Lab Automation Market Size, Share, Growth and Forecast (2025 - 2035)
Lab Automation Market Analysis By Product (Lab Automation Equipment, Microplate Readers, Automated ELISA Systems, Automated Nucleic Acid Purification Systems, and others), By Application (Drug Discovery, Clinical Diagnostics, Genomics Solutions, and Proteomics Solutions), By End User, and By Region - Market Insights 2025 to 2035
Analysis of Lab Automation Market Covering 30+ Countries Including Analysis of US, Canada, UK, Germany, France, Nordics, GCC countries, Japan, Korea and many more
Lab Automation Market Outlook (2025 to 2035)
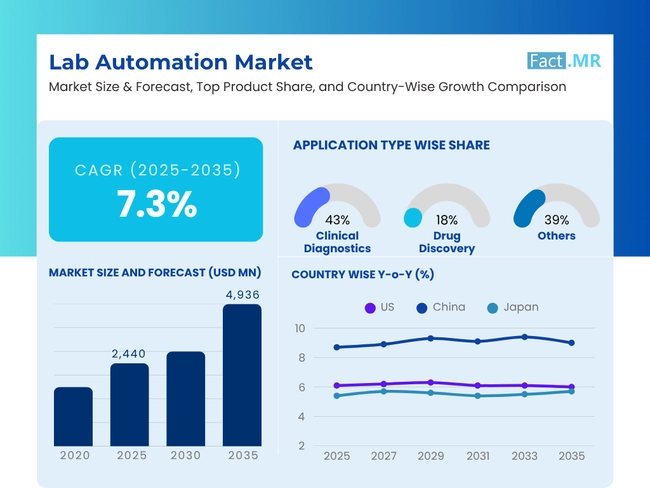
The global lab automation market is expected to reach USD 4,936 million by 2035, up from USD 2,274 million in 2024. During the forecast period (2025-2035), the industry is projected to expand at a CAGR of 7.3%, driven by rising technological advancements that have transformed laboratory functions. Advances in robotics, AI, and machine learning enhance precision and throughput, enabling labs to handle more work.
This is further promoted by increased labor expenses and escalated shortage of skilled personnel in laboratories. Automation enables organisations to maximise the potential of their workforce, and they achieve this by minimising the levels of manual intervention, allowing them to redeploy the workforce to more challenging tasks.
Most importantly, lab automation can go a long way toward addressing one of the most critical requirements: mitigating human error and ensuring reproducibility. Automated systems also lead to uniform and reproducible data, as the workflows have been standardized and the amount of manual work reduced, thereby eliminating potential errors in scientific accuracy and regulatory compliance.
What are the drivers of the Lab Automation Market?
One of the major drivers of the market for lab automation is the ever-changing pace of technology, which includes advanced robotics, specifically designed for sample and reagent handling, and incorporates artificial intelligence (AI) and machine learning (ML) to perform more complex data analysis and predictions.
The innovations enable labs to work more efficiently and accurately, without boundaries, and perform more complex tasks that have not been observed before in research, diagnostics, and drug discovery.
Manual laboratory operations are, by their nature, vulnerable to human error and lack reproducibility, which may decrease the dependability and replicability of the results. Automation standardizes work processes, allowing each to be performed accurately and with a consistent level of performance.
This dramatically reduces the risk of human error, such as mislabeling or incorrect measurements, resulting in more accurate data and highly reproducible experimental results, which is of utmost importance in validation and regulation.
What are the regional trends of the Lab Automation Market?
The regional territory of the lab automation market reflects the presence of various directions, including AI and Machine learning, Drug Discovery and Development, and the demand for personalized medicines. They control the market with their high demand for medicines and technological advancements, especially in North America, where diseases and infections are on the rise.
The high privacy drug-related requirement and the nature of the regulatory process, which is standardized, will raise the market growth in Europe. It is also expected that the trend of shifting towards lab automation technology in the region will be promoted by the need to keep up with changing healthcare regulations.
The transformational change being evidenced in Asia-Pacific is characterized by high data-driven products. The primary factors driving this growth include the emergence of the biotechnology and pharmaceutical industries, new and improved healthcare infrastructure, an increase in disposable income, and an increase in government funding for research activities.
Latin America is enjoying a smaller market share in the market, but is nonetheless expected to fuel the growth of the entire market, owing to the growing spending on research and health infrastructure development.
What are the challenges and restraining factors of the Lab Automation Market?
High investment cost is one of the greatest challenges of the lab automation market, and this requires a massive capital investment to initiate the business and then make an entry into the market. The cost of investing in a machine, particularly in terms of a tailored or specific delivery machine, is a deterrent or a cog in terms of overall expenditure.
The other structural limitation is that the knowledge and application of the technology or machines imply that professionals are not yet adequately trained to operate the machines effectively, which can lead to system failure due to misuse, resulting from the incorrect combination.
Additionally, compatibility issues and the lack of standards may hinder the integration of new automation systems with existing software systems and legacy systems that have already been implemented.
Country-Wise Insights
Lab automation market in USA is strengthened by the development of efficiency and precision
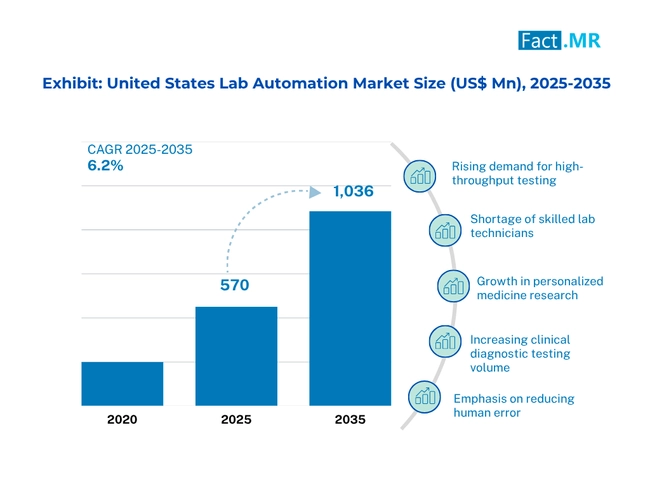
The rising demand of proper and efficient diagnostic and researching equipments are key reason behind the growth of the lab automation market in the United States. A lot of focus is given to the application of high technologies like robotics and artificial intelligent that results in the enhancement of efficiency and little likelihood of human error in laboratory functions.
Another significant trend and the factor that encourages differentiation of solutions to the demands of a lab is installation of modular scalable automation system. Cloud solutions are becoming increasingly popular in order to preserve the data in a better condition and to gain access to the data remotely. Such transformation can be transformed into better treatment of patients and advancement in science leading to laboratory with more volume of samples, shortened turnaround time, and free up personnel to add more difficult duties.
China Lab Automation Market accounts for a revenue of USD 267.3 Mn in 2025
The Chinese lab automation market is also growing due to the government's high focus on developing biotechnology and the healthcare sector. A significant driver is the increasing need in various industries, such as pharmaceuticals, diagnostics, and research, to effectively and accurately manage their laboratory activities.
Technologies such as artificial intelligence, machine learning, and robotics are being implemented to enhance the productivity of various processes, eliminate human error, and accelerate research and development. It is paramount to employ high-throughput screening and analysis applications.
Cost of investing might also be an issue, but the long-term advantages of enhanced quality control, quicker turnaround time and easier processes will see their usage achieve exponentially. It is also believed that business innovation is driven by collaboration between academic institutions and business companies in this rapidly evolving market.
Japan growth was speeded up by pioneering efficiency through advanced robotics and AI integration
The lab automation industry in Japan is growing in great part due to the unique demographic trends, especially the aging population and the loss of workforce. This creates a vast demand for the need to transform laboratories into more productive areas and minimize manual-driven labor. The economic growth of Japan, especially in the field of lab automation, is closely associated with its innovativeness concerning ultra-efficient robots and AI.
Small laboratory equipment, such as laboratory arms and liquid handlers, is becoming increasingly popular in Japan, offering space-saving solutions for small laboratories. The developments are decisive in maintaining high standards in research and diagnostic capabilities, aiming to minimize errors per task when humans compete with amplified range and direction, without necessarily involving specialized labor that has proven increasingly scarce in this era. Partnerships between life science institutes and the technology sector are accelerating the adoption of these innovative solutions in Japan.
Category-Wise Analysis
Lab automation equipment emerging to meet the requirements of high throughput analytical infrastructure
Technological core building blocks of automated lab equipment supplies instruments such as liquid handlers, microplate readers, robot arms, and multi-port storage systems. They are meant to reduce human interference but enhance speed, accuracy, and reproduction in the course of experiments. Automation is seen as vital in handling optimal and constantly rising sample size and complicated processes in life sciences, diagnostics, and chemistry labs.
The demand is on an increase as the labs seek efficiency and cost reduction by streamlining operations. The new direction is the introduction of AI and modular units of automation characterized by flexibility and adaptability. The leading manufactories are working on the development of innovations that redeem workflow integrity. Investment in the automated laboratory infrastructure is also growing around the world with the increased prevalence of chronic diseases and personalized medicine.
The adoption of Lab automation associated with drugs discovery is caused by faster and mistake-free molecule development.
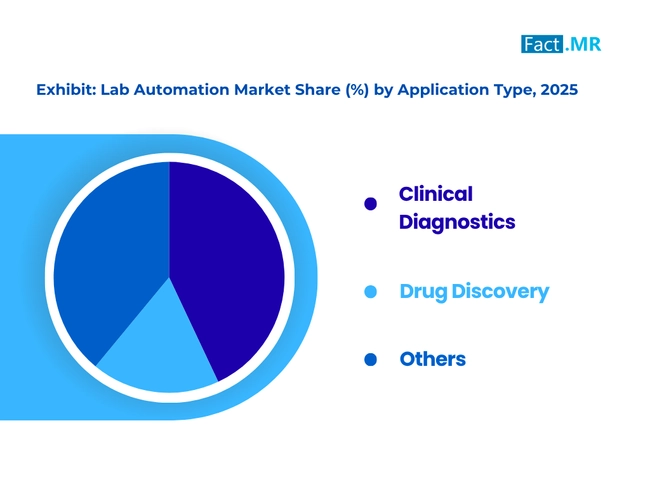
Drug discovery constitutes a significant portion of the utilization of the technologies in drug research in pharmaceuticals and biotechnology. Automation facilitates high-throughput screening, compound profiling and assay development processes in order to identify viable drug candidates at an accelerated rate.
These systems enhance reproducibility and reduce error in large-scale experiments, especially from human error. As the pressure to lower time-to-market and the cost of R&D is increasing, businesses are automating to perform repetitive workflows to optimize processes in early discovery. Automation facilitates the processing of information and data, as well as the combination of experiments, allowing for superior decision-making.
Targeted therapies and biologics have also increased, thus increasing the need for lab automation in drug discovery. Robotic platforms, liquid handlers, and AI-based systems are a growing staple of both startups and large companies as they use them to make discovery laboratories more efficient and innovative.
The Biotech and Pharma are pressuring lab automation to meet the requirements of precision and compliance.
The primary end-users of lab automation solutions are biotechnology and pharmaceutical companies seeking reproducibility, scalability, and compliance in their laboratory work. These fields depend on automation in drug discovery, clinical testing, the production of biologics, and quality assurance. Robotics, data analytics, and AI seamlessly integrate samples, and provide real-time data that can be tracked.
As R&D investment is increasing, especially in the cell and gene therapy sector, automation would result in the faster development and minimized human error. Laboratory practice regulations also promote standardized procedures that are automated. Leading participants are putting money in intelligent labs and IT infrastructure to enhance throughput and innovation. The demand is also supported by the necessity in pandemic response and expansion of the personalized medicine.
Competitive Analysis
The big companies in the markets are always pushing the envelope as they are working to achieve more through the employment of superior technologies into their systems to respond to the changing needs of customers. They are also not responding to needs; they are going ahead to recognize unmet needs and designing creative solutions.
This includes a significant investment into the research and development where scientific research and engineering prowess is used to develop ground breaking products and services. For instance, in June, 2025 OTI Lumionics, which is a Canadian company, enters the field of testing of next-gen lab automation.
In addition to internal innovation, the companies develop specific partnerships as a way of strategic maneuvering to widen their margin as well as capabilities. This will open new markets to them, enable them to leverage on their new found competencies, and also to speed the commercialization of their innovations when they are collaborating with specialized firms or with research institutions.
These businesses bring their latest goods into the market through intensive testing, aggressive marketing strategies, and effective supply lines. This is a multifaceted approach that combines a profound discovery process and prudent alliances with commercialization that is both effective and profit-enhancing, thus enabling them to not only meet immediate customer demands but also predict future needs and occupy a central marketplace at the expense of their competitors.
Key players in the lab automation industry include Agilent Technologies, Thermo Fisher Scientific, Siemens Healthineers, Danaher Corporation, AB Controls, Hamilton Company, Biolitic Lab, Automata, and PerkinElmer.
Recent Development
- In March 2025, Grifols, a global healthcare company and manufacturer of plasma-derived medicines and diagnostic solutions, has signed a strategic agreement with Inpeco, a leader in total laboratory automation technologies, to provide transfusion medicine labs with complete, tailored instrumentation, robotics and software capabilities to modernize their operations and make them more efficient.
- In January 2025, Agilent Technologies & ABB Robotics collaborate to produce automated laboratory solutions. This solution will help lab staff adopt novel techniques to improve workflows, making operations more efficient and flexible.
Fact.MR has provided detailed information about the price points of key manufacturers of Lthe ab Automation Market positioned across regions, sales growth, production capacity, and speculative technological expansion, in the recently published report.
Methodology and Industry Tracking Approach
The Fact.MR 2025 Global Lab Automation Market report incorporated the inputs of 12,500 stakeholders in 35 countries to achieve the minimum of 350 responses per regional market. Out of the total number of the participants accepted about 2/3 were end users/technology developers (e.g., pharmaceutical researchers, clinical lab managers, biotech engineers) and the rest represented the industry professionals (e.g., procurement heads, R&D directors, regulatory consultants).
Real time trends, demand patterns, investment profiles, market risks and market gap (unmet needs) were identified between June 2024 and May 2025 via survey data which was collected over this period. All answers were weighted basing on regional market share and their demographic importance.
The research was based on more than 300 data sets (peer-reviewed publications, patents, compliance, and financial statements) and made use of advanced statistical instruments (regression analysis) to provide accurate market intelligence.
With Fact.MR monitoring consumer behavior, product efficacy, industry trends, and market opportunities since 2018, this report is becoming an authoritative source of information that stakeholders can rely on.
Segmentation of Lab Automation Market
-
By Product Type :
- Lab Automation Equipment
- Automated Workstations
- Automated Liquid Handling
- Automated Integrated Workstations
- Pipetting Systems
- Reagent Dispensers
- Microplate Washers
- Automated Microplate Readers
- Multi-mode Microplate Readers
- Single-mode Microplate Readers
- Automated ELISA Systems
- Automated Nucleic Acid Purification Systems
- Lab Automation Software & Informatics
- Workstation/Unit Automation Software
- Laboratory Information Management Systems (LIMS)
- Electronic Laboratory Notebook
- Scientific Data Management System
- Lab Automation Equipment
-
By Application Type :
- Drug Discovery
- Clinical Diagnostics
- Genomics Solutions
- Proteomics Solutions
-
By End-User Type :
- Biotechnology & Pharmaceutical Industries
- Research & Academic Institutes
- Hospitals & Diagnostic Laboratories
- Forensic Laboratories
- Environmental Testing Labs
- Food and Beverage Industry
-
By Region :
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
- Frequently Asked Questions -
What was the Global Lab Automation Market Size Reported by Fact.MR for 2025?
The Global Lab Automation Market was valued at USD 2,441 million in 2025.
Who are the Major Players Operating in the Lab Automation Market?
Prominent players in the market are Agilent Technologies, Thermo Fisher Scientific, Siemens Healthineers, Danaher Corporation, AB Controls, Hamilton Company, Biolitic Lab, Automata, and Perkin Elmer among others.
What is the Estimated Valuation of the Lab Automation Market in 2035?
The market is expected to reach a valuation of USD 4,936 million in 2035.
What Value CAGR did the Lab Automation Market Exhibit Over the Last Five Years?
The historic growth rate of the Lab Automation Market was 7.3% from 2020 to 2024.
Table of Content
- Executive Summary
- Industry Introduction, including Taxonomy and Market Definition
- Market Trends and Success Factors, including Macro-economic Factors, Market Dynamics, and Recent Industry Developments
- Global Market Demand Analysis 2020 to 2024 and Forecast 2025 to 2035, including Historical Analysis and Future Projections
- Pricing Analysis
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035
- Product Type
- Application Type
- End User
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product Type
- Equipment
- Automated Workstations
- Automated Liquid Handling
- Automated Integrated Workstations
- Pipetting Systems
- Reagent Dispensers
- Microplate Washers
- Automated Microplate Readers
- Multi-mode Microplate Readers
- Single-mode Microplate Readers
- Automated ELISA Systems
- Automated Nucleic Acid Purification Systems
- Software & Informatics
- Workstation/Value (US$ Mn) & Tons Automation Software
- Laboratory Information Management Systems (LIMS)
- Electronic Laboratory Notebook
- Scientific Data Management System
- Equipment
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application Type
- Drug Discovery
- Clinical Diagnostics
- Genomics Solutions
- Proteomics Solutions
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Biotechnology & Pharmaceutical Industries
- Research & Academic Institutes
- Hospitals & Diagnostic Laboratories
- Forensic Laboratories
- Environmental Testing Labs
- Food and Beverage Industry
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- North America
- Latin America
- Western Europe
- South Asia
- East Asia
- Eastern Europe
- Middle East & Africa
- North America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Latin America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Western Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- South Asia Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- East Asia Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Eastern Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Middle East & Africa Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Sales Forecast 2025 to 2035 by Product Type, Application Type, and End-user Type for 30 Countries
- Competition Outlook, including Market Structure Analysis, Company Share Analysis by Key Players, and Competition Dashboard
- Company Profile
- Agilent Technologies
- Thermo Fisher Scientific
- Siemens Healthineers
- Danaher Corporation
- AB Controls
- Hamilton Company
- Biolitic Lab
- Automata
- Perkin Elmer