Heart Pump Devices Market Size, Share, Growth and Forecast (2026 - 2036)
Heart Pump Devices Market is segmented by Type Implanted Heart Pump Devices and Extracorporeal Heart Pump Devices, Product Ventricular Assist Devices including LVADs, RVADs, BiVADs and PVADs, Total Artificial Hearts, Intra-aortic Balloon Pumps, and Extracorporeal Membrane Oxygenation, End use Hospitals, Cardiac Centers and Others, and Region. Forecast period is 2026 to 2036.
Fact.MR believes the market is moving through a destination or bridge turning point. Traditional pulsatile pumps developed mainly as temporary bridge to transplant solutions are gradually being replaced by continuous flow LVADs and total artificial hearts built for long term destination therapy. This shift is being driven by donor organ shortages and broader regulatory approvals that position mechanical circulatory support as a definitive treatment rather than a temporary measure.
Segmental Analysis
Heart Pump Devices Market Analysis by Type
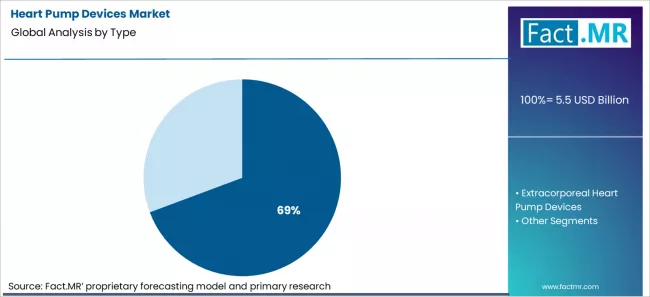
Implanted heart pump devices are expected to hold 69.3% of the market in 2026. These surgically placed systems allow patients to leave the hospital and return to daily activities. With durability ranging from 3 to 10 years, they offer meaningful quality-of-life improvement compared to extracorporeal systems that require intensive care unit support.
- Abbott reported USD 480 million in HeartMate 3 sales in 2024, representing roughly 1,600 implants [5]. Since the withdrawal of Medtronic’s HVAD, HeartMate 3 remains the only FDA-approved durable LVAD for destination therapy in the U.S., effectively creating a single-supplier market under regulatory oversight.
- Innovation is also focused on miniaturization. In September 2024, BiVACOR began first-in-human trials for a smaller total artificial heart designed to fit patients previously excluded due to body size constraints [6]. This could expand eligibility to thousands of additional patients.
- Outpatient care is improving as well. According to a 2024 scientific statement from the American Heart Association, 75% of HeartMate 3 patients are discharged within two weeks, with significantly fewer readmissions compared to earlier devices [7]. This supports the expansion of VAD programs beyond traditional transplant centers.
Heart Pump Devices Market Analysis by Product
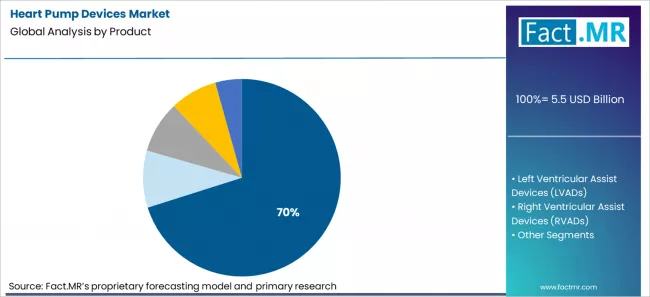
Left Ventricular Assist Devices are projected to account for 70.1% of the market in 2026. This reflects the fact that most advanced heart failure cases involve left ventricular dysfunction. Right ventricular and biventricular failure cases represent a smaller share of mechanical support needs.
- Five-year survival for Heartmate 3 patients reached 59.1% in the MOMENTUM 3 destination therapy cohort, with fewer adverse events compared to earlier systems [8]. These results led professional guidelines to upgrade LVAD destination therapy to a recommended standard of care.
- In the temporary support segment, Abiomed’s Impella devices reached 160,000 procedures in 2023 across 2,500 U.S. hospitals [9]. These percutaneous systems are typically used for short-term stabilization and complement rather than replace durable LVAD therapy.
- Total artificial hearts such as SynCardia’s CardioWest remain approved only for bridge-to-transplant use, with 79% survival to transplant or recovery since 2004 [10]. However, regulatory caution around permanent external drivers has limited their expansion into destination therapy, leaving room for future innovation in fully implantable systems.
Drivers, Restraints, and Opportunities
Fact.MR analysis shows the heart pump devices market is being shaped by two powerful forces. First, aging populations and rising diabetes rates are increasing the number of patients with advanced heart failure. Second, regulators are steadily expanding access to mechanical circulatory support, moving it from a temporary bridge-to-transplant solution to a recognized destination therapy. In the past, ventricular assist devices were limited to the sickest patients under INTERMACS Profile 1 and 2. This kept the U.S. market to roughly 2,000 to 3,000 procedures per year.
- A key challenge has been balancing durability with quality of life. Early pulsatile LVADs were bulky and lasted only 12 to 24 months. Second-generation continuous-flow devices improved durability but required intensive anticoagulation, increasing bleeding risk.
- CMS National Coverage Determination 20.9.1, revised in October 2022, expanded Medicare coverage for destination therapy to include INTERMACS Profile 3 patients [2]. Coverage is limited to centers meeting strict survival benchmarks and registry reporting requirements. As a result, implant volumes are increasingly concentrated in about 65 high-volume U.S. centers performing the majority of procedures.
- Clinical evidence has reinforced this shift. The MOMENTUM 3 trial, published in the New England Journal of Medicine in November 2019, showed superior outcomes for HeartMate 3 compared to earlier devices, with 86% freedom from disabling stroke or device failure at two years [3].
Regional Analysis
The heart pump devices market is analyzed across North America, Europe, Asia-Pacific, Latin America, and Middle East. Regional device adoption intensity is shaped by cardiac transplant infrastructure maturity, interventional cardiology versus cardiac surgery competitive dynamics, national health insurance reimbursement policy, and per-capita cardiac surgical case volumes. The report offers detailed market attractiveness analysis comparing destination therapy coverage status, VAD center certification requirements, and device approval timelines across regional regulatory jurisdictions.
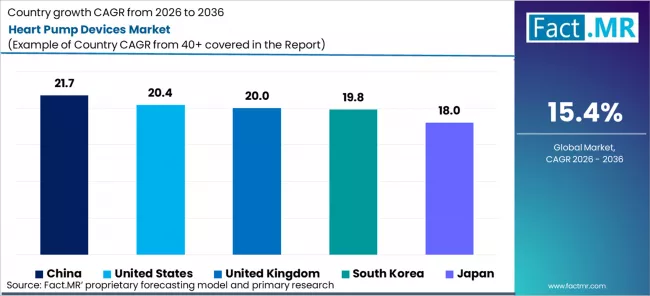
| Country | CAGR% |
|---|---|
| China | 21.7% |
| United States | 20.4% |
| United Kingdom | 20.0% |
| South Korea | 19.8% |
| Japan | 18.0% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
North America Heart Pump Devices Market Analysis
North America remains the global center for mechanical circulatory support innovation. Regulatory precedents from the FDA, Medicare coverage expansion, and the INTERMACS registry system have created the most advanced VAD adoption environment worldwide. Abbott Laboratories leads the U.S. durable LVAD market with HeartMate 3 following Medtronic’s HVAD withdrawal. Abiomed, now part of Johnson and Johnson, dominates the temporary percutaneous segment with Impella devices across more than 2,500 hospitals. Strong transplant infrastructure, high cardiac surgery volumes, and supportive reimbursement sustain regional leadership.
- United States: U.S. demand is projected to grow at 20.4% CAGR through 2036. In 2024, Abbott invested USD 150 million to expand production capacity in California to address backlog demand. The Joint Commission introduced VAD center certification in 2023, further formalizing quality standards.
Europe Heart Pump Devices Market Analysis
Europe offers an alternative regulatory pathway through CE Mark approval, often enabling earlier access for emerging technologies. Registry systems such as EUROMACS support data tracking, though reimbursement varies by country. Adoption remains more conservative than in the U.S., reflecting transplant preference and budget constraints.
- United Kingdom: The UK market is forecast to grow at 20.0% CAGR. NHS England centralized VAD implantation into 12 designated centers in 2024. NICE recommended HeartMate 3 as cost effective destination therapy in late 2023, expanding access beyond bridge to transplant. Clinical trials and registry partnerships continue to support broader adoption.
Asia Pacific Heart Pump Devices Market Analysis
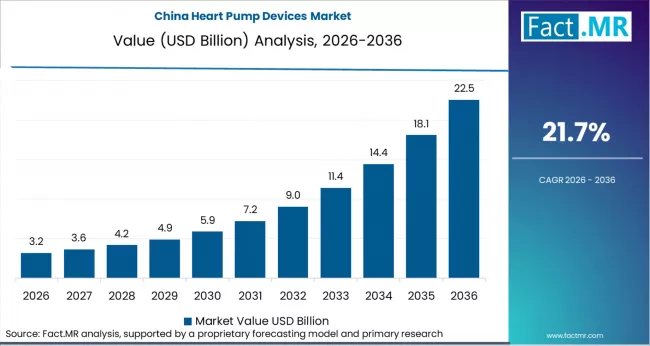
Asia Pacific represents the strongest long term growth opportunity. Aging populations, diabetes prevalence, and expanding cardiac infrastructure are increasing demand. Domestic manufacturers in China are introducing lower cost LVADs, reducing reliance on imports and supporting wider access.
- China: China is projected to grow at 21.7% CAGR. National reimbursement pilots launched in 2024 now cover destination therapy in major provinces. The National Medical Products Administration accelerated approvals for domestic LVADs, enabling more affordable alternatives to imported systems. Leading cardiac centers report rising destination therapy volumes.[4]
- Japan: Japan is expected to grow at 19.8% CAGR. HeartMate 3 received bridge to transplant approval in 2023, with destination therapy under review. Reimbursement expanded in 2024, and domestic manufacturers such as Terumo Corporation are advancing next generation systems.
- South Korea: South Korea is forecast to grow at 18.0% CAGR. National insurance expanded reimbursement to include destination therapy in 2024. Major hospitals have developed outpatient VAD management programs, strengthening long term care infrastructure.
Competitive Aligners for Market Players
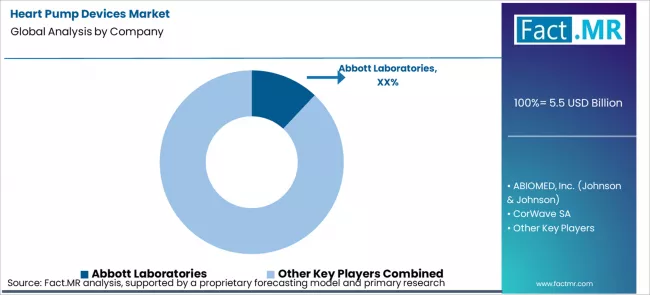
The heart pump devices market is highly concentrated in the durable implanted LVAD segment. FDA destination therapy approvals require long term randomized trials that can take over a decade and cost hundreds of millions of dollars, limiting participation to large, well-funded cardiovascular device companies.
Abbott’s leadership is reinforced by its long clinical history, with continuous FDA approvals from HeartMate XVE to HeartMate 3, and close alignment with evolving Medicare coverage policies. Its integrated approach, spanning device design, clinical trials, and post market registries, creates barriers that smaller innovators struggle to overcome. Medtronic’s exit highlights that clinical outcomes, not scale or investment size, ultimately determine market survival. [5]
Buyer behavior further strengthens supplier dominance. Surgeons prioritize outcomes, familiarity, and institutional protocols, with price playing a secondary role. Group purchasing organizations achieve only modest discounts, while high switching costs tied to training, registries, and consumables limit hospitals’ ability to change suppliers.
Recent Development
- In December 2025, Johnson & Johnson completed the acquisition of Abiomed, with the business continuing to operate independently under Johnson and Johnson’s MedTech division.
- In 2025, Abiomed announced that the U.S. Food and Drug Administration granted approval for two clinical research studies evaluating Impella heart pump systems in patients experiencing acute myocardial infarction and cardiogenic shock.
Key Players
- Abbott Laboratories
- ABIOMED, Inc. (Johnson & Johnson)
- CorWave SA
- LivaNova PLC
- Berlin Heart GmbH
- SynCardia Systems LLC
- Jarvik Heart, Inc.
- BiVACOR Inc.
- Medtronic plc
Bibliography
- [1] Abbott Laboratories. (2024, October). Q3 2024 Earnings Call Transcript Mechanical Circulatory Support Business Update.
- [2] Centers for Medicare & Medicaid Services. (2022, October). National Coverage Determination (NCD) 20.9.1 Ventricular Assist Devices as Destination Therapy.
- [3] Mehra, M. R., et al. (2019, November). Two-Year Outcomes with a Magnetically Levitated Cardiac Pump in Heart Failure. New England Journal of Medicine, 381(17), 1618-1627.
- [4] China National Medical Products Administration. (2024, March). Innovative Medical Device Priority Review Approval List LVAD Category 2020-2024.
- [5] Abbott Laboratories. (2025, January). Q4 2024 Earnings Release Cardiovascular & Neuromodulation Segment Revenue.
This Report Addresses
- Market intelligence for strategic planning: Clear analysis of mechanical circulatory support adoption trends. Review of bridge to transplant versus destination therapy shift. Comparison of regulatory timelines across FDA, CE Mark, and NMPA pathways. Assessment of clinical evidence requirements for approval and reimbursement.
- Market size and forecast: Global market valued at USD 4.8 billion in 2025. Projected to reach USD 23.2 billion by 2036 at 15.4% CAGR. Segment sizing by device type, product, end use, and region included.
- Growth opportunity mapping: Focus on destination therapy LVADs for INTERMACS Profile 3 to 4 patients. Coverage of miniaturized pumps for smaller body sizes. Analysis of total artificial hearts for biventricular failure. Review of percutaneous support in cardiogenic shock.
- Segment and regional forecasts: Country level CAGR for the United States, United Kingdom, China, Japan, South Korea, and additional markets. Breakdown by device type and indication mix. Evaluation of demographic and reimbursement drivers.
- Competition strategy assessment: Positioning analysis of Abbott, ABIOMED under Johnson and Johnson, LivaNova, and emerging Chinese players. Review of clinical trial timelines, surgeon training reach, and payer contracting models.
- Product and compliance tracking: Analysis of FDA indication expansion, CMS coverage updates, European MDR rules, and registry participation requirements.
- Clinical outcomes benchmarking: Survival comparisons across device generations. Review of stroke, bleeding, readmissions, and cost effectiveness versus transplant.
- Report delivery formats: Excel data tables with procedure and pricing forecasts. PowerPoint summary with milestones and competition insights. Full PDF report with verified clinical and regulatory references.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD 1.6 billion (2026) to USD 2.7 billion (2036), at a CAGR of 5.5% |
| Market Definition | Aquaculture lighting and environmental control systems encompass LED and spectrum-tunable lighting, aeration and oxygenation equipment, water quality sensors, and automated feeding systems deployed in fish and shellfish farming facilities to precisely manage the biological environment and optimize production performance. |
| System Type | LED Lighting, Aeration/Oxygenation, Water Quality Sensors, Feeding/Automation |
| Species | Salmonids, Tilapia, Shrimp, Others |
| Farm Type | Land-Based, Offshore Marine, RAS, Cage Farms |
| Buyer Type | Large Farms, SME Farms, Research/Other |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, Australia & New Zealand, India, ASEAN, Rest of Asia Pacific, Norway, Germany, United Kingdom, France, Spain, Netherlands, Nordics, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Ecuador, Rest of Latin America, Kingdom of Saudi Arabia, United Arab Emirates, South Africa, Turkey, Rest of Middle East & Africa |
| Key Companies Profiled | Signify, AKVA Group, ams OSRAM, Innovasea, Xylem, Pentair, Monitorfish, LumenAqua, ScaleAQ, OxyGuard |
| Forecast Period | 2026 to 2036 |
| Approach | Top-down and bottom-up market modeling validated through primary interviews with aquaculture operators, environmental control system vendors, RAS technology integrators, and fisheries ministry officials, supported by FAO production statistics, national aquaculture development plans, and company disclosed capex data. |
Heart Pump Devices Market by Segment
-
By Type :
- Implanted Heart Pump Devices
- Extracorporeal Heart Pump Devices
-
By Product :
- Ventricular Assist Devices (VADs)
- Left Ventricular Assist Devices (LVADs)
- Right Ventricular Assist Devices (RVADs)
- Bi-ventricular Assist Devices (BiVADs)
- Percutaneous Ventricular Assist Devices (PVADs)
- Ventricular Assist Devices (VADs)
-
By End-use :
- Hospitals
- Cardiac Centers
- Others
-
Region :
- Asia Pacific
- China
- Japan
- South Korea
- Australia & New Zealand
- India
- ASEAN
- Rest of Asia Pacific
- Europe
- Norway
- Germany
- United Kingdom
- France
- Spain
- Netherlands
- Nordics
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Ecuador
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
- Asia Pacific
- Frequently Asked Questions -
How large is the heart pump devices market in 2025?
The heart pump devices market was valued at USD 4.8 billion in 2025.
What will the market size be in 2026?
The market is estimated to grow to USD 5.5 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 23.2 billion by 2036.
What is the expected CAGR for the forecast period 2026 to 2036?
FACT.MR projects a CAGR of 15.4% from 2026 to 2036.
Which Type segment holds the largest market share?
Implanted Heart Pump Devices command 69.3% market share in 2026.
Which Product sub-segment leads within VADs?
Left Ventricular Assist Devices (LVADs) hold 70.1% of the VAD segment share.
Which End-use setting captures the largest share?
Hospitals capture 84.2% market share as surgical VAD implantation and ECMO cannulation require cardiac surgery operating rooms and cardiopulmonary bypass capability.
What is the absolute dollar growth from 2026 to 2036?
The absolute dollar growth from 2026 to 2036 represents a transformational gain of USD 17.7 billion.
What is driving China's growth acceleration?
China's 4.5% CAGR is driven by National Medical Products Administration approval of three domestically manufactured LVADs (Evaheart, Chio, Chindex)
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- Implanted Heart Pump Devices
- Extracorporeal Heart Pump Devices
- Implanted Heart Pump Devices
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Ventricular Assist Devices (VADs)
- Left Ventricular Assist Devices (LVADs)
- Right Ventricular Assist Devices (RVADs)
- Bi-ventricular Assist Devices (BiVADs)
- Percutaneous Ventricular Assist Devices (PVADs)
- Ventricular Assist Devices (VADs)
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Product
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- ABIOMED, Inc. (Johnson & Johnson)
- CorWave SA
- LivaNova PLC
- Berlin Heart GmbH
- SynCardia Systems LLC
- Jarvik Heart, Inc.
- BiVACOR Inc.
- Medtronic plc
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Product, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Product
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Type , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Type
- Figure 23: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Product, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Type , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Product, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Product
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Type , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Product, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Product
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Type , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Product, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Product
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Type , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Product, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Product
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Type , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Product, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Type , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Product, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis