Reprocessed Medical Devices Market Size, Share, Growth and Forecast (2026 - 2036)
Reprocessed Medical Devices Market is segmented by Device Type (Cardiovascular Devices, Blood Pressure Cuffs And Tourniquet Cuffs, Cardiac Stabilization And Positioning Devices, Compression Sleeves, Diagnostic Electrophysiology Catheters, Electrophysiology Cables, General Surgery Devices, Balloon Inflation Devices, Infusion Pressure Bags, Laparoscopic Devices, Endoscopic Trocars And Components, Harmonic Scalpel, Orthopedic External Fixation Devices, Gastroenterology Biopsy Forceps), Product (Reprocessed Medical Devices, Cardiovascular Devices, Interventional Cardiology Devices, Gastroenterology Devices, General Surgery Devices), Application (Minimally Invasive Procedures, Gastrointestinal Procedures, General Surgery, Orthopedic Surgery, Urological Procedures), and Region, with forecasts covering the period from 2026 to 2036.
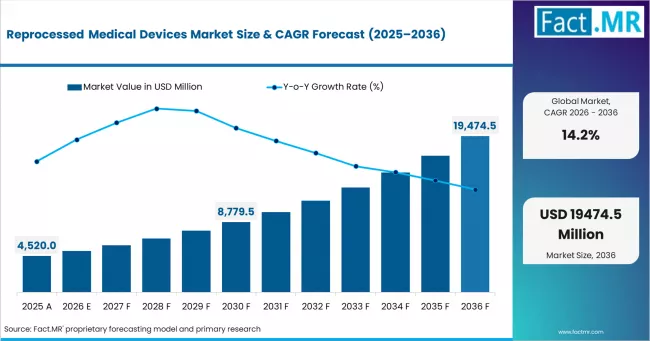
According to Fact.MR estimates, the global reprocessed medical devices market market was valued at USD 4,520.0 million in 2025. The market is projected to reach USD 5,161.8 million in 2026 and is expected to grow to USD 19,474.5 million by 2036, expanding at a CAGR of 14.2%. Cardiovascular Devices is anticipated to account for 25.0% of the product segment in 2026, while Reprocessed Medical Devices is expected to remain the leading application with around 36.0% share.
Reprocessed Medical Devices Market Analysis and Forecast by Fact.MR
The reprocessed medical devices market, valued at USD 4,520 million in 2025, is expanding at a 14.2% CAGR to reach USD 19,474.467 million by 2036. Reprocessed medical devices are single-use devices that have been collected, cleaned, sterilized, tested, and repackaged to meet original equipment manufacturer specifications for reuse in clinical procedures. Growth reflects intensifying hospital cost containment pressure, sustainability mandates on medical waste reduction, and expanding regulatory acceptance of reprocessing as a validated supply chain practice. The USD 14,312.627 million incremental opportunity between 2026 and 2036 is concentrated in minimally invasive procedure supply chains across North America, Europe, and East Asia.
Summary of Reprocessed Medical Devices Market
- Market Snapshot
- Market value in 2025: USD 4,520 million
- Projected value in 2026: USD 5,161.84 million; forecast to reach USD 19,474.467 million by 2036
- CAGR from 2026 to 2036: 14.2%
- Incremental opportunity (2026 to 2036): USD 14,312.627 million
- Cardiovascular devices lead the device-type segment with 25.0% share in 2025
- China and USA lead country-level growth, followed by South Korea and Germany
- Demand and Growth Drivers
- Hospital cost containment mandates are intensifying across both public and private healthcare systems, with reprocessed devices offering 40% to 60% cost savings compared to new single-use equivalents for high-volume procedure supplies.
- Sustainability mandates on medical waste reduction are being formalized by healthcare regulators and hospital accreditation bodies, creating procurement incentives for reprocessed alternatives to single-use devices.
- Regulatory acceptance is expanding as FDA, EU MDR, and national health authorities in Asia formalize validation requirements and quality standards for reprocessed devices, reducing institutional resistance to adoption.
- Minimally invasive procedure volumes continue to grow across cardiovascular, gastrointestinal, and orthopedic specialties, increasing the consumption of high-cost single-use devices that are prime candidates for reprocessing.
- Product and Segment View
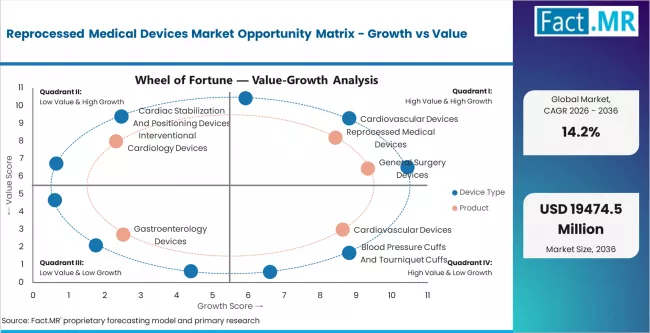
- Cardiovascular devices hold 25.0% of device-type demand, driven by the high unit cost of electrophysiology catheters, cardiac stabilization devices, and balloon inflation systems used in interventional cardiology.
- Minimally invasive procedures account for 42.1% of application share, reflecting the growth of catheter-based and laparoscopic interventions that consume costly single-use device inventories.
- Hospitals account for 56.8% of end-use demand, as the primary institutional buyers of reprocessed devices through GPO-negotiated supply contracts.
- Key segmentation includes:
- Device Type: Cardiovascular Devices, Blood Pressure Cuffs and Tourniquet Cuffs, Cardiac Stabilization and Positioning Devices, Compression Sleeves, Diagnostic Electrophysiology Catheters, General Surgery Devices, Laparoscopic Devices, Orthopedic External Fixation Devices, Gastroenterology Biopsy Forceps, Others
- Product: Reprocessed Medical Devices, Cardiovascular Devices, Interventional Cardiology Devices, Gastroenterology Devices, General Surgery Devices
- Application: Minimally Invasive Procedures, Gastrointestinal Procedures, General Surgery, Orthopedic Surgery, Urological Procedures
- End Use: Hospitals, Ambulatory Surgical Centers, Diagnostic Centers
- Technology: Reprocessing Technology, Sterilization Methods, Low Temperature Sterilization
- Geography and Competitive Outlook
- North America is the largest revenue region, anchored by the USA where FDA-regulated reprocessing has been established since the early 2000s and hospital cost pressure is accelerating adoption.
- East Asia is the fastest-growing region, with China recording the highest country CAGR as provincial governments pilot medical device reprocessing programs to control public hospital procurement costs.
- Europe is shaped by the EU Medical Device Regulation, which is formalizing reprocessing quality standards across member states and expanding the regulatory pathway for reprocessed device market access.
- Stryker leads the competitive field with a 15.0% market share, followed by Medline Industries, Johnson and Johnson, and a growing set of specialized reprocessing companies.
- Analyst Opinion
- The reprocessed medical devices market is at a structural inflection point where cost containment pressure, waste reduction mandates, and regulatory maturation are converging to accelerate adoption. The category has moved from a niche cost-saving tactic to a procurement strategy endorsed by hospital C-suites and sustainability officers. Fact.MR expects the fastest growth to occur in device categories with the highest new-device unit costs, particularly electrophysiology catheters and laparoscopic instruments, where reprocessing delivers the largest per-unit savings. Regulatory clarity in the EU and pilot programs in China are the catalysts for the next phase of geographic expansion beyond the established North American base.
Key Growth Drivers, Constraints, and Opportunities
Key Factors Driving Growth
- Hospital budget compression: Public and private hospitals facing flat or declining reimbursement rates are adopting reprocessed devices as a direct procurement cost reduction mechanism, with per-device savings of 40% to 60% on high-volume procedure supplies.
- Medical waste reduction mandates: Healthcare sustainability regulations in the EU, USA, and Australia are establishing medical waste reduction targets that incentivize reprocessing adoption as an alternative to landfill and incineration disposal of single-use devices.
- Regulatory pathway maturation: FDA establishment of reprocessed device quality standards, EU MDR formalization of reprocessing requirements, and pilot regulatory programs in China and Brazil are reducing institutional resistance by providing validated safety and efficacy frameworks.
Key Market Constraints
- OEM resistance and device design barriers: Original equipment manufacturers have financial incentives to maintain single-use designation and are designing devices with features that complicate third-party reprocessing, including embedded electronics and single-use lockout mechanisms.
- Clinician perception gaps: Surgeon and physician resistance to reprocessed devices persists in some institutions, driven by concerns about device performance equivalence and liability exposure, despite regulatory evidence of safety parity.
- Supply chain logistics complexity: Collecting used devices from hospital procedure rooms, sorting by reprocessing eligibility, and managing reverse logistics to reprocessing facilities requires specialized infrastructure that limits market entry for new competitors.
Key Opportunity Areas
- Electrophysiology catheter reprocessing: High-unit-cost diagnostic EP catheters are the most economically attractive reprocessing category, with per-device savings exceeding USD 1,000 and growing procedure volumes in atrial fibrillation ablation.
- Emerging market regulatory pilots: Government-sponsored reprocessing pilot programs in China, Brazil, and India are creating structured entry points for reprocessing companies to demonstrate safety, build regulatory relationships, and establish collection infrastructure.
- Hospital sustainability certification: Green hospital certification programs (Practice Greenhealth, NHS Net Zero) are incorporating medical device reprocessing into scoring criteria, creating procurement preference advantages for reprocessed device suppliers.
Segment-wise Analysis of the Reprocessed Medical Devices Market
- Cardiovascular devices lead device-type demand at 25.0%, driven by the high unit cost of EP catheters and cardiac stabilization systems.
- Minimally invasive procedures hold 42.1% of application share, reflecting the growth of catheter-based interventions that consume costly single-use device inventories.
- Hospitals account for 56.8% of end-use demand, as the primary institutional purchasers of reprocessed devices through GPO contracts.
Which device type dominates the Reprocessed Medical Devices Market?
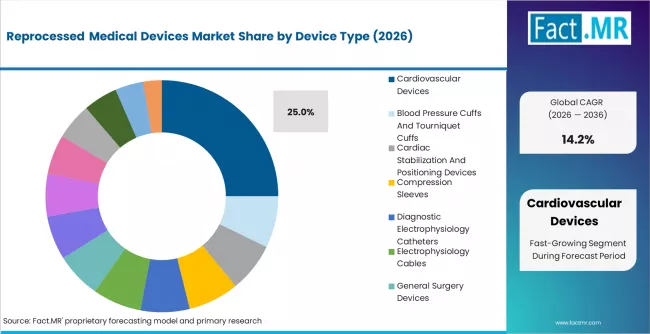
Cardiovascular devices hold a 25.0% share of device-type demand in 2025, the largest single device category. The dominance reflects the high unit cost of electrophysiology catheters, cardiac stabilization and positioning devices, and balloon inflation systems used in interventional cardiology and electrophysiology procedures. Reprocessing delivers the greatest per-unit dollar savings in this category, with reprocessed EP catheters priced at 40% to 60% below new-device equivalents while meeting identical FDA-cleared performance specifications.
General surgery devices and laparoscopic instruments are the next-largest categories, driven by growing procedure volumes and the expanding range of single-use laparoscopic trocars, harmonic scalpels, and energy-based devices that qualify for reprocessing. Gastroenterology biopsy forceps represent a smaller but growing segment, supported by endoscopy volume growth and the comparatively straightforward reprocessing validation requirements for these devices.
Which application dominates the Reprocessed Medical Devices Market?
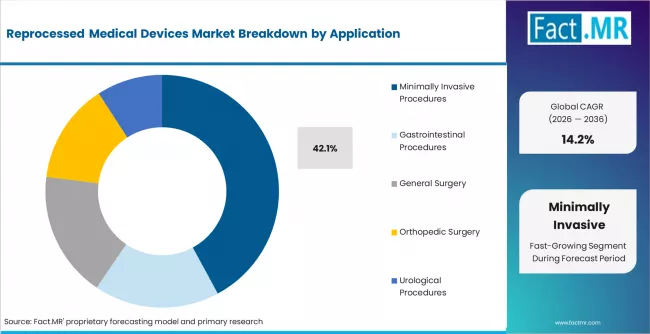
Minimally invasive procedures account for 42.1% of application demand in 2025, the largest single application category. The share reflects the high consumption of costly single-use devices in catheter-based cardiovascular interventions, laparoscopic surgery, and endoscopic procedures. These procedure types consume multiple single-use devices per case, creating substantial cumulative cost exposure that makes reprocessing economically compelling for hospital procurement teams.
General surgery and gastrointestinal procedures are the next-largest application categories. Orthopedic surgery is a growing reprocessing application as the range of reprocessable orthopedic external fixation devices and single-use instruments expands. Urological procedures contribute a smaller share, concentrated in cystoscopy and ureteroscopy device reprocessing.
Which end use dominates the Reprocessed Medical Devices Market?
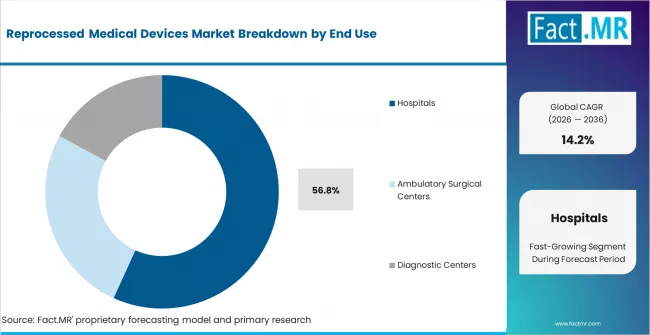
Hospitals account for 56.8% of end-use demand in 2025, reflecting the concentration of surgical volume, device procurement budgets, and GPO contract participation in acute care facilities. Hospital adoption of reprocessed devices is driven by procurement department cost targets, sustainability officer mandates, and supply chain standardization across multi-facility health systems.
Ambulatory surgical centers are the fastest-growing end-use segment, as these facilities face intense cost pressure from bundled payment reimbursement models that incentivize supply cost reduction per procedure. Diagnostic centers represent a smaller share, primarily in endoscopy and cardiac catheterization laboratory settings where device reprocessing is well-established.
Which Product Trend is Shaping the Next Phase of Growth in the Reprocessed Medical Devices Market?
The expansion of reprocessing into complex electrophysiology and energy-based surgical devices is shaping the next growth phase. Early reprocessing programs focused on comparatively simple devices such as blood pressure cuffs, compression sleeves, and basic surgical instruments. The category is now moving into higher-complexity, higher-value devices including diagnostic EP catheters, harmonic scalpels, and advanced laparoscopic instruments. This migration up the device complexity ladder is enabled by advances in reprocessing technology, including improved low-temperature sterilization methods that preserve electronic components and polymer materials that degrade under traditional autoclave conditions.
The economic case for reprocessing complex devices is significantly stronger than for simple devices, as the per-unit cost differential between new and reprocessed products widens with device complexity. A reprocessed EP catheter can save a hospital over USD 1,000 per device compared to a new equivalent, making the total savings material at institutional scale when multiplied across thousands of procedures annually. Fact.MR projects that reprocessing of complex electrophysiology and energy-based devices will account for the majority of incremental revenue growth in this market through 2036, as hospitals and reprocessing companies collaborate on expanding the range of FDA-cleared and EU MDR-compliant reprocessed device categories.
Regional Outlook Across Key Markets
.webp)
- North America is the largest revenue region, with the USA anchoring demand through two decades of FDA-regulated reprocessing and strong GPO-mediated procurement.
- East Asia is the fastest-growing region, with China recording the highest country CAGR as provincial governments establish medical device reprocessing frameworks.
- Europe is shaped by EU MDR formalization of reprocessing standards, expanding market access for validated reprocessed devices across member states.
- South Asia and Pacific is an emerging growth corridor, with South Korea and Australia-NZ recording solid growth driven by hospital cost containment and sustainability initiatives.
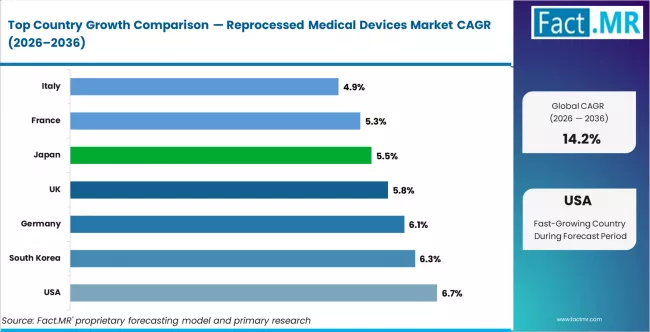
CAGR Table
| Country | CAGR (%) |
|---|---|
| China | 7.2% |
| USA | 6.7% |
| South Korea | 6.3% |
| Germany | 6.1% |
| UK | 5.8% |
| Australia-NZ | 5.7% |
| Japan | 5.5% |
| France | 5.3% |
| Italy | 4.9% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research.
China: Provincial reprocessing pilot programs and cost containment
China records the highest country-level CAGR in the reprocessed medical devices market, driven by provincial government initiatives to reduce public hospital procurement costs through medical device reprocessing pilot programs. The National Medical Products Administration has issued preliminary guidance on device reprocessing standards, creating a regulatory pathway that provincial health authorities are using to approve pilot reprocessing operations in selected hospital networks. Domestic companies are partnering with international reprocessing firms to acquire validation expertise and sterilization technology.
- Growth anchor: Provincial government pilot programs are creating structured entry points for reprocessing adoption within public hospital procurement systems.
USA: Established regulatory framework and GPO adoption
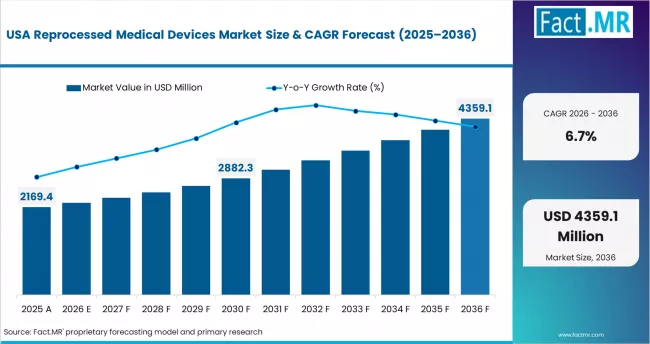
The USA is the most mature reprocessed medical devices market, with FDA regulation of third-party reprocessing established since 2000 and broad GPO-mediated distribution to hospital networks. Growth is driven by expanding the range of device categories cleared for reprocessing, with recent FDA 510(k) clearances for more complex electrophysiology and laparoscopic instruments. Hospital sustainability programs are adding reprocessing volume targets alongside traditional cost-saving objectives, creating dual procurement incentives.
- Growth anchor: FDA clearance expansion into complex device categories is increasing the addressable market while sustainability mandates add procurement motivation beyond cost savings.
South Korea: Hospital cost pressure and quality standards
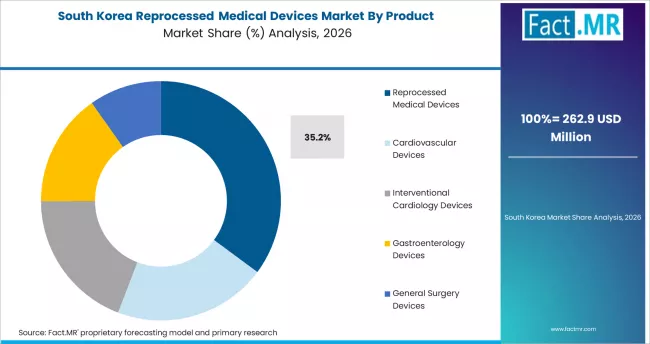
South Korea records strong growth driven by hospital cost containment pressure under the national health insurance system's fee schedule constraints. Korean hospitals, particularly large academic medical centers performing high volumes of cardiovascular and endoscopic procedures, are adopting reprocessed devices as a procurement cost reduction mechanism. The Korean Ministry of Food and Drug Safety has established quality standards for reprocessed devices that align with international benchmarks, reducing adoption barriers.
- Growth anchor: National health insurance fee schedule constraints are creating direct financial incentives for hospitals to adopt reprocessed device procurement.
Germany: EU MDR leadership and sustainability integration
Germany leads European reprocessed medical device growth, driven by early adoption of EU MDR reprocessing requirements and strong hospital sustainability commitments. German hospitals, particularly university medical centers with large surgical volumes, have integrated device reprocessing into procurement strategies that combine cost reduction with medical waste minimization targets. The regulatory environment in Germany provides clarity on reprocessing quality requirements, reducing institutional resistance compared to EU member states where implementation guidance remains less developed.
- Growth anchor: Mature EU MDR implementation and hospital sustainability integration create stable demand growth supported by regulatory and institutional frameworks.
Competitive Benchmarking and Company Positioning
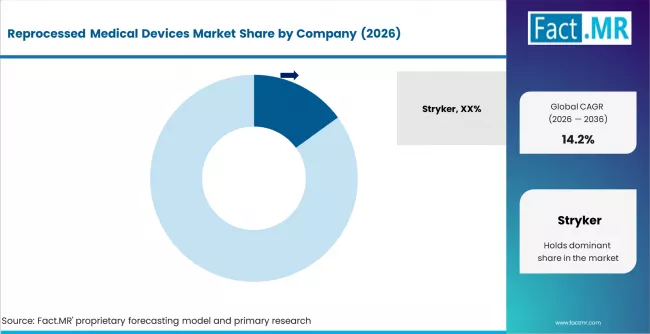
Reprocessed Medical Devices Market Analysis By Company
- Stryker leads with 15.0% market share, operating its Sustainability Solutions division as one of the largest third-party medical device reprocessing operations in North America with FDA clearance across multiple device categories.
- Medline Industries and Johnson and Johnson compete in the integrated supply tier, where reprocessed device offerings complement their broader hospital supply portfolios and GPO contract relationships.
- Specialized reprocessing companies including ReNu Medical, Sterilmed, and Hygia Health Services compete on processing expertise, device category breadth, and collection logistics infrastructure.
The competitive field combines large medical device companies that have entered reprocessing as a sustainability and customer retention strategy with specialized reprocessing firms that compete on processing expertise and reverse logistics capabilities. Stryker's Sustainability Solutions division has established the scale and FDA clearance portfolio to serve large hospital systems, positioning reprocessing as a value-added service that reinforces broader device supply relationships.
Specialized reprocessing companies compete by focusing exclusively on reprocessing operations, investing in sterilization technology development and regulatory clearance expansion for new device categories. These companies often target specific device niches, such as EP catheters or laparoscopic instruments, where they can build deep processing expertise and regulatory track records.
OEM resistance to third-party reprocessing is creating a competitive dynamic where device manufacturers design products to resist reprocessing while simultaneously offering their own reprocessing programs. This dual strategy aims to control the reprocessing value chain while maintaining new-device revenue streams, creating a complex competitive environment for independent reprocessing companies.
Key Companies in the Reprocessed Medical Devices Market
Stryker, Medline Industries Inc., ReNu Medical, LUMITOS AG, Vanguard AG, Johnson and Johnson, Sterilmed Inc., Hygia Health Services Inc., Centurion Medical Products Corporation, Siemens Healthcare, Philips Healthcare, GE Healthcare, and Suretech Medical Inc. are the principal companies operating in the global reprocessed medical devices market.
Recent Industry Developments
- Stryker Sustainability Solutions: EP catheter reprocessing clearance expansion (2025)
- Stryker's Sustainability Solutions division received FDA 510(k) clearance for reprocessing of additional diagnostic electrophysiology catheter models in 2025, expanding its reprocessable device portfolio in the highest-value cardiovascular device category.
- Medline Industries: reprocessing program launch for ASC networks (2025)
- Medline Industries launched a dedicated reprocessed device procurement program for ambulatory surgical center networks in 2025, targeting cost-sensitive ASCs operating under bundled payment reimbursement models.
- Johnson and Johnson: OEM reprocessing pilot in Europe (2025)
- Johnson and Johnson initiated an OEM-operated device reprocessing pilot program in select European markets during 2025, collecting and reprocessing its own single-use laparoscopic instruments through a controlled supply chain to maintain quality standards and device performance.
- ReNu Medical: expansion into Asian reprocessing markets (2026)
- ReNu Medical established a partnership with a Chinese hospital network in early 2026 to provide reprocessing technology transfer and validation consulting services, supporting pilot reprocessing operations targeting cardiovascular device categories.
Leading Companies Shaping the Reprocessed Medical Devices Market
-
Global Players
- Stryker
- Medline Industries Inc.
- Johnson and Johnson
- Siemens Healthcare
- Philips Healthcare
- GE Healthcare
-
Regional Players
- Sterilmed Inc.
- Hygia Health Services Inc.
- Centurion Medical Products Corporation
- Suretech Medical Inc.
-
Emerging / Start-up Players
- ReNu Medical
- LUMITOS AG
- Vanguard AG
Sources and Research References
- Stryker Corporation Annual Report 2024 and Sustainability Solutions Division Press Releases
- FDA 510(k) Premarket Notification Database for Reprocessed Single-Use Devices
- EU Medical Device Regulation (MDR) Annex I Requirements for Reprocessing of Single-Use Devices
- Practice Greenhealth Environmental Excellence Awards and Hospital Sustainability Benchmarking Data
- National Medical Products Administration of China, Medical Device Reprocessing Guidance Documents
- Johnson and Johnson ESG Report 2024 and Medical Device Sustainability Initiatives
All numerical data in this report is derived from Fact.MR proprietary research. Source references are provided for contextual and directional validation only.
Key Questions This Report Addresses
- What is the projected size of the reprocessed medical devices market by 2036?
- Which device type holds the largest share in the reprocessed medical devices market?
- What CAGR is the reprocessed medical devices market expected to register from 2026 to 2036?
- Which country is the fastest-growing market for reprocessed medical devices?
- Who is the leading company in the global reprocessed medical devices market?
- What is the incremental opportunity in the reprocessed medical devices market between 2026 and 2036?
- Which application dominates reprocessed medical device demand?
- How is sustainability regulation affecting reprocessed device adoption?
- What role do GPOs play in reprocessed medical device procurement?
- What savings do hospitals achieve by using reprocessed devices?
Reprocessed Medical Devices Market Definition
The reprocessed medical devices market covers single-use medical devices that have been collected after initial clinical use, subjected to validated cleaning, testing, and sterilization processes, and repackaged for reuse in clinical procedures. These devices must meet original equipment manufacturer performance specifications and regulatory standards established by the FDA, EU MDR, or equivalent national authorities. The market includes both third-party reprocessing services and OEM-operated reprocessing programs.
Reprocessed Medical Devices Market Inclusions
- Reprocessed single-use cardiovascular, general surgery, laparoscopic, and gastroenterology devices cleared for clinical reuse
- Third-party reprocessing services and OEM-operated collection and reprocessing programs
- Sterilization, testing, and repackaging services performed to regulatory validation standards
- Reprocessed devices sold to hospitals, ambulatory surgical centers, and diagnostic centers through direct and GPO-mediated channels
Reprocessed Medical Devices Market Exclusions
- New single-use medical devices sold for first-time clinical use
- Reusable medical devices designed for multi-use with standard sterilization between uses
- Medical device repair, refurbishment, and reconditioning services for capital equipment
- Pharmaceutical products, biologics, and implantable devices not subject to reprocessing
Reprocessed Medical Devices Market Research Methodology
- Bottom-up revenue modeling using reprocessed device volume, average selling prices, and hospital procurement contract data across 20+ countries
- Primary interviews with hospital supply chain directors, reprocessing company executives, and GPO category managers
- Cross-referencing FDA 510(k) clearance database for reprocessed device registrations and EU MDR notified body certifications
- Validation of segment shares through hospital procurement records, tender databases, and reprocessing company product catalogues
- Forecasting calibrated against surgical procedure volume projections, healthcare cost containment policy trajectories, and sustainability regulation implementation timelines
- Company-level revenue triangulation using annual reports, SEC filings, and investor presentations from listed medical device and reprocessing companies
Scope of Analysis
| Parameter | Details |
|---|---|
| Market size (2025) | USD 4,520 million |
| Forecast period | 2026 to 2036 |
| CAGR | 14.2% |
| Base year | 2025 |
| Units | USD Million |
| Segments covered | Device Type (Cardiovascular Devices, Blood Pressure Cuffs and Tourniquet Cuffs, Cardiac Stabilization Devices, Compression Sleeves, Diagnostic EP Catheters, General Surgery Devices, Laparoscopic Devices, Orthopedic External Fixation Devices, Gastroenterology Biopsy Forceps); Product (Reprocessed Medical Devices, Cardiovascular, Interventional Cardiology, Gastroenterology, General Surgery); Application (Minimally Invasive Procedures, GI Procedures, General Surgery, Orthopedic Surgery, Urological Procedures); End Use (Hospitals, ASCs, Diagnostic Centers); Technology (Reprocessing Technology, Sterilization Methods, Low Temperature Sterilization) |
| Regions covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries profiled | USA, China, South Korea, Germany, UK, France, Japan, Italy, Australia-NZ |
| Companies profiled | Stryker, Medline Industries Inc., ReNu Medical, Johnson and Johnson, Sterilmed Inc., Hygia Health Services Inc., GE Healthcare, Philips Healthcare, Siemens Healthcare |
| Forecasting approach | Bottom-up revenue modeling validated through primary research, FDA clearance database cross-referencing, and company-level financial triangulation |
Market Segmentation Analysis
-
Reprocessed Medical Devices Market Market Segmented by Device Type:
- Cardiovascular Devices
- Electrophysiology Catheters
- Cardiac Stabilization Devices
- Cardiac Positioning Devices
- Blood Pressure Cuffs And Tourniquet Cuffs
- Reusable Blood Pressure Cuffs
- Surgical Tourniquet Cuffs
- Cardiac Stabilization And Positioning Devices
- Off Pump Stabilizers
- Heart Positioning Systems
- Compression Sleeves
- Deep Vein Thrombosis Sleeves
- Intermittent Pneumatic Compression Sleeves
- Diagnostic Electrophysiology Catheters
- Conventional Electrophysiology Catheters
- Advanced Mapping Catheters
- Electrophysiology Cables
- Recording Cables
- Connector Cables
- General Surgery Devices
- Surgical Instruments
- Electrosurgical Devices
- Balloon Inflation Devices
- Manual Inflation Devices
- Digital Inflation Devices
- Infusion Pressure Bags
- Disposable Pressure Bags
- Reusable Pressure Bags
- Laparoscopic Devices
- Laparoscopic Graspers
- Laparoscopic Scissors
- Laparoscopic Dissectors
- Endoscopic Trocars And Components
- Trocars
- Cannulas
- Obturators
- Harmonic Scalpel
- Ultrasonic Scalpels
- Energy Based Surgical Devices
- Orthopedic External Fixation Devices
- Unilateral Fixators
- Ring Fixation Systems
- Gastroenterology Biopsy Forceps
- Disposable Biopsy Forceps
- Reusable Biopsy Forceps
- Cardiovascular Devices
-
Reprocessed Medical Devices Market Market Segmented by Product:
- Reprocessed Medical Devices
- Cardiovascular Devices
- Electrophysiology Devices
- Diagnostic Catheters
- Ablation Catheters
- Interventional Cardiology Devices
- Balloon Catheters
- Guiding Catheters
- Gastroenterology Devices
- Endoscopy Devices
- Endoscopes
- Biopsy Forceps
- General Surgery Devices
- Laparoscopic Devices
- Trocars
- Graspers
-
Reprocessed Medical Devices Market Market Segmented by Application:
- Minimally Invasive Procedures
- Cardiac Procedures
- Electrophysiology Procedures
- Angioplasty Procedures
- Gastrointestinal Procedures
- Endoscopy Procedures
- Diagnostic Endoscopy
- Therapeutic Endoscopy
- General Surgery
- Laparoscopic Surgery
- Abdominal Surgery
- Thoracic Surgery
- Orthopedic Surgery
- Joint Procedures
- Knee Surgery
- Hip Surgery
- Urological Procedures
- Urology Treatments
- Stone Management
- Bladder Procedures
- Minimally Invasive Procedures
-
Reprocessed Medical Devices Market Market Segmented by End Use:
- Hospitals
- Public Hospitals
- Tertiary Care Hospitals
- Specialty Hospitals
- Ambulatory Surgical Centers
- Outpatient Facilities
- Day Care Surgery Centers
- Specialty Clinics
- Diagnostic Centers
- Diagnostic Facilities
- Imaging Centers
- Endoscopy Centers
- Hospitals
-
Reprocessed Medical Devices Market Market Segmented by Technology:
- Reprocessing Technology
- Cleaning and Disinfection
- Manual Cleaning
- Automated Cleaning
- Sterilization Methods
- High Temperature Sterilization
- Steam Sterilization
- Dry Heat Sterilization
- Low Temperature Sterilization
- Ethylene Oxide Sterilization
- Hydrogen Peroxide Sterilization
- Reprocessing Technology
-
Reprocessed Medical Devices Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
What is the current size of the reprocessed medical devices market?
The global reprocessed medical devices market is valued at USD 4,520 million in 2025.
What growth rate is expected for the reprocessed medical devices market?
The market is projected to grow at a CAGR of 14.2% from 2026 to 2036.
What will the reprocessed medical devices market be worth by 2036?
The market is forecast to reach USD 19,474.467 million by 2036.
Which device type leads the reprocessed medical devices market?
Cardiovascular devices lead with a 25.0% share of device-type demand in 2025.
Which country is growing fastest in the reprocessed medical devices market?
China records the highest country-level CAGR among tracked markets.
Who is the market leader in reprocessed medical devices?
Stryker leads with a 15.0% market share in 2025.
What cost savings do reprocessed devices offer?
Reprocessed devices typically offer 40% to 60% cost savings compared to new single-use equivalents.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Device Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Device Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Device Type , 2026 to 2036
- Cardiovascular Devices
- Electrophysiology Catheters
- Cardiac Stabilization Devices
- Cardiac Positioning Devices
- Blood Pressure Cuffs And Tourniquet Cuffs
- Reusable Blood Pressure Cuffs
- Surgical Tourniquet Cuffs
- Cardiac Stabilization And Positioning Devices
- Off Pump Stabilizers
- Heart Positioning Systems
- Compression Sleeves
- Deep Vein Thrombosis Sleeves
- Intermittent Pneumatic Compression Sleeves
- Diagnostic Electrophysiology Catheters
- Conventional Electrophysiology Catheters
- Advanced Mapping Catheters
- Electrophysiology Cables
- Recording Cables
- Connector Cables
- General Surgery Devices
- Surgical Instruments
- Electrosurgical Devices
- Balloon Inflation Devices
- Manual Inflation Devices
- Digital Inflation Devices
- Infusion Pressure Bags
- Disposable Pressure Bags
- Reusable Pressure Bags
- Laparoscopic Devices
- Laparoscopic Graspers
- Laparoscopic Scissors
- Laparoscopic Dissectors
- Endoscopic Trocars And Components
- Trocars
- Cannulas
- Obturators
- Cardiovascular Devices
- Y to o to Y Growth Trend Analysis By Device Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Device Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Reprocessed Medical Devices
- Cardiovascular Devices
- Electrophysiology Devices
- Diagnostic Catheters
- Ablation Catheters
- Interventional Cardiology Devices
- Balloon Catheters
- Guiding Catheters
- Gastroenterology Devices
- Endoscopy Devices
- Endoscopes
- Biopsy Forceps
- General Surgery Devices
- Laparoscopic Devices
- Trocars
- Graspers
- Reprocessed Medical Devices
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Minimally Invasive Procedures
- Cardiac Procedures
- Electrophysiology Procedures
- Angioplasty Procedures
- Gastrointestinal Procedures
- Endoscopy Procedures
- Diagnostic Endoscopy
- Therapeutic Endoscopy
- General Surgery
- Laparoscopic Surgery
- Abdominal Surgery
- Thoracic Surgery
- Orthopedic Surgery
- Joint Procedures
- Knee Surgery
- Hip Surgery
- Urological Procedures
- Urology Treatments
- Stone Management
- Bladder Procedures
- Minimally Invasive Procedures
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Public Hospitals
- Tertiary Care Hospitals
- Specialty Hospitals
- Ambulatory Surgical Centers
- Outpatient Facilities
- Day Care Surgery Centers
- Specialty Clinics
- Diagnostic Centers
- Diagnostic Facilities
- Imaging Centers
- Endoscopy Centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Reprocessing Technology
- Cleaning and Disinfection
- Manual Cleaning
- Automated Cleaning
- Sterilization Methods
- High Temperature Sterilization
- Steam Sterilization
- Dry Heat Sterilization
- Low Temperature Sterilization
- Ethylene Oxide Sterilization
- Hydrogen Peroxide Sterilization
- Reprocessing Technology
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Device Type
- By Product
- By Application
- By End Use
- By Technology
- Competition Analysis
- Competition Deep Dive
- Stryker
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Medline Industries Inc.
- ReNu Medical
- LUMITOS AG
- Vanguard AG
- Johnson & Johnson
- Sterilmed Inc.
- Hygia Health Services Inc.
- Centurion Medical Products Corporation
- Siemens Healthcare
- Philips Healthcare
- GE Healthcare
- Suretech Medical Inc.
- Stryker
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Device Type,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Device Type
- Figure 6: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Product
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Technology
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 22: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Device Type,2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Device Type
- Figure 32: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Device Type,2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Device Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by Product
- Figure 51: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Application
- Figure 54: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by End Use
- Figure 57: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by Technology
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Device Type,2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by Device Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 66: Western Europe Market Attractiveness Analysis by Product
- Figure 67: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 69: Western Europe Market Attractiveness Analysis by Application
- Figure 70: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by End Use
- Figure 73: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Technology
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Device Type,2026 to 2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Device Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Product
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Application
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Device Type,2026 to 2036
- Figure 95: East Asia Market Attractiveness Analysis by Device Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 98: East Asia Market Attractiveness Analysis by Product
- Figure 99: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 101: East Asia Market Attractiveness Analysis by Application
- Figure 102: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 104: East Asia Market Attractiveness Analysis by End Use
- Figure 105: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 107: East Asia Market Attractiveness Analysis by Technology
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Device Type,2026 to 2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Device Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Device Type,2026 to 2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Device Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis