Antibody Drug Conjugates Contract Market Size, Share, Growth and Forecast (2026 - 2036)
Antibody Drug Conjugates Contract Market is segmented by Condition Type (Breast Cancer, Myeloma, Lymphoma, Other Condition Types), Application (Cleavable Linker, Non cleavable Linker), End Use (Pharmaceutical Companies, Biotechnology Companies, Research Institutes), and Region, with forecasts covering the period from 2026 to 2036.
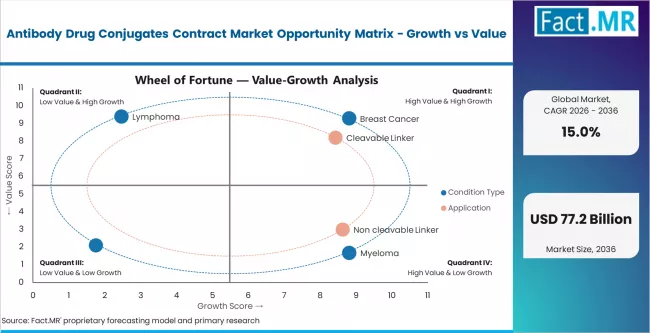
According to Fact.MR estimates, the global antibody drug conjugates contract market market was valued at USD 16.6 billion in 2025. The market is projected to reach USD 19.1 billion in 2026 and is expected to grow to USD 77.2 billion by 2036, expanding at a CAGR of 15.0%. Breast Cancer is anticipated to account for 42.6% of the product segment in 2026, while Cleavable Linker is expected to remain the leading application with around 58.3% share.
Antibody Drug Conjugates Contract Market Analysis and Forecast by Fact.MR
The antibody drug conjugates contract market, valued at USD 16.6 billion in 2025, is expanding at a 15.0% CAGR to reach USD 77.23 billion by 2036. ADC contract manufacturing covers the outsourced production of antibody-drug conjugate therapies, including antibody expression, linker chemistry, cytotoxin payload conjugation, and fill-finish operations provided by CDMOs to pharmaceutical and biotechnology companies. Growth reflects the rapidly expanding ADC clinical pipeline, increasing manufacturing complexity that favors specialist outsourcing, and capacity expansion investments by leading CDMOs. The USD 58.14 billion incremental opportunity between 2026 and 2036 is concentrated in clinical-scale and commercial-scale production for oncology ADC programs.

Summary of Antibody Drug Conjugates Contract Market
- Market Snapshot
- Market value in 2025: USD 16.6 billion
- Projected value in 2026: USD 19.09 billion; forecast to reach USD 77.23 billion by 2036
- CAGR from 2026 to 2036: 15.0%
- Incremental opportunity (2026 to 2036): USD 58.14 billion
- Breast cancer leads condition-type demand with 42.6% share in 2025
- China and India lead country-level growth, followed by Germany and Brazil
- Demand and Growth Drivers
- The ADC clinical pipeline has expanded rapidly, with over 100 ADC candidates in active clinical development across oncology indications, each requiring specialized contract manufacturing for clinical trial material and eventual commercial supply.
- Manufacturing complexity favors outsourcing, as ADC production requires integrated capabilities spanning antibody expression, linker-payload chemistry, conjugation, and high-containment fill-finish that few pharmaceutical companies maintain in-house.
- Regulatory approvals of ADC therapies are accelerating, with multiple new ADC products reaching commercial launch in 2024 and 2025, converting clinical-scale CDMO contracts into higher-volume commercial manufacturing agreements.
- CDMO capacity expansion investments, including high-containment suite construction and potent compound handling infrastructure, are enabling the contract manufacturing sector to absorb growing pipeline demand.
- Product and Segment View
- Breast cancer holds 42.6% of condition-type demand, driven by the commercial success of trastuzumab deruxtecan and other HER2-targeted ADC therapies.
- Cleavable linker technology accounts for 58.3% of application share, reflecting the preference for acid-labile and enzymatically cleavable linker systems in most commercially approved and late-stage ADC programs.
- Clinical-scale production holds 63.2% of deployment share, reflecting the concentration of ADC programs in clinical development phases where CDMO services are most heavily utilized.
- Key segmentation includes:
- Condition Type: Breast Cancer, Myeloma, Lymphoma, Other Condition Types
- Application: Cleavable Linker, Non-cleavable Linker
- End Use: Pharmaceutical Companies, Biotechnology Companies, Research Institutes
- Distribution Channel: Direct Contracting, Outsourcing Platforms, Partnership Agreements
- Deployment: Clinical Scale, Late Stage, Commercial Scale
- Geography and Competitive Outlook
- North America is the largest revenue region, with the USA contributing USD 6.884 billion in 2025 through the concentration of ADC drug development and CDMO infrastructure.
- East Asia is the fastest-growing region, with China recording the highest country CAGR driven by CDMO capacity expansion and domestic ADC pipeline growth.
- Europe is shaped by established CDMO operations in Germany and Switzerland, with Lonza and other European CDMOs serving global pharmaceutical clients.
- Lonza Group leads the competitive field with a 16.0% market share, followed by Thermo Fisher Scientific, Cambrex, AbbVie, and Novasep.
- Analyst Opinion
- The ADC contract manufacturing market is at a structural inflection point where clinical pipeline breadth and manufacturing complexity are converging to accelerate outsourcing demand. The technical requirements for ADC production, including potent compound handling, conjugation chemistry expertise, and high-containment fill-finish capabilities, create barriers to in-house manufacturing that favor specialized CDMOs. Fact.MR expects the fastest growth to concentrate in CDMOs that offer integrated end-to-end ADC manufacturing services, from antibody production through conjugation to finished drug product, as pharmaceutical companies increasingly prefer single-source manufacturing partners that reduce supply chain complexity and technology transfer risk.
Key Growth Drivers, Constraints, and Opportunities
Key Factors Driving Growth
- ADC pipeline acceleration: Over 100 ADC candidates are in clinical development globally, with the pipeline expanding faster than in-house manufacturing capacity, creating structural outsourcing demand for clinical trial material production.
- Manufacturing complexity favoring outsourcing: ADC production requires specialized potent compound handling, conjugation chemistry expertise, and high-containment manufacturing suites that most pharmaceutical companies find more cost-effective to outsource than to build and maintain internally.
- Commercial ADC approval momentum: The acceleration of ADC regulatory approvals is converting clinical-scale CDMO contracts into larger, longer-duration commercial manufacturing agreements with higher annual revenue per program.
Key Market Constraints
- CDMO capacity bottlenecks: High-containment manufacturing suite construction requires 18 to 36 months of lead time and significant capital investment, creating capacity constraints that limit the ability of CDMOs to absorb rapidly growing demand.
- Technology transfer complexity: Moving ADC manufacturing processes between CDMO sites or from CDMO to in-house facilities involves extensive process validation and regulatory filings, creating switching costs that slow supply chain optimization.
- Potent compound safety requirements: Handling cytotoxic payloads used in ADC conjugation requires specialized occupational health infrastructure and regulatory compliance, increasing manufacturing costs and limiting the number of qualified contract manufacturers.
Key Opportunity Areas
- Asia-Pacific CDMO capacity expansion: CDMOs in China, India, and South Korea are investing in ADC manufacturing capabilities at cost structures below Western European and North American competitors, creating cost-competitive capacity for both regional and global pharmaceutical clients.
- Next-generation ADC platform services: CDMOs that develop proprietary linker-payload platform technologies can offer differentiated manufacturing services and capture technology licensing revenue in addition to production fees.
- Integrated end-to-end ADC services: CDMOs offering single-source manufacturing from antibody production through fill-finish are capturing contract preference from pharmaceutical companies seeking to minimize technology transfer risk and supply chain complexity.
Segment-wise Analysis of the Antibody Drug Conjugates Contract Market
- Breast cancer leads condition-type demand at 42.6%, driven by the commercial success and pipeline depth of HER2-targeted and other breast cancer ADC therapies.
- Cleavable linker technology holds 58.3% of application share, reflecting the preference for targeted payload release mechanisms in most approved and late-stage ADC programs.
- Clinical-scale production accounts for 63.2% of deployment share, as the majority of ADC programs remain in clinical development phases.
Which condition type dominates the Antibody Drug Conjugates Contract Market?
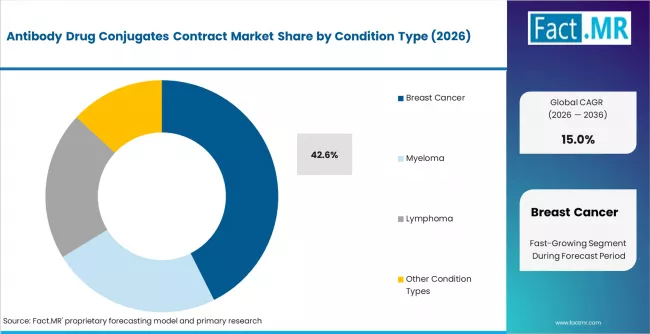
Breast cancer accounts for 42.6% of condition-type demand in 2025, the largest single indication category. The dominance reflects the commercial success of trastuzumab deruxtecan (Enhertu) and other HER2-targeted ADC therapies that have established breast cancer as the primary ADC treatment indication. The depth of the breast cancer ADC pipeline, with multiple candidates targeting HER2, TROP2, and other breast cancer antigens in late-stage trials, generates sustained clinical and commercial manufacturing demand.
Lymphoma and myeloma represent the next-largest condition categories, with brentuximab vedotin (Adcetris) and belantamab mafodotin establishing ADC therapy in hematological malignancies. The condition-type mix is diversifying as ADC programs expand into solid tumors beyond breast cancer, including lung, gastric, and urothelial cancers, broadening the manufacturing demand base.
Which linker technology dominates the Antibody Drug Conjugates Contract Market?
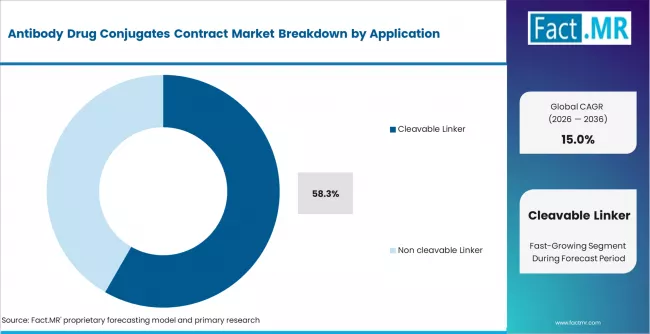
Cleavable linker technology holds 58.3% of application share in 2025, reflecting the predominance of acid-labile, protease-cleavable, and disulfide-based linker systems in commercially approved and late-stage ADC programs. Cleavable linkers are designed to release their cytotoxic payload selectively in the tumor microenvironment, offering a bystander killing effect that extends therapeutic activity to antigen-negative tumor cells.
Non-cleavable linkers retain a significant share, used in ADC designs where payload release is controlled through lysosomal degradation of the entire antibody-linker-payload complex. The linker technology choice is determined by the specific drug design and target biology of each ADC program, with both approaches maintaining active clinical pipeline representation.
Which deployment scale dominates the Antibody Drug Conjugates Contract Market?
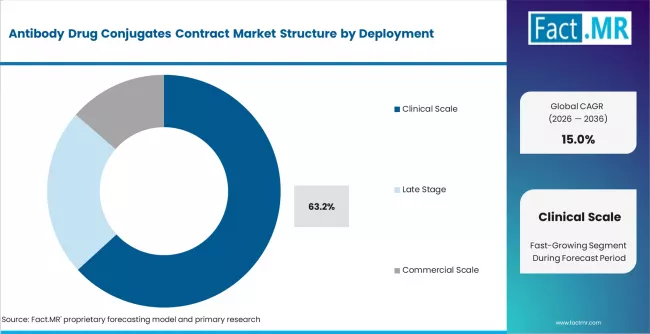
Clinical-scale production accounts for 63.2% of deployment demand in 2025, reflecting the concentration of ADC programs in Phase 1, Phase 2, and Phase 3 clinical trials where CDMO services are most heavily utilized. Clinical-scale contracts generate moderate per-program revenue but are high in number, as each ADC candidate in the pipeline requires dedicated manufacturing campaigns for clinical trial material supply.
Commercial-scale production is the fastest-growing deployment category, driven by the increasing number of ADC therapies reaching regulatory approval and requiring sustained commercial supply manufacturing. Late-stage production, covering pivotal trial material and commercial launch inventory, generates the highest per-program revenue and the longest contract durations.
Which Product Trend is Shaping the Next Phase of Growth in the Antibody Drug Conjugates Contract Market?
Integrated end-to-end ADC manufacturing platforms are shaping the next growth phase in the contract market. Pharmaceutical companies are increasingly seeking CDMOs that can provide the complete manufacturing value chain for ADC products, from monoclonal antibody cell line development and large-scale expression through linker-payload conjugation, analytical characterization, and high-containment fill-finish. This integrated approach reduces technology transfer events between multiple specialist providers, shortens manufacturing timelines, and simplifies regulatory filings by consolidating manufacturing site documentation.
CDMOs that have invested in building integrated ADC capabilities are capturing contract preference and commanding premium pricing relative to providers offering individual process steps. Lonza, Thermo Fisher Scientific, and Samsung Biologics have made significant capital investments in dedicated ADC manufacturing facilities designed to support end-to-end production at both clinical and commercial scale. Fact.MR projects that CDMOs offering integrated platforms will capture a disproportionate share of the incremental growth opportunity, as the ADC pipeline transitions from clinical to commercial production phases and pharmaceutical companies prioritize supply chain simplicity and manufacturing partner consolidation.
Regional Outlook Across Key Markets
.webp)
- North America is the largest revenue region, with the USA contributing USD 6.884 billion in 2025 through the concentration of ADC drug development activity and established CDMO infrastructure.
- East Asia is the fastest-growing region, with China recording the highest country CAGR driven by CDMO capacity investment and domestic ADC pipeline growth.
- Europe is shaped by established CDMO operations in Germany and Switzerland, with premium positioning in high-containment biologics manufacturing.
- Latin America is an emerging corridor, with Brazil recording strong CAGR as regional pharmaceutical companies begin outsourcing biologics manufacturing.
CAGR Table
| Country | CAGR (%) |
|---|---|
| China | 19.3% |
| India | 17.9% |
| Germany | 16.4% |
| Brazil | 15.0% |
| USA | 13.6% |
| UK | 12.1% |
| Japan | 10.7% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research.
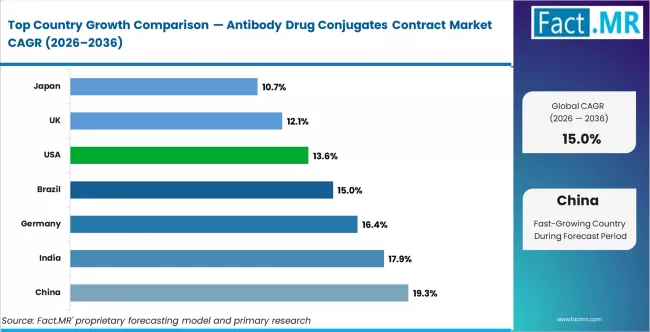
China: CDMO capacity investment and domestic pipeline growth
China records the highest country-level CAGR in the ADC contract manufacturing market, driven by massive CDMO capacity expansion and rapid growth of the domestic ADC clinical pipeline. Chinese CDMOs including WuXi Biologics, Asymchem, and Hengrui are investing in high-containment ADC manufacturing suites designed to serve both Chinese pharmaceutical companies and multinational sponsors seeking cost-competitive manufacturing capacity. The depth of China's oncology clinical pipeline, with dozens of ADC candidates in clinical development, generates local demand for clinical-scale contract manufacturing.
- Growth anchor: CDMO capacity expansion and the depth of the domestic ADC pipeline are creating a dual demand structure combining local and export-oriented manufacturing.
India: Cost-competitive CDMO entry and biosimilar integration
India records the second-highest country CAGR, driven by the entry of Indian CDMOs into ADC contract manufacturing as an extension of their established biologics and biosimilar production capabilities. Companies including Biocon and Piramal Pharma are investing in high-containment manufacturing infrastructure to capture ADC production contracts from global pharmaceutical companies seeking cost-optimized manufacturing partnerships. India's established regulatory relationship with the FDA and EMA for biologics manufacturing provides a credibility platform for ADC CDMO services.
- Growth anchor: Cost-competitive manufacturing infrastructure and established biologics regulatory credentials are enabling Indian CDMO entry into ADC contract production.
Germany: Premium CDMO positioning and European pipeline
Germany leads European ADC contract manufacturing growth, anchored by Lonza's operations and the concentration of European pharmaceutical company headquarters that generate regional outsourcing demand. German CDMOs compete on manufacturing quality, regulatory track record, and proximity to European pharmaceutical R&D centers. The EU regulatory framework for ADC manufacturing is well-established, providing a stable compliance environment for high-containment biologics production.
- Growth anchor: Premium CDMO positioning and proximity to European pharmaceutical headquarters create sustained contract demand for clinical and commercial ADC manufacturing.
USA: Pipeline concentration and integrated CDMO demand
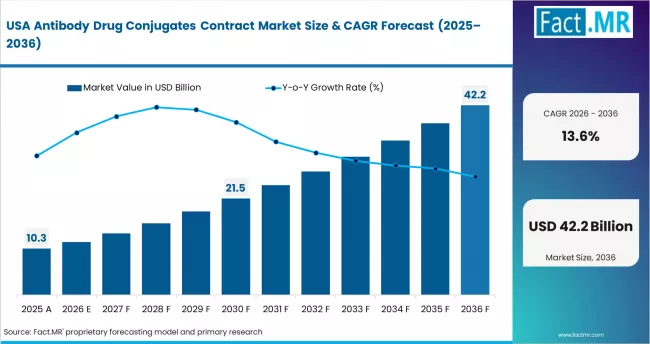
The USA contributes USD 6.884 billion to the global ADC contract market in 2025, the largest single-country revenue pool. Growth reflects the concentration of ADC drug development in US-based pharmaceutical and biotechnology companies, the presence of major CDMO operations including Catalent and Thermo Fisher Scientific, and strong venture capital funding for ADC-focused biotech companies that outsource manufacturing. The shift toward integrated end-to-end CDMO services is most advanced in the US market.
- Growth anchor: Pipeline concentration, established CDMO infrastructure, and venture-funded biotech demand create the deepest contract manufacturing market globally.
Competitive Benchmarking and Company Positioning
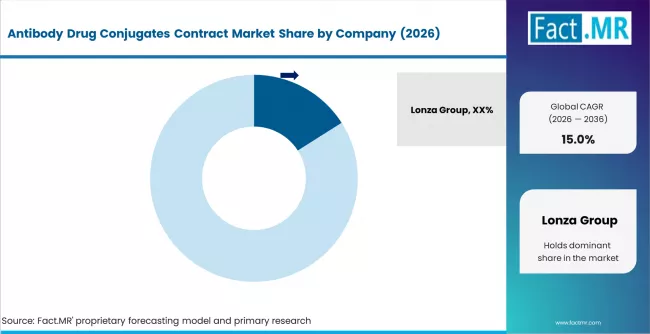
Antibody Drug Conjugates Contract Market Analysis By Company
- Lonza Group leads with 16.0% market share, anchored by its dedicated ADC manufacturing facilities in Europe and the USA offering integrated end-to-end production from antibody to fill-finish.
- Thermo Fisher Scientific and Cambrex compete in the broad CDMO tier, with Thermo Fisher leveraging its Patheon biologics network and Cambrex offering specialized small-molecule and conjugation chemistry services.
- AbbVie (through its contract manufacturing division), Novasep, and ReciPharm maintain positions in specific ADC manufacturing steps, with Novasep focused on payload and linker chemistry and AbbVie on biologics production.
The competitive field is moderately concentrated, with the top five CDMOs accounting for a combined share above 45%. Lonza's leadership reflects its early investment in dedicated ADC manufacturing infrastructure and its ability to offer the complete manufacturing value chain under one organization. The company's high-containment suites in Visp (Switzerland) and Portsmouth (USA) are purpose-built for potent compound handling, providing a capacity and capability advantage over CDMOs that have retrofitted existing facilities.
Competition is intensifying from Asian CDMOs, particularly WuXi Biologics and Samsung Biologics, that have invested in ADC-capable manufacturing at cost structures below Western competitors. These entrants are initially capturing clinical-scale manufacturing contracts and are progressively moving toward commercial-scale capabilities as their regulatory track records mature.
The market is also experiencing vertical integration pressure, as large pharmaceutical companies with successful ADC products consider building in-house manufacturing capacity to reduce long-term dependency on CDMOs. This creates a dynamic where CDMOs must demonstrate technical differentiation and cost competitiveness to retain commercial-scale contracts against in-sourcing alternatives.
Key Companies in the Antibody Drug Conjugates Contract Market
Lonza Group, Thermo Fisher Scientific Inc., Cambrex Corporation, AbbVie Inc., Novasep, ReciPharm, Pantheon, Corden Pharma, and Sterling Pharma Solutions are the principal companies operating in the global ADC contract manufacturing market.
Recent Industry Developments
- Lonza Group: ADC manufacturing capacity expansion (2025)
- Lonza Group completed construction of an expanded high-containment ADC manufacturing suite in 2025, increasing its commercial-scale production capacity to accommodate growing demand from pharmaceutical clients transitioning ADC programs from clinical to commercial supply.
- Thermo Fisher Scientific: integrated ADC service launch (2025)
- Thermo Fisher Scientific launched an integrated end-to-end ADC contract manufacturing service in 2025, consolidating antibody production, conjugation, and fill-finish capabilities under a single contract framework to reduce technology transfer complexity for pharmaceutical sponsors.
- Samsung Biologics: ADC facility investment (2025)
- Samsung Biologics announced a major investment in ADC-dedicated manufacturing facilities in 2025, targeting completion in 2027 to serve both Korean pharmaceutical companies and international ADC program sponsors seeking Asia-Pacific manufacturing capacity.
- WuXi Biologics: ADC clinical manufacturing milestone (2026)
- WuXi Biologics achieved a milestone of supporting over 50 ADC clinical manufacturing programs through its dedicated facilities in early 2026, demonstrating the scale of demand for contract manufacturing services from the Chinese and global ADC pipeline.
Leading Companies Shaping the Antibody Drug Conjugates Contract Market
-
Global Players
- Lonza Group
- Thermo Fisher Scientific Inc.
- Cambrex Corporation
- AbbVie Inc.
- Novasep
- Samsung Biologics
-
Regional Players
- ReciPharm
- Pantheon
- Corden Pharma
- Sterling Pharma Solutions
-
Emerging / Start-up Players
- WuXi Biologics
- Asymchem Laboratories
- Piramal Pharma Solutions
Sources and Research References
- Lonza Group Annual Report 2024 and Biologics Manufacturing Investor Presentations
- Thermo Fisher Scientific Inc. Annual Report 2024, Patheon Division Updates
- ClinicalTrials.gov ADC Clinical Trial Registry Data
- FDA Biologics License Application (BLA) Database for ADC Approvals
- EMA European Public Assessment Reports for ADC Therapies
- Beacon ADC Database: Pipeline and Clinical Development Tracker
All numerical data in this report is derived from Fact.MR proprietary research. Source references are provided for contextual and directional validation only.
Key Questions This Report Addresses
- What is the projected size of the ADC contract market by 2036?
- Which condition type holds the largest share in the ADC contract market?
- What CAGR is the ADC contract market expected to register from 2026 to 2036?
- Which country is the fastest-growing market for ADC contract manufacturing?
- Who is the leading company in the global ADC contract market?
- What is the incremental opportunity between 2026 and 2036?
- How is pipeline growth affecting ADC contract manufacturing demand?
- What role does integrated end-to-end manufacturing play in CDMO competition?
Antibody Drug Conjugates Contract Market Definition
The antibody drug conjugates contract market covers outsourced manufacturing services for ADC therapies, including monoclonal antibody production, linker-payload conjugation, analytical testing, and fill-finish operations provided by contract development and manufacturing organizations to pharmaceutical and biotechnology companies. Services span pre-clinical through commercial-scale production for ADC programs targeting oncology and other therapeutic indications.
Antibody Drug Conjugates Contract Market Inclusions
- Contract antibody expression, purification, and characterization services for ADC programs
- Linker-payload conjugation chemistry and process development services
- High-containment fill-finish operations for potent ADC drug products
- Analytical method development, stability testing, and regulatory filing support for ADC manufacturing
Antibody Drug Conjugates Contract Market Exclusions
- In-house ADC manufacturing performed by pharmaceutical companies within their own facilities
- Contract research services not directly related to ADC manufacturing (e.g., discovery biology, target identification)
- Commercial distribution, logistics, and cold chain services for finished ADC products
- Clinical trial management, patient recruitment, and regulatory submission consulting services
Antibody Drug Conjugates Contract Market Research Methodology
- Bottom-up revenue modeling using ADC pipeline program counts, manufacturing phase (clinical vs. commercial), and average contract values by production scale
- Primary interviews with CDMO business development executives, pharmaceutical outsourcing directors, and ADC program manufacturing leads
- Cross-referencing ADC clinical trial registrations (ClinicalTrials.gov, EMA) and commercial drug approval databases for pipeline-to-commercialization conversion rates
- Validation of segment shares through CDMO capacity utilization data, published contract announcements, and investor presentations
- Forecasting calibrated against ADC pipeline growth projections, regulatory approval timelines, and CDMO capacity expansion investment plans
- Company-level revenue triangulation using annual reports, SEC filings, and earnings call transcripts from listed CDMO and pharmaceutical companies
Scope of Analysis
| Parameter | Details |
|---|---|
| Market size (2025) | USD 16.6 billion |
| Forecast period | 2026 to 2036 |
| CAGR | 15.0% |
| Base year | 2025 |
| Units | USD Billion |
| Segments covered | Condition Type (Breast Cancer, Myeloma, Lymphoma, Others); Application (Cleavable Linker, Non-cleavable Linker); End Use (Pharmaceutical Companies, Biotechnology Companies, Research Institutes); Distribution Channel (Direct Contracting, Outsourcing Platforms, Partnership Agreements); Deployment (Clinical Scale, Late Stage, Commercial Scale) |
| Regions covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries profiled | USA, China, India, Germany, UK, Brazil, Japan |
| Companies profiled | Lonza Group, Thermo Fisher Scientific Inc., Cambrex Corporation, AbbVie Inc., Novasep, ReciPharm |
| Forecasting approach | Bottom-up revenue modeling validated through primary research, clinical pipeline analysis, and company-level financial triangulation |
Market Segmentation Analysis
-
Antibody Drug Conjugates Contract Market Market Segmented by Condition Type:
- Breast Cancer
- HER2 Positive Breast Cancer
- Triple Negative Breast Cancer
- Hormone Receptor Positive Breast Cancer
- Myeloma
- Multiple Myeloma
- Relapsed and Refractory Myeloma
- Smoldering Myeloma
- Lymphoma
- Hodgkin Lymphoma
- Non Hodgkin Lymphoma
- Cutaneous T Cell Lymphoma
- Other Condition Types
- Leukemia
- Lung Cancer
- Ovarian Cancer
- Breast Cancer
-
Antibody Drug Conjugates Contract Market Market Segmented by Application:
- Cleavable Linker
- Acid Cleavable Linker
- Protease Cleavable Linker
- Glutathione Cleavable Linker
- Non cleavable Linker
- Thioether Linker
- Stable Amide Linker
- Maleimide Linker
- Cleavable Linker
-
Antibody Drug Conjugates Contract Market Market Segmented by End Use:
- Pharmaceutical Companies
- Innovator Companies
- Large Pharma
- Mid Size Pharma
- Biotechnology Companies
- Emerging Biotech
- Clinical Stage Companies
- Research Focused Firms
- Research Institutes
- Academic Institutions
- Clinical Research Centers
- Government Research Labs
- Pharmaceutical Companies
-
Antibody Drug Conjugates Contract Market Market Segmented by Distribution Channel:
- Direct Contracting
- Big Pharma Contracts
- Long Term Agreements
- Strategic Partnerships
- Outsourcing Platforms
- Service Aggregators
- Project Based Contracts
- Flexible Engagement Models
- Partnership Agreements
- Collaborations
- Co Development
- Licensing Agreements
- Direct Contracting
-
Antibody Drug Conjugates Contract Market Market Segmented by Deployment:
- Clinical Scale
- Early Phase
- Phase One
- Phase Two
- Late Stage
- Phase Three
- Pre Commercial
- Commercial Scale
- Large Scale Manufacturing
- Global Supply
- High Volume Production
- Clinical Scale
-
Antibody Drug Conjugates Contract Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
What is the current size of the ADC contract market?
The global ADC contract market is valued at USD 16.6 billion in 2025.
What growth rate is expected?
The market is projected to grow at a CAGR of 15.0% from 2026 to 2036.
What will the market be worth by 2036?
The market is forecast to reach USD 77.23 billion by 2036.
Which condition type leads the ADC contract market?
Breast cancer leads with a 42.6% share of condition-type demand in 2025.
Which country is growing fastest?
China records the highest country-level CAGR among tracked markets.
Who is the market leader?
Lonza Group leads with a 16.0% market share in 2025.
What is the USA market value?
The USA contributes USD 6.884 billion to the global ADC contract market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Condition Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Condition Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Condition Type , 2026 to 2036
- Breast Cancer
- HER2 Positive Breast Cancer
- Triple Negative Breast Cancer
- Hormone Receptor Positive Breast Cancer
- Myeloma
- Multiple Myeloma
- Relapsed and Refractory Myeloma
- Smoldering Myeloma
- Lymphoma
- Hodgkin Lymphoma
- Non Hodgkin Lymphoma
- Cutaneous T Cell Lymphoma
- Other Condition Types
- Leukemia
- Lung Cancer
- Ovarian Cancer
- Breast Cancer
- Y to o to Y Growth Trend Analysis By Condition Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Condition Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Cleavable Linker
- Acid Cleavable Linker
- Protease Cleavable Linker
- Glutathione Cleavable Linker
- Non cleavable Linker
- Thioether Linker
- Stable Amide Linker
- Maleimide Linker
- Cleavable Linker
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical Companies
- Innovator Companies
- Large Pharma
- Mid Size Pharma
- Biotechnology Companies
- Emerging Biotech
- Clinical Stage Companies
- Research Focused Firms
- Research Institutes
- Academic Institutions
- Clinical Research Centers
- Government Research Labs
- Pharmaceutical Companies
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct Contracting
- Big Pharma Contracts
- Long Term Agreements
- Strategic Partnerships
- Outsourcing Platforms
- Service Aggregators
- Project Based Contracts
- Flexible Engagement Models
- Partnership Agreements
- Collaborations
- Co Development
- Licensing Agreements
- Direct Contracting
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- Clinical Scale
- Early Phase
- Phase One
- Phase Two
- Late Stage
- Phase Three
- Pre Commercial
- Commercial Scale
- Large Scale Manufacturing
- Global Supply
- High Volume Production
- Clinical Scale
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Condition Type
- By Application
- By End Use
- By Distribution Channel
- By Deployment
- Competition Analysis
- Competition Deep Dive
- Lonza Group
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Thermo Fisher Scientific Inc.
- Cambrex Corporation
- AbbVie Inc.
- Novasep
- ReciPharm
- Pantheon
- Corden Pharma
- Sterling Pharma Solutions
- Others
- Lonza Group
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Condition Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Condition Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Condition Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Condition Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Condition Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Condition Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Condition Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Condition Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Condition Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Condition Type,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Condition Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Distribution Channel
- Figure 15: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Deployment,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Deployment
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 22: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Condition Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Condition Type,2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Condition Type
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by End Use
- Figure 38: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by Distribution Channel
- Figure 41: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Deployment,2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Deployment
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Condition Type, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Condition Type,2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Condition Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by Application
- Figure 51: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by End Use
- Figure 54: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 57: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Deployment,2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by Deployment
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Condition Type, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Condition Type,2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by Condition Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 66: Western Europe Market Attractiveness Analysis by Application
- Figure 67: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 69: Western Europe Market Attractiveness Analysis by End Use
- Figure 70: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 73: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Deployment,2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Deployment
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Condition Type, 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Condition Type,2026 to 2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Condition Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Application
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Deployment,2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Condition Type, 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Condition Type,2026 to 2036
- Figure 95: East Asia Market Attractiveness Analysis by Condition Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 98: East Asia Market Attractiveness Analysis by Application
- Figure 99: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 101: East Asia Market Attractiveness Analysis by End Use
- Figure 102: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 104: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 105: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Deployment,2026 to 2036
- Figure 107: East Asia Market Attractiveness Analysis by Deployment
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Condition Type, 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Condition Type,2026 to 2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Condition Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment,2026 to 2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Condition Type, 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Condition Type,2026 to 2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Condition Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment,2026 to 2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis