. 2024. nmpa.gov.cn
- [9] Central Drugs Standard Control Organisation (CDSCO). (2024). Clinical Trial Site Registration and Approval Data 2023-2024: Growth in Approved Research Sites, International Sponsor Inclusion Trends, and INR Payment Processing Requirements for Cross-Border Clinical Trials. 2024. cdsco.gov.in
This Report Addresses
- Strategic intelligence on investigator payment management solutions demand across pharmaceutical sponsors, CROs, and academic research institutions managing multi-site global clinical trials.
- Market forecast from USD 0.7 billion in 2026 to USD 2.3 billion by 2036 at a CAGR of 13.4%.
- Opportunity mapping across India and China CRO-driven site network expansion, US FDA compliance-led platform adoption, EU EMA GCP-aligned payment documentation requirements, and UK NIHR-backed research infrastructure investment.
- Segment analysis by component (software, services), functionality (payment tracking and automation, multi-currency payments, others), and end user (pharma and biotech, CROs, academic institutes) across global and regional markets.
- Regional outlook covering Asia Pacific trial volume growth and cross-currency payment demand, North America FDA compliance-driven platform standardisation, and Europe EMA-aligned financial transparency investment.
- Competitive landscape of IQVIA, Medidata Solutions, Veeva Systems, Oracle Health Sciences, Parexel, and ICON plc, covering platform integration depth, audit capability, and CRO channel positioning.
- Regulatory analysis covering FDA 21 CFR Part 54 financial disclosure requirements, EMA GCP inspection findings, India CDSCO ICH E6(R3) adoption, China NMPA clinical trial expansion, and UK MHRA post-Brexit GCP guideline revisions.
- Report delivered in PDF, Excel, and presentation formats supported by primary interviews, FDA registry data, EMA inspection records, and CRO annual financial disclosures.
Investigator Payment Management Solutions Market Definition
The investigator payment management solutions market covers software platforms and associated services used to automate, track, and process payments to clinical trial sites and investigators. These solutions manage milestone-based disbursements, multi-currency transfers, tax withholding, and audit trail documentation.
Investigator Payment Management Solutions Market Inclusions
Covers global and regional forecasts from 2026 to 2036. Includes software and professional services. Segmentation covers functionality, end user, and component type. Encompasses payment automation, multi-currency processing, compliance reporting, and site payment reconciliation tools.
Investigator Payment Management Solutions MarketExclusions
Excludes generic accounts payable software not designed for clinical trial workflows. Omits clinical trial management systems unless they include dedicated payment modules. Excludes grant management platforms for non-clinical academic research and standalone payroll or HR payment tools.
Investigator Payment Management Solutions Market Research Methodology
- Primary Research:Interviews with clinical operations directors, site payment managers at CROs, and IT procurement leads at pharmaceutical and biotech sponsors across North America, Europe, and Asia Pacific.
- Desk Research:Uses FDA financial disclosure regulation guidance, EMA GCP inspection reports, and company disclosures from IQVIA, Medidata Solutions, Veeva Systems, and Oracle Health Sciences (2024–2025).
- Market-Sizing and Forecasting:Hybrid model combining software licence revenue, services revenue, active clinical trial site counts, and average payment management spend per site per trial. Includes end-user and functional segment splits.
- Data Validation and Update Cycle:Validated using CRO annual reports, FDA clinical trial registry data, and EMA transparency disclosures. Updated through expert interviews annually.
Scope of Report

| Attribute | Details |
|---|---|
| Quantitative Units | USD 0.7 billion (2026) to USD 2.3 billion (2036), at a CAGR of 13.4% |
| Market Definition | Software platforms and professional services for automating, tracking, and processing payments to clinical trial sites and investigators, including milestone disbursements, multi-currency transfers, tax compliance, and audit documentation. |
| Component Segmentation | Software, Services |
| Functionality Segmentation | Payment Tracking & Automation, Multi-Currency & Cross-Border Payments, Others |
| End User Segmentation | Pharmaceutical & Biotechnology Companies, Contract Research Organizations, Academic & Research Institutes |
| Application Coverage | Milestone-based investigator payment automation, multi-currency site disbursement, FDA 21 CFR Part 54 and EMA GCP-aligned audit trail documentation, budget reconciliation, and tax withholding management for global clinical trial programmes. |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Canada, Mexico, Germany, UK, France, Italy, Spain, Nordic, BENELUX, China, India, Japan, South Korea, ASEAN, Australia and New Zealand, Brazil, Argentina, Saudi Arabia, GCC, Turkey, South Africa, Rest of MEA |
| Key Companies Profiled | IQVIA Holdings Inc., Medidata Solutions Inc., Oracle Corporation (Oracle Health Sciences), Veeva Systems Inc., Parexel International Corporation, ICON plc |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using software licence revenue, services revenue benchmarks, active clinical trial site counts by region, average payment management spend per site, and primary interviews with clinical operations and IT procurement professionals. |
Investigator Payment Management Solutions Market by Segments
-
By Component:
- Software
- Services
-
By Functionality:
- Payment Tracking & Automation
- Multi-Currency & Cross-Border Payments
- Others
-
By End User:
- Pharmaceutical & Biotechnology Companies
- Contract Research Organizations (CROs)
- Academic & Research Institutes
-
By Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Western Europe
- Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia and New Zealand
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East and Africa
- Saudi Arabia
- GCC Countries
- Turkey
- South Africa
- Rest of MEA
- North America
- Frequently Asked Questions -
How large is the global investigator payment management solutions market in 2025?
The global investigator payment management solutions market was valued at USD 0.24 billion in 2025.
What will the market size be in 2026?
Based on Fact.MR analysis, demand for investigator payment management solutions is estimated to grow to USD 0.7 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 2.3 billion by 2036, generating USD 1.6 billion in absolute dollar opportunity over the forecast period.
What is the expected CAGR from 2026 to 2036?
FACT.MR projects a CAGR of 13.4% for the global investigator payment management solutions market during the 2026 to 2036 forecast period.
Which component segment is poised to lead the market?
Software leads with approximately 68% share in 2026, driven by recurring licence revenue and deep integration with CTMS and EDC systems at large pharmaceutical and biotech sponsors.
Which functionality segment shows the fastest growth?
Multi-currency and cross-border payments represent the fastest-growing sub-segment at approximately 16% CAGR, driven by increasing globalisation of clinical trial site networks into Asia Pacific and Latin America. Which country shows the fastest growth in the market?
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Type , 2026 to 2036
- Pharmaceutical Contract Manufacturing
- Medical Device Manufacturing
- Pharmaceutical Contract Manufacturing
- Y to o to Y Growth Trend Analysis By Service Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Service Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Business Line
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Business Line, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Business Line, 2026 to 2036
- API Manufacturing
- Finished Dosage Formulation
- Biologics & Advanced Therapies
- API Manufacturing
- Y to o to Y Growth Trend Analysis By Business Line, 2021 to 2025
- Absolute $ Opportunity Analysis By Business Line, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical & Biotech Companies
- Medical Device OEMs
- Pharmaceutical & Biotech Companies
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service Type
- By Business Line
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Business Line
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service Type
- By Business Line
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Business Line
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service Type
- By Business Line
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Business Line
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service Type
- By Business Line
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Business Line
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service Type
- By Business Line
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Business Line
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service Type
- By Business Line
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Business Line
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service Type
- By Business Line
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Business Line
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Business Line
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service Type
- By Business Line
- By End Use
- Competition Analysis
- Competition Deep Dive
- Lonza Group
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Catalent Inc.
- Samsung Biologics
- WuXi Biologics
- Thermo Fisher Scientific
- Recipharm AB
- Boehringer Ingelheim Biopharmaceuticals GmbH
- Jabil Inc.
- Lonza Group
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Business Line, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Business Line, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Business Line, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Business Line, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Business Line, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Business Line, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Business Line, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Business Line, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by ServiceType, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by ServiceType, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Service Type
- Figure 6: Global Market Value Share and BPS Analysis by Business Line, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Business Line, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Business Line
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by ServiceType, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by ServiceType, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Service Type
- Figure 26: North America Market Value Share and BPS Analysis by Business Line, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Business Line, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Business Line
- Figure 29: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End Use
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by ServiceType, 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by ServiceType, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Service Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Business Line, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Business Line, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Business Line
- Figure 39: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End Use
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by ServiceType, 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by ServiceType, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Service Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Business Line, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Business Line, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Business Line
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Use
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by ServiceType, 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by ServiceType, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Service Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Business Line, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Business Line, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Business Line
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by ServiceType, 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by ServiceType, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Service Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Business Line, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Business Line, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Business Line
- Figure 69: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End Use
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by ServiceType, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by ServiceType, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Service Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Business Line, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Business Line, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Business Line
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by ServiceType, 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by ServiceType, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Service Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Business Line, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Business Line, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Business Line
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis