Peripheral IV Infiltration Detection Devices Market Size, Share, Growth and Forecast (2026 - 2036)
The Peripheral IV Infiltration Detection Devices Market is segmented by Product Type (Wearable Infiltration Sensors, Integrated IV Monitoring Systems, and Standalone Detection Devices), Application (Chemotherapy Infusion Monitoring, Pediatric/Neonatal IV Monitoring, General Infusion Therapy, and Contrast Media Delivery), End User (Hospitals, Ambulatory Surgical Centers, and Homecare), and Region. Forecast for 2026 to 2036.
Fact.MR analysis indicates that the peripheral IV infiltration detection devices market is entering a phase defined by a widening technology-access divide. Hospitals that are integrating wired and wireless detection systems into infusion pumps and electronic medical record workflows are significantly reducing preventable infiltration events. These institutions are moving toward automated, continuous monitoring frameworks that enhance patient safety and clinical efficiency.
Peripheral IV Infiltration Detection Devices Market Forecast and Outlook By FACT.MR
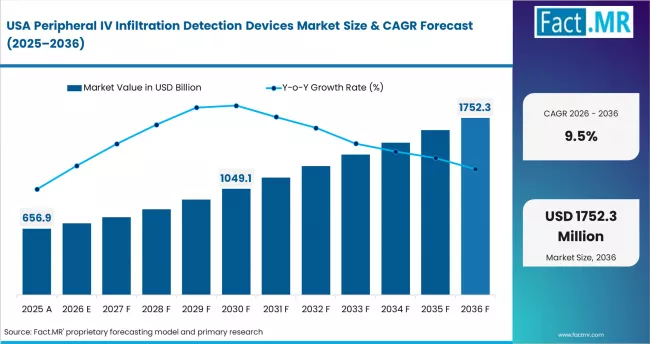
In 2025, the peripheral IV infiltration detection devices market was valued at USD 1.3 billion. Based on Fact.MR analysis, demand for peripheral IV infiltration detection devices is estimated to grow to USD 1.4 billion in 2026 and USD 3.7 billion by 2036. FACT.MR projects a CAGR of 9.9% during the forecast period.
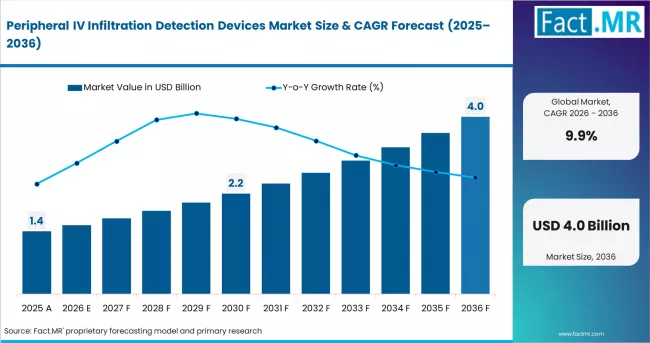
Summary of Peripheral IV Infiltration Detection Devices Market
- Market Definition
- Covers wearable infiltration sensors, integrated IV monitoring systems, and standalone detection devices for real-time peripheral IV site surveillance across hospital, ambulatory surgical, and homecare settings globally.
- Demand Drivers
- U.S. hospital quality programmes are mandating infiltration detection as part of IV therapy safety protocols, driving procurement of integrated monitoring systems across infusion and oncology units.
- NHS patient safety initiatives in the United Kingdom are accelerating adoption of automated IV infiltration detection in pediatric and general ward settings through national procurement frameworks.
- Rising chemotherapy infusion volumes globally are increasing demand for continuous infiltration monitoring to prevent extravasation injuries linked to vesicant drug administration.
- Key Segments Analyzed
- By Product Type: Integrated IV monitoring systems lead with approximately 48% share in 2026.
- By Application: General infusion therapy leads at approximately 41% in 2026.
- By End User: Hospitals dominate at approximately 64% in 2026.
- By Geography: India leads at 10.9% CAGR through 2036, driven by hospital infrastructure expansion and growing patient safety investment in tertiary care.
- Analyst Opinion at FACT.MR
- Shambhu Nath Jha, Principal Consultant at Fact.MR, opines, 'CXOs will find that peripheral IV infiltration detection is no longer a bedside accessory decision but a patient safety and liability management investment, and vendors delivering EMR-integrated, clinically validated detection platforms will capture disproportionate share of hospital group purchasing contracts through 2036.'
- Strategic Implications
- Invest in EMR integration and infusion pump connectivity to qualify for group purchasing organisation contracts at U.S. hospital networks replacing manual IV monitoring protocols.
- Build reimbursement evidence and clinical outcome data to support CMS and NHS coverage decisions for wearable infiltration sensors beyond ICU and chemotherapy settings.
- Develop regulatory dossiers for EU MDR certification and FDA 510(k) clearance to access premium European and U.S. hospital procurement without commodity pricing pressure.
- Methodology
- Market sizing based on annual IV procedure volumes, device penetration by product type, and average unit prices by detection technology grade. Validated with 2024 to 2025 procurement and clearance data.
- FDA 510(k) databases, NHS procurement records, EU MDR certification filings, and CMS reimbursement data supported demand and country-level analysis.
- Forecasts incorporate infiltration adverse event reporting trends, hospital quality programme mandates, and infusion therapy protocol standardisation from primary clinical expert interviews.
| Metric | Value |
|---|---|
| Estimated Value in 2026 | USD 1.4 billion |
| Forecast Value in 2036 | USD 3.7 billion |
| Forecast CAGR (2026 to 2036) | 9.9% |
Between 2026 and 2036, the market is expected to create USD 2.3 billion in new opportunity. Growth is transformational. Demand is driven by patient safety mandates and rising IV therapy volumes. Infiltration remains a top-five nursing-sensitive adverse event. Growth is constrained by device reimbursement gaps and limited clinical protocol standardization across low-income markets.
Hospitals are shifting procurement toward integrated detection systems. Standalone visual monitoring tools are losing institutional favour. Wireless sensor adoption is rising in chemotherapy and pediatric units. U.S. hospitals are embedding detection into infusion therapy quality programmes. Premium integrated systems command 30 to 50% price premiums over manual-check protocols. Vendors without EMR compatibility and clinical validation data are being excluded from group purchasing organization contracts.
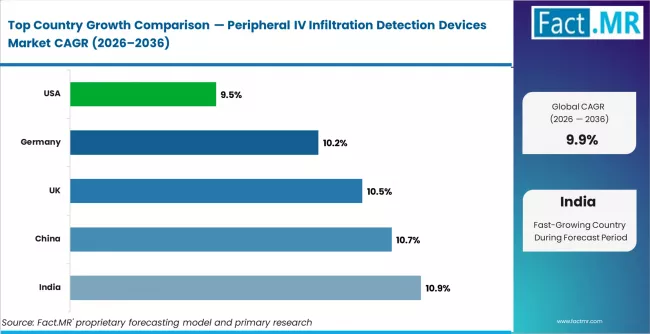
India leads with 10.9% CAGR through 2036, driven by rapid hospital infrastructure expansion and patient safety investment. China follows at 10.7%, supported by ICU modernisation and high-volume infusion therapy demand. The United Kingdom records 10.5%, anchored in NHS-driven patient safety initiatives and procurement frameworks. Germany grows at 10.2%, driven by advanced hospital device adoption and EU MDR compliance requirements. The United States records 9.5%, supported by strong patient safety technology adoption and regulatory pathway depth.
Segmental Analysis
Peripheral IV Infiltration Detection Devices Market Analysis by Product Type
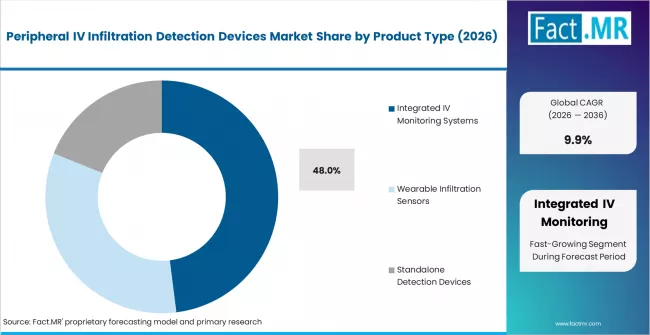
Based on FACT.MR, integrated IV monitoring systems hold 48% share in 2026. Hospitals prefer multi-parameter platforms over standalone tools. Integration with infusion pumps and EMR systems improves workflow and enables continuous monitoring.
- ivWatch LLC Expansion: Expanded U.S. hospital network in Q3 2024. Secured multi-site agreements with academic centers. Optical sensor platform is FDA-cleared across patient groups. [1]
- Becton Dickinson Development: Launched updated Alaris system in 2024. Integrated infiltration alert functionality. Connects with EMR systems for real-time monitoring. [2]
- Integrated System Procurement Trend: 61% of U.S. hospitals prioritised infiltration monitoring integration in 2024. Procurement is shifting to multi-function platforms. Group purchasing organisations are updating preferred supplier lists.
Peripheral IV Infiltration Detection Devices Market Analysis by Application
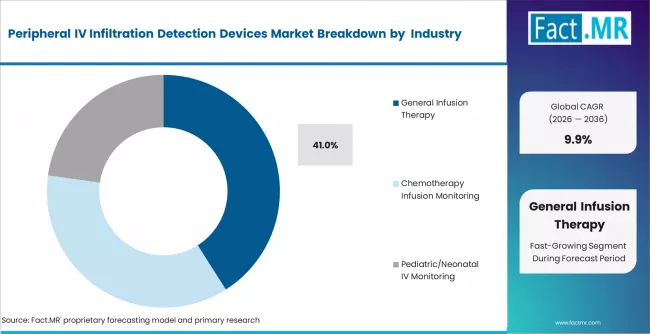
Based on FACT.MR, general infusion therapy holds 41% share in 2026. It is the largest segment by volume. High procedure frequency drives recurring device demand.
- Baxter International Investment: Expanded IV monitoring portfolio in 2024. Focus on sensor-compatible infusion systems. Supports integration with detection platforms. [4]
- Fresenius SE Development: Launched updated infusion system in early 2025. Enables peripheral monitoring compatibility. Integrates with hospital systems for reporting. [6]
Chemotherapy Application Trend: Oncology Nursing Society updated guidelines in 2024. Recommends continuous monitoring for vesicant infusions. Oncology centers are adopting wearable sensors. Pediatric units are leading adoption. [3]
Drivers, Restraints, and Opportunities
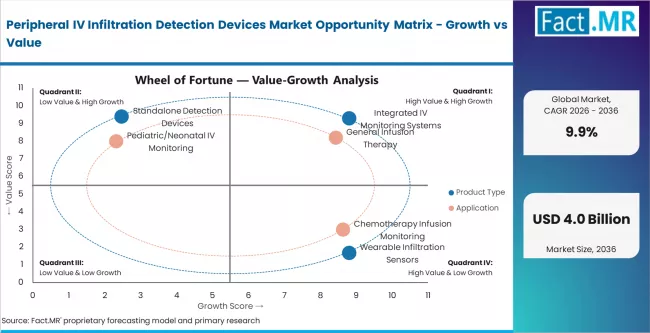
FACT.MR analysts note that growth is driven by rising IV therapy volumes and patient safety regulations. Peripheral IV infiltration affects 23 to 35% of insertions. It is one of the most common preventable hospital adverse events. This creates strong demand for automated detection across clinical settings.
Procurement is shifting rapidly. Manual visual inspection is losing clinical acceptance. Hospitals prefer integrated sensor-based systems with real-time alerts. FDA and EU MDR-compliant devices command premium pricing. Vendors without interoperability and clinical validation are being excluded from supplier lists.
- FDA Patient Safety Mandate: The FDA updated IV safety guidance in 2024. Hospitals are required to reduce infiltration-related harm. This expanded demand for automated detection systems. Joint Commission standards now include infiltration monitoring in quality scoring. [5]
- NHS Procurement Acceleration: NHS England issued a safety alert in 2024. Hospitals must review detection practices. Procurement of evidence-based monitoring tools is mandated. Pediatric and oncology wards are prioritised.
- India Hospital Infrastructure Expansion: National Health Mission funding in 2024 supports ICU and infusion upgrades. IV monitoring devices are included in procurement schemes. PLI incentives support domestic manufacturing and reduce costs.
Regional Analysis
The peripheral IV infiltration detection devices market is assessed across Asia Pacific, North America, Europe, Latin America, and Middle East and Africa, covering 40+ countries with distinct demand profiles shaped by healthcare infrastructure, hospital quality mandates, reimbursement frameworks, and regulatory certification requirements. The full report provides market attractiveness analysis by region and country.
.webp)
| Countrya | CAGR (2026 to 2036) |
|---|---|
| India | 10.9% |
| China | 10.7% |
| United Kingdom | 10.5% |
| Germany | 10.2% |
| United States | 9.5% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
Asia Pacific Peripheral IV Infiltration Detection Devices Market Analysis
Asia Pacific is the highest-growth region. It is driven by hospital infrastructure expansion and rising IV therapy volumes across India and China. Patient safety investment is increasing across state and provincial health systems. ivWatch LLC and Becton Dickinson are establishing regional presence through distributor partnerships and regulatory registrations.
- India: Infrastructure-led procurement supports 10.9% CAGR through 2036. It is the fastest among all tracked markets. National Health Mission issued procurement guidelines in 2024. These include IV therapy monitoring devices under ICU and infusion upgrades. State hospitals in Maharashtra and Tamil Nadu tendered IV monitoring equipment in Q2 2024. This was under Ayushman Bharat grants. Domestic manufacturers under PLI scheme are expanding IV sensor production. This reduces import costs.
- China: ICU modernisation supports 10.7% CAGR through 2036. National Health Commission updated hospital equipment standards in 2024. These include continuous IV monitoring recommendations. Mindray Medical expanded infusion monitoring products in Q3 2024. This aligns with domestic innovation mandates. Provincial procurement frameworks prioritise local devices. This limits imports. High infusion volumes in oncology and general wards drive demand. Growth is linked to health system modernisation and national quality programmes.
FACT.MR's analysis covers China, India, Japan, South Korea, ASEAN, and ANZ. It includes infrastructure trends, procurement frameworks, and adoption data.
North America Peripheral IV Infiltration Detection Devices Market Analysis
North America leads in device standards and procurement volume. It has strong clinical validation requirements. Demand is driven by hospital quality mandates and FDA pathways. ivWatch LLC, Becton Dickinson, and Baxter International dominate supply. EMR interoperability and outcome documentation are key requirements.
- United States: Patient safety adoption supports 9.5% CAGR through 2036. It has the largest IV therapy volume globally. FDA cleared three devices under 510(k) in 2024. This expanded available platforms. CMS updated hospital-acquired condition reporting in Q1 2025. Peripheral IV infiltration is now a quality metric. Veterans Health Administration launched procurement in late 2024. It covers 65 VA hospitals. Group purchasing organisations covering 4,500+ hospitals added IV monitoring in 2024. Growth is driven by regulation and quality compliance.
FACT.MR's analysis covers the United States, Canada, and Mexico. It includes FDA trends, CMS updates, and procurement frameworks.
Europe Peripheral IV Infiltration Detection Devices Market Analysis
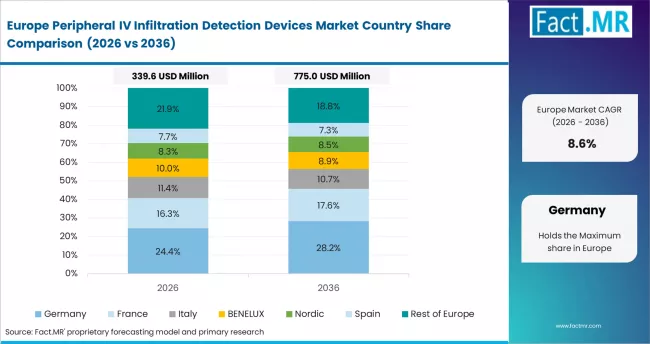
Europe is a compliance-driven market. Procurement is shaped by EU MDR and national safety mandates. Suppliers must meet CE marking and clinical evidence requirements. Markets are fragmented but high-value.
- Germany: Advanced adoption supports 10.2% CAGR through 2036. B. Braun and Fresenius SE lead domestic supply. German Hospital Association updated standards in 2024. IV complication reporting is now a quality indicator. Bundeswehr expanded procurement in 2024. It includes IV monitoring systems. EU MDR transition accelerated device replacement. Growth is driven by compliance and quality programmes.
- United Kingdom: NHS mandates support 10.5% CAGR through 2036. NHS England issued a safety alert in 2024. It targets IV infiltration detection. Trusts must procure monitoring tools. NICE updated IV therapy guidelines in Q3 2024. Electronic monitoring is recommended for high-risk infusions. Philips Healthcare expanded NHS contracts in 2024. Scotland piloted wearable sensors in neonatal units. Growth is driven by NHS frameworks and safety mandates.
FACT.MR's analysis covers Germany, France, the United Kingdom, Italy, Spain, and Rest of Europe. It includes EU MDR timelines, NHS frameworks, and adoption trends.
Competitive Aligners for Market Players
Competition in the peripheral IV infiltration detection devices market is increasingly shaped by regulatory depth, clinical validation, and system integration rather than pricing alone. Market entry is tightly controlled by FDA 510(k) clearance in the United States and EU MDR certification in Europe. Hospitals are prioritizing devices that demonstrate measurable reductions in adverse events and can integrate seamlessly with electronic medical record systems. As a result, only a limited group of compliant manufacturers is securing the majority of large-scale institutional contracts.
ivWatch LLC holds a leading position in continuous optical IV monitoring. Its advantage comes from strong clinical validation across adult, pediatric, and neonatal use cases, making it a preferred choice for hospital safety programs. Established players such as Becton Dickinson and Baxter International compete by embedding infiltration detection into existing infusion systems. This approach reduces switching costs and supports faster adoption within hospitals already using their platforms.
Companies like ICU Medical and B. Braun Melsungen AG leverage their extensive installed base across global hospital networks. They introduce monitoring upgrades within existing infrastructure, avoiding the need for full system replacement. Meanwhile, Fresenius SE focuses on Europe with EU MDR-compliant systems integrated into hospital IT environments. Below the top tier, smaller sensor-focused companies target niche applications such as oncology and neonatal care. However, they rely on partnerships with larger system providers for scale. Hospital procurement trends favor established vendors with proven clinical outcomes, regulatory compliance, and strong integration capabilities, reinforcing their long-term competitive advantage.
Key Players
- ivWatch LLC
- Baxter International Inc.
- Becton Dickinson and Company
- B. Braun Melsungen AG
- Fresenius SE & Co. KGaA
- ICU Medical Inc.
Bibliography
- ivWatch LLC. (Q3 2024). Clinical Validation Update and Hospital Network Expansion: Multi-Site Installation Agreements with U.S. Academic Medical Centre Networks Following FDA-Cleared Optical IV Infiltration Monitoring Platform Validation Across Adult, Pediatric, and Neonatal Patient Populations. Q3 2024. ivwatch.com/news
- Becton Dickinson and Company. (2024). Product Update: Alaris Infusion System Integrated Infiltration Alert Module with EMR Connectivity for Real-Time Peripheral IV Site Status Monitoring at U.S. and European Hospital IV Therapy Units. 2024. bd.com/newsroom
- American Society of Health-System Pharmacists. (2024). IV Therapy Monitoring Technology Survey 2024: 61% of U.S. Hospital Pharmacies Cite Infiltration Detection Integration as Priority Criterion in Infusion System Procurement Decisions. 2024. ashp.org
- Baxter International Inc. (2024). Portfolio Expansion: IV Therapy Patient Monitoring Sensor-Compatible Infusion Sets for Integration with Third-Party Peripheral Infiltration Detection Platforms Across U.S., European, and Asia Pacific Hospital Markets. 2024. baxter.com/newsroom
- U.S. Food and Drug Administration. (2024). Updated IV Device Safety Guidance: Medical Device Safety Action Plan Reinforcement of Hospital Obligations for Infiltration-Reducing Technologies in Peripheral IV Therapy Under Nursing-Sensitive Adverse Event Reduction Frameworks. 2024. fda.gov/medicaldevices
- Fresenius SE & Co. KGaA. (Early 2025). Product Launch: Updated IV Infusion System with Peripheral Site Monitoring Compatibility for EU MDR-Compliant General Ward and Oncology Infusion Centre Applications Across German and European Hospital Networks. January 2025. fresenius.com/press
This Report Addresses
- Strategic intelligence on demand across wearable infiltration sensors, integrated IV monitoring systems, and standalone detection devices for peripheral IV site safety globally.
- Market forecast from USD 1.4 billion in 2026 to USD 3.7 billion by 2036 at a CAGR of 9.9%.
- Growth opportunity mapping across India National Health Mission procurement, China ICU modernisation, U.S. CMS quality metric mandates, UK NHS patient safety alerts, and Germany EU MDR compliance replacement cycles.
- Segment analysis by product type (wearable sensors, integrated systems, standalone devices), application (chemotherapy, pediatric/neonatal, general infusion, contrast media), and end user (hospitals, ASC, homecare).
- Regional outlook covering Asia Pacific infrastructure-led adoption, North America FDA-regulated procurement, and Europe NHS and EU MDR compliance-driven demand cycles.
- Competitive analysis of ivWatch LLC, Baxter International, Becton Dickinson, B. Braun, Fresenius SE, and ICU Medical. Focus on FDA clearance depth, EMR interoperability, and clinical outcome validation.
- Regulatory analysis covering FDA 510(k) clearance requirements, EU MDR certification obligations, CMS quality metric reporting, NHS patient safety alert procurement implications, and national IV therapy guideline updates.
- Report delivered in PDF, Excel, and presentation formats. Supported by primary interviews, hospital procurement records, FDA clearance filings, NHS framework data, and manufacturer revenue disclosures.
Peripheral IV Infiltration Detection Devices Market Definition
The market covers wearable sensors, integrated monitoring systems, and standalone detection devices used to identify peripheral IV infiltration and extravasation in real time. Devices detect fluid leakage from IV sites using impedance, optical, or acoustic-based sensing technologies.
Peripheral IV Infiltration Detection Devices Market Inclusions
Covers global forecasts (2026 to 2036) by product type, application, and end user. Includes wearable sensors, integrated IV systems, and standalone detectors. Covers hospital, ambulatory surgical center, and homecare procurement across infusion therapy, chemotherapy, pediatric, and contrast media delivery settings.
Peripheral IV Infiltration Detection Devices Market Exclusions
Excludes general IV infusion pumps unless equipped with integrated infiltration detection modules. Omits standalone EMR software platforms and IV catheter consumables. Focus remains on active detection devices and their immediate clinical monitoring intermediates used at the peripheral IV site.
Peripheral IV Infiltration Detection Devices Market Research Methodology
- Primary Research
- Interviews with IV therapy nurses, infusion center directors, hospital procurement officers, and device manufacturers across the United States, United Kingdom, Germany, China, and India.
- Desk Research
- Uses FDA 510(k) clearance databases, NHS procurement frameworks, EU MDR certification records, CMS reimbursement data, and company filings from ivWatch LLC, Baxter International, and Becton Dickinson from 2024 to 2025.
- Market-Sizing and Forecasting
- Hybrid model using annual IV procedure volumes, device penetration by product type and end-user segment, and average contract unit prices by detection technology grade across all covered geographies.
- Data Validation and Update Cycle
- Validated through hospital procurement records, FDA clearance volumes, and company revenues. Cross-checked with clinical expert interviews. Updated annually.
Scope of Report

| Attribute | Details |
|---|---|
| Quantitative Units | USD 1.4 billion (2026) to USD 3.7 billion (2036), at a CAGR of 9.9% |
| Market Definition | Wearable infiltration sensors, integrated IV monitoring systems, and standalone detection devices for real-time peripheral IV site surveillance across hospital, ambulatory surgical, and homecare settings, procured by hospitals, ambulatory surgical centers, and homecare providers globally. |
| Product Type Segmentation | Wearable Infiltration Sensors, Integrated IV Monitoring Systems, Standalone Detection Devices |
| Application Segmentation | Chemotherapy Infusion Monitoring, Pediatric/Neonatal IV Monitoring, General Infusion Therapy, Contrast Media Delivery |
| End User Segmentation | Hospitals, Ambulatory Surgical Centers, Homecare |
| Application Coverage | Hospital IV therapy infiltration surveillance, chemotherapy and vesicant extravasation prevention, pediatric and neonatal IV site monitoring, ambulatory surgical center infusion safety, and homecare IV therapy monitoring. |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Canada, Mexico, Germany, UK, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, ASEAN, Australia and New Zealand, Brazil, Argentina, Chile, Saudi Arabia, GCC, Turkey, South Africa, Rest of MEA |
| Key Companies Profiled | ivWatch LLC, Baxter International Inc., Becton Dickinson and Company, B. Braun Melsungen AG, Fresenius SE & Co. KGaA, ICU Medical Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using annual IV procedure volume estimates, device penetration by product type and end-user segment, average contract unit prices by detection technology grade, and primary interviews with IV therapy nurses, hospital procurement officers, and device manufacturers. |
Peripheral IV Infiltration Detection Devices Market by Segments
-
By Product Type:
- Wearable Infiltration Sensors
- Integrated IV Monitoring Systems
- Standalone Detection Devices
-
By Application:
- Chemotherapy Infusion Monitoring
- Pediatric/Neonatal IV Monitoring
- General Infusion Therapy
- Contrast Media Delivery
-
By End User:
- Hospitals
- Ambulatory Surgical Centers
- Homecare
-
By Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Mexico
- Argentina
- Chile
- Rest of Latin America
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Rest of Middle East and Africa
- North America
- Frequently Asked Questions -
How large is the peripheral IV infiltration detection devices market in 2025?
The peripheral IV infiltration detection devices market was valued at USD 1.3 billion in 2025, with demand estimated to grow to USD 1.4 billion in 2026.
What will the market size be by 2036?
Based on Fact.MR analysis, the market is projected to reach USD 3.7 billion by 2036.
What is the expected CAGR from 2026 to 2036?
FACT.MR projects a CAGR of 9.9% for the peripheral IV infiltration detection devices market during the 2026 to 2036 forecast period.
Which product type segment is poised to lead the market?
Integrated IV monitoring systems lead with approximately 48% share in 2026, driven by hospital preference for multi-parameter platforms with EMR and infusion pump connectivity over standalone detection tools.
Which end-user segment is expected to dominate?
Hospitals dominate with approximately 64% share in 2026, representing the largest procurement base across all product and application categories globally.
Which application holds the largest share?
General infusion therapy holds approximately 41% share in 2026, driven by the highest global IV procedure volume across hospital and ambulatory surgical settings.
Which country shows the fastest growth?
India leads at 10.9% CAGR through 2036, driven by National Health Mission hospital infrastructure investment, Ayushman Bharat equipment grants, and rising tertiary care IV therapy volumes.
What is driving demand for peripheral IV infiltration detection devices?
Demand is driven by hospital patient safety mandates, rising IV therapy procedure volumes, FDA and EU MDR regulatory requirements, NHS patient safety alerts, and CMS quality metric obligations for IV site complication reporting.
What are the major restraints?
Key restraints include reimbursement gaps for detection devices beyond ICU settings, limited clinical protocol standardisation in low-income markets, and high integration costs for EMR connectivity at smaller hospital systems.
What is the main challenge in this market?
Demonstrating measurable clinical and financial return on investment from automated infiltration detection is the primary barrier to widespread adoption, as procurement decisions at institutional buyers require outcome data linking device use to adverse event reduction and liability cost savings.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Integrated IV Monitoring Systems
- Wearable Infiltration Sensors
- Standalone Detection Devices
- Integrated IV Monitoring Systems
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- General Infusion Therapy
- Chemotherapy Infusion Monitoring
- Pediatric/Neonatal IV Monitoring
- General Infusion Therapy
- General Infusion Therapy
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- Competition Analysis
- Competition Deep Dive
- ivWatch LLC
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Baxter International Inc.
- Becton Dickinson and Company
- B. Braun Melsungen AG
- Fresenius SE & Co. KGaA
- ICU Medical Inc.
- ivWatch LLC
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis