MRI-Safe Infusion Monitoring Devices Market Size, Share, Growth and Forecast (2026 - 2036)
The MRI-Safe Infusion Monitoring Devices Market is segmented by Product Type (MRI-compatible Infusion Pumps, Shielded Pump Monitoring Stations, Remote Monitoring Interfaces, MRI-safe Syringe Pump Accessories), Application (Oncology Imaging Procedures, Neurology Imaging, Cardiac MRI Procedures, Interventional MRI Procedures, Pediatric MRI Procedures), End User (Hospitals, Diagnostic Imaging Centers, Ambulatory Surgical Centers, Research Institutes) and Region. Forecast for 2026 to 2036.
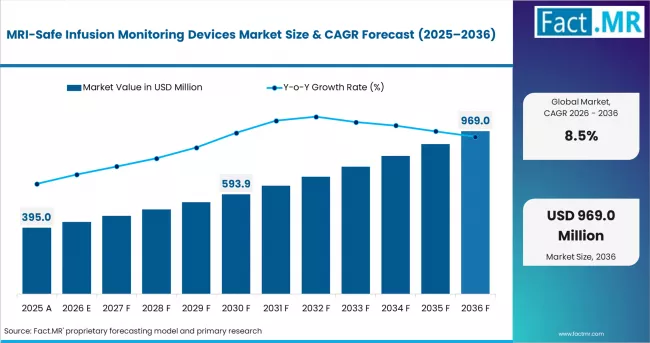
Fact MR states that the MRI-safe infusion monitoring devices market was valued at USD 360 million in 2025 and is forecast to reach USD 395 million in 2026 and USD 890 million by 2036, expanding at a CAGR of 8.5%. Hospitals are projected to maintain leadership with 54%, while MRI-compatible infusion pumps are expected to represent 44%.
MRI-Safe Infusion Monitoring Devices Market Size, Share and Forecast and Outlook By FACT.MR
In 2025, the MRI-safe infusion monitoring devices market was valued at USD 360 million. Fact MR analysis indicates demand for MRI-safe infusion monitoring devices is expected to reach USD 395 million in 2026 and USD 890 million by 2036. FMR estimates the market will expand at a CAGR of 8.5% across the forecast period.
MRI-Safe Infusion Monitoring Devices Market
| Metric | Details |
|---|---|
| Industry Size (2026E) | USD 395 million |
| Industry Value (2036F) | USD 890 million |
| CAGR (2026 to 2036) | 8.5% |
Summary of the MRI-Safe Infusion Monitoring Devices Market
- Market Definition
- The MRI-safe infusion monitoring devices market comprises specialized infusion delivery and monitoring equipment engineered to operate safely within magnetic resonance imaging environments. These devices utilize non-ferromagnetic materials, shielded electronics, and interference-resistant components to ensure continuous and accurate administration of fluids, contrast agents, anesthetics, and medications during diagnostic imaging procedures without compromising image quality or patient safety.
- Demand Drivers
- Increasing MRI procedure volumes requiring uninterrupted administration of contrast agents, sedatives, and critical care medications during imaging workflows.
- Growing emphasis on patient safety compliance standards mandating MRI-compatible monitoring equipment in high-field magnetic environments.
- Expansion of hospital imaging infrastructure supporting integration of MRI-safe infusion systems within radiology departments.
- Rising adoption of advanced MRI procedures in oncology, neurology, and cardiac diagnostics requiring precise infusion control during imaging sessions.
- Development of non-ferromagnetic engineering components enabling safe device operation within strong magnetic fields.
- Replacement demand for legacy infusion pumps not compatible with MRI environments across established healthcare facilities.
- Key Segments Analyzed
- Product Type: MRI-compatible infusion pumps lead with 44% share due to essential role in controlled drug delivery during imaging procedures.
- End User: Hospitals dominate with 54% share supported by concentration of MRI suites and critical care imaging workflows.
- Application: Oncology and neurology imaging procedures represent major usage environments requiring stable medication infusion during prolonged scanning sessions.
- Geography: China and India demonstrate higher growth rates supported by hospital imaging infrastructure expansion, while the United States, Germany, and the United Kingdom reflect replacement-driven procurement demand.
- Analyst Opinion at Fact MR
- Shambhu Nath Jha, Principal Consultant, Fact MR, opines, 'In this updated edition of the MRI-Safe Infusion Monitoring Devices Market report, industry participants observe a specialized clinical device segment shaped by strict patient safety requirements in magnetic resonance environments. Market participants are aligning engineering capabilities with electromagnetic compatibility standards and precision dosing requirements across radiology workflows. Adoption patterns indicate sustained demand for MRI-compatible infusion systems capable of maintaining operational stability without interfering with imaging accuracy. Competitive positioning is influenced by reliability of non-ferromagnetic components, calibration precision, and compliance with evolving clinical safety frameworks through 2036.'
- Strategic Implications/Executive Takeaways
- Strengthen development of non-ferromagnetic device components ensuring safe operation within high-strength magnetic environments.
- Expand product portfolios aligned with MRI-compatible infusion systems supporting oncology, neurology, and cardiac imaging procedures.
- Improve device accuracy and calibration stability supporting continuous medication delivery during extended imaging workflows.
- Support interoperability between infusion monitoring devices and hospital digital infrastructure systems.
- Maintain compliance with regulatory standards governing electromagnetic compatibility and medical device safety certifications.
- Invest in engineering innovation improving shielding efficiency and reliability of electronic monitoring interfaces used in MRI suites.
- Methodology
- Built on primary interviews with medical device manufacturers, biomedical engineers, radiology equipment suppliers, and hospital procurement specialists.
- Benchmarked against MRI procedure volume trends, hospital imaging infrastructure expansion patterns, and clinical infusion device utilization statistics.
- Applied hybrid market estimation combining manufacturer revenue analysis with top down evaluation of MRI diagnostic procedure demand.
- Validated through triangulation using regulatory approvals, clinical device specifications, and company product portfolio disclosures.
- Periodically updated to reflect changes in imaging technology deployment, medical device certification requirements, and hospital procurement cycles.
A CAGR of 8.5%, increasing from USD 395 million to USD 890 million, reflects a relatively stable but elevated growth trajectory with moderate volatility risk tied to hospital capital budgeting cycles and MRI suite expansion rates. Adoption is supported by patient safety compliance requirements and compatibility needs in high-field imaging environments, while variability may arise from procurement delays, device qualification standards, and integration costs within existing radiology infrastructure.
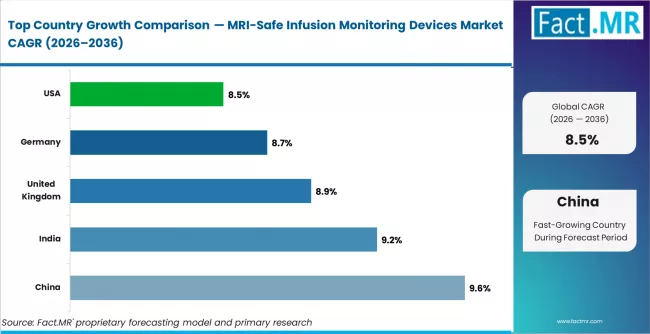
China leads growth at 9.6%, supported by expansion of hospital imaging infrastructure and rising MRI procedure volumes. India follows at 9.2%, driven by increasing installation of advanced diagnostic imaging systems across multispecialty hospitals. The United Kingdom records 8.9% growth, reflecting adoption of MRI-compatible patient monitoring systems in critical care imaging workflows. Germany at 8.7% and the United States at 8.5% represent mature markets where demand is largely replacement-driven due to established installed base of MRI monitoring equipment. A structural constraint across mature markets is longer procurement cycles within regulated healthcare purchasing systems.
Segmental Analysis
MRI-Safe Infusion Monitoring Devices Market Analysis by End User
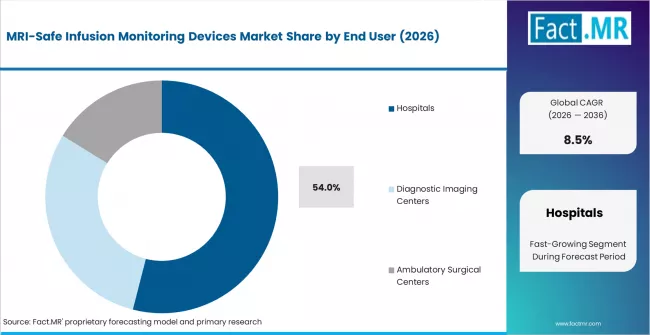
- Market Overview: Hospitals are projected to hold 54% share of the MRI-safe infusion monitoring devices market in 2026. Demand concentration reflects the presence of advanced diagnostic imaging infrastructure and the requirement for continuous patient monitoring during MRI procedures. Clinical environments prioritize infusion monitoring systems capable of operating safely within strong magnetic fields without interfering with imaging accuracy or patient safety protocols.
- Demand Drivers:
- Imaging Procedure Volume: Hospitals perform a high volume of MRI scans requiring uninterrupted administration of contrast agents, sedatives, and critical care medications during imaging sessions.
- Compliance Requirements: Healthcare facilities adopt MRI-safe monitoring equipment designed to meet electromagnetic compatibility standards and patient safety guidelines applicable to radiology departments.
- Critical Care Integration: Intensive care units within hospitals require infusion monitoring devices capable of supporting complex medication regimens during diagnostic imaging procedures.
MRI-Safe Infusion Monitoring Devices Market Analysis by Product Type
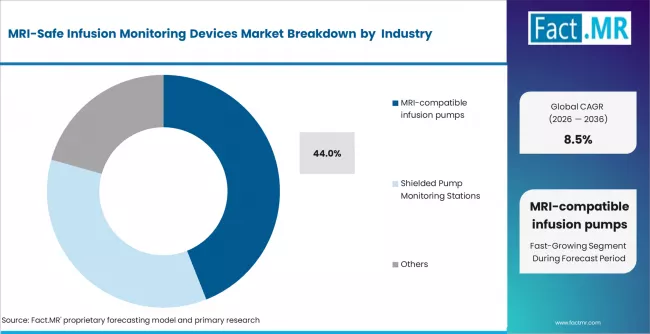
- Market Overview: MRI-compatible infusion pumps are estimated to account for 44% share in 2026, supported by their role in delivering controlled medication dosing within MRI suites where conventional electronic equipment cannot be safely operated. Equipment specifications focus on non-ferromagnetic construction and interference-resistant performance characteristics.
- Demand Drivers:
- Magnetic Field Compatibility: MRI-compatible infusion pumps are designed using non-magnetic components to ensure safe operation within high-strength magnetic environments.
- Dose Precision Requirements: Controlled infusion accuracy supports administration of contrast agents, anesthesia, and continuous medication delivery during extended imaging procedures.
- Hospital Equipment Standardization: Healthcare providers prioritize standardized MRI-safe device portfolios to maintain operational consistency across radiology departments and reduce workflow disruption during patient transfer between diagnostic units.
Key Dynamics
MRI-Safe Infusion Monitoring Devices Market Drivers, Restraints, and Opportunities
Fact MR analysis indicates that the market reflects a structurally specialized medical device niche shaped by the increasing procedural intensity of magnetic resonance imaging workflows. Market size exists because conventional infusion pumps contain ferromagnetic components that cannot operate safely within high magnetic field environments, creating a mandatory substitution requirement for MRI-safe monitoring systems. The current valuation is supported by hospital investment cycles in diagnostic imaging infrastructure, where uninterrupted drug delivery during imaging procedures is required to maintain patient stability and workflow efficiency.
Current market dynamics show declining reliance on conventional infusion pumps requiring patient repositioning outside MRI suites, while integrated MRI-compatible monitoring devices command higher per-unit pricing due to non-ferrous components, electromagnetic shielding, and software-enabled alarm safety protocols. Higher equipment costs are offset by operational efficiency gains, including reduced scan delays and minimized risk of medication interruption during imaging procedures. The market is transitioning toward integrated smart monitoring systems that align with digital hospital infrastructure and safety compliance protocols.
- Magnetic Safety Compliance: FDA 510(k) clearance pathways and IEC 60601 standards define baseline safety requirements for electromagnetic compatibility, creating structural barriers for non-compliant infusion technologies entering MRI environments.
- Imaging Procedure Expansion: North America and Western Europe lead adoption due to higher MRI scan volumes and investment in advanced imaging infrastructure supporting continuous patient monitoring workflows.
- Non-Ferrous Engineering Shift: Transition toward ultrasonic motors, fiber-optic sensors, and shielded electronics increases unit pricing while improving reliability in high-field MRI environments.
Regional Analysis
The MRI-safe infusion monitoring devices market is assessed across Asia Pacific, Europe, and North America, segmented by country-level demand for non-magnetic infusion pumps, MRI-compatible monitoring systems, and critical care drug delivery devices used during imaging procedures. Regional demand reflects hospital imaging infrastructure expansion, patient safety protocols, and regulatory approval pathways for MRI-compatible medical equipment. The full report offers market attractiveness analysis.
.webp)
CAGR Table
| Country | CAGR (2026–2036) |
|---|---|
| China | 9.6% |
| India | 9.2% |
| United Kingdom | 8.9% |
| Germany | 8.7% |
| United States | 8.5% |
Source: Fact MR analysis, based on proprietary forecasting model and primary research
Asia Pacific
Asia Pacific functions as the hospital imaging infrastructure expansion hub supported by increasing MRI installation rates and modernization of critical care monitoring systems. Shenzhen Mindray Bio-Medical Electronics Co., Ltd. strengthens MRI-compatible monitoring technology capability. B. Braun SE expands infusion safety device portfolio availability. Terumo Corporation supports precision drug delivery system innovation.
- China: China is projected to record 9.6% CAGR in MRI-safe infusion monitoring devices through 2036. National Health Commission imaging infrastructure development plan update (March 2023) supports MRI-compatible device procurement. Shenzhen Mindray Bio-Medical Electronics Co., Ltd. expanded MRI monitoring device production capability (July 2023).
- India: India is expected to observe 9.2% CAGR in MRI-safe infusion monitoring devices through 2036. National Medical Device Policy framework (Department of Pharmaceuticals, April 2023) supports domestic manufacturing of critical care equipment. B. Braun India expanded infusion technology manufacturing capacity (June 2023).
Europe
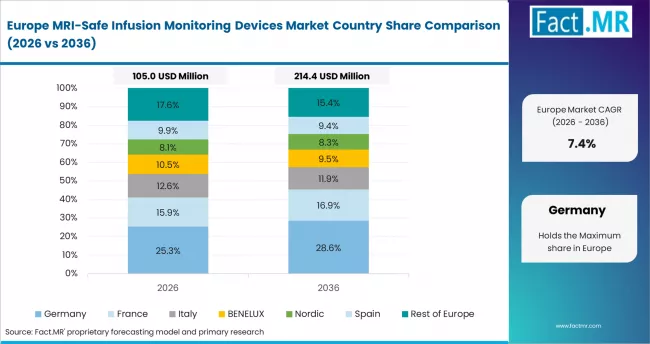
Europe operates as the patient safety compliance center supported by strict medical device regulatory pathways and hospital imaging protocol standardization. B. Braun SE strengthens MRI-compatible infusion technology expertise. Fresenius Kabi AG expands precision infusion system portfolio depth. Smiths Medical supports specialized monitoring equipment supply capability.
- United Kingdom: The United Kingdom is anticipated to expand at 8.9% CAGR in MRI-safe infusion monitoring devices through 2036. NHS medical technology procurement framework update (September 2023) supports adoption of MRI-compatible infusion systems. Smiths Medical expanded infusion monitoring product availability (May 2023).
- Germany: Germany is forecast to grow at 8.7% CAGR in MRI-safe infusion monitoring devices through 2036. EU Medical Device Regulation compliance implementation (European Commission, ongoing) supports safety-certified monitoring device usage. B. Braun SE expanded MRI infusion technology development investment (March 2023).
North America
North America functions as the MRI patient monitoring commercialization center supported by advanced hospital imaging capacity and strict clinical safety requirements for drug delivery during scanning procedures. Baxter International Inc. strengthens infusion device engineering capability. ICU Medical Inc. expands MRI-compatible pump portfolio depth. Medtronic plc supports precision monitoring technology innovation.
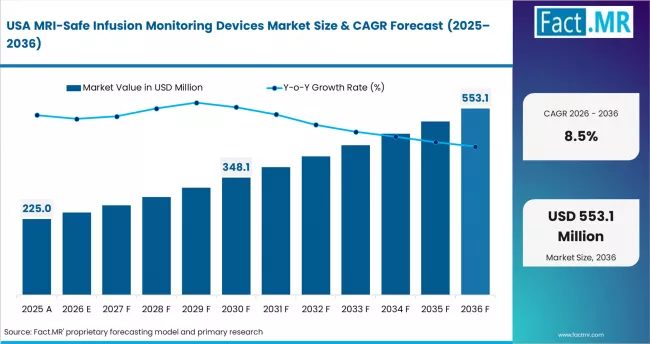
- United States: The United States is projected to register 8.5% CAGR in MRI-safe infusion monitoring devices through 2036. U.S. Food and Drug Administration device safety guidance update (February 2023) supports MRI-compatible equipment approval pathways. Baxter International Inc. expanded MRI infusion pump product development program (August 2023).
Fact MR's analysis of MRI-safe infusion monitoring devices market in global regions consists of country-wise assessment that includes China, India, United Kingdom, Germany, and United States. Readers can find MRI-compatible device adoption trends, regulatory compliance developments, hospital procurement patterns, and competitive technology positioning insights.
Competitive Landscape
What is the Competitive Structure of the MRI-Safe Infusion Monitoring Devices Market?
The MRI-Safe Infusion Monitoring Devices Market demonstrates a moderately concentrated competitive structure, supported by global medical device manufacturers with expertise in infusion therapy and imaging-compatible equipment. Leading companies including B. Braun Melsungen AG, Baxter International Inc., Becton, Dickinson and Company, Medtronic plc, ICU Medical Inc., and Fresenius Kabi AG collectively account for nearly 60–70% of global market share, particularly in hospital environments requiring MRI-compatible infusion safety systems. Competitive positioning is primarily determined by electromagnetic shielding capability, infusion accuracy, device reliability, and compliance with MRI safety standards. High regulatory approval requirements and technical design constraints create barriers to entry, limiting participation to companies with established clinical engineering capabilities and proven infusion system performance.
Structural advantages are observed among companies possessing established infusion therapy portfolios and long-term relationships with hospitals and diagnostic imaging centers. Baxter International Inc. and B. Braun Melsungen AG benefit from integrated product ecosystems covering infusion pumps, consumables, and monitoring accessories compatible with MRI environments. Buyers typically manage supplier dependency through multi-vendor procurement strategies and compatibility evaluation across installed infusion infrastructure. Procurement decisions often consider lifecycle maintenance reliability, calibration stability, and compliance with hospital safety protocols governing imaging procedures. Pricing power remains relatively balanced, though suppliers retain moderate leverage where validated MRI-safe certifications and interoperability requirements create switching constraints for healthcare providers operating complex diagnostic imaging workflows.
Key Players of the MRI-Safe Infusion Monitoring Devices Market
- IRadimed Corporation
- B. Braun Melsungen AG
- Fresenius Kabi AG
- Becton, Dickinson and Company
- Baxter International Inc.
- Medtronic plc
- ICU Medical Inc.
- Mindray Medical International Ltd.
- Arcomed AG
- Smiths Medical
Bibliographies
- [1] National Health Commission (China). (2023, March). Imaging infrastructure development plan update.
- [2] Department of Pharmaceuticals (India). (2023, April). National Medical Device Policy framework.
- [3] National Health Service (United Kingdom). (2023, September). Medical technology procurement framework update.
- [4] European Commission. (ongoing). EU Medical Device Regulation compliance implementation.
- [5] U.S. Food and Drug Administration. (2023, February). Device safety guidance update.
- [6] Shenzhen Mindray Bio-Medical Electronics Co., Ltd. (2023, July). MRI monitoring device production capability expansion.
- [7] B. Braun India. (2023, June). Infusion technology manufacturing capacity expansion.
- [8] Smiths Medical. (2023, May). Infusion monitoring product availability expansion.
- [9] B. Braun Melsungen AG. (2023, March). MRI infusion technology development investment expansion.
- [10] Baxter International Inc. (2023, August). MRI infusion pump product development program expansion.
This Report Addresses
- Market size estimation and revenue forecasts from 2026 to 2036, supported by validated MRI procedure volume benchmarks and hospital imaging infrastructure expansion indicators.
- Growth opportunity mapping across MRI-compatible infusion pumps, shielded monitoring stations, and non-ferromagnetic delivery components aligned with patient safety compliance requirements.
- Segment and regional revenue forecasts covering MRI-compatible infusion pumps, syringe pump accessories, and remote monitoring interfaces across oncology, neurology, cardiac, interventional, and pediatric MRI applications.
- Competition strategy assessment including technology positioning among infusion device manufacturers and benchmarking of electromagnetic shielding capability, infusion precision accuracy, and MRI safety certification compliance.
- Regulatory impact analysis covering FDA 510(k) clearance frameworks, IEC 60601 electromagnetic compatibility standards, and hospital safety protocol requirements influencing MRI-compatible device adoption.
- Market report delivery in PDF, Excel, PPT, and structured data formats designed for hospital procurement teams, biomedical engineers, and diagnostic imaging infrastructure planners.
- Supply chain vulnerability assessments identifying concentration risks in non-ferrous component manufacturing, shielded electronic modules, and precision infusion control system production networks.
MRI-Safe Infusion Monitoring Devices Market Definition
The MRI-safe infusion monitoring devices market includes specialized medical devices designed to safely deliver and monitor fluids, medications, or anesthetics during magnetic resonance imaging procedures. These systems use non-magnetic materials and shielded components to prevent interference with imaging accuracy while ensuring continuous and precise drug administration in radiology and critical care settings.
MRI-Safe Infusion Monitoring Devices Market Inclusions
The report covers global and regional market size, historical performance, and forecast outlook across the study period. It includes segmentation by product type such as MRI-compatible infusion pumps, syringe pumps, and monitoring systems, application areas including oncology and neurology imaging, and end users such as hospitals and diagnostic imaging centers. Pricing trends and supply chain dynamics are evaluated.
MRI-Safe Infusion Monitoring Devices Market Exclusions
The scope excludes standard infusion pumps not certified for MRI environments and general patient monitoring equipment lacking magnetic shielding. Implantable drug delivery systems not intended for active MRI use are excluded. Downstream diagnostic imaging equipment and unrelated radiology devices are not included, focusing strictly on MRI-compatible infusion delivery and monitoring technologies.
MRI-Safe Infusion Monitoring Devices Market Research Methodology
- Primary Research: Interviews were conducted with medical device manufacturers, radiology equipment suppliers, biomedical engineers, and hospital procurement specialists involved in MRI-compatible clinical technologies.
- Desk Research: Public regulatory filings, clinical device specifications, hospital procurement data, and manufacturer technical documentation were reviewed to assess adoption patterns and technology development.
- Market-Sizing and Forecasting: A hybrid modeling approach combining bottom-up analysis of manufacturer revenues and top-down evaluation of MRI procedure volumes was used to estimate market size and growth potential.
- Data Validation and Update Cycle: Data outputs were verified using multiple technical sources, regulatory approvals, and expert consultation. Updates reflect technological improvements in non-magnetic components and increased installation of advanced MRI suites globally.
Report Scope

| Metric | Value |
|---|---|
| Quantitative Units | USD 395 million (2026) to USD 890 million (2036), at a CAGR of 8.5% |
| Market Definition | The MRI-safe infusion monitoring devices market includes specialized infusion delivery and monitoring equipment designed for safe operation within magnetic resonance imaging environments. These systems ensure controlled administration of fluids, contrast agents, and medications during imaging procedures while preventing electromagnetic interference and maintaining patient safety in high magnetic field conditions. |
| Product Type Segmentation | MRI-compatible infusion pumps, Shielded pump monitoring stations, Remote monitoring interfaces, MRI-safe syringe pump accessories |
| Application Segmentation | Oncology imaging procedures, Neurology imaging, Cardiac MRI procedures, Interventional MRI procedures, Pediatric MRI procedures |
| End User Segmentation | Hospitals, Diagnostic imaging centers, Ambulatory surgical centers, Research institutes |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia, and 40+ countries |
| Key Companies Profiled | IRadimed Corporation, B. Braun Melsungen AG, Fresenius Kabi AG, Becton, Dickinson and Company, Baxter International Inc., Medtronic plc, ICU Medical Inc., Mindray Medical International Ltd., Arcomed AG, Smiths Medical |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market estimation validated through primary interviews with medical device manufacturers, hospital procurement specialists, imaging equipment providers, and regulatory compliance experts, supported by procedure volume analysis, healthcare infrastructure assessment, and product certification benchmarking across MRI-compatible medical equipment |
MRI Safe Infusion Monitoring Devices Market Key Segments
-
Product Type:
- MRI Compatible Infusion Pumps
- Shielded Pump Monitoring Stations
- Remote Monitoring Interfaces
- MRI Safe Syringe Pump Accessories
-
Application:
- Oncology Imaging Procedures
- Neurology Imaging
- Cardiac MRI Procedures
- Interventional MRI Procedures
- Pediatric MRI Procedures
-
End User
- Hospitals
- Diagnostic Imaging Centers
- Ambulatory Surgical Centers
- Research Institutes
-
Region:
- North America
- USA
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East and Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Rest of Middle East and Africa
- Other Regions
- Oceania
- Central Asia
- Other Markets
- North America
- Frequently Asked Questions -
How large is the demand for MRI Safe Infusion Monitoring Devices in the global market in 2026?
Demand for MRI safe infusion monitoring devices in the global market is estimated to be valued at USD 395 million in 2026.
What will be the market size of MRI Safe Infusion Monitoring Devices in the global market by 2036?
Market size for MRI safe infusion monitoring devices is projected to reach USD 890 million by 2036.
What is the expected demand growth for MRI Safe Infusion Monitoring Devices in the global market between 2026 and 2036?
Demand for MRI safe infusion monitoring devices in the global market is expected to grow at a CAGR of 8.5% between 2026 and 2036.
Which End User is poised to lead global demand by 2026?
Hospitals are expected to dominate demand, accounting for approximately 54% share in 2026 due to concentration of MRI diagnostic infrastructure and critical care monitoring requirements.
What is Driving MRI Safe Infusion Monitoring Device Demand in China?
Demand is influenced by expansion of hospital imaging infrastructure and increasing adoption of MRI compatible patient monitoring equipment.
What is the China Growth Outlook in this Report?
China is projected to expand at a CAGR of 9.6% between 2026 and 2036 reflecting increased installation of MRI systems across tertiary care facilities.
What is Driving MRI Safe Infusion Monitoring Device Demand in India?
Demand is supported by growth in diagnostic imaging capacity and procurement of MRI compatible infusion systems across multispecialty hospitals.
What is the India Growth Outlook in this Report?
India is projected to grow at a CAGR of 9.2% between 2026 and 2036 reflecting expansion of hospital infrastructure and imaging accessibility.
What is Driving MRI Safe Infusion Monitoring Device Demand in the United Kingdom?
Demand is associated with modernization of radiology departments and replacement of legacy infusion monitoring systems with MRI compatible devices.
What is the United Kingdom Growth Outlook in this Report?
The United Kingdom is projected to expand at a CAGR of 8.9% between 2026 and 2036 reflecting continued procurement of MRI compatible clinical equipment.
What is Driving MRI Safe Infusion Monitoring Device Demand in Germany?
Demand is linked to hospital equipment upgrades and integration of MRI safe monitoring technologies across diagnostic imaging workflows.
What is the Germany Growth Outlook in this Report?
Germany is projected to grow at a CAGR of 8.7% between 2026 and 2036 supported by replacement demand in clinical imaging environments.
What is Driving MRI Safe Infusion Monitoring Device Demand in the United States?
Demand is influenced by adoption of MRI compatible patient monitoring devices aligned with safety standards for magnetic resonance environments.
What is the United States Growth Outlook in this Report?
The United States is projected to expand at a CAGR of 8.5% between 2026 and 2036 reflecting continued investment in advanced imaging infrastructure.
Which Companies are Identified as Leading Participants in the MRI Safe Infusion Monitoring Devices Market?
IRadimed Corporation is identified as a leading manufacturer due to its specialization in MRI compatible patient monitoring and infusion system technologies.
What are MRI Safe Infusion Monitoring Devices and What are They Mainly Used For?
MRI safe infusion monitoring devices are specialized medical systems designed to safely deliver and monitor intravenous fluids within magnetic resonance imaging environments.
What does MRI Safe Infusion Monitoring Devices Market Mean in this Report?
MRI safe infusion monitoring devices market refers to global production, procurement, and clinical utilization of MRI compatible infusion systems across hospital imaging departments.
What is Included in the Scope of this MRI Safe Infusion Monitoring Devices Market Report?
Scope includes device demand by end user, hospital based imaging applications, and integration of infusion monitoring systems within MRI environments.
What is Excluded from the Scope of this Report?
Standard infusion pumps not designed for MRI compatibility and non imaging patient monitoring equipment are excluded unless specifically configured for MRI safe operation.
What does Market Forecast Mean on this Page?
Market forecast represents a modeled projection developed using defined assumptions to support strategic planning and medical device demand estimation.
How is the MRI Safe Infusion Monitoring Devices Market Forecast Developed in this Report?
Forecast modeling is based on hospital procurement patterns, imaging infrastructure expansion, and validation through manufacturer level production assessment.
What does Data Validation Approach Mean in this Report?
Primary clinical inputs, company disclosures, and publicly available healthcare infrastructure statistics are used to verify market sizing assumptions.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User , 2026 to 2036
- Hospitals
- Diagnostic Imaging Centers
- Ambulatory Surgical Centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End User , 2021 to 2025
- Absolute $ Opportunity Analysis By End User , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- MRI-compatible infusion pumps
- Shielded Pump Monitoring Stations
- Others
- MRI-compatible infusion pumps
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By End User
- By Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Product Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End User
- By Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Product Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End User
- By Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Product Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End User
- By Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Product Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By End User
- By Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Product Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End User
- By Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Product Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End User
- By Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By End User
- By Product Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By End User
- By Product Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End User
- By Product Type
- Competition Analysis
- Competition Deep Dive
- IRadimed Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- B. Braun Melsungen AG
- Fresenius Kabi AG
- Becton, Dickinson and Company
- Baxter International Inc.
- IRadimed Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by End User , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by End User
- Figure 6: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Product Type
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by End User
- Figure 23: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by End User
- Figure 30: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Product Type
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by End User
- Figure 37: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Product Type
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by End User
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by End User
- Figure 51: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Product Type
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis