Peripheral Intravenous Catheter Market Size, Share, Growth and Forecast (2026 - 2036)
The Peripheral Intravenous Catheter Market is segmented by Product Type (Short Peripheral IV Catheter, Integrated/Closed Peripheral IV Catheter, and Others), Technology (Safety PIVC and Conventional PIVC), End User (Hospitals, Clinics, Ambulatory Surgical Centers, and Others), and Region. Forecast for 2026 to 2036.
According to FACT.MR, the peripheral intravenous catheter market is entering a decisive shift toward safety-focused innovation. Standard PIVC products are gradually losing share as healthcare providers move toward safety-engineered and integrated catheter systems. The shift is fueled by stricter needlestick prevention mandates and rising hospital-acquired infection reduction targets that conventional catheter designs cannot effectively address.
Summary of Peripheral Intravenous Catheter Market
- Market Definition
- Peripheral intravenous catheters are short vascular access devices inserted into peripheral veins to deliver medications, fluids, and blood products across acute, surgical, and ambulatory care settings.
- Demand Drivers
- OSHA blood borne pathogen standards and EU needlestick prevention directive enforcement is mandating hospital adoption of safety-engineered PIVC designs across regulated markets.
- Hospital-acquired infection reduction targets under CMS value-based purchasing programmes are driving procurement of integrated closed-system catheters that reduce catheter-related bloodstream infection risk.
- Expanding hospital infrastructure under India’s Ayushman Bharat programme and China’s 14th Five-Year Plan is increasing PIVC procedure volumes and first-time premium catheter adoption across new facility openings.
- Key Segments Analyzed
- By Product Type: Short peripheral IV catheters hold approximately 38% share in 2026 due to their dominance in routine short-term IV therapy across all healthcare settings.
- By Technology: Safety PIVCs lead at approximately 33% share in 2026, reflecting regulatory compliance requirements and hospital prioritization of healthcare worker needlestick protection.
- By End User: Hospitals dominate at approximately 56% share in 2026, driven by high patient volumes, complex treatment regimens, and centralized procurement budgets.
- By Geography: Asia Pacific leads growth through hospital infrastructure expansion and increasing procedure volumes.
- Analyst Opinion at FACT.MR
- Shambhu Nath Jha, Principal Consultant at Fact.Mr, opines that CXOs will find the market’s 7.1% CAGR through 2036 anchored by needlestick prevention mandates and hospital infection control investment, with integrated closed-system catheter adoption creating a durable premium revenue stream.
- Strategic Implications
- Manufacturers should accelerate development of integrated PIVC platforms combining passive safety mechanisms, antimicrobial coatings, and securement features targeting hospital infection control procurement committees.
- Distributors in India and China should offer clinical in-service training and protocol development support alongside catheter supply to drive conversion from conventional to safety-engineered devices.
- Companies should engage NHS, SSN, and national hospital procurement agencies with health economics data demonstrating CRBSI reduction and total cost-per-patient advantages of closed-system catheters.
- Methodology
- Market sizing used hospital admission volumes and IV catheter consumption per admission by care setting, validated against OEM revenue data and distributor shipment records.
- FDA, EU MDR, and WHO injection safety publications from 2024 to 2025 supported regulatory compliance demand analysis across regions.
- Forecasts incorporated hospital infrastructure investment data, procedure volume statistics, safety device adoption surveys, and primary interviews with procurement managers and IV therapy nurses.
Segmental Analysis
Peripheral Intravenous Catheter Market Analysis by Product Type
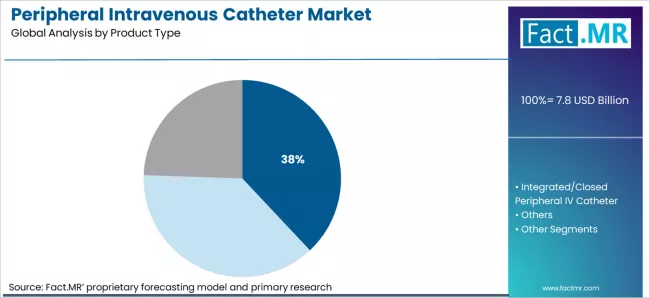
Based on FACT.MR’s peripheral intravenous catheter market report, short peripheral IV catheters are estimated to hold approximately 38% share in 2026. Short PIVCs dominate because they address the broadest range of routine IV therapy indications, are compatible with all standard IV administration sets, and are stocked universally across hospital and clinic care settings, making them the default procurement choice for general-purpose IV access.
- BD Nexiva Closed IV Catheter Expansion: In 2025, Becton, Dickinson and Company expanded commercial distribution of its Nexiva Closed IV Catheter System into Asian and Latin American hospital markets, positioning integrated closed-system products against conventional PIVC procurement contracts. [5]
- B. Braun Introcan Safety 3 Development: In 2024, B. Braun Melsungen AG received CE Mark renewal for its Introcan Safety 3 passive safety PIVC with an updated needle retraction mechanism designed to reduce accidental needlestick events during catheter insertion and removal. [6]
- Integrated PIVC Adoption Trend: A 2024 survey published by the Association for Vascular Access showed integrated closed IV catheters among the fastest-growing device categories in U.S. hospital vascular access programme procurement, reflecting hospital system-wide conversion from conventional to closed-system platforms. [7]
Peripheral Intravenous Catheter Market Analysis by Technology
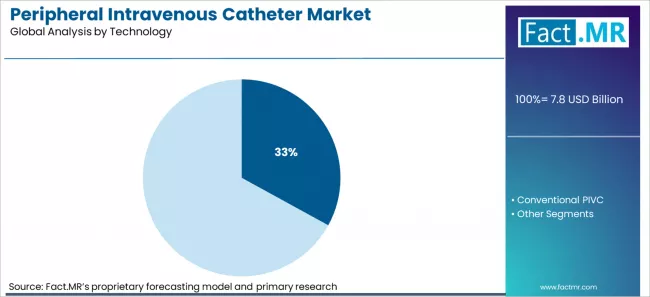
Based on FACT.MR’s peripheral intravenous catheter market report, safety PIVCs are projected to hold approximately 33% share in 2026. Safety-engineered PIVCs hold their leading position because needle stick prevention regulations in North America, Europe, and increasingly Asia Pacific require documented use of safety-activated devices, making conventional non-safety catheters a regulatory and liability risk for hospital procurement officers.
- Teleflex Arrow Safety PIVC Launch: In 2025, Teleflex Incorporated launched its Arrow Nanocath safety PIVC with an integrated passive needle protection mechanism, targeting the European hospital market where EU Directive 2010/32/EU safety device requirements are fully enforced. [8]
- Terumo Surflo Safety Technology: In 2024, Terumo Corporation updated its Surflo safety PIVC line with a new single-handed activation safety mechanism for the North American market, receiving FDA 510(k) clearance in August 2024. [9]
- Safety PIVC Conversion Programme Trend: NHS Supply Chain in the United Kingdom issued a 2025 procurement framework update requiring all new PIVC tenders to specify passive safety activation as a mandatory product criterion, accelerating conventional-to-safety conversion across NHS trusts. [10]
Peripheral Intravenous Catheter Market Analysis by End User
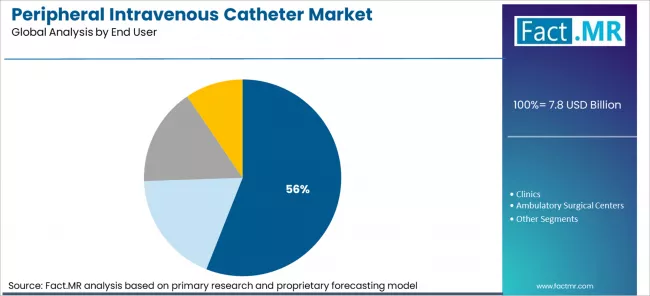
Based on FACT.MR’s peripheral intravenous catheter market report, hospitals are projected to hold approximately 56% share in 2026. Hospitals dominate because they concentrate the highest IV therapy procedure volumes, manage the most complex patient populations requiring frequent catheter replacement, and operate centralised procurement systems that negotiate bulk supply contracts directly with PIVC manufacturers and distributors.
- HCA Healthcare Procurement Standardization: In 2025, HCA Healthcare standardised safety PIVC procurement across its 186 hospitals in the United States and United Kingdom to BD Nexiva and Smiths Group Venflon Pro Safety, consolidating suppliers to two approved vendors per catheter category. [11]
- Ambulatory Surgical Center PIVC Adoption: In 2024, the Ambulatory Surgery Center Association reported accelerating PIVC procurement among U.S. ASCs as outpatient surgical volumes grew 8% versus 2023, driven by CMS approval of new outpatient procedure codes increasing IV therapy requirements. [12]
Clinic Segment Growth Trend: Vygon’s 2025 distributor network expansion in Southeast Asia targeted clinic and day care centre accounts, reflecting rising demand for PIVC devices in outpatient infusion therapy outside traditional hospital settings. [13]
Regional Analysis
The peripheral intravenous catheter market is analysed across Asia Pacific, Europe, North America, Latin America, and Middle East and Africa, covering 40-plus countries with distinct demand profiles shaped by hospital infrastructure investment, needlestick prevention regulation enforcement, and surgical and IV therapy procedure volumes. The full report offers market attractiveness analysis based on regulatory enforcement timelines and hospital capacity expansion pipelines.
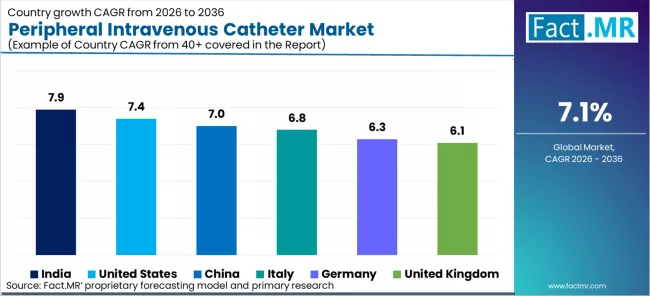
| Country | CAGR (2026-2036) |
|---|---|
| India | 7.9% |
| United States | 7.4% |
| China | 7.0% |
| Italy | 6.8% |
| Germany | 6.3% |
| United Kingdom | 6.1% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
Asia Pacific Peripheral Intravenous Catheter Market Analysis
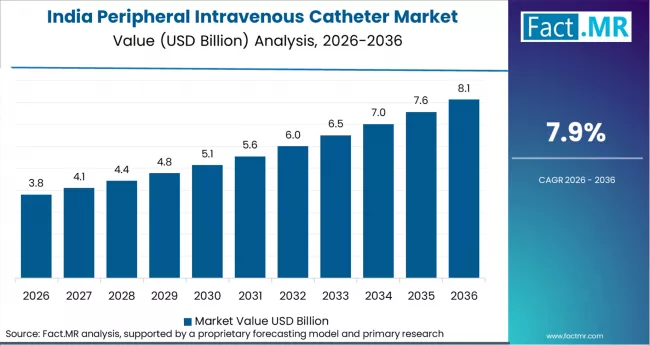
Asia Pacific is the fastest-growing region for peripheral intravenous catheters, driven by hospital infrastructure expansion in India and rising IV therapy procedure volumes across China’s county-level hospital network. Global OEMs including BD and Terumo supply premium safety PIVC products, while regional manufacturers compete in conventional catheter segments through cost-competitive pricing on public hospital tenders.
- India: Demand for peripheral intravenous catheters in India is projected to rise at 7.9% CAGR through 2036. The Ayushman Bharat Health Infrastructure Mission continued funding 150 new critical care hospital blocks and 600 district hospital upgrades through 2025, directly expanding IV therapy procedure capacity. The National Medical Devices Policy 2023 also incentivised domestic PIVC manufacturing, with HLL Lifecare Limited expanding its catheter production line in Kerala in February 2025 to meet public hospital procurement demand under GeM portal tenders.
- China: Demand for peripheral intravenous catheters in China is expected to grow at 7.0% CAGR through 2036. The National Health Commission’s 14th Five-Year Plan for healthcare infrastructure approved construction of 1,200 county-level hospitals through 2025, each requiring standardized IV device supply under national healthcare procurement rules. Jiangsu Province’s 2025 centralized medical device procurement tender included safety PIVC as a mandatory specification category for the first time, signaling a regulatory shift from conventional to safety-engineered devices in public hospital purchasing.
FACT.MR’s Asia Pacific analysis of the peripheral intravenous catheter market covers China, Japan, South Korea, India, Australia and New Zealand, ASEAN, and Rest of South Asia and Pacific. Readers can find country CAGR forecasts, hospital infrastructure investment data, regulatory safety device timelines, and OEM competitive positioning.
Europe Peripheral Intravenous Catheter Market Analysis
Europe is a mature, regulation-driven peripheral intravenous catheter market anchored by EU Directive 2010/32/EU needlestick prevention requirements and EU MDR compliance obligations. B. Braun Melsungen AG and Smiths Group hold strong competitive positions through broad safety PIVC portfolios, while Vygon and regional distributors serve mid-market clinical and ambulatory accounts.
- Italy: Demand for peripheral intravenous catheters in Italy is forecast to grow at 6.8% CAGR through 2036. Italy’s implementation of Decreto Legislativo 19/2014 continued generating documented safety device procurement requirements across SSN hospital networks, with CONSIP’s 2025 national health device framework contract mandating passive-activation safety PIVCs across all contracted public hospitals.
- Germany: Demand for peripheral intravenous catheters in Germany is projected to rise at 6.3% CAGR through 2036. Germany’s TRBA 250 biological agents technical rule, updated in 2024, tightened documentation requirements for safety-engineered sharp device usage in hospital and outpatient clinical settings, accelerating safety PIVC procurement across statutory health insurer-contracted facilities.
- United Kingdom: Demand for peripheral intravenous catheters in the United Kingdom is expected to grow at 6.1% CAGR through 2036. NHS Supply Chain’s 2025 Medical Devices Framework for vascular access set passive safety activation as a mandatory criterion in all PIVC category tenders, effectively excluding conventional non-safety catheters from NHS procurement.
FACT.MR’s European analysis of the peripheral intravenous catheter market covers Germany, France, the United Kingdom, Italy, Spain, Nordic countries, BENELUX, and Rest of Western Europe. Readers can find EU MDR compliance impact data, needle stick prevention directive enforcement trends, and country-level forecasts by product type and technology.
North America Peripheral Intravenous Catheter Market Analysis
North America is the largest revenue market for peripheral intravenous catheters, sustained by OSHA blood borne pathogen standard enforcement, CMS hospital-acquired condition penalties, and strong adoption of integrated closed-system catheter platforms. Becton, Dickinson and Company and Smiths Group dominate premium PIVC supply, supported by broad hospital group purchasing organisation contracts.
- United States: Demand for peripheral intravenous catheters in the United States is projected to rise at 7.4% CAGR through 2036. OSHA’s Bloodborne Pathogens Standard (29 CFR 1910.1030) continued requiring annual exposure control plan reviews at all healthcare facilities in 2025, sustaining safety PIVC procurement as a compliance obligation.
FACT.MR’s North American analysis of the peripheral intravenous catheter market covers the United States, Canada, and Mexico. Readers can find OSHA enforcement impact data, CMS value-based purchasing trends, hospital group purchasing organization contract dynamics, and country forecasts by product type and technology.
Competitive Aligners for Market Players
The global peripheral intravenous catheter market is moderately concentrated in the premium safety-engineered device tier and highly fragmented in the conventional catheter segment. Becton, Dickinson and Company, B. Braun Melsungen AG, Smiths Group, and Terumo Corporation hold the largest global positions by revenue across safety PIVC, integrated catheter, and conventional catheter categories. Their competitive strength derives from FDA-cleared and EU MDR-compliant product portfolios covering all major PIVC types, established hospital group purchasing organisation contract positions in North America, and distributorships embedded in European national health system procurement frameworks.
Teleflex, Vygon, and C. R. Bard (now part of BD) compete in mid-range and specialized PIVC categories through application-specific product designs and regional distribution strength. NIPRO Medical and Argon Medical Devices maintain competitive positions in the conventional PIVC segment in Asia and Latin America by offering certified products at pricing competitive with regional manufacturers. Technology differentiation through integrated passive safety mechanisms, antimicrobial catheter coatings, and visibility-enhanced insertion technologies represents the primary competitive lever in premium segments where BD and B. Braun are building hospital system platform relationships beyond individual device transactions.
Large hospital system and group purchasing organisation customers manage supplier dependency by maintaining two to three approved PIVC vendors per product category, limiting single-supplier pricing power in standard conventional catheter segments. However, hospitals that complete system-wide conversion to integrated closed-system PIVC platforms develop higher switching costs due to staff training investment and infection control protocol documentation, creating a durable competitive advantage for first-mover platform providers in large health system accounts.
Key Players
- Becton, Dickinson and Company
- B. Braun Melsungen AG
- Smith’s Group Plc.
- Terumo Corporation
- Venner Medical
- Vygon
- Teleflex Incorporated
- C. R. Bard, Inc.
- NIPRO Medical Corporation
- Argon Medical Devices, Inc.
Bibliography
[1] Becton, Dickinson and Company. (2025). Q1 Fiscal Year 2025 Earnings Call Transcript: Medication Delivery Solutions Segment Performance Commentary. February 2025. investors.bd.com
[2] Ministero della Salute, Italy. (2024). Implementation Review: Decreto Legislativo 19/2014 Needlestick Prevention Enforcement Audit Requirements for SSN-Contracted Hospitals. October 2024. salute.gov.it
[3] Centers for Medicare and Medicaid Services. (2025). Hospital-Acquired Conditions Reduction Programme Fiscal Year 2025 Payment Adjustment: Affected Hospital List and Penalty Methodology. January 2025. cms.gov
[4] Ministry of Health, People’s Republic of China. (2024). 14th Five-Year Plan Healthcare Infrastructure Progress Report: County-Level Hospital Construction Approvals and Medical Device Procurement Framework. November 2024. nhc.gov.cn
[5] Becton, Dickinson and Company. (2025). Press Release: Nexiva Closed IV Catheter System International Market Expansion into Asia Pacific and Latin America. January 2025. bd.com
[6] B. Braun Melsungen AG. (2024). Product Regulatory Filing: Introcan Safety 3 Peripheral IV Catheter CE Mark Renewal under EU MDR 2017/745 with Updated Safety Mechanism Documentation. October 2024. bbraun.com
[7] Association for Vascular Access. (2024). Annual Device Survey Report: Safety and Integrated PIVC Adoption Trends in U.S. Hospital Vascular Access Programmes. September 2024. avainfo.org
[8] Teleflex Incorporated. (2025). Product Launch Release: Arrow Nanocath Safety PIVC with Integrated Passive Needle Protection for European Hospital Market. February 2025. teleflex.com
[9] Terumo Corporation. (2024). Regulatory Clearance Notice: Surflo Safety PIVC Single-Handed Activation Mechanism FDA 510(k) Clearance. August 2024. terumo.com
[10] NHS Supply Chain. (2025). Medical Devices Category Framework Update: Vascular Access Peripheral IV Catheter Passive Safety Activation Mandatory Specification. March 2025. nhssupplychain.nhs.uk
This Report Addresses
- Strategic intelligence on needle stick prevention regulation enforcement, integrated closed-system catheter adoption, hospital infrastructure expansion in India and China, and CMS value-based purchasing impact on PIVC procurement decisions.
- Market forecast from USD 7.8 billion in 2026 to USD 15.5 billion by 2036 at 7.1% CAGR, segmented by product type, technology, end user, and region.
- Growth opportunity mapping across India’s Ayushman Bharat hospital expansion, China’s county-level hospital procurement, Italy and Germany EU MDR safety device compliance, and U.S. CMS CRBSI reduction incentive programmes.
- Country CAGR outlook for India 7.9%, United States 7.4%, China 7.0%, Italy 6.8%, Germany 6.3%, and United Kingdom 6.1%, with key regulation and hospital sector demand drivers per country.
- Competitive analysis of BD, B. Braun, Smiths Group, Terumo, Teleflex, and Vygon covering product portfolios, safety device approvals, hospital GPO contract positions, and integrated catheter platform strategies.
- Technology tracking including passive safety needle retraction mechanisms, antimicrobial catheter coatings, closed-system integrated PIVC designs, and digital IV insertion tracking platforms.
- Regulatory analysis covering OSHA 29 CFR 1910.1030, EU Directive 2010/32/EU, EU MDR 2017/745, NICE Medical Technologies Guidance, NHS Supply Chain frameworks, and TRBA 250 biological agent’s regulation.
- Report delivered in PDF, Excel datasets, PowerPoint summary, and dashboard formats supported by FDA clearance records, EU MDR filings, NHS procurement data, and primary research interviews.
Scope of Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 7.8 billion (2026) to USD 15.5 billion (2036), at a CAGR of 7.1% |
| Market Definition | Safety-engineered and conventional peripheral IV catheter devices for vascular access across hospitals, clinics, and ambulatory surgical centers delivering medications, fluids, and blood products. |
| Product Type Segmentation | Short Peripheral IV Catheter (38%), Integrated/Closed Peripheral IV Catheter, Others |
| Technology Segmentation | Safety PIVC (33%), Conventional PIVC |
| End User Segmentation | Hospitals (56%), Clinics, Ambulatory Surgical Centers, Others |
| Application Coverage | Acute IV therapy, surgical fluid administration, oncology infusion, blood product delivery, antibiotic therapy, and parenteral nutrition access. |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | China, Japan, South Korea, India, Australia and New Zealand, ASEAN, Rest of South Asia and Pacific, Germany, UK, France, Italy, Spain, Nordic, BENELUX, Rest of Western Europe, Russia, Poland, Hungary, Balkan and Baltic, USA, Canada, Mexico, Brazil, Chile, Ecuador, Rest of Latin America, Saudi Arabia, GCC, Turkey, South Africa, Rest of MEA |
| Key Companies Profiled | Becton Dickinson, B. Braun, Smiths Group, Terumo, Venner Medical, Vygon, Teleflex, C. R. Bard, NIPRO Medical, Argon Medical Devices |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using hospital admission volumes, IV therapy procedure rates, catheter consumption per admission, average selling prices by technology, and primary interviews with procurement managers and IV therapy specialists. |
Peripheral Intravenous Catheter Market by Segments
-
By Product Type:
- Short Peripheral IV Catheter
- Integrated/Closed Peripheral IV Catheter
- Others
-
By Technology:
- Safety PIVC
- Conventional PIVC
-
By End User:
- Hospitals
- Clinics
- Ambulatory Surgical Centers
- Others
-
By Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Ecuador
- Rest of Latin America
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East and Africa
- North America
- Frequently Asked Questions -
How large is the peripheral intravenous catheter market in 2025?
The market was valued at USD 7.3 billion in 2025.
What will the market size be in 2026?
The market is estimated to reach USD 7.8 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 15.5 billion by 2036, creating a USD 7.7 billion absolute dollar opportunity over the forecast period.
What is the expected CAGR?
The forecast CAGR from 2026 to 2036 is 7.1%.
Which product type leads the market?
Short peripheral IV catheters lead with approximately 38% share in 2026. Safety PIVCs lead at approximately 33% share in 2026.
Which end user segment holds the largest share?
Hospitals dominate with approximately 56% share in 2026.
Which country shows the fastest growth?
India leads at 7.9% CAGR through 2036, driven by Ayushman Bharat hospital infrastructure expansion.
What is the primary restraint?
Price sensitivity in public hospital procurement in lower-income markets and long national tender cycles that delay premium safety PIVC technology adoption are the main structural constraints on market growth.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Short Peripheral IV Catheter
- Integrated/Closed Peripheral IV Catheter
- Others
- Short Peripheral IV Catheter
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Safety PIVC
- Conventional PIVC
- Safety PIVC
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Clinics
- Ambulatory Surgical Centers
- Others
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Technology
- By End User
- Competition Analysis
- Competition Deep Dive
- Becton, Dickinson and Company
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- B. Braun Melsungen AG
- Smith’s Group Plc.
- Terumo Corporation
- Venner Medical
- Vygon
- Teleflex Incorporated
- C. R. Bard, Inc.
- NIPRO Medical Corporation
- Argon Medical Devices, Inc.
- Becton, Dickinson and Company
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Product Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Technology, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Technology
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Product Type, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Technology, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Technology
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Product Type, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Technology, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Technology
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Product Type, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Technology, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Technology
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Product Type, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Technology, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Product Type, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Technology, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Technology
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Product Type, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Technology, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Product Type, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Technology, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis