Prefilled Auto Injectors Market Size, Share, Growth and Forecast (2026 - 2036)
Prefilled Auto Injectors Market is segmented by Mechanism (Spring-Based, Gas/Other, Disposable Wearable & Others), Therapy (Autoimmune, Anaphylaxis, Oncology & Others), End User (At-Home/Self, Clinics, Hospitals), and Region. Forecast for 2026 to 2036.
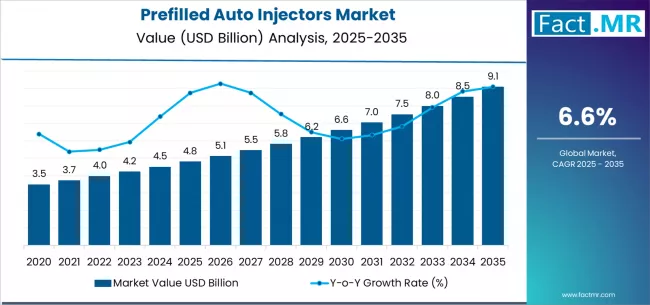
FACT.MR opines the prefilled auto injectors market was valued at USD 4.80 billion in 2025. Sales are expected to reach USD 5.12 billion in 2026 and USD 9.70 billion by 2036. Spring-Based is poised to lead by Mechanism with 69.0% share in 2026.
Prefilled Auto Injectors Market Size, Market Forecast and Outlook By FACT.MR
The prefilled auto injectors market was valued at USD 4.80 billion in 2025, projected to reach USD 5.12 billion in 2026, and is forecast to expand to USD 9.70 billion by 2036 at a 6.6% CAGR. The absolute dollar opportunity of approximately USD 4.58 billion added over the forecast decade reflects the accelerating shift of biologic drug administration from clinical settings to patient self-injection at home. FACT.MR opines that biosimilar market entry for autoimmune therapies and expanding indications for subcutaneous biologics are the two forces sustaining above-average growth in the prefilled auto injector segment.
Pharmaceutical companies developing biologic drugs for autoimmune conditions, oncology supportive care, and anaphylaxis treatment increasingly specify prefilled auto injectors as the primary delivery format for patient self-administration. Device selection decisions during drug development involve rigorous human factors engineering studies to meet FDA and EMA combination product regulatory requirements. Spring-based mechanical actuation dominates the market, though gas-powered and electronic mechanisms are emerging for higher-viscosity biologic formulations. FACT.MR analysts note that the patent cliff for several major biologic drugs is creating biosimilar competition that expands the total addressable device market as multiple manufacturers require auto injector platforms for their respective formulations.
The United States represents the largest market for prefilled auto injectors, where a combination of high biologic drug adoption rates, favorable insurance reimbursement, and patient preference for self-administration sustains demand. Germany and the broader EU market operate under medical device regulation (MDR) frameworks that require notified body certification for combination products. Japan's Pharmaceuticals and Medical Devices Agency (PMDA) maintains separate device registration pathways that add development timeline complexity for global launches. Based on FACT.MR's analysis, India's expanding biosimilar manufacturing capacity is creating a cost-accessible growth corridor for auto injector device platforms. China's biologics market is rapidly adopting subcutaneous self-injection formats as healthcare system reforms encourage outpatient treatment pathways.
Prefilled Auto Injectors Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 5.12 billion |
| Industry Value (2036) | USD 9.70 billion |
| CAGR (2026 to 2036) | 6.6% |
Prefilled Auto Injectors Market Definition
Prefilled auto injectors are single-use or reusable medical devices pre-loaded with a measured dose of pharmaceutical medication, designed to enable patients or caregivers to self-administer subcutaneous or intramuscular injections outside clinical settings. The devices incorporate automated needle insertion, dose delivery, and needle retraction mechanisms. Primary therapeutic applications include autoimmune disease management, anaphylaxis emergency treatment, and oncology supportive care.
Prefilled Auto Injectors Market Inclusions
Market scope covers global and regional sales volumes and values for prefilled auto injectors, forecast period from 2026 to 2036, segment breakdowns by mechanism, therapy area, and end user. Combination product regulatory analysis and human factors engineering trend assessments are included.
Prefilled Auto Injectors Market Exclusions
The scope excludes manually operated prefilled syringes without auto-injection mechanisms, insulin pen injectors, infusion pumps, and implantable drug delivery systems. Vial-based drug reconstitution devices and traditional hypodermic syringes are excluded.
Prefilled Auto Injectors Market Research Methodology
- Primary Research: Analysts engaged with pharmaceutical device development leads, contract device manufacturing executives, and specialty pharmacy procurement managers across 30 countries.
- Desk Research: Data collection aggregated FDA combination product guidance documents, EMA device regulation frameworks, and pharmaceutical pipeline databases for injectable biologic drugs.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of device shipment data linked to biologic drug prescription volumes, applying therapy area growth curves and biosimilar market entry projections.
- Data Validation and Update Cycle: Projections are cross-validated against publicly reported pharmaceutical company device sourcing agreements and contract manufacturing revenue disclosures.
Summary of Prefilled Auto Injectors Market
- Prefilled Auto Injectors Market Definition
- Prefilled auto injectors encompass single-use or reusable devices pre-loaded with pharmaceutical medication for patient self-administration of subcutaneous or intramuscular injections, used primarily in autoimmune disease management, anaphylaxis treatment, and oncology supportive care.
- Demand Drivers in the Market
- Pharmaceutical companies developing biologic drugs for chronic conditions specify auto injectors as the preferred self-administration format, linking device demand directly to biologic prescription growth.
- Biosimilar market entry for major autoimmune biologic drugs expands the total addressable device market as each new manufacturer requires a compatible auto injector platform.
- Healthcare system cost optimization efforts globally encourage self-injection at home over clinic-administered infusion, shifting biologic delivery formats toward patient-operable devices.
- Key Segments Analyzed in the FACT.MR Report
- Spring-Based mechanism holds 69.0% share in 2026, reflecting proven reliability, lower manufacturing complexity, and established regulatory acceptance for combination product approvals.
- Autoimmune therapy accounts for 56.0% of therapy-area demand, anchored by subcutaneous biologic treatments for rheumatoid arthritis, psoriasis, and inflammatory bowel disease.
- At-Home/Self administration represents 72.0% of end-user demand, reflecting the structural shift from clinic-based infusion to patient self-injection for chronic disease management.
- Analyst Opinion at FACT.MR
- FACT.MR analysts observe that the prefilled auto injector market operates at the intersection of pharmaceutical pipeline expansion and medical device engineering, where device platform selection during drug development creates multi-year supply commitments. Manufacturers that cannot demonstrate human factors validation data compliant with both FDA and EU MDR combination product requirements face exclusion from pharmaceutical partnership opportunities. The biosimilar wave for autoimmune biologics amplifies total device demand but simultaneously introduces pricing pressure as multiple device platforms compete for formulary inclusion.
- Strategic Implications / Executive Takeaways
- Device manufacturers must invest in platform flexibility to accommodate varying viscosity ranges and fill volumes as pharmaceutical partners expand biologic formulation pipelines.
- Contract device manufacturers should establish dual regulatory submission capabilities for both FDA and EU MDR combination product pathways to serve global pharmaceutical clients.
- Pharmaceutical companies selecting auto injector platforms must prioritize human factors engineering validation early in development to avoid regulatory delays during combination product submission.
- Methodology
- Primary Research: FACT.MR conducted interviews with device development leads, contract manufacturers, and specialty pharmacy procurement managers across 30 countries.
- Desk Research: Analysts aggregated FDA guidance documents, EMA device regulations, and pharmaceutical pipeline databases for injectable biologics.
- Market-Sizing and Forecasting: Models apply a bottom-up methodology linking device shipments to biologic prescription volumes and biosimilar market entry projections.
Prefilled Auto Injectors Market Key Takeaways
| Metric | Value |
|---|---|
| Estimated Value in (2026E) | USD 4.8 billion |
| Forecast Value in (2036F) | USD 9.1 billion |
| Forecast CAGR (2026 to 2036) | 6.6% |
From 2030 to 2036, the market is forecast to grow from USD 6.7 billion to USD 9.1 billion, adding another USD 2.4 billion, which constitutes 55.8% of the overall ten-year expansion. This period is expected to be characterized by the expansion of connected device systems, the integration of digital health platforms, and the development of wearable injection technologies for chronic disease management. The growing adoption of personalized medicine programs and smart drug delivery initiatives will drive demand for prefilled auto injectors with enhanced connectivity capabilities and improved patient monitoring.
Between 2020 and 2026, the prefilled auto injectors market experienced steady recovery growth, driven by increasing chronic disease awareness and growing recognition of auto injectors as essential tools for patient convenience and medication adherence improvement. The market developed as healthcare providers and patients recognized the potential for automated devices to enhance treatment compliance while reducing injection anxiety and improving self-care capabilities. Technological advancement in injection mechanisms and user interface design began emphasizing the critical importance of maintaining ease of use and safety in patient self-administration operations.
Why is the Prefilled Auto Injectors Market Growing?
Market expansion is being supported by the increasing global chronic disease burden and the corresponding need for convenient drug delivery systems that can maintain treatment adherence and patient safety while supporting diverse therapeutic applications across various healthcare environments.
Modern healthcare providers and patients are increasingly focused on implementing delivery solutions that can improve medication compliance, reduce administration complexity, and provide consistent performance in home healthcare settings. Prefilled auto injectors' proven ability to deliver enhanced patient convenience, reliable drug delivery capabilities, and versatile therapeutic applications make them essential devices for contemporary healthcare operations and patient self-care solutions.
The growing emphasis on home healthcare modernization and patient empowerment is driving demand for prefilled auto injectors that can support chronic disease management, reduce healthcare facility visits, and enable efficient medication administration across varying patient needs. Patient preference for devices that combine safety with convenience and effectiveness is creating opportunities for innovative delivery implementations.
The rising influence of personalized medicine and connected health technologies is also contributing to increased adoption of prefilled auto injectors that can provide advanced monitoring capabilities without compromising performance or patient safety.
Opportunity Pathways - Prefilled Auto Injectors Market
The prefilled auto injectors market is poised for robust growth and transformation. As healthcare providers and patients across both developed and emerging markets seek drug delivery systems that are convenient, safe, connected, and patient-friendly, auto injector systems are gaining prominence not just as medical devices but as strategic solutions for treatment adherence, patient empowerment, healthcare cost reduction, and therapeutic outcome improvement.
Rising chronic disease prevalence and home healthcare adoption in North America, Europe, and Asia Pacific amplify demand, while manufacturers are picking up on innovations in connected devices and smart delivery technologies.
Pathways like connected health integration, wearable systems, and personalized medicine promise strong margin uplift, especially in developed markets. Geographic expansion and therapeutic diversification will capture volume, particularly where chronic disease management is growing or home healthcare infrastructure requires modernization. Patient care pressures around treatment adherence, safety optimization, convenience enhancement, and outcome improvement give structural support.
- Pathway A - Connected & Smart Injection Systems. Healthcare providers increasingly require devices with digital connectivity and data monitoring capabilities. Device manufacturers who specialize in IoT integration or enhance smart capabilities can command a premium. Expected revenue pool: USD 1.2-1.6 billion
- Pathway B - Wearable & Patch Delivery Systems. Advanced delivery systems -- wearable patches, continuous administration, automated dosing - improve patient convenience and treatment outcomes. Opportunity: USD 1.0-1.4 billion
- Pathway C - Personalized Medicine Integration. Customized dosing, patient-specific devices, and therapy optimization enhance treatment effectiveness and patient satisfaction. Systems with stronger personalization capabilities will allow premium positioning. Revenue lift: USD 0.8-1.2 billion
- Pathway D - Specialized Therapeutic Applications. Extending reach into rare diseases, pediatric treatments, and emergency medications. Healthcare systems will look for device suppliers who provide specialized, reliable delivery solutions. Pool: USD 0.9-1.3 billion
- Pathway E - Emerging Market Expansion. Strong growth in developing healthcare markets, chronic disease awareness, and home healthcare adoption. Local partnerships and training programs help penetration. Expected upside: USD 0.6-1.0 billion
- Pathway F - Comprehensive Patient Services. Training programs, adherence monitoring, and patient support services help optimize treatment outcomes and device utilization. Service approaches benefit from long-term patient relationships. USD 0.5-0.9 billion
- Pathway G - Safety Enhancement Technologies. Improved safety features, needle protection, and user-friendly design create differentiation and regulatory compliance advantages. Pool: USD 0.4-0.8 billion
Segmental Analysis
The market is segmented by mechanism, therapy, and end user. By mechanism, the market is divided into spring-based, gas/other, and disposable wearable & others. By therapy, it covers autoimmune, anaphylaxis, and oncology & others. By end user, it is segmented into at-home/self, clinics, and hospitals. Regionally, the market is divided into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa.
By Mechanism, the Spring-Based Segment Accounts for 69.0% Market Share
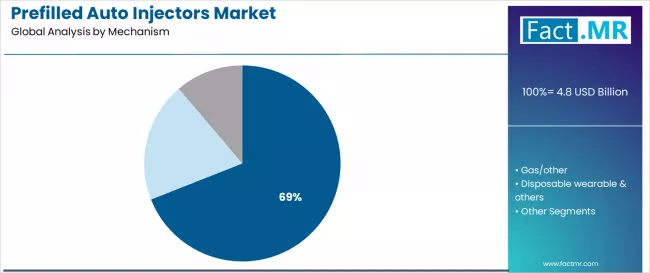
The spring-based mechanism segment is projected to account for 69.0% of the prefilled auto injectors market in 2025, reaffirming its position as the leading mechanism category. Healthcare providers and patients increasingly utilize spring-based auto injectors for their proven reliability, established safety profile, and consistent delivery performance in drug administration across chronic disease management, emergency treatment, and self-care applications. Spring-based technology's established activation procedures and reliable injection output directly address the medical requirements for dependable drug delivery and patient safety in diverse healthcare environments.
This mechanism segment forms the foundation of current auto injector operations, as it represents the technology with the greatest mechanical reliability and established manufacturing infrastructure across multiple applications and therapeutic scenarios. Device investments in enhanced spring systems and activation optimization continue to strengthen adoption among manufacturers and healthcare providers. With users prioritizing device reliability and consistent performance, spring-based mechanisms align with both safety objectives and manufacturing requirements, making them the central component of comprehensive drug delivery strategies.
By Therapy, the Autoimmune Segment Accounts for 56.0% Market Share
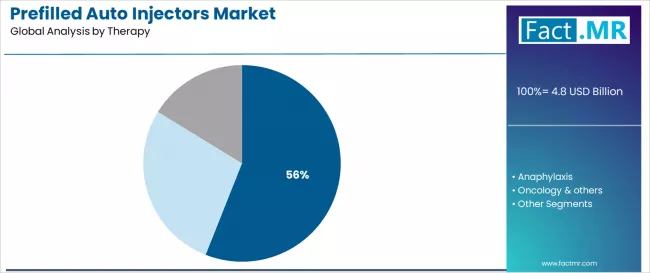
Autoimmune therapy applications are projected to represent 56.0% of prefilled auto injectors utilization in 2025, underscoring their critical role as the primary therapeutic area for chronic disease management and patient self-administration. Healthcare providers prefer prefilled auto injectors for autoimmune conditions due to their treatment consistency, established clinical protocols, and ability to support long-term therapy regimens while maintaining patient compliance and treatment effectiveness requirements. Positioned as essential delivery systems for modern autoimmune care, auto injectors offer both therapeutic advantages and patient care benefits.
The segment is supported by continuous innovation in autoimmune drug development and the growing availability of specialized therapeutic formulations that enable effective treatment with enhanced patient convenience. Additionally, healthcare providers are investing in patient education to support self-administration adoption and treatment optimization. As autoimmune disease prevalence becomes more recognized and treatment requirements increase, autoimmune applications will continue to dominate the therapy market while supporting advanced treatment protocols and patient care strategies.
By End User, the At-Home/Self Segment Accounts for 72.0% Market Share
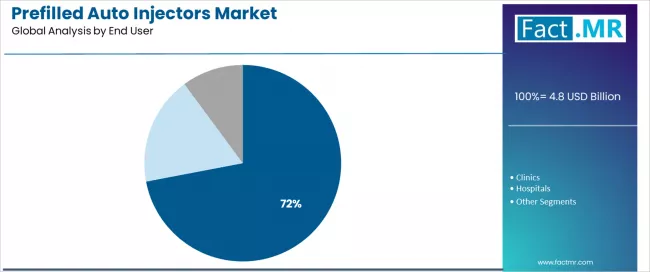
At-home/self administration is projected to represent 72.0% of prefilled auto injectors usage in 2025, underscoring its critical role as the primary administration setting for patient convenience and healthcare cost optimization. Patients prefer at-home auto injector use for their convenience benefits, reduced healthcare facility dependence, and ability to maintain treatment privacy while supporting medication adherence and lifestyle integration requirements. Positioned as essential self-care tools for modern healthcare, at-home devices offer both patient empowerment advantages and cost benefits.
The segment is supported by continuous innovation in device user-friendliness and the growing availability of patient support programs that enable safe self-administration with enhanced confidence building. Additionally, patients are investing in home healthcare optimization to support treatment independence and quality of life improvement. As home healthcare becomes more prevalent and patient empowerment requirements increase, at-home/self administration will continue to dominate the end user market while supporting advanced patient autonomy and healthcare accessibility strategies.
What are the Drivers, Restraints, and Key Trends of the Prefilled Auto Injectors Market?
The prefilled auto injectors market is advancing steadily due to increasing chronic disease prevalence and growing adoption of patient-centered healthcare infrastructure that provides enhanced treatment convenience and medication adherence across diverse therapeutic applications. However, the market faces challenges, including device development costs, regulatory approval complexities, and varying patient acceptance across different demographic environments. Innovation in connected health technologies and user interface design continues to influence device development and market expansion patterns.
Rising Global Chronic Disease Burden and Patient Empowerment
The growing expansion of chronic disease management and patient-centered care is enabling device manufacturers to develop prefilled auto injector systems that provide superior convenience, enhanced safety, and reliable performance in home healthcare environments. Advanced delivery systems provide improved patient capacity while allowing more effective treatment administration and consistent therapeutic outcomes across various applications and patient requirements. Manufacturers are increasingly recognizing the competitive advantages of modern delivery capabilities for patient satisfaction and healthcare provider positioning.
Connected Health Integration and Smart Technology Drive Device Innovation
Modern prefilled auto injector manufacturers are incorporating digital connectivity features and smart monitoring systems to enhance treatment adherence, improve patient outcomes, and ensure consistent performance delivery to healthcare stakeholders. These technologies improve treatment management while enabling new applications, including adherence tracking and remote patient monitoring solutions. Advanced technology integration also allows manufacturers to support premium device positioning and healthcare optimization beyond traditional drug delivery device supply.
Analysis of the Prefilled Auto Injectors Market by Key Countries
.webp)
| Country | CAGR (2025-2035) |
|---|---|
| USA | 7.1% |
| Mexico | 6.6% |
| Germany | 6.3% |
| France | 6.2% |
| South Korea | 6.1% |
| United Kingdom | 6.0% |
| Japan | 5.9% |
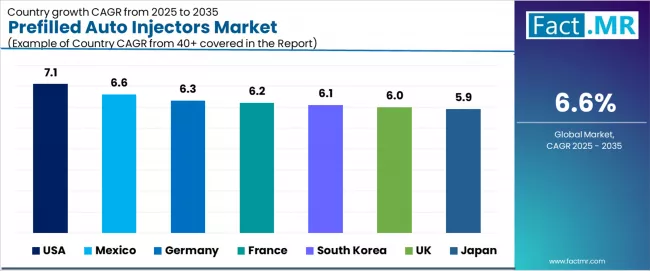
The prefilled auto injectors market is experiencing strong growth globally, with the USA leading at a 7.1% CAGR through 2035, driven by extensive chronic disease management infrastructure, advanced healthcare technology adoption, and significant implementation of connected health solutions. Mexico follows at 6.6%, supported by growing healthcare investment, rapid chronic disease awareness, and growing adoption of patient-centered care technologies. Germany shows growth at 6.3%, emphasizing regulatory excellence and advanced manufacturing capabilities. France records 6.2%, focusing on healthcare innovation and patient care optimization. South Korea demonstrates 6.1% growth, supported by technology advancement and healthcare modernization. United Kingdom holds 6.0%, supported by healthcare system requirements and patient empowerment initiatives. Japan demonstrates 5.9% growth, supported by aging population needs but constrained by healthcare cost pressures.
The report covers an in-depth analysis of 40+ countries; seven top-performing countries are highlighted below.
USA Leads Global Market Growth with Healthcare Technology Infrastructure
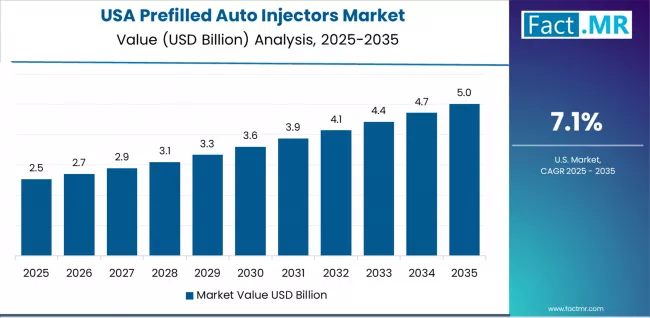
Revenue from prefilled auto injectors in the USA is projected to exhibit exceptional growth with a CAGR of 7.1% through 2035, driven by extensive healthcare technology infrastructure and rapidly growing adoption of connected health solutions supported by patient empowerment initiatives and chronic disease management programs. The country's comprehensive medical device ecosystem and increasing investment in digital health technologies are creating substantial demand for advanced auto injector solutions. Major pharmaceutical companies and healthcare providers are establishing comprehensive delivery systems to serve both patient care operations and treatment adherence requirements.
- Strong pharmaceutical industry presence and healthcare technology leadership is driving demand for sophisticated delivery technologies throughout major healthcare systems and patient care programs.
- Robust chronic disease management infrastructure and expanding network of specialty care providers are supporting the rapid adoption of advanced delivery technologies among providers seeking enhanced patient outcomes and treatment effectiveness.
Mexico Demonstrates Strong Market Potential with Healthcare Infrastructure Development
Revenue from prefilled auto injectors in Mexico is expanding at a CAGR of 6.6%, supported by the country's growing healthcare investment, extensive chronic disease awareness development, and increasing adoption of patient-centered care systems. The country's comprehensive healthcare modernization and expanding pharmaceutical access are driving sophisticated delivery requirements. Healthcare providers and pharmaceutical companies are establishing extensive distribution operations to address the growing demand for convenient drug delivery and treatment accessibility.
- Rising healthcare infrastructure investment and expanding pharmaceutical access are creating opportunities for device adoption across hospitals, clinics, and patient care programs in major metropolitan regions.
- Growing government focus on chronic disease management and healthcare modernization is driving adoption of advanced delivery technologies among providers seeking enhanced patient care capabilities and treatment outcomes.
Europe Market Split by Countries
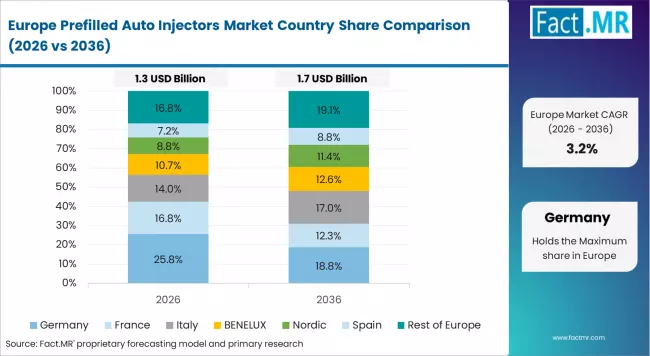
The prefilled auto injectors market in Europe is projected to grow from USD 1.4 billion in 2025 to USD 2.5 billion by 2035, registering a CAGR of 6.0% over the forecast period. Germany is expected to maintain its leadership position with a 28.6% market share in 2025, increasing to 29.2% by 2035, supported by its advanced pharmaceutical infrastructure, comprehensive regulatory capabilities, and major manufacturing facilities serving European and international markets.
France follows with a 21.4% share in 2025, projected to ease to 20.9% by 2035, driven by healthcare innovation programs, patient care initiatives, and established pharmaceutical capabilities, but facing challenges from competitive pressures and healthcare cost constraints. United Kingdom holds a 18.7% share in 2025, expected to decline to 18.3% by 2035, supported by healthcare system requirements and patient empowerment programs but facing challenges from regulatory changes and market competition. Italy commands a 14.3% share in 2025, projected to reach 14.5% by 2035, while Spain accounts for 10.5% in 2025, expected to reach 10.6% by 2035. The Rest of Europe region, including Nordic countries, Eastern European markets, Netherlands, Belgium, and other European countries, is anticipated to gain momentum, expanding its collective share from 6.5% to 6.5% by 2035, attributed to increasing healthcare development across Nordic countries and growing chronic disease management across various European markets implementing patient care enhancement programs.
Competitive Landscape of the Prefilled Auto Injectors Market
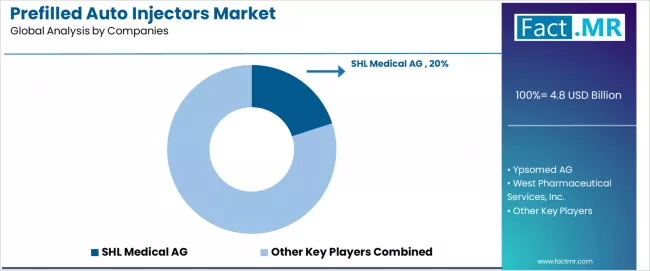
The prefilled auto injectors market is characterized by competition among established medical device manufacturers, specialized drug delivery companies, and integrated pharmaceutical service providers. Companies are investing in advanced mechanism research, connected health development, user experience optimization, and comprehensive device portfolios to deliver convenient, safe, and technologically integrated auto injector solutions. Innovation in digital connectivity, patient interface design, and therapeutic specialization is central to strengthening market position and competitive advantage.
SHL Medical leads the market with a 20.0% market share, offering specialized auto injector devices with a focus on manufacturing excellence and customizable design for pharmaceutical operations. Ypsomed provides comprehensive drug delivery portfolios with an emphasis on innovation, patient-centricity, and European market services. West delivers diverse pharmaceutical packaging solutions with a focus on safety and global manufacturing capabilities. Gerresheimer specializes in pharmaceutical packaging with emphasis on glass and plastic technologies. BD focuses on medical technology with global healthcare leadership and large-scale production capabilities. Owen Mumford offers specialized delivery devices with emphasis on user-friendly design and safety features. Haselmeier provides auto injector solutions with pharmaceutical industry partnerships and customization expertise.
Key Players in the Prefilled Auto Injectors Market
- SHL Medical AG
- Ypsomed AG
- West Pharmaceutical Services Inc.
- Gerresheimer AG
- Becton, Dickinson and Company (BD)
- Owen Mumford Ltd.
- Haselmeier GmbH
- Nemera SA
- AptarGroup Inc.
- Phillips-Medisize Corporation
Bibliography
- United States Food and Drug Administration. (2024). Guidance for industry: Human factors studies and related clinical study considerations in combination product design and development. FDA.
- European Medicines Agency. (2024). Guideline on the quality requirements for combination products. EMA.
- International Society for Pharmaceutical Engineering. (2024). ISPE good practice guide: Combination products. ISPE.
- World Health Organization. (2024). Prequalification of medicines: Injectable delivery devices technical specifications. WHO.
- Japan Pharmaceuticals and Medical Devices Agency. (2024). Guidance on combination product review. PMDA.
- European Commission. (2024). Medical Devices Regulation (EU) 2017/745: Implementation status report. EC.
This bibliography is provided for reader reference. The full FACT.MR report contains the complete reference list with primary research documentation.
This Report Addresses
- Market sizing and quantitative forecast metrics detailing global prefilled auto injector sales volumes and values through 2036.
- Segmentation analysis mapping adoption patterns across mechanisms, therapy areas, and end-user categories.
- Regional deployment intelligence comparing US biologic self-injection adoption against emerging market biosimilar device growth.
- Regulatory compliance assessment analyzing FDA and EU MDR combination product requirements and human factors validation standards.
- Competitive landscape evaluation tracking market positioning of major device manufacturers and their pharmaceutical partnership portfolios.
- Strategic platform engineering guidance identifying viscosity range, fill volume, and connectivity investment priorities.
- Pharmaceutical pipeline analysis identifying upcoming biologic drug launches that will drive new device platform demand.
- Custom data delivery formats encompassing interactive dashboards, raw Excel datasets, and comprehensive PDF narrative reports.
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 5.12 billion to USD 9.70 billion, at a CAGR of 6.6% |
| Market Definition | Prefilled auto injectors encompass single-use or reusable devices pre-loaded with pharmaceutical medication for patient self-administration of subcutaneous or intramuscular injections, used primarily in autoimmune disease management, anaphylaxis treatment, and oncology supportive care. |
| Mechanism Segmentation | Spring-Based, Gas/Other, Disposable Wearable & Others |
| Therapy Segmentation | Autoimmune, Anaphylaxis, Oncology & Others |
| End User Segmentation | At-Home/Self, Clinics, Hospitals |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | India, China, USA, Canada, UK, Germany, Japan, and 40 plus countries |
| Key Companies Profiled | SHL Medical AG, Ypsomed AG, West Pharmaceutical Services Inc., Gerresheimer AG, Becton, Dickinson and Company (BD), Owen Mumford Ltd., Haselmeier GmbH, Nemera SA, AptarGroup Inc., Phillips-Medisize Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with device shipment data linked to biologic drug prescription volumes, integrating therapy area growth curves, biosimilar entry projections, and combination product regulatory timelines. |
Prefilled Auto Injectors Market by Segments
-
Mechanism :
- Spring-based
- Gas/other
- Disposable wearable & others
-
Therapy :
- Autoimmune
- Anaphylaxis
- Oncology & others
-
End User :
- At-home/self
- Clinics
- Hospitals
-
Region :
-
North America
- United States
- Canada
- Mexico
-
Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
-
Asia Pacific
- China
- Japan
- India
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
-
Latin America
- Brazil
- Chile
- Rest of Latin America
-
Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
-
- Frequently Asked Questions -
How large is the demand for prefilled auto injectors in the global market in 2026?
Demand for prefilled auto injectors in the global market is estimated to be valued at USD 5.12 billion in 2026.
What will be the market size of prefilled auto injectors by 2036?
Market size for prefilled auto injectors is projected to reach USD 9.70 billion by 2036.
What is the expected demand growth for prefilled auto injectors between 2026 and 2036?
Demand for prefilled auto injectors is expected to grow at a CAGR of 6.6% between 2026 and 2036.
Which mechanism is poised to lead by 2026?
Spring-Based accounts for 69.0% share in 2026.
How significant is the role of Autoimmune in driving prefilled auto injectors demand?
Autoimmune represents 56.0% of therapy demand in 2026.
What end user segment leads in 2026?
At-Home/Self accounts for 72.0% of end user demand in 2026.
What is the prefilled auto injectors market and what is it mainly used for?
Refer to the market definition section for a complete description of product scope and primary applications.
What is included in the scope of this report?
The market covers global and regional sales volumes and values, segment breakdowns, and forecast projections from 2026 to 2036.
What is excluded from the scope of this report?
Refer to the market exclusions section for specific product types and applications that fall outside the analytical boundaries.
How does FACT.MR build and validate this forecast?
Forecasting models apply a bottom-up methodology and cross-validate projections against publicly reported data from industry sources and regulatory agencies.
Does the report cover USA in its regional analysis?
Yes, the USA is included within North America under the regional scope of analysis.
Does the report cover Germany in its regional analysis?
Yes, Germany is included within Europe under the regional scope of analysis.
Does the report cover India in its regional analysis?
Yes, India is included within South Asia under the regional scope of analysis.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Mechanism
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Mechanism , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Mechanism , 2026 to 2036
- Spring-Based
- Gas/Other
- Disposable Wearable & Others
- Spring-Based
- Y to o to Y Growth Trend Analysis By Mechanism , 2021 to 2025
- Absolute $ Opportunity Analysis By Mechanism , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapy
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapy, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapy, 2026 to 2036
- Autoimmune
- Anaphylaxis
- Oncology & Others
- Autoimmune
- Y to o to Y Growth Trend Analysis By Therapy, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapy, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- At-Home/Self
- Clinics
- Hospitals
- At-Home/Self
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Mechanism
- By Therapy
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Mechanism
- By Therapy
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Mechanism
- By Therapy
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Mechanism
- By Therapy
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Mechanism
- By Therapy
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Mechanism
- By Therapy
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Mechanism
- By Therapy
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Mechanism
- By Therapy
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Mechanism
- By Therapy
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Mechanism
- By Therapy
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Mechanism
- By Therapy
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Mechanism
- By Therapy
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Mechanism
- By Therapy
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Mechanism
- By Therapy
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Mechanism
- By Therapy
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Mechanism
- By Therapy
- By End User
- Competition Analysis
- Competition Deep Dive
- SHL Medical AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Ypsomed AG
- West Pharmaceutical Services Inc.
- Gerresheimer AG
- Becton, Dickinson and Company (BD)
- Owen Mumford Ltd.
- Haselmeier GmbH
- Nemera SA
- AptarGroup Inc.
- Phillips-Medisize Corporation
- SHL Medical AG
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Mechanism , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Therapy, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Mechanism , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Therapy, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Mechanism , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Therapy, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Mechanism , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Therapy, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Mechanism , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Therapy, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Mechanism , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Therapy, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Mechanism , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Therapy, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Mechanism , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Therapy, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Mechanism , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Mechanism , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Mechanism
- Figure 6: Global Market Value Share and BPS Analysis by Therapy, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Therapy, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Therapy
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Mechanism , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Mechanism , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Mechanism
- Figure 26: North America Market Value Share and BPS Analysis by Therapy, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Therapy, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Therapy
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Mechanism , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Mechanism , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Mechanism
- Figure 36: Latin America Market Value Share and BPS Analysis by Therapy, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Therapy, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Therapy
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Mechanism , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Mechanism , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Mechanism
- Figure 46: Western Europe Market Value Share and BPS Analysis by Therapy, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Therapy, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Therapy
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Mechanism , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Mechanism , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Mechanism
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Therapy, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Therapy, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Therapy
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Mechanism , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Mechanism , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Mechanism
- Figure 66: East Asia Market Value Share and BPS Analysis by Therapy, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Therapy, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Therapy
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Mechanism , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Mechanism , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Mechanism
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Therapy, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Therapy, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Therapy
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Mechanism , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Mechanism , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Mechanism
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Therapy, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Therapy, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Therapy
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis