Prefilled Syringes Market Size, Share, Growth and Forecast (2026 - 2036)
The global prefilled syringes market is segmented by Product Type (Conventional Prefilled Syringes and Safety Prefilled Syringes), Material (Glass and Plastic), Design (Single-Chamber, Dual-Chamber, and Customized / Specialty), Application (Diabetes, Rheumatoid Arthritis & Autoimmune Diseases, Oncology, and Others), Route of Administration (Subcutaneous (SC), Intramuscular (IM), and Intravenous (IV)), End User (Hospitals, Clinics, and others), and Region (North America, Latin America, East Asia, South Asia-Pacific, Western Europe, Eastern Europe, Middle East & Africa), with forecasts covering the period from 2026 to 2036.
Prefilled Syringes Market Forecast and Outlook By Fact.MR
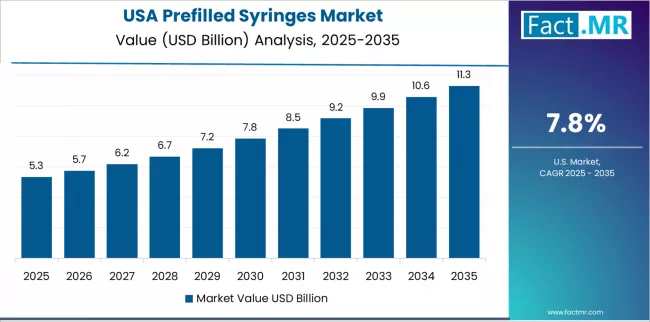
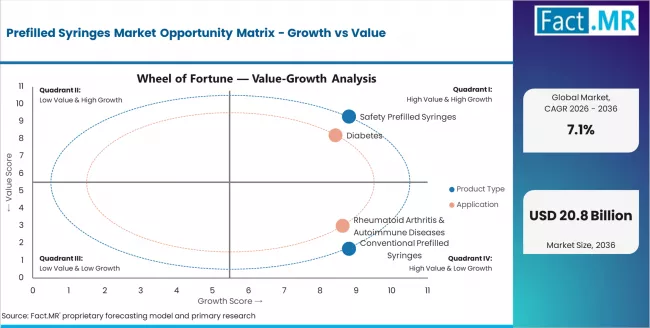
In 2025, the Prefilled Syringes market was valued at USD 9.8 billion. Based on Fact.MR analysis, demand for prefilled syringes is estimated to grow to USD 10.5 billion in 2026 and USD 20.8 billion by 2036. Fact.MR projects a CAGR of 7.1% during the forecast period.
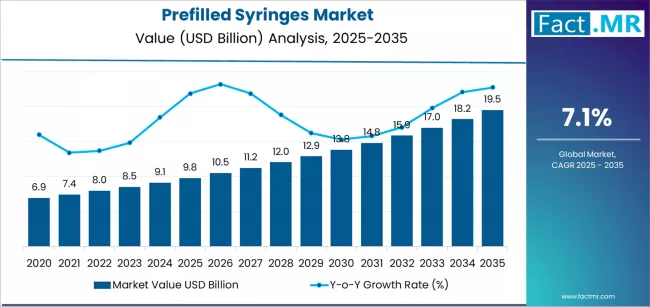
This USD 11.1 billion increase reflects steady, incremental growth rather than transformational expansion, supported by rising adoption of biologics and increased shift toward self-administration. Growth remains moderated by structural constraints such as high regulatory and sterility compliance costs, fill-finish capacity limitations, material supply sensitivities (glass and advanced polymers), and substitution pressure from autoinjectors and other advanced drug delivery systems. As Tom Polen, Chairman, CEO, and President of Becton, Dickinson and Company, noted regarding hospital efficiency and technology adoption, 'solutions that drive automation and efficiency' help providers manage costs.
Summary of Prefilled Syringes Market
- Market Snapshot
- Global prefilled syringes market revenue stood at USD 9.8 billion in 2025 and is forecast to reach USD 20.8 billion by 2036.
- At a 7.1% CAGR from 2025 to 2036, this market is set to expand 2.0x in value, adding USD 9.7 billion in absolute opportunity.
- Growth is being driven by increasing demand for advanced drug delivery solutions and rising adoption in pharmaceutical manufacturing systems.
- Prefilled syringes are gaining traction as efficient drug delivery systems, improving patient safety, dosing accuracy, and reducing administration complexity.
- Demand and Growth Drivers
- Increasing demand for drug delivery solutions across pharmaceutical and healthcare sectors is driving adoption.
- Rising focus on patient safety optimization and reduction of medication errors is accelerating market growth.
- Growing penetration of prefilled syringes in specialized healthcare applications is expanding the addressable market.
- Expansion of pharmaceutical manufacturing capabilities and advanced drug delivery technologies is supporting long-term growth.
- Product and Segment View
- Safety Prefilled Syringes holds 63.4% of material share in 2026, emerging as the leading segment due to strong compatibility and regulatory acceptance.
- Key product categories include:
- Glass Prefilled Syringes
- Plastic Prefilled Syringes
- Geography and Competitive Outlook
- Growth is supported across major regions including North America, Europe, and Asia Pacific.
- Market expansion is closely tied to:
- Increasing pharmaceutical production
- Expansion of healthcare infrastructure
- Rising adoption of advanced drug delivery systems
- Key companies active in this market include: Gerresheimer, SCHOTT, BD, Nipro, West Pharma, Terumo, Stevanato, Weigao, Ompi.
Prefilled Syringes Market — At a Glance
| Attribute | Details |
|---|---|
| Market Value 2025 | USD 9.8 billion |
| Market Value 2036 | USD 20.8 billion |
| Absolute Dollar Opportunity 2025-2036 | USD 11.1 billion |
| Total Growth 2025-2036 | 99.0% |
| CAGR 2026-2036 | 7.1% |
| Growth Multiple | ~2.0x |
| Key Demand Theme | Growing adoption of advanced, safe, and efficient drug delivery systems |
| Leading Segment by Material (2025) | Glass |
| Segment Share (2025) | 72.0% |
| Key Growth Regions | North America, Europe, Asia Pacific |
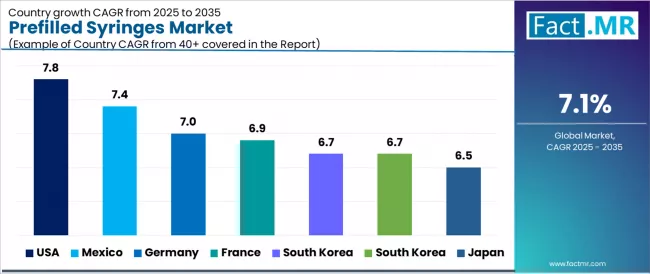
| Country CAGRs | U.S. (6.4%), China (8.6%), India (10.9%), Germany (4.4%), France (5.4%), U.K. (5.1%), Canada (7.2%) |
| Top Companies | Gerresheimer, SCHOTT, BD, Nipro, West Pharma, Terumo, Stevanato, Weigao, Ompi |
| Segmentation by Material | Glass, Plastic |
| Segmentation by Region | North America, Latin America, Western Europe, Eastern Europe, East Asia, south Asia and ASEAN, Middle East & Africa |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research
Segmental Analysis
Prefilled Syringes Market Analysis by Product Type
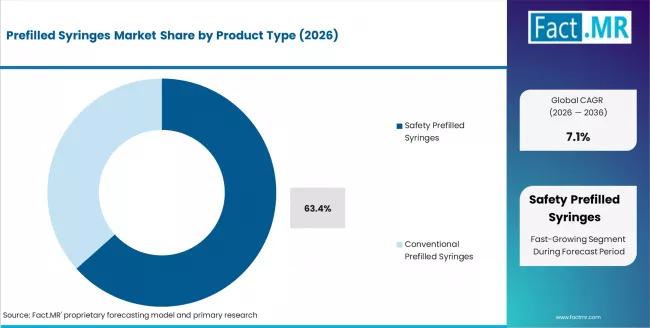
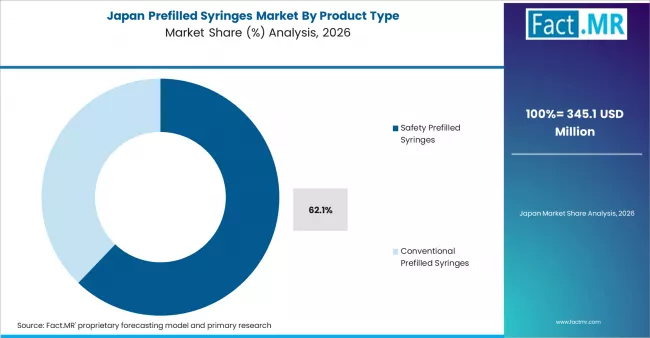
Based on Fact.MR's Prefilled Syringes market report, consumption of safety prefilled syringes holds 63.4% share in 2025. The reason behind the dominance is that it has natural safeguards against needlesticks. This is one of the most important factors when it comes to regulation and operations in the healthcare sector. It is also more structurally sound as it helps maintain safety standards without any liabilities.
- Capacity Investment: Becton, Dickinson and Company (BD) announced an USD 110 million investment in August 2026 to expand its U.S. manufacturing footprint for prefillable syringes, including BD Neopak production in Nebraska [2]. This investment highlights ongoing efforts to strengthen domestic supply chains and meet rising demand for biologics-compatible delivery systems.
- Technology Advancement: In January 2026, BD and Ypsomed expanded their partnership to develop a 5.5 mL BD Neopak XtraFlow glass prefillable syringe compatible with the YpsoMate autoinjector platform, specifically targeting large-volume biologics [3]. The system incorporates thinner cannulas and shorter needles to improve flow and injection time, addressing limitations of conventional syringe formats.
- Supply Chain Expansion: Gerresheimer is expanding its Wertheim site with a new production facility for ready-to-fill pharmaceutical containers, strengthening its capacity to serve global pharma customers and biologics demand [4]. This reflects a broader industry shift toward scaling high-quality primary packaging infrastructure to support long-term supply agreements.
Prefilled Syringes (PFS) Production and Supply
Prefilled syringe production in the world is being carried out more by a consortium of large pharmaceuticals CDMOs and special device producers with the top 10 manufacturers controlling around 55% of total capacity. Capital investment in manufacturing is huge due to aseptic filling lines, which costs between USD 8-15 million per line depending on automation and size of the machine (nest-and-tub vs. bulk). Glass prefilled syringes remain the dominant form of delivery, but polymeric forms are growing in significance due to breakable nature and biological products compatibility.
The manufacturing process of prefilled syringes is very complex, and there is no single player involved; instead, there are primary packaging makers (barrel, plunger, stopper), fill-finish companies, and pharmaceutical companies working in a synchronized production cycle. As indicated in the survey by Fact.MR, post-COVID situation, lead times in PFS manufacturing are around 4-6 months although capacity tightness still occurs because of biological demand growth. North America and Europe contribute around 60% to worldwide production, but now Asia (China and India) is becoming a fast-growing region, with 10-15% yearly capacity increase.
Regulatory Complexity & Approval Timelines (US FDA, EMA, CDSCO)
Regulatory pathways are one of the top barriers to success in the prefilled syringes (PFS) market, affecting product time-to-market, cost structures, and partnerships. For instance, in the U.S., the PFS should be approved by the USFDA, being a combination of drug and device, therefore following strict guidelines related to both CDER and CDRH. Typically, such approval requires 12–18 months, which can be even prolonged for biologic drugs due to the necessity of providing stability studies and E&L data.
In Europe, the PFS is required to go through the central procedure regulated by the European Medicines Agency. However, the variability associated with the presence of device-related parts leads to extended approval processes for an average of 12–24 months. The new Medical Device Regulation (MDR) is expected to bring additional challenges, including increased documentation requirements and higher costs, accounting for approximately 15–25%. The Indian regulator of prefilled syringes, the Central Drugs Standard Control Organisation- provides faster paths to approval, up to 6–12 months. However, more stringent demands can be expected concerning locally manufactured biologic drugs and those targeting international markets. According to the study, more than 65% of the market's producers view regulatory alignment as one of the biggest obstacles on their way to the global stage.
Prefilled Syringes Market Analysis by Application
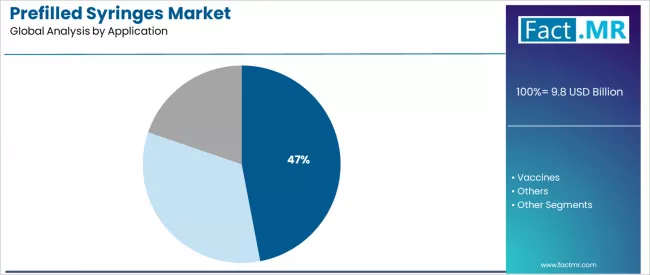
Based on Fact.MR's Prefilled Syringes market report, consumption of diabetes is estimated to hold around 41.2% share in 2025. In this regard, this segment dominates because of the recurrent and high requirement for insulin shots, which demand a handy and accurate way of administering the drug. One of the structures that can facilitate this process is the pre-filled syringe, which is structured to solve this problem efficiently.
- Demand for Diabetic Care: As there is an increased presence of diabetes globally, the demand for prefilled syringes is being fueled because patients have to take frequent doses of the medicine. This makes it necessary for them to administer insulin injections regularly, which are easy with prefilled syringes.
- Patient Preference for Self-Administration: Since more and more people want self-administration for their diseases, it is leading to greater adoption of prefilled syringes. There is greater convenience associated with these as patients have to go through less trouble of getting trained on their use.
- Compatibility with Biologics: Injectable biologic drugs have been introduced as treatments for metabolic diseases, and hence there is a growing need for compatibility with such medicines.
Prefilled Syringes Market Analysis by Material
Glass is projected to account for 72.0% of the prefilled syringes market in 2025 by material, reaffirming its position as the category's dominant material type. Medical device manufacturers increasingly recognize the optimal balance of performance and cost-effectiveness offered by glass systems for most pharmaceutical applications, particularly in drug delivery and healthcare optimization processes.
Glass forms the foundation of most pharmaceutical protocols for healthcare applications, as it represents the most widely accepted and commercially viable level of drug delivery technology in the industry. Performance control standards and extensive medical testing continue to strengthen confidence in glass formulations among pharmaceutical and healthcare providers.
With increasing recognition of the cost-performance optimization requirements in pharmaceutical production, glass systems align with both operational efficiency and drug delivery reliability goals, making them the central growth driver of comprehensive healthcare strategies.
Regional Analysis
The report covers North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and the Middle East & Africa. Geographic segmentation of the market can be done considering regional consumption trends, healthcare framework, and regulatory scenarios. In addition to that, the report includes the market attractiveness study.
.webp)
CAGR Table Prefilled Syringes Market
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 8.6% |
| India | 10.9% |
| Germany | 4.4% |
| Brazil | 7.4% |
| USA | 6.4% |
| U.K. | 5.1% |
| Japan | 6.6% |
North America
This is one of the leading markets in the application of advanced drug delivery, owing to the presence of robust pipelines of biologics and stringent safety norms. Some of the prominent companies in this market include Becton, Dickinson and Company – market leader with diversified range of prefillable syringes and safety systems, West Pharmaceutical Services, Inc., key player offering quality injectables and containment solutions, Catalent, Inc.
- United States: Demand for prefilled syringes in the United States is projected to rise at 6.4% CAGR through 2036. Growth is supported by continued regulatory momentum, including FDA approvals of new biologics in prefilled syringe formats (multiple approvals, 2025), reinforcing adoption of ready-to-use delivery systems.
- Canada: Demand for prefilled syringes in Canada is projected to rise at 7.2% CAGR through 2036. Growth is mainly attributed to the increase in utilization of biologics and preference for self-injection by patients suffering from chronic diseases. Other factors contributing to the growth include harmonization of Canada's device standards with international standards and stable public health-care procurement systems in Canada despite slow growth because of an established healthcare system.
Western Europe
The region plays a central role in setting stringent safety and quality standards for prefilled syringes, driven by strong regulatory frameworks and advanced pharmaceutical manufacturing. Key players include Gerresheimer AG, a leader in high-quality glass and polymer syringe systems, SCHOTT Pharma AG, known for premium drug containment solutions for biologics, and Stevanato Group, which holds a strong position in integrated drug delivery and fill-finish solutions.
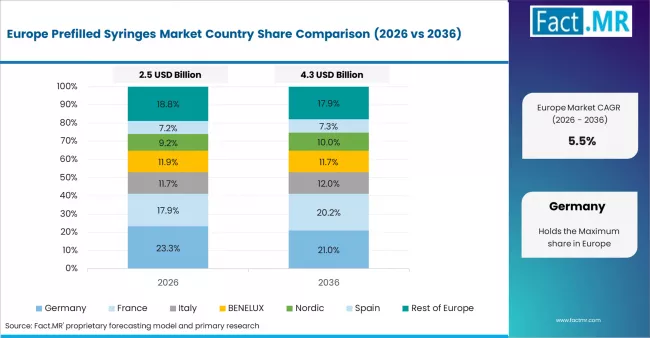
- Germany: Demand for prefilled syringes in Germany is projected to rise at 4.4% CAGR through 2036. Growth is supported by the country’s Statutory Health Insurance (SHI) system, which continues to reimburse advanced injectable therapies and associated delivery formats, ensuring steady adoption of prefilled systems. EU Medical Device Regulation (MDR) updates impacting Germany (March 2026) are reinforcing stricter safety and quality requirements, driving preference for high-quality, compliant prefilled syringe systems.
- United Kingdom: Demand for prefilled syringes in the United Kingdom is projected to rise at 5.1% CAGR through 2036. Growth is driven by public healthcare deployment, including NHS Accelerated Access Collaborative programs that fast-track adoption of advanced drug delivery technologies. UK government updates to medical device regulations aligned with EU frameworks (March 2026) are strengthening compliance requirements, supporting uptake of safer and more efficient prefilled systems.
- France: Demand for prefilled syringes in France is projected to rise at 5.4% CAGR through 2036. Expansion will be driven by robust reimbursement structures for biologics and injectables within France’s national healthcare system, promoting usage of cutting-edge dosage forms. The adherence of manufacturers to the scientific guidelines of the European Medicines Agency (EMA) with respect to drug/device combinations is expected to continue to drive growth in the market.
South Asia and ASEAN
India leads with a CAGR of 10.9%, driven by rapid expansion of biologics access and a large patient pool, followed by China at 8.6% due to strong domestic manufacturing and rising chronic disease burden, Brazil at 7.4% supported by expanding public healthcare and vaccine demand, and Japan at 6.6% owing to its aging population increasing injectable drug usage, meanwhile, mature markets including the U.S. (6.4%), France (5.4%), the UK (5.1%), and Germany (4.4%) exhibit comparatively slower growth as they are largely driven by replacement demand rather than new adoption, with growth constrained by high regulatory and compliance costs.
According to Fact.MR, the market analysis of prefilled syringes in North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East & Africa comprises individual country analysis including the USA, Canada, Mexico, Brazil, Germany, the UK, France, Russia, China, Japan, India, and GCC countries. The readers will get detailed information regarding the regional demand patterns and growth rate of each individual market segment.
Key Takeaways
- Market Definition: The prefilled syringes market comprises ready-to-use injection devices preloaded with precise doses of medication, designed to improve safety, dosing accuracy, and ease of administration across clinical and homecare settings.
- Demand Drivers:
- Rising use of biologic drugs for chronic conditions such as rheumatoid arthritis and cancer is increasing demand for precise, sterile delivery formats like prefilled syringes.
- Growth in self-administration trends, especially among diabetes and autoimmune patients, is driving adoption of user-friendly, ready-to-inject systems.
- Expansion of global vaccination programs and emergency stockpiling is boosting demand for prefilled formats that reduce preparation time and dosing errors.
- Key Segments Analyzed:
- By Product Type: Safety prefilled syringes lead, accounting for over 60% share due to regulatory push for needlestick injury prevention.
- By Application: Diabetes and autoimmune diseases dominate, supported by high-frequency injection requirements.
- By Geography: North America leads with ~40% share, driven by advanced biologics adoption and healthcare infrastructure.
- Analyst Opinion at Fact.MR:
- Senior Consultants at Fact.MR opines that, 'CXOs will find that long-term value lies in integrating safety features and aligning with biologics-driven demand while managing cost and compliance pressures'.'
- Strategic Implications / Executive Takeaways:
- Invest in safety-engineered and autoinjector-compatible syringe platforms to align with evolving regulatory and patient usability requirements.
- Expand fill-finish and sterile manufacturing capacity to address supply bottlenecks in biologics delivery.
- Strengthen partnerships with pharmaceutical companies to integrate drug-device combination offerings and secure long-term contracts.
- Methodology:
- Data triangulated using primary interviews with industry stakeholders and secondary sources such as company filings and regulatory databases.
- Market estimates validated through cross-verification of production capacities, pricing trends, and regional consumption patterns.
- Forecasts updated periodically based on new product launches, regulatory changes, and shifts in end-use demand.
Drivers, Restraints, and Opportunities
Transformation from Generic to Compliant Driven Industry Unfolds New Opportunity
According to Fact.MR analysts, the global market for prefilled syringes has transformed from a specialized packing type to an organically developing and regulatory-compliant segment amid the larger landscape of the injectable drugs delivery industry. The present market value of this product category is driven by the emergence of biological drugs and treatments for chronic diseases, necessitating high precision, sterility, and repeatability in dosage forms. Moreover, the need for protecting healthcare professionals against needlestick injuries and ensuring the safety of medications has spurred the migration from traditional vial packs to prefilled syringes. Adoption accelerates when pharmaceutical companies shift from vial-based formats to ready-to-use delivery systems to reduce preparation time, contamination risk, and operational complexity.
Fact.MR analysts observe a clear shift from standard, low-cost glass prefilled syringes toward advanced, safety-engineered and biologics-compatible systems. Demand for conventional formats is gradually softening, particularly in mature hospital settings where safety regulations and automation are reducing their relevance. Prefilled syringes face substitution pressure from autoinjectors and pen injectors, particularly in homecare settings requiring ease of use.
On the other hand, there are increased growth being recorded in value-added products like dual chamber syringes, polymeric syringes, and auto-injector compatible syringes due to the application in biological drugs and home care applications. Even though these syringes cost more on a unit basis, they are used in smaller amounts because of their unique applications. As a result, the revenue increase offsets the limited volume increase.
- Biologics Expansion: The fast proliferation of monoclonal antibodies and other biologics products creates more structural demand for syringes that are able to accommodate highly viscous medicines with specific dosing needs. As distinct from small molecules, biologics need special mechanisms for delivery, thus forcing companies to manufacture syringes capable of this task. As a result, the per-unit value grows dramatically.
- Regulatory Safety Mandates: Policies such as the U.S. Needlestick Safety and Prevention Act and the EU Sharps Directive (2010/32/EU) are accelerating adoption of safety-engineered syringes [1]. These regulations require healthcare providers to reduce needle-related injuries, forcing a transition away from conventional syringes. As compliance becomes mandatory, safety syringes command higher pricing and dominate procurement decisions.
- Asia Manufacturing Shift: Developing countries such as China and India have emerged as vital production and consumer centers for several reasons, including cost savings and growth in their respective pharmaceutical markets. The rise in local production facilities and health care accessibility has led to an increase in regional consumption of prefilled syringe systems.
Where Opportunities Underlying in Global Prefilled Syringes Market
The prefilled syringes market represents a specialized growth opportunity, expanding from USD 9.8 billion in 2025 to USD 20.8 billion by 2036 at a 7.1% CAGR. As healthcare providers prioritize patient safety, regulatory compliance, and treatment efficiency in complex drug delivery processes, prefilled syringe systems have evolved from a niche pharmaceutical technology to an essential component enabling healthcare optimization, drug monitoring, and multi-stage delivery across pharmaceutical operations and specialized medical applications. Fill-finish capacity constraints act as a structural bottleneck, creating pricing power for suppliers with sterile manufacturing capabilities.
The convergence of healthcare expansion, increasing drug delivery optimization adoption, specialized pharmaceutical organization growth, and patient safety monitoring requirements creates sustained momentum in demand. High-efficiency formulations offering superior prefilled syringe performance, cost-effective glass material systems balancing performance with economics, and specialized plastic variants for critical applications will capture market premiums, while geographic expansion into high-growth Asian pharmaceutical markets and emerging market penetration will drive volume leadership. Regulatory emphasis on healthcare adoption and drug delivery performance provides structural support.
- Glass Material Dominance: Leading with 72.0% market share, glass materials drive primary demand through complex pharmaceutical workflows requiring specialized prefilled syringe systems for healthcare optimization. Advanced formulations enabling improved drug stability, reduced contamination risks, and enhanced patient safety command premium pricing from healthcare providers requiring stringent performance specifications and regulatory compliance. Expected revenue pool: USD 11.5-15.2 billion.
- Diabetes Market Leadership: Dominating with 41.2% market share through an optimal balance of performance and cost-effectiveness, biologics serve most pharmaceutical applications while meeting prefilled syringe requirements. This specification addresses both performance standards and economic considerations, making it the preferred choice for pharmaceutical production and healthcare operations seeking reliable performance. Opportunity: USD 7.0-9.0 billion.
- American Market Acceleration: USA (6.4% CAGR) and Canada (7.2% CAGR) lead global growth through pharmaceutical infrastructure expansion, drug delivery capability development, and domestic prefilled syringe demand. Strategic partnerships with local pharmaceutical manufacturers, regulatory compliance expertise, and supply chain localization enable the expansion of drug delivery technology in major healthcare hubs. Geographic expansion upside: USD 3.5-5.0 billion.
- Single-chamber Design Growth: Accounting for 74.0% of design demand, single-chamber systems represent high-growth opportunities in standard drug delivery markets. Companies developing advanced single-chamber formulations, supporting pharmaceutical initiatives, and expanding into healthcare-focused markets capture premium demand from pharmaceutical manufacturers and specialized healthcare organizations. Market expansion potential: USD 4.8-6.5 billion.
- European Market Leadership & Supply Chain Excellence: Germany (4.4% CAGR) and U.K. (5.1% CAGR) lead European expansion through specialized distribution networks, strategic inventory management, and reliable supply systems. Companies offering guaranteed supply security, technical support, and regulatory documentation gain preferred supplier status with compliance-focused healthcare providers across developed markets. European opportunity: USD 2.8-4.0 billion.
Competitive Aligners for Market Players
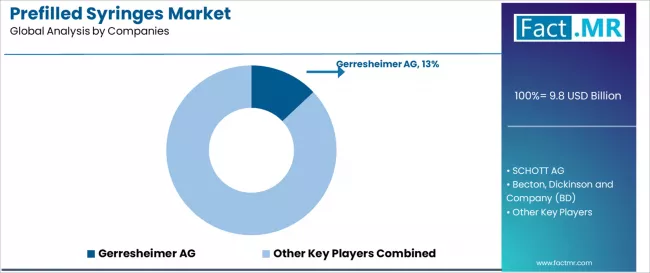
The prefilled syringes market exhibits a moderately consolidated structure, with the top 5 players accounting for an estimated approximately 55% of global revenue. Companies such as Becton, Dickinson and Company, Gerresheimer AG, and SCHOTT Pharma AG enjoy the competitive edge of size and experience in their pharmaceutical partnerships. Quality and compliance emerge as the key factor in competition, followed closely by supply reliability and compatibility with biologicals, not price.
The structural advantage of the industry lies in firms that offer the combination of design and production as well as fill-finish expertise. Such companies as Stevanato Group possess a vertical advantage by having integrated capabilities for packaging and engineering. Companies with stable contracts with major pharmaceutical firms also have the competitive edge due to their assured demand and the difficulty involved in switching, especially when biological products require lengthy and expensive validation processes.
Switching costs are high, and buyers exhibit risk aversion since pharmaceutical firms favor suppliers who have been tested. Buyers can reduce their dependency on suppliers through dual sourcing and long-term contracts. Although this constrains the supplier from increasing prices quickly, suppliers that have shown their ability to comply and possess special skills maintain some degree of pricing power, particularly in the expensive biologics category where there are limited qualified substitutes.
Recent Developments
- Gerresheimer Product & Platform Launch (January 2026): Gerresheimer AG showcased its EZ-fill Smart ready-to-fill platform and Gx Elite syringe systems at Pharmapack 2026, targeting biologics and homecare delivery applications [Gerresheimer].
- Fresenius Kabi expanded its Simplist® portfolio of prefilled syringes in December 2025 with the launch of ephedrine sulfate and succinylcholine chloride injections, extending its use into operating room and anesthesia settings [Fresenius Kabi].
- BD Capacity Investment (August 2025): Becton, Dickinson and Company announced a USD 35 million investment to expand prefilled syringe production at its Nebraska facility, strengthening U.S. supply capacity for injectable therapies [BD].
Key Players in the Global Prefilled Syringes Market
- Becton, Dickinson and Company (BD)
- Gerresheimer AG
- SCHOTT Pharma AG
- West Pharmaceutical Services, Inc.
- Stevanato Group
- Terumo Corporation
- Nipro Corporation
- Baxter International Inc.
- AptarGroup, Inc.
- B. Braun SE
- Catalent, Inc.
- Vetter Pharma International GmbH
- Credence MedSystems, Inc.
- Weigao Group
- Fresenius Kabi
Report Scope

| Metric | Value |
|---|---|
| Quantitative Units | USD 10.5 Bn to USD 20.8 Bn |
| Market Definition | Prefilled syringes are ready-to-use injection devices preloaded with a fixed dose of medication, designed to ensure accurate dosing, reduce contamination risk, and simplify drug administration in clinical and homecare settings. |
| By Product Type | Conventional Prefilled Syringes and Safety Prefilled Syringes |
| By Material | Glass and Plastic |
| By Design | Single-Chamber, Dual-Chamber, and Customized / Specialty |
| By Application | Diabetes, Rheumatoid Arthritis & Autoimmune Diseases, Oncology, Cardiovascular Diseases, Infectious Diseases (Vaccines), and Others |
| By Route of Administration | Subcutaneous (SC), Intramuscular (IM), and Intravenous (IV) |
| By End User | Hospitals, Clinics, Homecare Settings, and Ambulatory Surgical Centers |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia-Pacific, and Middle East & Africa |
| Countries Covered | U.S., Germany, China, India, U.K., and France among 40 others |
| Key Companies | Becton, Dickinson and Company (BD), Gerresheimer AG, SCHOTT Pharma AG, West Pharmaceutical Services, Inc., Stevanato Group, Terumo Corporation, Nipro Corporation, Baxter International Inc., AptarGroup, Inc. B. Braun SE, Catalent, Inc. and others |
| Forecast Period | 2026 to 2036 |
| Additional Attributes | Dollar sales by material and application, regional demand trends, competitive landscape, manufacturer preferences for specific drug delivery systems, integration with specialty pharmaceutical supply chains, innovations in drug delivery technologies, performance monitoring, and healthcare optimization |
Bibliography
- [1] European Commission
- “Directive 2010/32/EU on prevention of sharps injuries in the hospital and healthcare sector”
- (Referenced regulatory framework, active in current compliance context)
- [2] Becton, Dickinson and Company (BD) - “BD announces $110 million investment to support U.S. pharmaceutical supply chain”, August 2026
- [3] Ypsomed Group - “Ypsomed and BD expand partnership to advance self-injection systems for large volume biologics”, January 2026
- [4] Gerresheimer AG (EQS News) - “Gerresheimer expands Wertheim site with new production facility for ready-to-fill vials”, January 2026
- [5] Gerresheimer AG - “Gerresheimer at Pharmapack 2026: Innovative solutions for biopharmaceuticals and adherence support”, January 2026
- [6] Contract Pharma - “Fresenius Kabi expands Simplist portfolio of prefilled syringes”, December 2025
- [7] Becton, Dickinson and Company (BD) - “BD to invest $35 million in Nebraska facility to expand prefilled flush syringe manufacturing”, August 2025
Prefilled Syringes Market Definition
The global prefilled syringes market covers a predetermined dosage of medicine without the requirement for filling manually prior to administration. The aim of the products are mainly focused on the delivery of injectable medicines with ease and safety. The main purpose of using the devices is to deliver medications through injection. In addition, prefilled syringes can be used for biopharmaceuticals and long-term illness treatment. Main applications include diabetes management, autoimmune disorders, vaccination, among others.
Prefilled Syringes Market Inclusion
The report covers global and regional market sizes for the prefilled syringes market, with forecasts spanning 2026 to 2036. It presents segment analysis by different categories such as product type, applications, end-users, and others. The report also offers other analytical parameters like pricing analysis, import-export, and competition analysis. It helps in understanding the overall structure of the market and future growth potential in the market.
Prefilled Syringes Market Exclusion
The report does not include devices that deliver drugs independently, including autoinjectors, pen injectors, and wearable injectors where they are not attached to prefilled syringes. In addition, empty syringes, bulk containers, ampoules, and downstream finished products do not fall within the research boundaries after primary packaging. Also excluded are syringes in veterinary use or other non-therapeutic purposes such as laboratory use, since this research confines itself to prefilled syringes for human therapeutic drug delivery.
Prefilled Syringes Market Research Methodology
In order to ensure accurate estimates for the global prefilled syringes market size and forecast, Fact.MR uses a comprehensive research methodology, taking into account all aspects of the pharmaceutical delivery system ecosystem. Secondary sources such as company annual reports/SEC filings, FDA/EMA drug approval databases, clinical trial databases and registrations, pharmaceutical journals, and publications from industry experts have been considered as sources of information for this study. Some of the key points covered through secondary research include the market sizing by type of syringe (glass vs. polymer), type of prefilled syringes (single vs. dual chambered), therapeutic areas (biologics, vaccines, insulin, anticoagulants), and target end-users.
Secondary research findings have been validated with the help of a bottom-up strategy based on sales analysis of the leading syringe manufacturers, including their unit shipments, along with a top-down approach relying on macro factors like rise in adoption of biologics drugs, number of injectable drug approvals, increasing vaccination rates, prevalence of self-administration. The models developed for forecasting take into consideration the impact of rising trend towards biologics drugs, rising acceptance of safety-engineered syringes, and growing trend towards home-based medical care. Data triangulation techniques were adopted while analyzing both supply-side and demand-side data sets to enhance the accuracy of findings.
Primary / Field Interviews
It can be mentioned that around 130 primary interviews are being conducted throughout the period 2025-2026 covering various stakeholders involved in the value chain of the global prefilled syringes market. Among these respondents, around 30% comprised of syringe manufacturers and component suppliers, while another 30% consisted of pharmaceutical & biotechnology companies. Around 20% of the total respondents were from CDMOs and fill-finish services, along with 20% from regulatory experts, procurement heads of hospitals and clinicians. Interviewees were contacted across the geographic markets of North America, Europe, Asia Pacific, and few other emerging countries.
Segment Taxonomy
-
By Product Type:
- Conventional Prefilled Syringes
- Safety Prefilled Syringes
- Passive Safety
- Active Safety
-
By Material:
- Glass
- Plastic
- Cyclic Olefin Polymer (COP)
- Cyclic Olefin Copolymer (COC)
-
By Design:
- Single-Chamber
- Dual-Chamber
- Customized / Specialty
-
By Application:
- Diabetes
- Rheumatoid Arthritis & Autoimmune Diseases
- Oncology
- Cardiovascular Diseases
- Infectious Diseases (Vaccines)
- Others
-
By Route of Administration:
- Subcutaneous (SC)
- Intramuscular (IM)
- Intravenous (IV)
-
By End Users:
- Hospitals
- Clinics
- Homecare Settings
- Ambulatory Surgical Centers
-
By Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia-Pacific
- Middle East & Africa
- Frequently Asked Questions -
How large is the global Prefilled Syringes market in 2026?
The global Prefilled Syringes market is estimated to be valued at USD 10.5 billion in 2026.
What will be the market size by 2036?
The market is projected to reach USD 20.8 billion by 2036.
What is the expected growth rate between 2026 and 2036?
The market is expected to grow at a CAGR of 7.1%, driven by increasing adoption of biologics, vaccines, and self-administration drug delivery systems.
Which segment dominates the market?
Safety Prefilled Syringes are expected to dominate, accounting for 63.4% of total market share in 2026, due to enhanced needlestick injury prevention and regulatory emphasis on healthcare worker safety.
Which country shows the highest growth rate?
India is projected to register the highest CAGR at 10.9%, supported by expanding pharmaceutical manufacturing, vaccination programs, and healthcare access.
How does China perform in this market?
China is expected to grow at a CAGR of 8.6%, driven by strong domestic pharmaceutical production and rising biologics demand.
What is the growth outlook for Brazil and Canada?
• Brazil: 7.4% CAGR • Canada: 7.2% CAGR Growth is supported by increased healthcare spending and biologics adoption.
How does the United States perform?
The United States is projected to grow at a CAGR of 6.4%, reflecting high penetration of advanced drug delivery systems and biologic therapies.
What is the projected growth for Japan and the U.K.?
• Japan: 6.6% CAGR • United Kingdom: 5.1% CAGR These markets are driven by aging populations and increasing chronic disease management.
How does Germany perform in the market?
Germany is projected to grow at a CAGR of 4.4%, reflecting a mature healthcare system with stable demand for prefilled delivery formats.
What are prefilled syringes used for?
Prefilled syringes are single-dose delivery systems used for administering vaccines, biologics, and injectable drugs. They improve dosing accuracy, reduce contamination risk, and enhance patient convenience.
What does the market scope include?
The market includes disposable and safety-engineered prefilled syringes used across hospitals, clinics, and home care settings, covering applications such as immunization, chronic disease treatment, and emergency care.
Table of Content
- Executive Summary
- Global Market Outlook
- Key Demand Drivers & Adoption Trends
- Supply Landscape & Capacity Dynamics
- Pricing, Value Chain & Strategic Outlook
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background and Foundation Data Points
- End-Use Demand & Consumption Analysis
- Injectable drug consumption (units/doses)
- Patient population & disease prevalence
- Therapeutic segmentation and usage intensity
- PFS vs vial/ampoule adoption rates (%)
- Self-administration vs clinical administration trends
- Biologics & biosimilars pipeline impact
- Regional demand distribution
- Manufacturing Capacity & Supply Ecosystem Analysis
- Manufacturing capacity (units/year) by region and key players
- Fill-finish infrastructure (number of lines, utilization rates)
- Material mix analysis (glass vs polymer)
- CDMO vs in-house manufacturing share (%)
- Capacity expansion pipeline and announced investments
- Value Chain & Cost Structure
- End-to-end value chain mapping
- Component-level cost breakdown (barrel, plunger, stopper)
- Conversion economics (vial → PFS transition rates)
- Yield losses and efficiency metrics
- Packaging and cold chain costs
- 7. Trade, Distribution & Access
- Import/export dynamics (PFS and components)
- Regional supply-demand gaps
- Distributor and hospital procurement patterns
- Channel margins and logistics structure
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- End-Use Demand & Consumption Analysis
- Global Prefilled Syringes Market Analysis 2021-2025 and Forecast, 2026-2036
- Historical Market Size Value (USD Bn) & Volume (Units) Analysis, 2021-2025
- Current and Future Market Size Value (USD Bn) & Volume (Units) Projections, 2026-2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Pricing & Revenue Analysis
- Average Selling Price (ASP) by region and product type
- Price benchmarking (glass vs polymer, safety vs standard)
- Fill-finish service pricing
- Bundled pricing models (device + drug)
- Net pricing adjustments (rebates, contracts)
- Global Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Bn) & Volume (Units) Analysis By Product Type, 2021-2025
- Current and Future Market Size Value (USD Bn) & Volume (Units) Analysis and Forecast By Product Type, 2026-2036
- Conventional Prefilled Syringes
- Safety Prefilled Syringes
- Passive Safety
- Active Safety
- Y-o-Y Growth Trend Analysis By Product Type, 2021-2025
- Absolute $ Opportunity Analysis By Product Type, 2026-2036
- Global Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Material
- Introduction / Key Findings
- Historical Market Size Value (USD Bn) & Volume (Units) Analysis By Material, 2021-2025
- Current and Future Market Size Value (USD Bn) & Volume (Units) Analysis and Forecast By Material, 2026-2036
- Glass
- Plastic
- Cyclic Olefin Polymer (COP)
- Cyclic Olefin Copolymer (COC)
- Y-o-Y Growth Trend Analysis By Material, 2021-2025
- Absolute $ Opportunity Analysis By Material, 2026-2036
- Global Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Design
- Introduction / Key Findings
- Historical Market Size Value (USD Bn) & Volume (Units) Analysis By Design, 2021-2025
- Current and Future Market Size Value (USD Bn) & Volume (Units) Analysis and Forecast By Design, 2026-2036
- Single-Chamber
- Dual-Chamber
- Customized / Specialty
- Y-o-Y Growth Trend Analysis By Design, 2021-2025
- Absolute $ Opportunity Analysis By Design, 2026-2036
- Global Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Bn) & Volume (Units) Analysis By Application, 2021-2025
- Current and Future Market Size Value (USD Bn) & Volume (Units) Analysis and Forecast By Application, 2026-2036
- Diabetes
- Rheumatoid Arthritis & Autoimmune Diseases
- Oncology
- Cardiovascular Diseases
- Infectious Diseases (Vaccines)
- Others
- Y-o-Y Growth Trend Analysis By Application, 2021-2025
- Absolute $ Opportunity Analysis By Application, 2026-2036
- Global Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Route of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Bn) & Volume (Units) Analysis By Route of Administration, 2021-2025
- Current and Future Market Size Value (USD Bn) & Volume (Units) Analysis and Forecast By Route of Administration, 2026-2036
- Subcutaneous (SC)
- Intramuscular (IM)
- Intravenous (IV)
- Y-o-Y Growth Trend Analysis By Route of Administration, 2021-2025
- Absolute $ Opportunity Analysis By Route of Administration, 2026-2036
- Global Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Bn) & Volume (Units) Analysis By End User, 2021-2025
- Current and Future Market Size Value (USD Bn) & Volume (Units) Analysis and Forecast By End User, 2026-2036
- Hospitals
- Clinics
- Homecare Settings
- Ambulatory Surgical Centers
- Y-o-Y Growth Trend Analysis By End User, 2021-2025
- Absolute $ Opportunity Analysis By End User, 2026-2036
- Global Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Region
- Introduction
- Historical Market Size Value (USD Bn) & Volume (Units) Analysis By Region, 2021-2025
- Current Market Size Value (USD Bn) & Volume (Units) Analysis and Forecast By Region, 2026-2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Country
- Historical Market Size Value (USD Bn) & Volume (Units) Trend Analysis By Market Taxonomy, 2021-2025
- Market Size Value (USD Bn) & Volume (Units) Forecast By Market Taxonomy, 2026-2036
- By Country
- U.S.
- Canada
- Mexico
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Key Takeaways
- Latin America Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Country
- Historical Market Size Value (USD Bn) & Volume (Units) Trend Analysis By Market Taxonomy, 2021-2025
- Market Size Value (USD Bn) & Volume (Units) Forecast By Market Taxonomy, 2026-2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Key Takeaways
- Western Europe Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Country
- Historical Market Size Value (USD Bn) & Volume (Units) Trend Analysis By Market Taxonomy, 2021-2025
- Market Size Value (USD Bn) & Volume (Units) Forecast By Market Taxonomy, 2026-2036
- By Country
- Germany
- U.K.
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Key Takeaways
- Eastern Europe Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Country
- Historical Market Size Value (USD Bn) & Volume (Units) Trend Analysis By Market Taxonomy, 2021-2025
- Market Size Value (USD Bn) & Volume (Units) Forecast By Market Taxonomy, 2026-2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Key Takeaways
- East Asia Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Country
- Historical Market Size Value (USD Bn) & Volume (Units) Trend Analysis By Market Taxonomy, 2021-2025
- Market Size Value (USD Bn) & Volume (Units) Forecast By Market Taxonomy, 2026-2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Key Takeaways
- South Asia and Pacific Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Country
- Historical Market Size Value (USD Bn) & Volume (Units) Trend Analysis By Market Taxonomy, 2021-2025
- Market Size Value (USD Bn) & Volume (Units) Forecast By Market Taxonomy, 2026-2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Key Takeaways
- Middle East & Africa Prefilled Syringes Market Analysis 2021-2025 and Forecast 2026-2036, By Country
- Historical Market Size Value (USD Bn) & Volume (Units) Trend Analysis By Market Taxonomy, 2021-2025
- Market Size Value (USD Bn) & Volume (Units) Forecast By Market Taxonomy, 2026-2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Key Takeaways
- Key Countries Prefilled Syringes Market Analysis
- U.S.
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Canada
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Mexico
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Brazil
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Chile
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Germany
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- U.K.
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Italy
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Spain
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- France
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- India
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- ASEAN Countries
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- China
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Japan
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- South Korea
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Russia
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Poland
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Hungary
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Turkiye
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- South Africa
- Pricing Analysis
- Market Value and Volume Analysis, 2026
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- U.S.
- Market Competition Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Material
- By Design
- By Application
- By Route of Administration
- By End User
- Company Profiles
-
- Becton, Dickinson and Company (BD)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Gerresheimer AG
- SCHOTT Pharma AG
- West Pharmaceutical Services, Inc.
- Stevanato Group
- Terumo Corporation
- Nipro Corporation
- Baxter International Inc.
- AptarGroup, Inc.
- B. Braun SE
- Catalent, Inc.
- Vetter Pharma International GmbH
- Credence MedSystems, Inc.
- Weigao Group
- Fresenius Kabi
- Becton, Dickinson and Company (BD)
-
- Assumptions & Acronyms Used
- Research Methodology
List Of Table
- Table 1: Global Market Value (USD Bn) Forecast by Region, 2021 to 2036
- Table 2: Global Market Volume (Units) Forecast by Region, 2021 to 2036
- Table 3: Global Market Value (USD Bn) Forecast by Product Type, 2021 to 2036
- Table 4: Global Market Volume (Units) Forecast by Product Type, 2021 to 2036
- Table 5: Global Market Value (USD Bn) Forecast by Material, 2021 to 2036
- Table 6: Global Market Volume (Units) Forecast by Material, 2021 to 2036
- Table 7: Global Market Value (USD Bn) Forecast by Design, 2021 to 2036
- Table 8: Global Market Volume (Units) Forecast by Design, 2021 to 2036
- Table 9: Global Market Value (USD Bn) Forecast by Application, 2021 to 2036
- Table 10: Global Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 11: Global Market Value (USD Bn) Forecast by Route of Administration, 2021 to 2036
- Table 12: Global Market Volume (Units) Forecast by Route of Administration, 2021 to 2036
- Table 13: Global Market Value (USD Bn) Forecast by End User, 2021 to 2036
- Table 14: Global Market Volume (Units) Forecast by End User, 2021 to 2036
- Table 15: North America Market Value (USD Bn) Forecast by Country, 2021 to 2036
- Table 16: North America Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 17: North America Market Value (USD Bn) Forecast by Product Type, 2021 to 2036
- Table 18: North America Market Volume (Units) Forecast by Product Type, 2021 to 2036
- Table 19: North America Market Value (USD Bn) Forecast by Material, 2021 to 2036
- Table 20: North America Market Volume (Units) Forecast by Material, 2021 to 2036
- Table 21: North America Market Value (USD Bn) Forecast by Design, 2021 to 2036
- Table 22: North America Market Volume (Units) Forecast by Design, 2021 to 2036
- Table 23: North America Market Value (USD Bn) Forecast by Application, 2021 to 2036
- Table 24: North America Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 25: North America Market Value (USD Bn) Forecast by Route of Administration, 2021 to 2036
- Table 26: North America Market Volume (Units) Forecast by Route of Administration, 2021 to 2036
- Table 27: North America Market Value (USD Bn) Forecast by End User, 2021 to 2036
- Table 28: North America Market Volume (Units) Forecast by End User, 2021 to 2036
- Table 29: Latin America Market Value (USD Bn) Forecast by Country, 2021 to 2036
- Table 30: Latin America Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 31: Latin America Market Value (USD Bn) Forecast by Product Type, 2021 to 2036
- Table 32: Latin America Market Volume (Units) Forecast by Product Type, 2021 to 2036
- Table 33: Latin America Market Value (USD Bn) Forecast by Material, 2021 to 2036
- Table 34: Latin America Market Volume (Units) Forecast by Material, 2021 to 2036
- Table 35: Latin America Market Value (USD Bn) Forecast by Design, 2021 to 2036
- Table 36: Latin America Market Volume (Units) Forecast by Design, 2021 to 2036
- Table 37: Latin America Market Value (USD Bn) Forecast by Application, 2021 to 2036
- Table 38: Latin America Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 39: Latin America Market Value (USD Bn) Forecast by Route of Administration, 2021 to 2036
- Table 40: Latin America Market Volume (Units) Forecast by Route of Administration, 2021 to 2036
- Table 41: Latin America Market Value (USD Bn) Forecast by End User, 2021 to 2036
- Table 42: Latin America Market Volume (Units) Forecast by End User, 2021 to 2036
- Table 43: Western Europe Market Value (USD Bn) Forecast by Country, 2021 to 2036
- Table 44: Western Europe Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 45: Western Europe Market Value (USD Bn) Forecast by Product Type, 2021 to 2036
- Table 46: Western Europe Market Volume (Units) Forecast by Product Type, 2021 to 2036
- Table 47: Western Europe Market Value (USD Bn) Forecast by Material, 2021 to 2036
- Table 48: Western Europe Market Volume (Units) Forecast by Material, 2021 to 2036
- Table 49: Western Europe Market Value (USD Bn) Forecast by Design, 2021 to 2036
- Table 50: Western Europe Market Volume (Units) Forecast by Design, 2021 to 2036
- Table 51: Western Europe Market Value (USD Bn) Forecast by Application, 2021 to 2036
- Table 52: Western Europe Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 53: Western Europe Market Value (USD Bn) Forecast by Route of Administration, 2021 to 2036
- Table 54: Western Europe Market Volume (Units) Forecast by Route of Administration, 2021 to 2036
- Table 55: Western Europe Market Value (USD Bn) Forecast by End User, 2021 to 2036
- Table 56: Western Europe Market Volume (Units) Forecast by End User, 2021 to 2036
- Table 57: Eastern Europe Market Value (USD Bn) Forecast by Country, 2021 to 2036
- Table 58: Eastern Europe Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 59: Eastern Europe Market Value (USD Bn) Forecast by Product Type, 2021 to 2036
- Table 60: Eastern Europe Market Volume (Units) Forecast by Product Type, 2021 to 2036
- Table 61: Eastern Europe Market Value (USD Bn) Forecast by Material, 2021 to 2036
- Table 62: Eastern Europe Market Volume (Units) Forecast by Material, 2021 to 2036
- Table 63: Eastern Europe Market Value (USD Bn) Forecast by Design, 2021 to 2036
- Table 64: Eastern Europe Market Volume (Units) Forecast by Design, 2021 to 2036
- Table 65: Eastern Europe Market Value (USD Bn) Forecast by Application, 2021 to 2036
- Table 66: Eastern Europe Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 67: Eastern Europe Market Value (USD Bn) Forecast by Route of Administration, 2021 to 2036
- Table 68: Eastern Europe Market Volume (Units) Forecast by Route of Administration, 2021 to 2036
- Table 69: Eastern Europe Market Value (USD Bn) Forecast by End User, 2021 to 2036
- Table 70: Eastern Europe Market Volume (Units) Forecast by End User, 2021 to 2036
- Table 71: East Asia Market Value (USD Bn) Forecast by Country, 2021 to 2036
- Table 72: East Asia Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 73: East Asia Market Value (USD Bn) Forecast by Product Type, 2021 to 2036
- Table 74: East Asia Market Volume (Units) Forecast by Product Type, 2021 to 2036
- Table 75: East Asia Market Value (USD Bn) Forecast by Material, 2021 to 2036
- Table 76: East Asia Market Volume (Units) Forecast by Material, 2021 to 2036
- Table 77: East Asia Market Value (USD Bn) Forecast by Design, 2021 to 2036
- Table 78: East Asia Market Volume (Units) Forecast by Design, 2021 to 2036
- Table 79: East Asia Market Value (USD Bn) Forecast by Application, 2021 to 2036
- Table 80: East Asia Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 81: East Asia Market Value (USD Bn) Forecast by Route of Administration, 2021 to 2036
- Table 82: East Asia Market Volume (Units) Forecast by Route of Administration, 2021 to 2036
- Table 83: East Asia Market Value (USD Bn) Forecast by End User, 2021 to 2036
- Table 84: East Asia Market Volume (Units) Forecast by End User, 2021 to 2036
- Table 85: South Asia and Pacific Market Value (USD Bn) Forecast by Country, 2021 to 2036
- Table 86: South Asia and Pacific Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 87: South Asia and Pacific Market Value (USD Bn) Forecast by Product Type, 2021 to 2036
- Table 88: South Asia and Pacific Market Volume (Units) Forecast by Product Type, 2021 to 2036
- Table 89: South Asia and Pacific Market Value (USD Bn) Forecast by Material, 2021 to 2036
- Table 90: South Asia and Pacific Market Volume (Units) Forecast by Material, 2021 to 2036
- Table 91: South Asia and Pacific Market Value (USD Bn) Forecast by Design, 2021 to 2036
- Table 92: South Asia and Pacific Market Volume (Units) Forecast by Design, 2021 to 2036
- Table 93: South Asia and Pacific Market Value (USD Bn) Forecast by Application, 2021 to 2036
- Table 94: South Asia and Pacific Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 95: South Asia and Pacific Market Value (USD Bn) Forecast by Route of Administration, 2021 to 2036
- Table 96: South Asia and Pacific Market Volume (Units) Forecast by Route of Administration, 2021 to 2036
- Table 97: South Asia and Pacific Market Value (USD Bn) Forecast by End User, 2021 to 2036
- Table 98: South Asia and Pacific Market Volume (Units) Forecast by End User, 2021 to 2036
- Table 99: Middle East & Africa Market Value (USD Bn) Forecast by Country, 2021 to 2036
- Table 100: Middle East & Africa Market Volume (Units) Forecast by Country, 2021 to 2036
- Table 101: Middle East & Africa Market Value (USD Bn) Forecast by Product Type, 2021 to 2036
- Table 102: Middle East & Africa Market Volume (Units) Forecast by Product Type, 2021 to 2036
- Table 103: Middle East & Africa Market Value (USD Bn) Forecast by Material, 2021 to 2036
- Table 104: Middle East & Africa Market Volume (Units) Forecast by Material, 2021 to 2036
- Table 105: Middle East & Africa Market Value (USD Bn) Forecast by Design, 2021 to 2036
- Table 106: Middle East & Africa Market Volume (Units) Forecast by Design, 2021 to 2036
- Table 107: Middle East & Africa Market Value (USD Bn) Forecast by Application, 2021 to 2036
- Table 108: Middle East & Africa Market Volume (Units) Forecast by Application, 2021 to 2036
- Table 109: Middle East & Africa Market Value (USD Bn) Forecast by Route of Administration, 2021 to 2036
- Table 110: Middle East & Africa Market Volume (Units) Forecast by Route of Administration, 2021 to 2036
- Table 111: Middle East & Africa Market Value (USD Bn) Forecast by End User, 2021 to 2036
- Table 112: Middle East & Africa Market Volume (Units) Forecast by End User, 2021 to 2036
List Of Figures
- Figure 1: Global Market Volume (Units) Forecast 2021–2036
- Figure 2: Global Market Pricing Analysis
- Figure 3: Global Market Value (USD Bn) Forecast 2021–2036
- Figure 4: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 5: Global Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 6: Global Market Attractiveness Analysis by Product Type
- Figure 7: Global Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 8: Global Market Y-o-Y Growth Comparison by Material, 2026 to 2036
- Figure 9: Global Market Attractiveness Analysis by Material
- Figure 10: Global Market Value Share and BPS Analysis by Design, 2026 and 2036
- Figure 11: Global Market Y-o-Y Growth Comparison by Design, 2026 to 2036
- Figure 12: Global Market Attractiveness Analysis by Design
- Figure 13: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 14: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 15: Global Market Attractiveness Analysis by Application
- Figure 16: Global Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 17: Global Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 18: Global Market Attractiveness Analysis by Route of Administration
- Figure 19: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 20: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 21: Global Market Attractiveness Analysis by End User
- Figure 22: Global Market Value (USD Bn) Share and BPS Analysis by Region, 2026 and 2036
- Figure 23: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 24: Global Market Attractiveness Analysis by Region
- Figure 25: North America Market Incremental $ Opportunity, 2026 to 2036
- Figure 26: Latin America Market Incremental $ Opportunity, 2026 to 2036
- Figure 27: Western Europe Market Incremental $ Opportunity, 2026 to 2036
- Figure 28: Eastern Europe Market Incremental $ Opportunity, 2026 to 2036
- Figure 29: East Asia Market Incremental $ Opportunity, 2026 to 2036
- Figure 30: South Asia and Pacific Market Incremental $ Opportunity, 2026 to 2036
- Figure 31: Middle East & Africa Market Incremental $ Opportunity, 2026 to 2036
- Figure 32: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 34: North America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 35: North America Market Attractiveness Analysis by Product Type
- Figure 36: North America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 37: North America Market Y-o-Y Growth Comparison by Material, 2026 to 2036
- Figure 38: North America Market Attractiveness Analysis by Material
- Figure 39: North America Market Value Share and BPS Analysis by Design, 2026 and 2036
- Figure 40: North America Market Y-o-Y Growth Comparison by Design, 2026 to 2036
- Figure 41: North America Market Attractiveness Analysis by Design
- Figure 42: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 43: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 44: North America Market Attractiveness Analysis by Application
- Figure 45: North America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 46: North America Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 47: North America Market Attractiveness Analysis by Route of Administration
- Figure 48: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 49: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 50: North America Market Attractiveness Analysis by End User
- Figure 51: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 53: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 54: Latin America Market Attractiveness Analysis by Product Type
- Figure 55: Latin America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 56: Latin America Market Y-o-Y Growth Comparison by Material, 2026 to 2036
- Figure 57: Latin America Market Attractiveness Analysis by Material
- Figure 58: Latin America Market Value Share and BPS Analysis by Design, 2026 and 2036
- Figure 59: Latin America Market Y-o-Y Growth Comparison by Design, 2026 to 2036
- Figure 60: Latin America Market Attractiveness Analysis by Design
- Figure 61: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 62: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 63: Latin America Market Attractiveness Analysis by Application
- Figure 64: Latin America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 65: Latin America Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 66: Latin America Market Attractiveness Analysis by Route of Administration
- Figure 67: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 68: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 69: Latin America Market Attractiveness Analysis by End User
- Figure 70: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 71: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 72: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 73: Western Europe Market Attractiveness Analysis by Product Type
- Figure 74: Western Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 75: Western Europe Market Y-o-Y Growth Comparison by Material, 2026 to 2036
- Figure 76: Western Europe Market Attractiveness Analysis by Material
- Figure 77: Western Europe Market Value Share and BPS Analysis by Design, 2026 and 2036
- Figure 78: Western Europe Market Y-o-Y Growth Comparison by Design, 2026 to 2036
- Figure 79: Western Europe Market Attractiveness Analysis by Design
- Figure 80: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 81: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 82: Western Europe Market Attractiveness Analysis by Application
- Figure 83: Western Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 84: Western Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 85: Western Europe Market Attractiveness Analysis by Route of Administration
- Figure 86: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 88: Western Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 90: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 91: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 92: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 93: Eastern Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 94: Eastern Europe Market Y-o-Y Growth Comparison by Material, 2026 to 2036
- Figure 95: Eastern Europe Market Attractiveness Analysis by Material
- Figure 96: Eastern Europe Market Value Share and BPS Analysis by Design, 2026 and 2036
- Figure 97: Eastern Europe Market Y-o-Y Growth Comparison by Design, 2026 to 2036
- Figure 98: Eastern Europe Market Attractiveness Analysis by Design
- Figure 99: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 100: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 101: Eastern Europe Market Attractiveness Analysis by Application
- Figure 102: Eastern Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 103: Eastern Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 104: Eastern Europe Market Attractiveness Analysis by Route of Administration
- Figure 105: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 106: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 107: Eastern Europe Market Attractiveness Analysis by End User
- Figure 108: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 110: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 111: East Asia Market Attractiveness Analysis by Product Type
- Figure 112: East Asia Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 113: East Asia Market Y-o-Y Growth Comparison by Material, 2026 to 2036
- Figure 114: East Asia Market Attractiveness Analysis by Material
- Figure 115: East Asia Market Value Share and BPS Analysis by Design, 2026 and 2036
- Figure 116: East Asia Market Y-o-Y Growth Comparison by Design, 2026 to 2036
- Figure 117: East Asia Market Attractiveness Analysis by Design
- Figure 118: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 119: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 120: East Asia Market Attractiveness Analysis by Application
- Figure 121: East Asia Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 122: East Asia Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 123: East Asia Market Attractiveness Analysis by Route of Administration
- Figure 124: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 125: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 126: East Asia Market Attractiveness Analysis by End User
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 128: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 129: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 130: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 131: South Asia and Pacific Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 132: South Asia and Pacific Market Y-o-Y Growth Comparison by Material, 2026 to 2036
- Figure 133: South Asia and Pacific Market Attractiveness Analysis by Material
- Figure 134: South Asia and Pacific Market Value Share and BPS Analysis by Design, 2026 and 2036
- Figure 135: South Asia and Pacific Market Y-o-Y Growth Comparison by Design, 2026 to 2036
- Figure 136: South Asia and Pacific Market Attractiveness Analysis by Design
- Figure 137: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 138: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 139: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 140: South Asia and Pacific Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 141: South Asia and Pacific Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 142: South Asia and Pacific Market Attractiveness Analysis by Route of Administration
- Figure 143: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 144: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 145: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 147: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 148: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 149: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 150: Middle East & Africa Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 151: Middle East & Africa Market Y-o-Y Growth Comparison by Material, 2026 to 2036
- Figure 152: Middle East & Africa Market Attractiveness Analysis by Material
- Figure 153: Middle East & Africa Market Value Share and BPS Analysis by Design, 2026 and 2036
- Figure 154: Middle East & Africa Market Y-o-Y Growth Comparison by Design, 2026 to 2036
- Figure 155: Middle East & Africa Market Attractiveness Analysis by Design
- Figure 156: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 157: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 158: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 159: Middle East & Africa Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 160: Middle East & Africa Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 161: Middle East & Africa Market Attractiveness Analysis by Route of Administration
- Figure 162: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 163: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 164: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 165: Global Market – Tier Structure Analysis
- Figure 166: Global Market – Company Share Analysis