Peptide-based Infection Therapeutics Market Size, Share, Growth and Forecast (2026 - 2036)
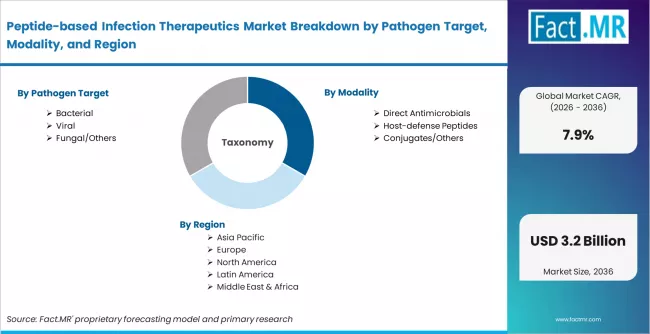
Peptide-based Infection Therapeutics Market is segmented by Pathogen Target (Bacterial, Viral, Fungal/Others), Modality (Direct Antimicrobials, Host-defense Peptides, Conjugates/Others), Setting (Hospital IV, Outpatient Oral, Topical), and Region, with forecasts covering the period from 2026 to 2036.
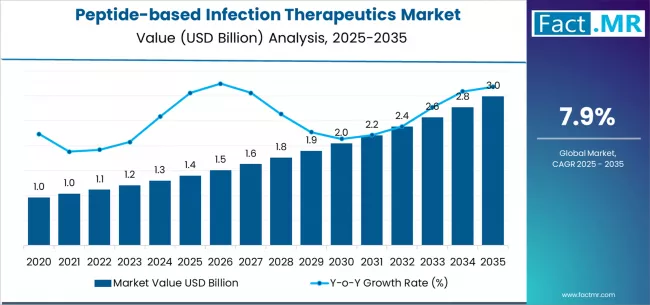
According to Fact.MR estimates, the global peptide-based infection therapeutics market market was valued at USD 1.4 billion in 2025. The market is projected to reach USD 1.5 billion in 2026 and is expected to grow to USD 3.2 billion by 2036, expanding at a CAGR of 7.9%. Bacterial is anticipated to account for 54.0% of the product segment in 2026, while Direct Antimicrobials is expected to remain the leading application with around 35.8% share.
Peptide-based Infection Therapeutics Market Analysis and Forecast by Fact.MR
The peptide-based infection therapeutics market, valued at USD 1.4 billion in 2025, is projected to expand at a 7.9% CAGR to reach USD 3.231 billion by 2036. Peptide therapeutics in this market include antimicrobial peptides (AMPs), host-defence peptides, and peptide-drug conjugates developed to treat bacterial, viral, and fungal infections. Growth reflects the global antimicrobial resistance crisis, the declining efficacy of conventional antibiotics against multidrug-resistant pathogens, and regulatory incentives for novel antimicrobial development. Demand is concentrated in hospital critical care units and infectious disease departments where resistant infections carry high mortality rates.
Summary of Peptide-based Infection Therapeutics Market
- Market Snapshot
- The market is valued at USD 1.4 billion in 2025.
- Revenue is estimated to reach USD 1.511 billion in 2026 and USD 3.231 billion by 2036.
- A CAGR of 7.9% is forecast for the 2026 to 2036 assessment period.
- Incremental opportunity of USD 1.72 billion is projected between 2026 and 2036.
- Bacterial-targeted peptides hold a 54.0% share of the pathogen target segment.
- The USA (8.4% CAGR) and Mexico (7.9% CAGR) are the fastest-expanding country markets.
- Demand and Growth Drivers
- Antimicrobial resistance (AMR) is rendering conventional antibiotics ineffective against an expanding range of pathogens, with WHO estimating 1.27 million deaths directly attributable to AMR in 2019, creating an urgent clinical need for novel mechanisms of action.
- Hospital-acquired infections caused by multidrug-resistant organisms such as MRSA, carbapenem-resistant Enterobacteriaceae, and Acinetobacter are driving demand for peptide-based agents in intensive care and surgical settings.
- Regulatory incentives including FDA Qualified Infectious Disease Product (QIDP) designation and EMA adaptive pathway approvals are accelerating development timelines and extending market exclusivity for novel antimicrobial peptides.
- Government-funded antimicrobial stewardship programmes in the USA, EU, and Japan are allocating procurement budgets specifically for novel antimicrobial agents, creating pull-through demand for peptide therapeutics.
- Product and Segment View
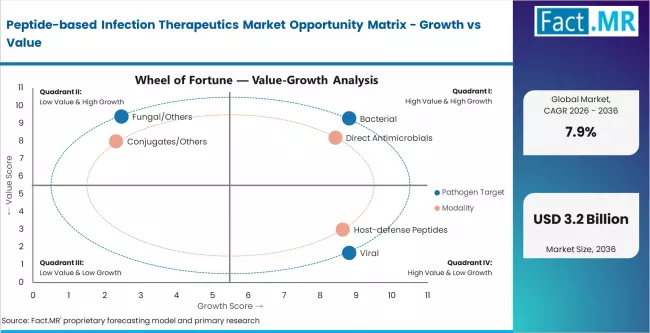
- Bacterial-targeted peptides command 54.0% of the pathogen target segment, reflecting the concentration of AMR burden in bacterial infections.
- Direct antimicrobials account for 35.8% of the modality segment, with host-defence peptides and conjugates comprising the balance.
- Critical care end use represents 48.9% of demand, driven by high-acuity infections in ICU and surgical ward settings.
- Key segmentation includes:
- Pathogen Target: Bacterial, Viral, Fungal/Others
- Modality: Direct Antimicrobials, Host-defense Peptides, Conjugates/Others
- Setting: Hospital IV, Outpatient Oral, Topical
- End Use: Critical Care, Infectious Disease Management, Dermatology, Wound Care
- Geography and Competitive Outlook
- North America accounts for the largest regional share, with the USA generating USD 0.559 billion in 2025 revenue.
- East Asia contributes steady demand, with Japan (USD 0.082 billion) and South Korea (USD 0.052 billion) in 2025.
- Pfizer Inc. leads the competitive field with an estimated 25.0% revenue share.
- Merck and Co., Roche Holding AG, and Basilea Pharmaceutica are principal challengers across global markets.
- Analyst Opinion
- The peptide-based infection therapeutics market occupies a high-urgency, high-barrier competitive space where clinical failure rates remain elevated but successful products command premium pricing and long market exclusivity periods. The AMR crisis is shifting hospital procurement from cost-minimisation toward clinical efficacy prioritisation for resistant infections, creating favourable pricing dynamics for novel peptide agents. Companies that can navigate the complex regulatory pathway and demonstrate clear superiority over last-resort antibiotics such as colistin will capture significant value. Pipeline attrition remains the primary commercial risk, requiring diversified clinical portfolios.
Key Growth Drivers, Constraints, and Opportunities
Key Factors Driving Growth
- The global antimicrobial resistance burden, with the WHO projecting AMR-attributable deaths to reach 10 million annually by 2050 under current trajectories, is creating sustained and growing demand for novel mechanisms of action that bypass conventional resistance pathways.
- Hospital formulary committees are increasingly reserving budget lines for novel antimicrobial agents with activity against carbapenem-resistant organisms, creating dedicated procurement channels for peptide therapeutics that demonstrate clinical superiority in resistant infection management.
- Regulatory incentives, including 10 to 12 year market exclusivity under QIDP/GAIN Act provisions in the USA and priority review pathways in the EU, improve the commercial return profile for peptide antimicrobials and attract pharmaceutical investment into this historically under-funded therapeutic area.
Key Market Constraints
- High clinical development costs and elevated Phase III failure rates for antimicrobial peptides (estimated at 40 to 50% for novel mechanism agents) create significant financial risk for companies pursuing peptide-based infection therapeutics.
- Peptide stability challenges, including susceptibility to proteolytic degradation in gastrointestinal and systemic environments, limit oral bioavailability and constrain delivery route options, with many peptide antimicrobials restricted to IV administration in hospital settings.
- Limited physician familiarity with peptide-based antimicrobials outside specialist infectious disease settings creates adoption friction, as prescribers in general medicine and surgery may default to conventional antibiotics even when resistance patterns warrant novel agents.
Key Opportunity Areas
- Combination therapy protocols pairing peptide antimicrobials with conventional antibiotics are demonstrating synergistic effects that restore activity against resistant organisms, creating treatment paradigms that position peptides as adjuncts rather than replacements for existing antibiotic portfolios.
- Topical peptide antimicrobials for wound care and dermatological infections represent a high-volume, lower-regulatory-barrier opportunity where peptide stability challenges are reduced and patient access is simplified through outpatient delivery.
- Government-funded pull incentive programmes, including the proposed PASTEUR Act in the USA and the EU Joint Action on Antimicrobial Resistance, are creating guaranteed procurement volumes for novel antimicrobials that would de-risk commercial returns and attract investment into peptide-based development programmes.
Segment-wise Analysis of the Peptide-based Infection Therapeutics Market
- Bacterial-targeted peptides hold 54.0% of the pathogen target segment, reflecting the concentration of clinical AMR burden in bacterial infections.
- Direct antimicrobials account for 35.8% of the modality segment, with host-defence peptides and conjugates comprising the balance.
- Critical care end use represents 48.9% of total demand, driven by high-acuity resistant infections in ICU settings.
Which pathogen target dominates the Peptide-based Infection Therapeutics Market?
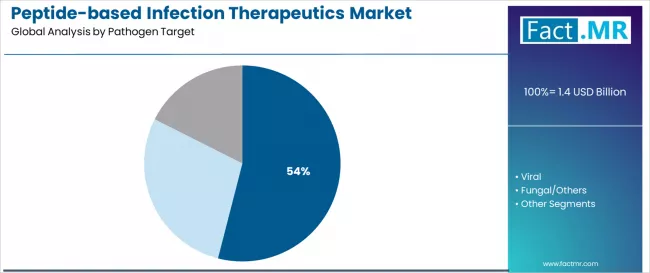
Bacterial-targeted peptides control 54.0% of the pathogen target segment. This dominance reflects the clinical reality that antimicrobial resistance is most acute and commercially consequential in bacterial infections, particularly those caused by ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species). Peptide antimicrobials with activity against these organisms command premium pricing in hospital settings where conventional antibiotics have failed.
Viral-targeted peptides hold a secondary position, with applications in HIV entry inhibition (enfuvirtide) and emerging antiviral peptide candidates for respiratory viruses. Fungal and other pathogen targets represent a smaller but growing niche, driven by rising invasive fungal infection rates in immunocompromised patient populations.
Which modality segment leads the Peptide-based Infection Therapeutics Market?
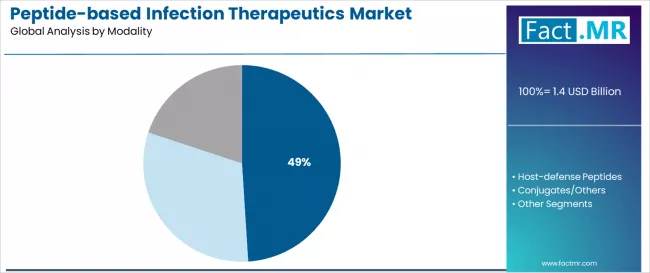
Direct antimicrobial peptides account for 35.8% of the modality segment. These agents act through membrane disruption mechanisms that physically compromise pathogen cell walls, making them less susceptible to the resistance mutations that defeat conventional antibiotics targeting intracellular processes. The rapid bactericidal activity of direct AMPs is clinically valued in sepsis and bacteraemia cases where time-to-pathogen-clearance directly correlates with patient survival.
Host-defence peptides, which modulate the patient's innate immune response to enhance pathogen clearance, hold a significant share with growing clinical interest. Conjugate and hybrid modalities combine peptide targeting with conventional drug payloads, offering selectivity advantages that reduce off-target toxicity.
Which end-use setting captures the largest share in the Peptide-based Infection Therapeutics Market?
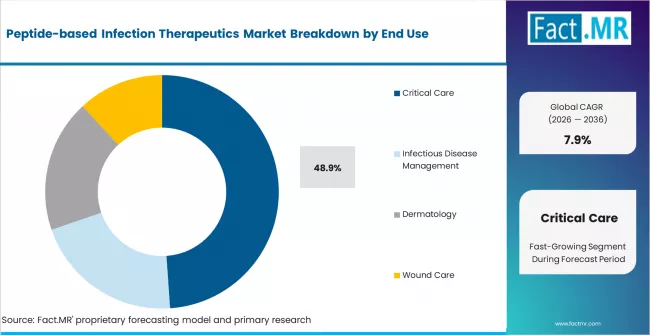
Critical care accounts for 48.9% of end-use demand. ICU patients with ventilator-associated pneumonia, catheter-related bloodstream infections, and surgical site infections caused by multidrug-resistant organisms are the primary clinical population for peptide antimicrobials. The willingness of hospital systems to pay premium prices for agents that reduce ICU length of stay and mortality in resistant infections creates favourable unit economics.
Infectious disease management departments, dermatology, and wound care represent the balance of demand. Wound care applications are growing fastest among non-critical-care end uses, driven by topical peptide formulations for chronic wound infections, diabetic ulcers, and burn wound management.
Which Product Trend is Shaping the Next Phase of Growth in the Peptide-based Infection Therapeutics Market?
The most commercially significant trend in peptide-based infection therapeutics is the development of resistance-resistant antimicrobial peptides that exploit membrane disruption mechanisms. Unlike conventional antibiotics that target specific intracellular enzymes or ribosomal subunits (where single-point mutations can confer resistance), AMPs act through physical disruption of bacterial cell membranes, a mechanism that requires fundamental changes in membrane composition for resistance to develop. Clinical data from peptide programmes targeting MRSA and carbapenem-resistant Enterobacteriaceae show that resistance emergence rates during treatment are 10 to 100 times lower than for conventional last-resort antibiotics such as colistin.
A parallel development is the clinical validation of peptide-antibiotic combination protocols. Companies including Spero Therapeutics and Summit Therapeutics are advancing programmes that pair peptide agents with beta-lactam antibiotics to restore susceptibility in resistant organisms. These combination approaches position peptide therapeutics not as standalone replacements for the existing antibiotic arsenal but as enablers that extend the useful life of approved antibiotics. This strategy reduces the regulatory burden (combination approvals with an established agent) and addresses prescriber familiarity barriers by maintaining conventional antibiotics as the backbone of treatment while adding peptide agents as potentiators. For hospital formulary committees, combination protocols simplify adoption by integrating into existing treatment guidelines rather than requiring entirely new prescribing patterns.
Regional Outlook Across Key Markets
.webp)
- North America holds the largest regional revenue share, anchored by the USD 0.559 billion USA market in 2025.
- East Asia contributes steady demand, with Japan at USD 0.082 billion and South Korea at USD 0.052 billion in 2025.
- Europe maintains strong growth driven by AMR surveillance programmes and antimicrobial stewardship initiatives.
- Latin America is emerging as a growth corridor, with Mexico recording a 7.9% CAGR.
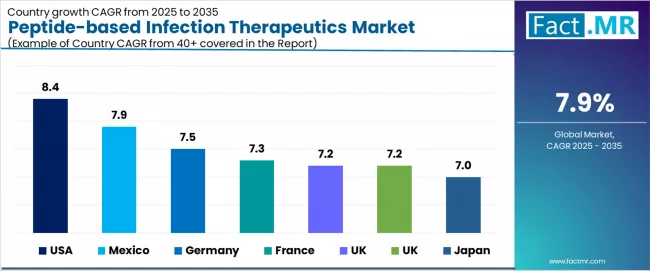
CAGR Table
| Country | CAGR (%) |
|---|---|
| USA | 8.4% |
| Mexico | 7.9% |
| Germany | 7.5% |
| France | 7.3% |
| UK | 7.2% |
| Japan | 7.0% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research.
USA: AMR crisis response and regulatory incentives position the largest market
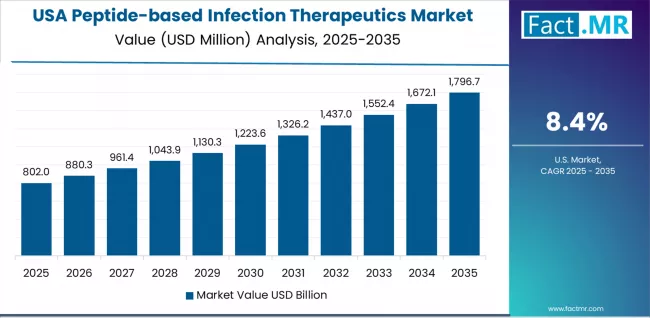
The USA generates USD 0.559 billion in 2025, the largest single-country market, growing at an 8.4% CAGR. Growth is driven by the scale of the AMR burden in US hospitals (over 2.8 million antibiotic-resistant infections annually per CDC data), combined with regulatory pull incentives under the GAIN Act that provide QIDP-designated products with 5 additional years of market exclusivity. Hospital systems are allocating dedicated antimicrobial stewardship budgets for novel agents, and CMS reimbursement changes under the New Technology Add-on Payment (NTAP) programme are improving hospital-level economics for premium-priced peptide antimicrobials.
- The CDC reports that antibiotic-resistant infections cost the US healthcare system over USD 4.6 billion annually in excess hospital costs, creating a strong economic case for novel antimicrobial procurement.
Mexico: Healthcare modernisation and infectious disease burden create demand
Mexico records a 7.9% CAGR, driven by a high infectious disease burden, growing hospital infrastructure investment, and increasing AMR awareness among healthcare providers. Government procurement programmes through the Mexican Institute of Social Security (IMSS) are creating institutional demand for novel antimicrobials, though market access remains constrained by formulary inclusion timelines and pricing negotiations.
- Mexico's national AMR surveillance programme, established in coordination with PAHO, identified carbapenem-resistant infections in over 15% of hospital isolates tested in 2024.
Germany: AMR surveillance leadership and hospital procurement structures support adoption
Germany grows at 7.5% CAGR, supported by one of Europe's most developed AMR surveillance systems (the German Antibiotic Resistance Surveillance programme, ARS) and structured hospital procurement processes that evaluate novel antimicrobials on clinical efficacy rather than cost alone. University hospitals in Berlin, Munich, and Hamburg serve as early adoption centres for peptide antimicrobials, with infectious disease departments conducting post-marketing studies that inform broader formulary inclusion.
- Germany's Robert Koch Institute reported that MRSA rates in hospital settings declined to 8.5% in 2024, though carbapenem-resistant gram-negative infections increased by 12%, shifting demand toward novel antimicrobial mechanisms.
France: National antibiotic stewardship and research investment drive growth
France records a 7.3% CAGR, reflecting the national Plan National Antibiotiques programme that directs hospital procurement toward novel antimicrobial agents for resistant infections. French research institutions, including INSERM and Institut Pasteur, maintain active peptide antimicrobial research programmes that feed into clinical development pipelines, creating a domestic innovation ecosystem.
- France allocated EUR 40 million in 2024 to antimicrobial resistance research under the national Plan d'Investissement d'Avenir, supporting peptide antimicrobial clinical development.
UK: NHS antimicrobial subscription model creates a procurement template
The UK grows at 7.2% CAGR, distinguished by the NHS subscription-based antimicrobial procurement model that pays manufacturers a fixed annual fee independent of unit volume. This model directly addresses the commercial viability challenge for novel antimicrobials (where low unit volumes at launch typically produce inadequate returns) and creates a predictable revenue stream for peptide antimicrobial developers entering the UK market.
- The NHS England subscription model for novel antimicrobials, piloted from 2022, committed GBP 10 million annually per product for two initial antimicrobial agents, establishing a replicable pull incentive framework.
Japan: Ageing population and hospital infection control priorities sustain demand
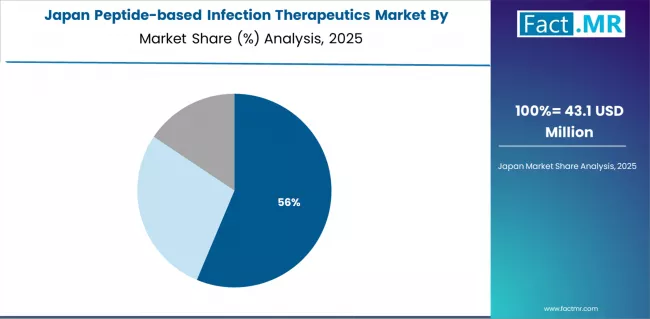
Japan contributes USD 0.082 billion in 2025, growing at 7.0% CAGR. An ageing population with high rates of hospitalisation and surgical procedures creates elevated exposure to hospital-acquired resistant infections. Japan's Pharmaceutical and Medical Devices Agency (PMDA) has established expedited review pathways for novel antimicrobials addressing unmet resistance needs, reducing time-to-market for approved peptide therapeutics.
- Japan's AMR Action Plan 2023 to 2027 set a target to reduce antimicrobial use in human medicine by 20% while increasing adoption of novel antimicrobials for resistant infections.
Competitive Benchmarking and Company Positioning
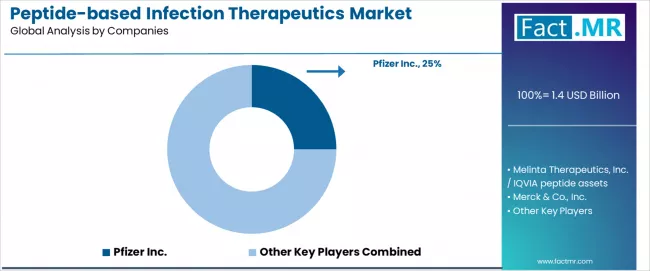
Peptide-based Infection Therapeutics Market Analysis By Company
- Pfizer Inc. holds approximately 25.0% of global revenue through its portfolio of approved peptide-based and peptide-derived antimicrobial agents, supported by hospital sales infrastructure and infectious disease specialist relationships.
- Merck and Co., Roche Holding AG, and Basilea Pharmaceutica collectively hold significant positions, competing on pipeline depth, clinical evidence, and geographic coverage.
- Specialised antimicrobial companies including Spero Therapeutics, Destiny Pharma, and Summit Therapeutics operate as clinical-stage or early-commercial players targeting specific resistance mechanisms.
The competitive structure is moderately concentrated, with Pfizer commanding the largest single-company share at 25.0% through its broad anti-infective portfolio that includes peptide-derived agents for resistant bacterial infections. Pfizer's hospital sales force and established relationships with infectious disease departments provide distribution advantages that smaller companies cannot replicate.
Merck and Co. competes through its anti-infective franchise, including agents with activity against multidrug-resistant gram-negative organisms. Roche Holding AG brings oncology-derived peptide conjugate expertise to infection therapeutics, exploring dual-targeting approaches. Basilea Pharmaceutica International holds a focused position in hospital-based anti-infective therapeutics, with a portfolio designed specifically for resistant infections.
The clinical-stage competitive layer is populated by specialised companies: Spero Therapeutics (potentiator-based approaches), Destiny Pharma (XF drug platform for biofilm-associated infections), and Summit Therapeutics (ridinilazole for C. difficile). These companies represent potential disruptive entrants if their clinical programmes achieve approval, though pipeline attrition risk remains elevated.
Key Companies in the Peptide-based Infection Therapeutics Market
The peptide-based infection therapeutics competitive field includes Pfizer Inc., Merck and Co. Inc., Roche Holding AG, Polyphor Ltd. (legacy assets), Spero Therapeutics Inc., Destiny Pharma plc, Summit Therapeutics plc, ContraFect Corporation, Basilea Pharmaceutica International Ltd., and Melinta Therapeutics Inc.
Recent Industry Developments
- Summit Therapeutics: ridinilazole Phase III completion (2025)
- Summit Therapeutics completed Phase III trials for ridinilazole, a targeted-spectrum peptide antimicrobial for Clostridioides difficile infection, reporting superior sustained clinical response rates compared to vancomycin and filing for regulatory approval in the USA and EU.
- Spero Therapeutics: tebipenem HBr FDA review (2025)
- Spero Therapeutics received an FDA Complete Response Letter for tebipenem pivoxil hydrobromide, the first oral carbapenem, and initiated additional clinical work to address regulatory queries for treatment of complicated urinary tract infections caused by resistant gram-negative organisms.
- Destiny Pharma: XF-73 Phase II results in surgical site infection prevention (2025)
- Destiny Pharma reported positive Phase II results for XF-73, a dicationic porphyrin-based antimicrobial, in prevention of post-surgical staphylococcal nasal carriage and surgical site infections, advancing toward Phase III planning.
- Pfizer Inc.: novel peptide antimicrobial portfolio expansion (2026)
- Pfizer expanded its anti-infective pipeline through licensing agreements for two late-stage peptide antimicrobial candidates targeting carbapenem-resistant Acinetobacter baumannii, strengthening its portfolio against WHO critical-priority pathogens.
Leading Companies Shaping the Peptide-based Infection Therapeutics Market
-
Global Players
- Pfizer Inc.
- Merck and Co., Inc.
- Roche Holding AG
- Basilea Pharmaceutica International Ltd.
- Melinta Therapeutics, Inc.
-
Regional Players
- Summit Therapeutics plc
- Spero Therapeutics, Inc.
- ContraFect Corporation
-
Emerging / Start-up Players
- Destiny Pharma plc
- Polyphor Ltd. (legacy assets)
- Cidara Therapeutics Inc.
Sources and Research References
- U.S. Centers for Disease Control and Prevention (CDC) Antibiotic Resistance Threats report
- World Health Organization (WHO) Global Antimicrobial Resistance and Use Surveillance System (GLASS) reports
- U.S. FDA QIDP designation and GAIN Act approval records
- European Medicines Agency (EMA) adaptive pathway and accelerated assessment records
- NHS England antimicrobial subscription model programme documentation
- ClinicalTrials.gov registry for peptide antimicrobial clinical programmes
- Robert Koch Institute German Antibiotic Resistance Surveillance (ARS) data
All numerical data in this report is derived from Fact.MR proprietary forecasting models, validated through primary research interviews and cross-referenced with the public-domain sources listed above. No data from syndicated market research publishers has been used.
Key Questions This Report Addresses
- What is the projected size of the peptide-based infection therapeutics market by 2036?
- Which pathogen target holds the largest share of the market?
- What CAGR is forecast for the peptide-based infection therapeutics market between 2026 and 2036?
- Which modality segment generates the most revenue?
- Which country is expected to grow fastest in the market?
- Who is the leading company in the global peptide-based infection therapeutics market?
- What is the incremental opportunity between 2026 and 2036?
- How is antimicrobial resistance affecting demand for peptide therapeutics?
- What regulatory incentives support peptide antimicrobial development?
Peptide-based Infection Therapeutics Market Definition
The peptide-based infection therapeutics market encompasses the development, regulatory approval, manufacture, and commercialisation of peptide-derived pharmaceutical products designed to treat infectious diseases caused by bacterial, viral, and fungal pathogens. Products in scope include natural and synthetic antimicrobial peptides, host-defence peptide modulators, and peptide-drug conjugates delivered via intravenous, oral, and topical routes. The market covers approved commercial products and late-stage pipeline candidates with near-term commercialisation potential.
Peptide-based Infection Therapeutics Market Inclusions
- All approved and late-stage (Phase III) peptide-based therapeutics targeting bacterial, viral, and fungal infections.
- Revenue from hospital IV formulations, outpatient oral products, and topical peptide antimicrobials sold through institutional and pharmacy channels.
- Products used in critical care, infectious disease management, dermatology, and wound care settings.
- Both direct antimicrobial peptides and host-defence peptide modulators that enhance innate immune response to infection.
Peptide-based Infection Therapeutics Market Exclusions
- Peptide-based vaccines and immunomodulators not primarily indicated for active infection treatment.
- Small-molecule antibiotics and antifungals that do not contain a peptide-based active pharmaceutical ingredient.
- Diagnostic peptide reagents and biomarkers used for infection detection rather than treatment.
- Preclinical and Phase I peptide antimicrobial candidates without near-term commercial viability.
Peptide-based Infection Therapeutics Market Research Methodology
- Primary research with infectious disease physicians, hospital pharmacists, and antimicrobial stewardship programme directors across 8 countries.
- Revenue modelling based on hospital procurement data, national formulary listings, and manufacturer-reported sales from annual filings.
- Pricing analysis using hospital group purchasing organisation (GPO) contract databases and national health system reimbursement schedules.
- Pipeline assessment through ClinicalTrials.gov registry data, FDA/EMA approval timelines, and QIDP designation tracking.
- Competitive share estimation through company annual reports, investor presentations, and SEC filings for publicly traded companies.
- Forecast calibration against AMR prevalence trends, hospital-acquired infection surveillance data, and antimicrobial stewardship programme budget allocations.
Scope of Analysis
| Parameter | Details |
|---|---|
| Report Coverage | Global market analysis with country-level forecasts for 6 countries and 40+ additional markets |
| Base Year | 2025 |
| Forecast Period | 2026 to 2036 |
| Market Size (2025) | USD 1.4 Billion |
| Units | USD Billion |
| Segments Covered | Pathogen Target (Bacterial, Viral, Fungal/Others), Modality (Direct Antimicrobials, Host-defense Peptides, Conjugates/Others), Setting (Hospital IV, Outpatient Oral, Topical), End Use (Critical Care, Infectious Disease Management, Dermatology, Wound Care) |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Profiled | USA, Mexico, Germany, France, UK, Japan |
| Companies Profiled | Pfizer, Merck, Roche, Polyphor (legacy), Spero Therapeutics, Destiny Pharma, Summit Therapeutics, ContraFect, Basilea Pharmaceutica, Melinta Therapeutics |
| Forecast Methodology | Bottom-up revenue modelling validated through primary research, company filings, and clinical trial registry data |
Market Segmentation Analysis
-
Peptide-based Infection Therapeutics Market Market Segmented by Pathogen Target:
- Bacterial
- Cell Wall Targeting Peptides
- Membrane Disruption Peptides
- Biofilm Disrupting Peptides
- Viral
- Viral Entry Inhibitors
- Viral Replication Inhibitors
- Envelope Disruption Peptides
- Fungal/Others
- Cell Membrane Targeting Peptides
- Antifungal Enzyme Inhibitors
- Broad Spectrum Antimicrobial Peptides
- Bacterial
-
Peptide-based Infection Therapeutics Market Market Segmented by Modality:
- Direct Antimicrobials
- Lytic Peptides
- Membrane Permeabilizing Peptides
- Broad Spectrum Synthetic Peptides
- Host-defense Peptides
- Cationic Antimicrobial Peptides
- Immunomodulatory Peptides
- Neutrophil Derived Peptides
- Conjugates/Others
- Peptide Drug Conjugates
- Nanoparticle Conjugated Peptides
- Stabilized Peptide Analogues
- Direct Antimicrobials
-
Peptide-based Infection Therapeutics Market Market Segmented by Setting:
- Hospital IV
- Inpatient Infusion Therapy
- Intensive Care Administration
- Emergency Infection Treatment
- Outpatient Oral
- Oral Peptide Formulations
- Self Administered Therapies
- Chronic Infection Management
- Topical
- Dermal Creams
- Wound Sprays
- Burn Care Applications
- Hospital IV
-
Peptide-based Infection Therapeutics Market Market Segmented by End Use:
- Critical Care
- Sepsis Management
- Multidrug Resistant Infections
- Post Surgical Infections
- Infectious Disease Management
- Respiratory Infections
- Bloodstream Infections
- Systemic Viral Infections
- Dermatology
- Skin Infection Treatment
- Acne Related Bacterial Therapy
- Fungal Skin Conditions
- Wound Care
- Chronic Wounds
- Diabetic Ulcers
- Surgical Wound Healing
- Critical Care
-
Peptide-based Infection Therapeutics Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
What is the current size of the peptide-based infection therapeutics market?
The market is valued at USD 1.4 billion in 2025.
What CAGR is expected for the peptide-based infection therapeutics market?
The market is forecast to grow at a 7.9% CAGR from 2026 to 2036.
Which pathogen target leads the market?
Bacterial-targeted peptides hold a 54.0% share of the pathogen target segment.
Which end-use setting is largest?
Critical care accounts for 48.9% of total market demand.
Which country will grow fastest?
The USA is projected to grow at an 8.4% CAGR through 2036.
Who is the leading company?
Pfizer Inc. holds an estimated 25.0% global revenue share.
What is the forecast value by 2036?
The market is projected to reach USD 3.231 billion by 2036.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Pathogen Target
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Pathogen Target , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Pathogen Target , 2026 to 2036
- Bacterial
- Cell Wall Targeting Peptides
- Membrane Disruption Peptides
- Biofilm Disrupting Peptides
- Viral
- Viral Entry Inhibitors
- Viral Replication Inhibitors
- Envelope Disruption Peptides
- Fungal/Others
- Cell Membrane Targeting Peptides
- Antifungal Enzyme Inhibitors
- Broad Spectrum Antimicrobial Peptides
- Bacterial
- Y to o to Y Growth Trend Analysis By Pathogen Target , 2021 to 2025
- Absolute $ Opportunity Analysis By Pathogen Target , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Modality
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Modality, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Modality, 2026 to 2036
- Direct Antimicrobials
- Lytic Peptides
- Membrane Permeabilizing Peptides
- Broad Spectrum Synthetic Peptides
- Host-defense Peptides
- Cationic Antimicrobial Peptides
- Immunomodulatory Peptides
- Neutrophil Derived Peptides
- Conjugates/Others
- Peptide Drug Conjugates
- Nanoparticle Conjugated Peptides
- Stabilized Peptide Analogues
- Direct Antimicrobials
- Y to o to Y Growth Trend Analysis By Modality, 2021 to 2025
- Absolute $ Opportunity Analysis By Modality, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Setting
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Setting, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Setting, 2026 to 2036
- Hospital IV
- Inpatient Infusion Therapy
- Intensive Care Administration
- Emergency Infection Treatment
- Outpatient Oral
- Oral Peptide Formulations
- Self Administered Therapies
- Chronic Infection Management
- Topical
- Dermal Creams
- Wound Sprays
- Burn Care Applications
- Hospital IV
- Y to o to Y Growth Trend Analysis By Setting, 2021 to 2025
- Absolute $ Opportunity Analysis By Setting, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Critical Care
- Sepsis Management
- Multidrug Resistant Infections
- Post Surgical Infections
- Infectious Disease Management
- Respiratory Infections
- Bloodstream Infections
- Systemic Viral Infections
- Dermatology
- Skin Infection Treatment
- Acne Related Bacterial Therapy
- Fungal Skin Conditions
- Wound Care
- Chronic Wounds
- Diabetic Ulcers
- Surgical Wound Healing
- Critical Care
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Pathogen Target
- By Modality
- By Setting
- By End Use
- Competition Analysis
- Competition Deep Dive
- Pfizer Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Merck & Co., Inc.
- Roche Holding AG
- Polyphor Ltd. (legacy assets)
- Spero Therapeutics, Inc.
- Destiny Pharma plc
- Summit Therapeutics plc
- ContraFect Corporation
- Basilea Pharmaceutica International Ltd.
- Melinta Therapeutics, Inc. / IQVIA peptide assets
- Pfizer Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Pathogen Target , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Pathogen Target , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Pathogen Target , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Pathogen Target , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Pathogen Target , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Pathogen Target , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Pathogen Target , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Pathogen Target , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Modality, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Pathogen Target, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Pathogen Target,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Pathogen Target
- Figure 6: Global Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Modality,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Modality
- Figure 9: Global Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Setting,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Setting
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 19: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 22: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Pathogen Target, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Pathogen Target,2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Pathogen Target
- Figure 29: North America Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Modality,2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Modality
- Figure 32: North America Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Setting,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Setting
- Figure 35: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by End Use
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Pathogen Target, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Pathogen Target,2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by Pathogen Target
- Figure 42: Latin America Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Modality,2026 to 2036
- Figure 44: Latin America Market Attractiveness Analysis by Modality
- Figure 45: Latin America Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Setting,2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Setting
- Figure 48: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by End Use
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Pathogen Target, 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Pathogen Target,2026 to 2036
- Figure 54: Western Europe Market Attractiveness Analysis by Pathogen Target
- Figure 55: Western Europe Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Modality,2026 to 2036
- Figure 57: Western Europe Market Attractiveness Analysis by Modality
- Figure 58: Western Europe Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Setting,2026 to 2036
- Figure 60: Western Europe Market Attractiveness Analysis by Setting
- Figure 61: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by End Use
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Pathogen Target, 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Pathogen Target,2026 to 2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Pathogen Target
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Modality,2026 to 2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Modality
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Setting,2026 to 2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Setting
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Pathogen Target, 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Pathogen Target,2026 to 2036
- Figure 80: East Asia Market Attractiveness Analysis by Pathogen Target
- Figure 81: East Asia Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Modality,2026 to 2036
- Figure 83: East Asia Market Attractiveness Analysis by Modality
- Figure 84: East Asia Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Setting,2026 to 2036
- Figure 86: East Asia Market Attractiveness Analysis by Setting
- Figure 87: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 89: East Asia Market Attractiveness Analysis by End Use
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Pathogen Target, 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Pathogen Target,2026 to 2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Pathogen Target
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Modality,2026 to 2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Modality
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Setting,2026 to 2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Setting
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Pathogen Target, 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Pathogen Target,2026 to 2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Pathogen Target
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Modality, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Modality,2026 to 2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Modality
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Setting,2026 to 2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Setting
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis