Stem Cell Therapy Market Size, Share, Growth and Forecast (2026 - 2036)
Stem Cell Therapy Market is segmented by Cell Type (Embryonic Stem Cells, Induced Pluripotent Stem Cells, Mesenchymal Stem Cells, Hematopoietic Stem Cells, and Neural Stem Cells), Application (Regenerative Medicine, Drug Discovery and Development, Toxicity Testing, Disease Modeling, and Others), End Use (Hospitals and Clinics, Research Institutes, Biotechnology Companies, and Others), and Region. Forecast for 2026 to 2036.
Fact.MR analysis suggests the stem cell therapy market is undergoing a decisive regulatory and commercialization shift. As iPSC technologies mature and allogeneic manufacturing scales, the longstanding divide between experimental cell therapies and approved clinical products is narrowing. Reimbursement coverage is expanding in the United States, Japan, and Germany, converting pipeline assets into commercial revenue streams. This transition is driving firms to move from single-indication development to platform-based approaches that address multiple diseases, creating higher-margin recurring treatment revenues and long-term manufacturing partnership opportunities for cell therapy enablers.
Summary of Stem Cell Therapy Market
- Market Definition
- Stem cell therapy includes treatment and research uses of mesenchymal, hematopoietic, embryonic, iPSC, and neural stem cells across hospitals, research centres, and biotech companies.
- Demand Drivers
- Faster regulatory approvals in the US and Europe are accelerating commercial launch of stem cell therapies.
- Japan’s regenerative medicine framework and Sakigake pathway support faster therapy approvals and attract biotech investment.
- Government funding programmes in the US and EU are supporting cell therapy research and manufacturing expansion.
- Key Segments Analyzed
- By Cell Type: Mesenchymal Stem Cells hold about 47% share in 2026 due to wider clinical applications and established manufacturing.
- By Application: Regenerative Medicine leads with about 62% share driven by cardiovascular, orthopaedic, and neurological therapies.
- By End User: Hospitals and clinics hold the largest share as approved therapies must be administered in regulated clinical settings.
- By Geography: Asia Pacific leads, contributing the highest expansion rates.
- Analyst Opinion at FACT.MR
- Shambhu Nath Jha, Principal Consultant at Fact.MR opines, “CXOs will find this report useful as regulatory approval acceleration, allogeneic manufacturing scale up, and expanding reimbursement coverage in the United States, Japan, and Germany are transforming stem cell therapy into a commercial market.
- Strategic Implications
- Developers should focus on allogeneic MSC and iPSC therapies which enable scalable off the shelf treatments.
- CDMOs should expand GMP compliant manufacturing facilities to meet future commercial demand.
- Companies should engage early with FDA RMAT, EMA ATMP, and PMDA Sakigake programmes to accelerate approvals.
- Methodology
- Market sizing combined therapy pipeline estimates with pricing data by cell type and indication.
- Regulatory timelines, funding programmes, and CDMO pricing trends were analysed using global regulatory databases.
- Regional forecasts used clinical trial data, national funding programmes, reimbursement expansion, and expert interviews.
Segmental Analysis
Stem Cell Therapy Market Analysis by Cell Type
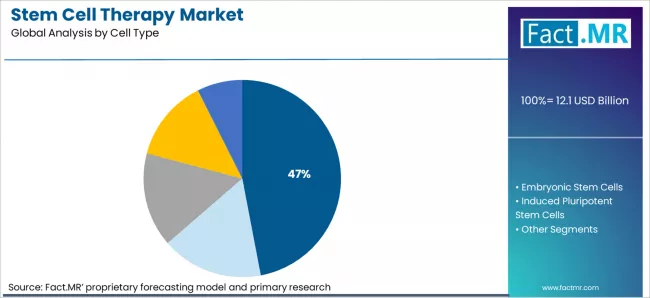
Based on FACT.MR's stem cell therapy market report, Mesenchymal Stem Cells are estimated to hold about 47% share in 2026. MSCs lead due to wider approved products, multiple clinical uses, and established GMP manufacturing supporting large scale production.
- Mesoblast Pipeline Advancement: In early 2025, Mesoblast received FDA acceptance of its BLA for remestemcel L targeting steroid refractory acute GvHD in paediatric patients, strengthening the commercial pathway for allogeneic MSC therapies. [1]
- JCR Pharmaceuticals MSC Expansion: In 2024, JCR Pharmaceuticals reported rising sales of TEMCELL HS Inj. for acute GvHD in Japan supported by national reimbursement coverage. The company also plans to expand TEMCELL into new indications. [2]
- Allogeneic Manufacturing Scale Up Trend: In 2024, Lonza expanded cGMP MSC manufacturing capacity at its Houston facility to support commercial production for multiple cell therapy developers. [3]
Stem Cell Therapy Market Analysis by Application
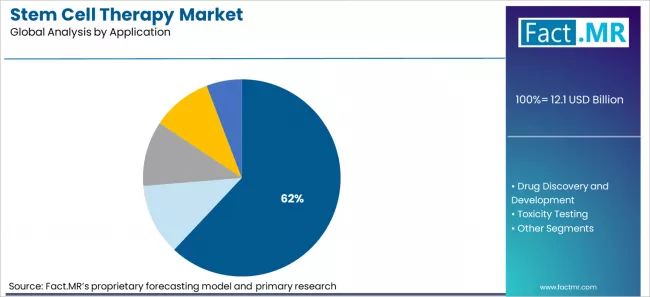
Based on FACT.MR's stem cell therapy market report, Regenerative Medicine is estimated to hold about 62% share in 2026. The segment leads due to high value therapies for cardiovascular, orthopaedic, and neurological diseases where treatment alternatives are limited.
- Bio Cardia Cardiovascular Programme: In 2024, Bio Cardia continued enrolment for its CardiAMP Heart Failure II trial evaluating autologous cell therapy for heart failure patients. [4]
- Neurological iPSC Application Development: In 2025, ReNeuron reported progress in its hRPC stem cell therapy programme for retinal diseases with preparation for IND enabling studies. [5]
- Drug Discovery Revenue Growth Trend: In 2024, STEMCELL Technologies reported rising demand for iPSC derived cell models used in drug testing and disease research by pharmaceutical companies. [6]
Key Dynamics
Drivers, Restraints, and Opportunities
FACT.MR analysts observe that the stem cell therapy market has shifted from a research focused field to an emerging commercial therapy segment. Growth reflects regulatory approvals, early MSC product launches, and expanding clinical research across multiple therapeutic areas.
Market development is shaped by high clinical potential alongside manufacturing cost and regulatory challenges. Autologous therapies offer personalised treatment but face scalability limits, while allogeneic platforms support larger scale production.
- FDA RMAT Designation Driving U.S. Commercial Pipeline: The FDA RMAT programme has granted over 270 designations by 2024, accelerating development timelines for regenerative therapies. Updated guidance in 2024 clarified manufacturing requirements for allogeneic products. [7]
- Japan Sakigake Conditional Approval Commercialization: Japan's Sakigake and regenerative medicine framework allow conditional approval of stem cell therapies with post market evidence requirements, supporting faster commercialization. [8]
- EU ATMP Hospital Exemption Expanding European Access: The EU ATMP Hospital Exemption allows hospitals to produce and use advanced therapies before full EMA approval, supporting early clinical adoption across European academic centres. [9]
Regional Analysis
The stem cell therapy market is analysed across North America, Europe, Asia Pacific, Latin America, and Middle East and Africa, covering 40+ countries with distinct demand profiles shaped by regulatory approval frameworks, government funding programmes, clinical infrastructure maturity, and reimbursement coverage for advanced therapy products. The full report offers market attractiveness analysis based on regulatory pipeline density and commercial launch timelines.
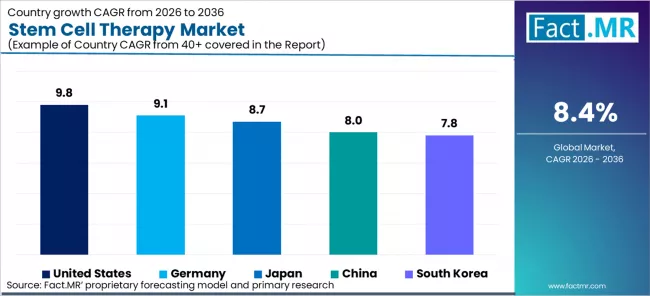
| Country | CAGR (2026-2036) |
|---|---|
| United States | 9.8% |
| Germany | 9.1% |
| Japan | 8.7% |
| China | 8.0% |
| South Korea | 7.8% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
North America Stem Cell Therapy Market Analysis
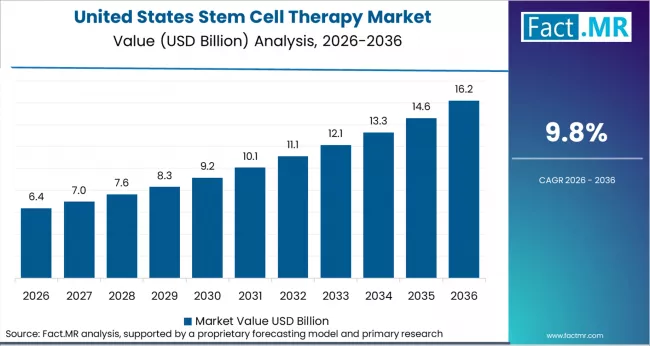
- United States: The United States stem cell therapy market is expected to expand at a CAGR of 9.8% through 2036. FDA RMAT designations exceeded 50 new requests in 2024, highlighting a strong development pipeline. Updated FDA guidance in 2024 clarified manufacturing requirements for allogeneic therapies, supporting Mesoblast’s BLA acceptance in 2025. NIH regenerative medicine grants exceeded USD 200 million in 2024, strengthening the clinical research ecosystem.
FACT.MR's North American analysis covers the United States, Canada, and Mexico. The report includes RMAT pipeline data, government funding programmes, CDMO investment trends, and country level CAGR forecasts.
Europe Stem Cell Therapy Market Analysis
- Germany: Germany is anticipated to witness around 9.1% CAGR in stem cell therapy demand through 2036. The Paul Ehrlich Institut updated ATMP hospital exemption oversight in 2024, enabling expanded hospital manufacturing programmes. Germany’s GKV insurance system updated reimbursement assessment rules in 2025, improving market access for developers. University hospitals such as Charité Berlin and Frankfurt expanded cell therapy programmes.
FACT.MR's European analysis covers Germany, France, the United Kingdom, Italy, Spain, the Netherlands, the Nordics, and Rest of Europe. The report includes ATMP approvals, hospital exemption adoption, reimbursement policies, and country growth forecasts.
Asia Pacific Stem Cell Therapy Market Analysis
- China: China is projected to record about 8.0% CAGR in the stem cell therapy market through 2036. Updated NMPA guidance in 2024 clarified clinical trial pathways for MSC and iPSC therapies. The National Health Commission supported over 100 active stem cell clinical trials under the national biomedical innovation programme. Domestic developers including Cellular Biomedicine Group advanced regulatory submissions.
- South Korea: South Korea is likely to experience close to 7.8% CAGR growth in stem cell therapy adoption through 2036. The MFDS updated conditional approval rules in 2024, expanding eligibility for stem cell therapy products. Domestic companies such as Medytox and HLB Life Science increased investment in MSC therapy programmes. The national pharmaceutical strategy in 2025 identified regenerative medicine as a priority export sector.
- Japan: Japan’s stem cell therapy sector is forecast to grow at nearly 8.7% CAGR by 2036. Updated PMDA guidance in 2024 clarified post market data requirements for regenerative therapies. JCR Pharmaceuticals reported growing TEMCELL sales supported by national reimbursement coverage. Japan’s AMED programme continued funding regenerative medicine research in 2024.
FACT.MR's Asia Pacific analysis covers China, Japan, South Korea, India, Australia and New Zealand, ASEAN, and Rest of Asia Pacific. The report includes regulatory approval timelines, funding programmes, and regional growth forecasts.
Competitive Aligners for Market Players
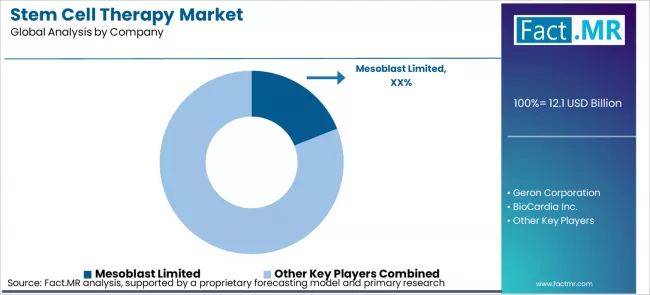
The global stem cell therapy market remains moderately fragmented, with only a few companies holding approved commercial products while many others are still advancing through clinical development. Large pharmaceutical players such as Novartis, Bristol Myers Squibb, and JCR Pharmaceuticals currently lead in approved product revenue. At the same time, several independent biotechnology companies including Mesoblast, BioCardia, Geron Corporation, and ReNeuron are progressing clinical programmes toward regulatory approval. In this market, success depends heavily on the ability to navigate complex regulatory pathways and scale up GMP compliant manufacturing for allogeneic therapies.
Companies with strong manufacturing infrastructure and regulatory experience hold a clear advantage. Lonza Group plays a key role as a leading contract manufacturer supporting commercial scale cell therapy production. Meanwhile, Takeda Pharmaceutical Company has strengthened its position in Europe through its TiGenix acquisition and the approved therapy Alofisel.
Hospitals and treatment centres often form long term partnerships with specific therapy providers, integrating certain products into their treatment protocols. These relationships create high switching costs. Research organisations and biotech developers purchasing stem cell tools and CDMO services, however, maintain stronger pricing leverage by comparing suppliers such as STEMCELL Technologies and Lonza Group through competitive procurement processes.
Recent Developments
- In 2025, Mesoblast Limited received FDA BLA acceptance for remestemcel-L in paediatric steroid-refractory acute graft-versus-host disease, representing the first allogeneic MSC therapy to reach the FDA BLA review stage and establishing a commercial milestone for the MSC cell therapy segment globally.
- In January 2025, STEMCELL Technologies announced expanded clinical-grade iPSC differentiation product portfolio for cardiac, neural, and hematopoietic cell applications, targeting growing pharmaceutical and biotech customer demand for standardized human cell models in drug discovery toxicity testing and disease modelling workflows.
Key Players
- Mesoblast Limited
- Geron Corporation
- BioCardia Inc.
- Lonza Group AG
- Novartis AG
- Bristol Myers Squibb
- JCR Pharmaceuticals Co.
- STEMCELL Technologies
- ReNeuron Group PLC
- Takeda Pharmaceutical Company
Bibliography
- [1] Mesoblast Limited. (2025). Corporate Update: FDA Biologics License Application Acceptance for Remestemcel-L in Paediatric Steroid-Refractory Acute GvHD. February 2025. mesoblast.com
- [2] JCR Pharmaceuticals Co. Ltd. (2024). Annual Report FY2024: TEMCELL HS Commercial Sales Growth and Clinical Programme Extension Plans under Japan ASRM Act. November 2024. jcrpharm.co.jp
- [3] Lonza Group AG. (2024). Press Release: Expanded cGMP Bioreactor-Based MSC Manufacturing Capacity Investment at Houston Facility. September 2024. lonza.com
- [4] BioCardia Inc. (2024). Clinical Programme Update: CardiAMP Heart Failure II Pivotal Trial Patient Enrolment Status. October 2024. biocardia.com
- [5] ReNeuron Group PLC. (2025). Pre-Clinical Programme Update: hRPC Stem Cell Therapy Manufacturing Process Development for IND-Enabling Studies. January 2025. reneuron.com
- [6] STEMCELL Technologies Inc. (2024). Commercial Market Update: Expanding iPSC-Derived Cell Model Demand for Pharmaceutical Drug Toxicity Testing. November 2024. stemcell.com
- [7] U.S. Food and Drug Administration. (2024). Guidance for Industry: Regenerative Medicine Advanced Therapy Designation Manufacturing Comparability Requirements for Allogeneic Cell Therapies. November 2024. fda.gov
- [8] Pharmaceuticals and Medical Devices Agency, Japan. (2024). Guidance Update: Real-World Evidence Data Collection Requirements for Conditionally Approved Regenerative Medicine Products under ASRM Act. 2024. pmda.go.jp
- [9] Paul-Ehrlich-Institut, Germany. (2024). Guidance Update: Hospital Exemption ATMP Oversight Framework GMP Requirements for Hospital-Manufactured Stem Cell Products. 2024. pei.de
This Report Addresses
- Strategic insights on RMAT and ATMP approval pipelines, MSC and iPSC manufacturing scale up, Sakigake commercialization, CDMO capacity expansion, and reimbursement growth.
- Market forecast from USD 12.1 billion in 2026 to USD 27.4 billion by 2036 at 8.4% CAGR by cell type, application, end use, and region.
- Growth opportunities across U.S. RMAT MSC pipelines, EU ATMP hospital exemption programmes, Japan Sakigake approvals, China NMPA framework, and CDMO manufacturing services.
- Regional outlook with CAGR for United States 9.8%, Germany 9.1%, Japan 8.7%, China 8.0%, and South Korea 7.8%.
- Competitive analysis of Mesoblast, BioCardia, Geron, Lonza, Novartis, Bristol Myers Squibb, JCR Pharmaceuticals, STEMCELL Technologies, ReNeuron, and Takeda.
- Technology tracking covering MSC bioreactor manufacturing, iPSC differentiation platforms, clinical grade cell products, GMP cell banking, and AI based cell quality analysis.
- Regulatory analysis including FDA RMAT updates, EMA ATMP approvals, Germany hospital exemption rules, Japan Sakigake framework, China IND pathway, and Korea MFDS approvals.
- Report delivered in Excel datasets, PowerPoint summary, and detailed PDF supported by regulatory data, clinical trial records, funding disclosures, and primary interviews.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD 12.1 billion (2026) to USD 27.4 billion (2036), at a CAGR of 8.4% |
| Market Definition | The stem cell therapy market includes therapies, research tools, and CDMO services using mesenchymal, hematopoietic, embryonic, iPSC, and neural stem cells for regenerative medicine, drug discovery, toxicity testing, and disease modelling across hospitals, research institutes, and biotech companies. |
| Cell Type Segmentation | Embryonic Stem Cells, Induced Pluripotent Stem Cells, Mesenchymal Stem Cells, Hematopoietic Stem Cells, Neural Stem Cells |
| Application Segmentation | Regenerative Medicine, Drug Discovery and Development, Toxicity Testing, Disease Modeling, Others |
| End Use Segmentation | Hospitals and Clinics, Research Institutes, Biotechnology Companies, Others |
| Application Coverage | MSC therapies for GvHD, cardiovascular and orthopaedic treatment; iPSC derived neural and cardiac therapies; haematopoietic stem cell transplants; iPSC cell models for drug discovery; stem cell CDMO manufacturing services for clinical and commercial developers. |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Netherlands, Nordics, Rest of Europe, China, Japan, South Korea, India, Australia and New Zealand, ASEAN, Rest of Asia Pacific, Brazil, Chile, Ecuador, Rest of Latin America, Kingdom of Saudi Arabia, UAE, South Africa, Turkey, Rest of Middle East and Africa |
| Key Companies Profiled | Mesoblast Limited, Geron Corporation, BioCardia Inc., Lonza Group AG, Novartis AG, Bristol Myers Squibb, JCR Pharmaceuticals Co., STEMCELL Technologies, ReNeuron Group PLC, Takeda Pharmaceutical Company |
| Forecast Period | 2026 to 2036 |
| Approach | A hybrid top down and bottom up model was applied. Top down inputs included clinical pipeline estimates by cell type, government regenerative medicine funding, and reimbursement expansion timelines. Bottom up inputs considered average therapy pricing, CDMO manufacturing cost trends, regulatory milestones, and clinical trial data from interviews with developers and hospital procurement teams. |
Stem Cell Therapy Market by Segment
-
By Cell Type :
- Embryonic Stem Cells
- Induced Pluripotent Stem Cells
- Mesenchymal Stem Cells
- Hematopoietic Stem Cells
- Neural Stem Cells
-
By Application :
- Regenerative Medicine (cardiovascular, orthopedic, neurological)
- Drug Discovery and Development
- Toxicity Testing
- Disease Modeling
- Others (cosmetic, veterinary applications)
-
By End Use :
- Hospitals and Clinics
- Research Institutes
- Biotechnology Companies
- Others
-
By Region :
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordics
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia and New Zealand
- ASEAN
- Rest of Asia Pacific
- Latin America
- Brazil
- Chile
- Ecuador
- Rest of Latin America
- Middle East and Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East and Africa
- North America
- Frequently Asked Questions -
How large is the stem cell therapy market in 2025?
The market surpassed USD 11.2 billion in 2025.
What will the market size be in 2026?
The market is estimated at USD 12.1 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 27.4 billion by 2036, creating a USD 15.3 billion absolute opportunity.
What is the expected CAGR from 2026 to 2036?
The market is forecast to grow at a CAGR of 8.4% during the period.
Which cell type segment is poised to lead the market?
Mesenchymal Stem Cells are expected to lead with approximately 47% share in 2026.
Which application segment holds the largest share?
Regenerative Medicine holds approximately 62% share in 2026.
Which country shows the fastest growth rate?
The United States leads with a projected CAGR of 9.8% through 2036.
How fast is the market expected to expand in Germany?
Germany is projected to grow at 9.1% CAGR through 2036.
What is the forecast CAGR for Japan?
Japan is expected to grow at 8.7% CAGR through 2036.
What growth rate is expected for China?
China is projected to grow at 8.0% CAGR through 2036.
What are the main growth drivers of the market?
Growth is driven by expanding regulatory approval pipelines, allogeneic manufacturing scale-up reducing per-treatment costs, and reimbursement coverage expansion in the United States, Japan, and Germany.
What is the primary constraint in this market?
High autologous manufacturing costs, complex regulatory requirements for novel cell types, and reimbursement coverage gaps.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Cell Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Cell Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Cell Type , 2026 to 2036
- Mesenchymal Stem Cells
- Embryonic Stem Cells
- Induced Pluripotent Stem Cells
- Hematopoietic Stem Cells
- Neural Stem Cells
- Mesenchymal Stem Cells
- Y to o to Y Growth Trend Analysis By Cell Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Cell Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Regenerative Medicine
- Drug Discovery and Development
- Toxicity Testing
- Disease Modeling
- Others (cosmetic, veterinary applications)
- Regenerative Medicine
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Cell Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Cell Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Cell Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Cell Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Cell Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Cell Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Cell Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Cell Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Cell Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Cell Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Cell Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Cell Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Cell Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Cell Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Cell Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Cell Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Mesoblast Limited
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Geron Corporation
- BioCardia Inc.
- Lonza Group AG
- Novartis AG
- Bristol Myers Squibb
- JCR Pharmaceuticals Co.
- STEMCELL Technologies
- ReNeuron Group PLC
- Takeda Pharmaceutical Company
- Mesoblast Limited
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Cell Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Cell Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Cell Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Cell Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Cell Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Cell Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Cell Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Cell Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Cell Type , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Cell Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Cell Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Cell Type , 2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Cell Type , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Cell Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Cell Type , 2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Cell Type , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Cell Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Cell Type , 2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Cell Type , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Cell Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Cell Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Cell Type , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Cell Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Cell Type , 2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Cell Type , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Cell Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Cell Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Cell Type , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Cell Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Cell Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Cell Type , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Cell Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Application, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis