Preclinical Antibody Development Market Size, Share, Growth and Forecast (2026 - 2036)
Preclinical Antibody Development Market Size and Share Forecast Outlook 2026 to 2036
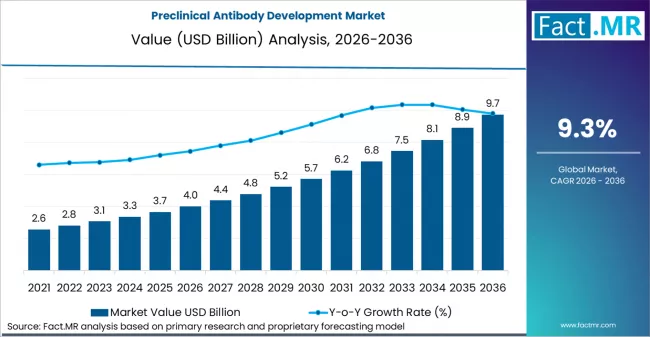
Preclinical antibody development market is projected to grow from USD 4.0 billion in 2026 to USD 9.7 billion by 2036, at a CAGR of 9.3%. Antibody discovery (hybridoma/phage) will dominate with a 24.0% market share, while oncology will lead the therapeutic area segment with a 36.0% share.
Preclinical Antibody Development Market Forecast and Outlook 2026 to 2036
The global demand for preclinical antibody development is expected to expand rapidly over the forecast period, with market value projected to increase from USD 4 billion in 2026 to approximately USD 9.7 billion by 2036, reflecting a CAGR of 9.3%. This growth is being driven by the rapid diversification of biopharmaceutical pipelines toward complex antibody modalities such as bispecific antibodies and antibody drug conjugates, which require deeper functional validation and higher resolution preclinical assessment than traditional monoclonal formats.
Key Takeaways from the Preclinical Antibody Development Market
- Preclinical Antibody Development Market Value (2026): USD 4 billion
- Preclinical Antibody Development Market Forecast Value (2036): USD 9.7 billion
- Preclinical Antibody Development Market Forecast CAGR (2026-2036): 9.3%
- Leading Service Type: Antibody discovery including hybridoma and phage display platforms (24%)
- Key Market Participants: Charles River Laboratories, WuXi Biologics, Labcorp Drug Development, GenScript Biotech, Abzena.
- Key Growth Regions: North America, Europe, Asia Pacific, Middle East and Africa
The market is also undergoing a structural transformation as regulatory and scientific framework increasingly favor predictive, human relevant models over conventional animal testing. The adoption of the FDA Modernization Act 2.0 has accelerated investment in advanced in vitro and in silico systems, including patient derived organoids, microphysiological platforms, and computational modeling approaches. These technologies enable more accurate prediction of pharmacokinetics, safety, and target engagement earlier in development.
At the same time, the integration of artificial intelligence into antibody discovery has reshaped preclinical demand. AI driven platforms are generating large volumes of candidate molecules, increasing reliance on high throughput wet lab validation to confirm biological relevance. As a result, preclinical antibody development is evolving from empirical screening toward predictive translational modeling, where mechanistic clarity, biomarker integration, and data quality are central to advancing candidates into clinical development.
Preclinical Antibody Development Market
| Metric | Value |
|---|---|
| Estimated Value in (2026E) | USD 4 billion |
| Forecast Value in (2036F) | USD 9.7 billion |
| Forecast CAGR (2026 to 2036) | 9.3 % |
Category
| Category | Segments |
|---|---|
| Service Type | Antibody discovery (hybridoma/phage), Humanization & engineeringAffinity maturation, Developability assessment (liability screening), Expression & purification (preclinical), In vitro functional characterization |
| Therapeutic Area | Oncology, Autoimmune & inflammation, Infectious diseases, NeurologyRare diseases |
| End User | Biotech & startups, Large pharma, Academic / tech transfer, Government & non-profit, Diagnostics developers |
| Region | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
Segmental Analysis
How are the shift toward complex antibody modalities positioning antibody discovery services as the leading segment in the Preclinical Antibody Development Market?
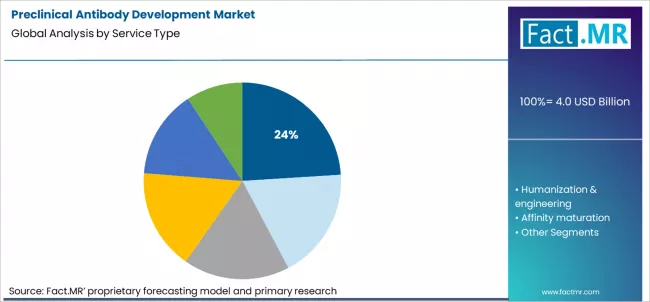
Antibody discovery services account for the 24% share of the preclinical antibody development market because they sit at the critical entry point of increasingly complex biologic pipelines. With the rapid expansion of bispecific antibodies, antibody drug conjugates, and multispecific formats, early stage discovery now requires far more than simple target binding confirmation. Hybridoma and phage display platforms are being used not only to identify binders but also to generate candidates suitable for downstream engineering, manufacturability, and safety optimization. This has elevated discovery from a screening activity to a strategic risk reduction step. As biopharmaceutical companies seek to compress timelines while increasing success probability, outsourcing high fidelity discovery to specialized providers has become a foundational requirement rather than an optional service.
Why does oncology remain the dominant therapeutic focus within preclinical antibody development?
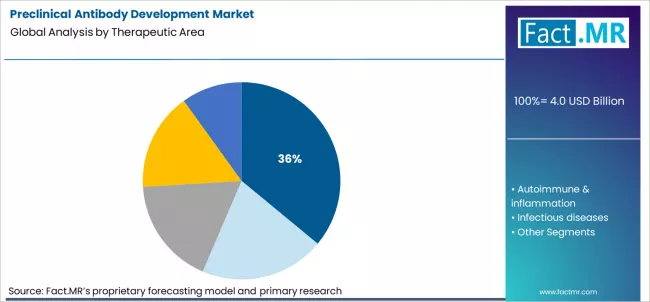
Oncology represents the 36 % share in therapeutic area because cancer pipelines are increasingly dependent on antibody based modalities that demand rigorous preclinical validation. Tumor heterogeneity, complex immune interactions, and narrow therapeutic windows require extensive functional characterization before clinical entry. Preclinical programs in oncology rely heavily on advanced in vitro systems, biomarker driven assays, and translational modeling to predict efficacy and toxicity. The continuous flow of novel targets in immuno oncology sustains high demand for discovery, engineering, and developability services, reinforcing oncology’s structural dominance in preclinical antibody development.
Market Dynamics
- Drivers: A primary driver shaping the preclinical antibody development market is the rising complexity of biologic drug modalities. As pipelines move beyond conventional monoclonal antibodies into bispecific, conjugates, and engineered formats, the need for deep preclinical characterization has intensified. Sponsors are prioritizing early assessment of binding kinetics, stability, aggregation risk, and off target effects to reduce costly late stage failures. This has expanded demand for integrated service platforms that combine discovery, engineering, and functional validation under a single workflow. Regulatory evolution is another major driver. The implementation of the FDA Modernization Act 2.0 has accelerated the shift toward human relevant, non-animal testing strategies. Patient derived organoids, microphysiological systems, and advanced in vitro assays are increasingly used to support regulatory submissions. These models require specialized expertise and infrastructure, driving outsourcing to CROs and CDMOs with translational capabilities. The rapid adoption of artificial intelligence in antibody discovery is further fueling market growth. AI platforms are generating large volumes of in silico candidates, creating a validation bottleneck that only scalable preclinical services can resolve. High throughput screening and AI compatible data generation have become essential to translate computational insights into viable drug candidates.
- Restraints: Despite strong demand, market growth is constrained by cost and technical complexity. Comprehensive preclinical programs require significant investment, which can strain early stage biotech budgets. Smaller companies may limit the scope of characterization, increasing development risk. Additionally, the interpretation of complex preclinical data requires multidisciplinary expertise, creating dependence on a limited pool of specialized providers. Data standardization remains a challenge. Variability across assay platforms and modeling systems can complicate cross program comparisons and regulatory alignment. This increases the burden on sponsors to ensure data robustness and reproducibility.
- Key Trend: Predictive Translational Modeling Preclinical antibody development is increasingly centered on predictive translational modeling rather than empirical screening. Sponsors are prioritizing mechanistic clarity, biomarker integration, and human relevant data to improve clinical translation. This trend is reshaping service demand toward platforms that can generate high resolution, decision ready datasets capable of supporting both regulatory confidence and AI driven discovery pipelines.
Analysis of the Preclinical Antibody Development Market by Key Country
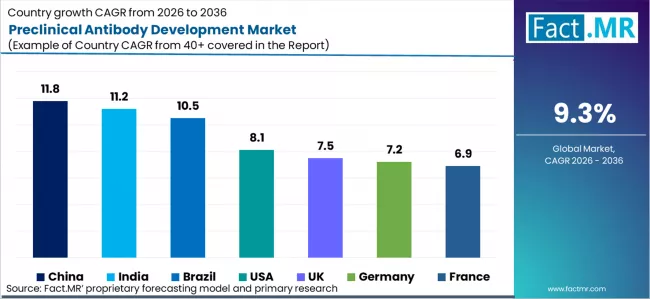
| Country | CAGR 2026 to 2036 |
|---|---|
| China | 11.8% |
| India | 11.2% |
| Brazil | 10.5% |
| USA | 8.1% |
| UK | 7.5% |
| Germany | 7.2% |
| France | 6.9% |
| Japan | 5.4% |
Why is China emerging as the fastest growing center for preclinical antibody development globally?
China is leading growth in the preclinical antibody development market with a CAGR of 11.8%, driven by large scale government investment and rapid infrastructure expansion. WuXi Biologics has integrated ADC specialized conjugation and screening suites to support bispecific and antibody drug conjugate pipelines. Backed by a multiyear national stimulus for best in class biologics, China is becoming a global hub for high throughput preclinical screening. This has accelerated the shift toward industrialized discovery and validation platforms capable of supporting both domestic and global antibody programs.
How is India positioning itself as a strategic alternative for outsourced preclinical antibody services?
India is expanding at a CAGR of 11.2%, supported by a structural realignment of global outsourcing strategies. Pressure from the US Biosecure Act has encouraged sponsors to adopt a China plus one approach, redirecting discovery and validation contracts to Indian CROs. Companies such as Aragen Life Sciences are building integrated discovery services that combine antibody engineering with medicinal chemistry. This has institutionalized end to end preclinical platforms and strengthened India’s role as a trusted partner for global biologics pipelines.
Why is Brazil gaining momentum in preclinical antibody development despite being a smaller base market?
Brazil is growing at a CAGR of 10.5%, driven by rising regional demand for biologics and biosimilar across Latin America. Ongoing expansion of local biologics manufacturing capacity is increasing the need for preclinical bioequivalence, comparability, and immunogenicity testing to support regional regulatory submissions. As medicine usage growth in Latin America continues to outpace that of developed markets, Brazil is emerging as a strategic hub for localized preclinical development. This positioning enables faster market entry, reduced dependency on overseas testing, and closer alignment with regional regulatory and healthcare requirements.
What is driving steady but selective growth in the United States preclinical antibody market?
The United States is expanding at a CAGR of 8.1%, shaped by regulatory reform and rapid adoption of advanced discovery technologies. The FDA Modernization Act 2.0, combined with increased NIH funding, has accelerated the shift toward human first in vitro models and reduced reliance on animal testing. Providers such as Sino Biological and GenScript are scaling recombinant antibody platforms, AI assisted discovery, and high resolution screening tools. This evolution is strengthening translational validation, improving lead selection accuracy, and supporting more predictive preclinical development pathways. How is the United Kingdom differentiating itself in advanced antibody discovery?
Why is the United Kingdom emerging as an innovation led market in preclinical antibody development?
The UK market is growing at a CAGR of 7.5%, driven primarily by scientific innovation rather than procedural volume. The Cambridge and London research corridor has evolved into a global center for next generation antibody formats, including IgA, IgE, and nanobody based oncology assets. Strong collaboration between academic institutions, biotech startups, and specialized CROs is accelerating early stage discovery and translational research. Strategic acquisitions and venture funding are concentrating high value intellectual property within the region, reinforcing the UK’s position as a leading hub for novel antibody scaffolds and advanced preclinical experimentation.
Why does Germany remain a regulation led preclinical antibody market?
Germany is advancing at a CAGR of 7.2%, with growth anchored in regulatory compliance and manufacturing excellence. Merck KGaA has launched single use ADC reactors to support rapid prototyping under strict ESG and animal welfare mandates. This has accelerated adoption of animal free recombinant antibody workflows and positioned Germany as a leader in high purity, compliant discovery platforms within the DACH region.
What factors are supporting moderate but stable growth in France?
France is recording a CAGR of 6.9%, supported by strong public research institutions and government backed translational programs. The market emphasizes early stage validation, academic spinouts, and collaborative preclinical research, particularly in oncology and rare diseases. While growth is more measured, France remains a key contributor to foundational antibody science and translational readiness.
Why is Japan showing the most conservative growth profile in preclinical antibody development?
Japan is growing at a CAGR of 5.4%, reflecting a mature and highly regulated market. Accelerated PMDA review timelines for orphan drugs have increased demand for first in human ready preclinical packages, particularly in rare diseases and oncology. However, conservative adoption of outsourced discovery and strong in house capabilities among large pharmaceutical companies have kept overall growth steady rather than rapid.
Competitive Landscape in Preclinical Antibody Development Market
Competition in the preclinical antibody development market has shifted from fragmented, service specific offerings toward integrated, translation focused platforms. In 2024, many providers operated within narrow functional boundaries, supplying discrete services such as antibody discovery, screening, or expression as stand-alone engagements. Sponsors often managed multiple vendors across development stages, with preclinical work viewed as a procedural requirement rather than a strategic value driver. Investment during this period prioritized capacity expansion and cost efficiency, while scientific differentiation remained limited.
The competitive landscape began to realign around end to end capability and predictive relevance. Leading CROs and CDMOs started consolidating discovery, engineering, developability assessment, and functional characterization into unified workflows. This shift was supported by rising demand for complex antibody formats, including bispecific and antibody drug conjugates, which require tighter integration between early discovery and downstream validation. Providers increasingly positioned themselves as translational partners, offering decision ready datasets rather than isolated experimental outputs.
External pressures have further accelerated this evolution. Regulatory acceptance of non-animal models and the growing influence of AI driven discovery have raised expectations for data depth, reproducibility, and clinical relevance. As a result, competition is increasingly defined by the ability to generate high fidelity, human relevant preclinical evidence that reduces downstream risk, rather than by throughput or cost alone.
Recent Developments
- In October 2025, Novartis acquired Avidity Biosciences, gaining access to Antibody-Oligonucleotide Conjugates (AOCs). This represents a new frontier where antibodies are used as precise delivery vehicles for gene-modulatory tools.
- In June 2025, BioNTech and BMS formed an $11.1 billion partnership for BNT327, a bispecific candidate. This deal underscores the industry’s bet on "tumor-targeted" antibodies that can redirect immune effector functions.
Key Players in Preclinical Antibody Development Market
- Charles River Laboratories
- WuXi Biologics
- Labcorp Drug Development
- GenScript Biotech
- Abzena
Bibliography
- European Commission. 2022. Regulation of advanced therapeutic medicinal products and biologics development under EU pharmaceutical and clinical research frameworks. Brussels: European Union.
- International Organization for Standardization (ISO). 2023. Biotechnology and biopharmaceutical development: Quality, safety, and analytical standards for antibody discovery and preclinical evaluation. ISO Technical Report. Geneva: ISO.
- U.S. Food and Drug Administration. 2022. FDA Modernization Act 2.0: Advancing non animal testing approaches for drug and biologics development. Silver Spring, MD: FDA.
- Journal of Pharmaceutical Sciences. 2024. Preclinical evaluation strategies for bispecific antibodies and antibody drug conjugates. Journal of Pharmaceutical Sciences 113(6): 1562-1578.
- Nature Reviews Drug Discovery. 2024. Translational modeling and human relevant systems in preclinical antibody development. Nature Reviews Drug Discovery 23(4): 241-259.
- IQVIA Institute for Human Data Science. 2025. Global biologics and antibody pipeline trends: Implications for preclinical research services. Durham, NC: IQVIA.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 4 Billion |
| Product Type | Exfoliating, Hydrating, and Brightening |
| Therapy Type | PLLA-based collagen stimulators, CaHA-based biostimulators, PCL-based stimulators, PMMA/collagen fillers (biostim), Energy-based collagen induction (RF/US), Autologous biostimulation (PRP/biologic kits) |
| Aesthetic Use | Facial volume restoration, Skin laxity tightening, Acne scar remodelingBody contouring & cellulite, Post-procedure regeneration |
| End User | Dermatology clinics, Plastic surgery centers, Med-spas / aesthetic chainsHospitals & ambulatory centers, Independent injectors |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | U.S., Canada, Germany, Italy, UK, France, Japan, Australia, and other countries |
| Key Companies Profiled | Galderma, Merz Aesthetics, AbbVie through Allergan Aesthetics, Sinclair, and Suneva Medical |
| Additional Attributes | Dollar revenue in the bio-stimulation therapies market is segmented by therapy or technology type, aesthetic use case, end user, treatment protocol, price tier, and region. Regional demand patterns are shaped by rising adoption of regenerative aesthetic procedures, increasing emphasis on collagen induction and tissue quality restoration, growing integration of injectable biostimulators with energy based devices, and premiumization of minimally invasive aesthetic treatments. |
Preclinical Antibody Development Market by Segment
-
Service Type :
- Antibody discovery (hybridoma/phage)
- Humanization & engineering
- Affinity maturation
- Developability assessment (liability screening)
- Expression & purification (preclinical)
- In vitro functional characterization
-
Therapeutic Use :
- Oncology
- Autoimmune & inflammation
- Infectious diseases
- Neurology
- Rare diseases
-
End User :
- Biotech & startups
- Large pharma
- Academic / tech transfer
- Government & non-profit
- Diagnostics developers
-
Region :
- Asia Pacific
- Japan
- South Korea
- China
- Australia
- New Zealand
- Rest of Asia Pacific
- North America
- United States
- Canada
- Mexico
- Europe
- France
- Italy
- Spain
- Norway
- UK
- Rest of Europe
- Latin America
- Chile
- Peru
- Argentina
- Rest of Latin America
- Asia Pacific
- Frequently Asked Questions -
How big is the preclinical antibody development market in 2026?
The global preclinical antibody development market is estimated to be valued at USD 4.0 billion in 2026.
What will be the size of preclinical antibody development market in 2036?
The market size for the preclinical antibody development market is projected to reach USD 9.7 billion by 2036.
How much will be the preclinical antibody development market growth between 2026 and 2036?
The preclinical antibody development market is expected to grow at a 9.3?GR between 2026 and 2036.
What are the key product types in the preclinical antibody development market?
The key product types in preclinical antibody development market are antibody discovery (hybridoma/phage), humanization & engineering, affinity maturation, developability assessment (liability screening), expression & purification (preclinical) and in vitro functional characterization.
Which therapeutic area segment to contribute significant share in the preclinical antibody development market in 2026?
In terms of therapeutic area, oncology segment to command 36.0% share in the preclinical antibody development market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Type, 2026 to 2036
- Antibody discovery (hybridoma/phage)
- Humanization & engineering
- Affinity maturation
- Developability assessment (liability screening)
- Expression & purification (preclinical)
- In vitro functional characterization
- Antibody discovery (hybridoma/phage)
- Y to o to Y Growth Trend Analysis By Service Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Service Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapeutic Area
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapeutic Area, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapeutic Area, 2026 to 2036
- Oncology
- Autoimmune & inflammation
- Infectious diseases
- Neurology
- Rare diseases
- Oncology
- Y to o to Y Growth Trend Analysis By Therapeutic Area, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapeutic Area, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By EndUser
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By EndUser, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By EndUser, 2026 to 2036
- Biotech & startups
- Large pharma
- Academic / tech transfer
- Government & non-profit
- Diagnostics developers
- Biotech & startups
- Y to o to Y Growth Trend Analysis By EndUser, 2021 to 2025
- Absolute $ Opportunity Analysis By EndUser, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service Type
- By Therapeutic Area
- By EndUser
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapeutic Area
- By EndUser
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service Type
- By Therapeutic Area
- By EndUser
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapeutic Area
- By EndUser
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service Type
- By Therapeutic Area
- By EndUser
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapeutic Area
- By EndUser
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service Type
- By Therapeutic Area
- By EndUser
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapeutic Area
- By EndUser
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service Type
- By Therapeutic Area
- By EndUser
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapeutic Area
- By EndUser
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service Type
- By Therapeutic Area
- By EndUser
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapeutic Area
- By EndUser
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service Type
- By Therapeutic Area
- By EndUser
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Therapeutic Area
- By EndUser
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Therapeutic Area
- By EndUser
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service Type
- By Therapeutic Area
- By EndUser
- Competition Analysis
- Competition Deep Dive
- Charles River Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- WuXi Biologics
- Labcorp Drug Development
- GenScript Biotech
- Abzena
- Charles River Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by EndUser, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by EndUser, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by EndUser, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by EndUser, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by EndUser, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by EndUser, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by EndUser, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Service Type, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by EndUser, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Service Type, 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Service Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Service Type
- Figure 6: Global Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Therapeutic Area
- Figure 9: Global Market Value Share and BPS Analysis by EndUser, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by EndUser, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by EndUser
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Service Type, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Service Type, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Service Type
- Figure 26: North America Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Therapeutic Area
- Figure 29: North America Market Value Share and BPS Analysis by EndUser, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by EndUser, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by EndUser
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Service Type, 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Service Type, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Service Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Therapeutic Area
- Figure 39: Latin America Market Value Share and BPS Analysis by EndUser, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by EndUser, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by EndUser
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Service Type, 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Service Type, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Service Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Therapeutic Area
- Figure 49: Western Europe Market Value Share and BPS Analysis by EndUser, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by EndUser, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by EndUser
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Service Type, 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Service Type, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Service Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Therapeutic Area
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by EndUser, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by EndUser, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by EndUser
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Service Type, 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Service Type, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Service Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Therapeutic Area
- Figure 69: East Asia Market Value Share and BPS Analysis by EndUser, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by EndUser, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by EndUser
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Service Type, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Service Type, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Service Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Therapeutic Area
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by EndUser, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by EndUser, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by EndUser
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Service Type, 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Service Type, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Service Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Therapeutic Area
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by EndUser, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by EndUser, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by EndUser
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis