Drug Eluting Balloon Catheter Market Size, Share, Growth and Forecast (2025 - 2035)
Drug Eluting Balloon Catheter Market Size and Share Forecast Outlook 2025 to 2035
The Drug Eluting Blood Catheter Market Will Total USD 1.8 Billion In 2025, Forecasted To Rise To USD 9.4 Billion By 2035, Registering A CAGR Of 18.0%. By Indication, Peripheral Vascular Disease Remains Dominant While By End User, Ambulatory Surgical Centers Will Be The Dominant Segment
Drug Eluting Balloon Catheter Market Outlook 2025 to 2035
The global drug eluting balloon catheter market is forecast to reach USD 9.4 billion by 2035, up from USD 1.8 billion in 2025. During the forecast period, the industry is projected to register at a CAGR of 18.0%.
The drug-coated balloon catheters are also known as drug-eluted balloon catheters which are designed to dilate the stenotic coronary artery and used in the treatment of in-stent restenosis (ISR).
Quick Stats on Drug Eluting Balloon Catheter Market
- Drug Eluting Balloon Catheter Market Size (2025):USD 1.8 billion
- Projected Drug Eluting Balloon Catheter Market Size (2035):USD 9.4 billion
- Forecast CAGR of Drug Eluting Balloon Catheter Market (2025 to 2035):18.0%
- Leading Indication Segment of Drug Eluting Balloon Catheter Market:Peripheral Vascular Disease
- Leading End User Segment of Drug Eluting Balloon Catheter Market: Ambulatory Surgical Centers
- Key Growth Regions of Drug Eluting Balloon Catheter Market:United States, China, Japan
- Prominent Players in the Drug Eluting Balloon Catheter Market: Cardionovum GmbH, Cook Medical Inc., Cordis Corporation, Abbott Laboratories, Meril Life Sciences Pvt Ltd., Others
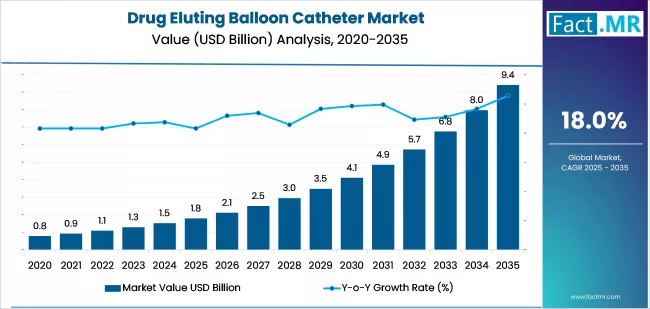
| Metric | Value |
|---|---|
| Industry Size (2025E) | USD 1.8 billion |
| Industry Size (2035F) | USD 9.4 billion |
| CAGR (2025-2035) | 18.0% |
The global drug eluting balloon catheter market is positioned for significant expansion, projected to surge from USD 1.8 billion in 2025 to USD 9.4 billion by 2035, at a CAGR of 18%.
This trajectory highlights the strong preference for minimally invasive cardiovascular solutions, as these devices integrate drug delivery with angioplasty procedures, effectively reducing restenosis rates and minimizing the need for repeat interventions.
Drug eluting balloon catheters are being increasingly recognized in interventional cardiology, not only as an alternative to stenting but also as a complementary therapy in complex vascular cases.
Clinical adoption is expected to intensify as physicians favor devices that balance procedural efficiency with improved patient outcomes. The growing incidence of peripheral artery disease, coupled with rising cases of coronary artery blockages, is creating a sizable patient pool that benefits from reduced vessel trauma and shorter recovery times.
Drug eluting balloon catheters are also gaining acceptance in patient subsets that are not well-suited for stents, such as those with small vessel disease or bifurcation lesions, widening their addressable base.
Advances in polymer coatings, controlled drug release mechanisms, and compatibility with multiple drug formulations are reinforcing product differentiation across suppliers.
Reimbursement improvements in key markets and expanding clinical guidelines recommending their use are strengthening the regulatory and commercial environment. Regional momentum is also shaped by infrastructure readiness.
North America remains the early adopter due to advanced healthcare systems, while Europe leads in clinical trial volume and evidence-based expansion. Asia Pacific is emerging as a high-growth territory, driven by large untreated patient populations, expanding catheter labs, and accelerated regulatory clearances.
Competitive intensity is rising as major medtech firms integrate drug eluting balloon technology into broader cardiovascular portfolios. Mergers, collaborations with hospitals for real-world evidence, and expanded manufacturing capabilities are reshaping the strategic landscape.
Going forward, the most influential trends will include heightened focus on reducing restenosis rates, development of catheters with enhanced drug delivery precision, and strategic penetration into emerging economies where patient volumes are climbing rapidly.
Analyzing Key Dynamics of the Global Drug Eluting Balloon Catheter Market
The drug eluting balloon catheter market is influenced by a mix of drivers that accelerate adoption and challenges that restrict expansion. While clinical evidence and technological innovations are fostering growth, regulatory hurdles and safety concerns continue to weigh on the long-term trajectory. Understanding both sides provides a balanced view of how the sector will evolve over the coming years.
Rising Demand for Minimally Invasive Procedures and Aging Population
The rise in demand for minimally invasive procedures has created a strong foundation for the adoption of drug eluting balloon catheters. These devices are increasingly used in treating coronary atherosclerosis and reducing restenosis rates, particularly in patients with complex vascular conditions. Their ability to deliver targeted therapy without leaving behind permanent implants has made them highly attractive for clinical use.
The growing older population has further influenced the market, as elderly individuals are more prone to cardiovascular and peripheral artery diseases. This demographic is benefiting significantly from drug-coated and drug-eluting balloons, which offer corrective solutions for blocked vessels and reduce the need for repeat interventions. The combination of an expanding geriatric base and preference for non-invasive techniques continues to drive uptake worldwide.
Technological Advancements and Clinical Evidence
Technological progress has been a major accelerator, with manufacturers investing in advanced coatings, drug formulations, and delivery mechanisms. Cardiovascular Systems, for instance, introduced improved balloon catheters for complex lesions, while Terumo launched trials for next-generation guide catheters. Such innovations enhance procedural outcomes and increase confidence among physicians.
Clinical data is reinforcing adoption, with randomized trials showing drug eluting balloons reduce target lesion failure compared to uncoated balloons. Evidence from small vessel disease and BTK (below-the-knee) applications has highlighted benefits, while sirolimus-based platforms with micro-reservoir technologies have emerged as promising alternatives to paclitaxel. This clinical validation has opened opportunities in new indications and broadened physician acceptance.
Safety Concerns and Mortality Risks
Despite clinical progress, safety concerns pose a significant restraint. Several studies have linked paclitaxel-coated balloons to late mortality in femoropopliteal interventions. Reports in the American Heart Association Journal highlighted deaths within one to five years post-treatment, creating hesitation in both clinical and regulatory circles.
Companies themselves have acknowledged risks, as seen with Medtronic’s warnings over late mortality for its IN.PACT AV product. These safety concerns have cast doubt on the long-term viability of paclitaxel platforms, pushing the industry toward sirolimus alternatives but slowing overall adoption rates.
Regulatory Barriers and Limited Approvals
Stringent regulatory frameworks also restrict growth. While Europe has embraced drug eluting balloons with established approvals, the United States remains a challenging market, with no coronary indication approvals currently granted. This divergence has created a geographical imbalance, limiting commercial expansion in North America.
Approval delays, coupled with lengthy commercialization pathways, continue to deter rapid uptake. Without broader regulatory acceptance, particularly in large healthcare markets like the U.S., the global potential of drug eluting balloon catheters remains partly unrealized, tempering otherwise strong growth fundamentals.
Regional Trends of the Drug Eluting Balloon Catheter Market
North America accounted for the largest share of the market in 2025. The significant market share in North America can be attributed to several factors, the availability of various major manufacturers, the launch of new, technologically advanced devices, and favorable reimbursement regulations are all important factors.
The American Heart Association estimates that over 500,000 percutaneous coronary procedures, including percutaneous transluminal coronary interventions, are carried out annually in the US. Additionally, rising government expenditure on health care in the U.S. is promoting market expansion.
Asia Pacific is anticipated to have rapid expansion. This can be ascribed to the rise in healthcare expenditure as well as an increase in the incidence of cardiovascular disorders. As per Biomedcentral, cardiovascular disease affects approximately one out of every five individuals in China.
Demand will be driven by an aging population in countries like China & Japan and rising disposable income. The expansion of the industry in the area is also being aided by the rise of health tourism in emerging countries.
Country-Wise Outlook
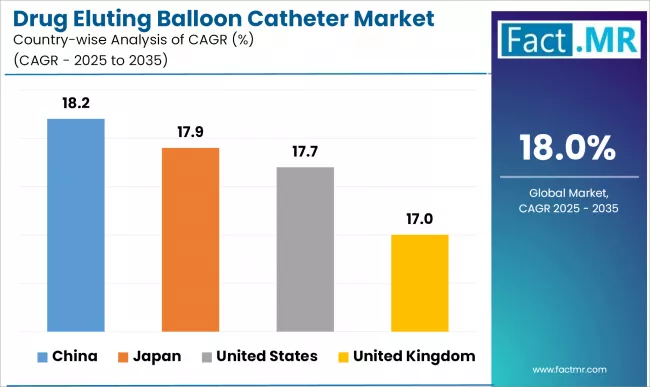
| Countries | CAGR (2025-2035) |
|---|---|
| United States | 17.7% |
| China | 18.2% |
| Japan | 17.9% |
United States Drug Eluting Balloon Catheter Market’s Growth Driven by Technological Advancements in Surgical Procedures
The United States drug eluting balloon catheter market is expanding on the back of high cardiovascular disease prevalence, covering coronary artery and peripheral artery conditions. A growing preference for minimally invasive treatments among patients and clinicians supports adoption, given shorter recovery times and fewer complications compared to traditional methods.
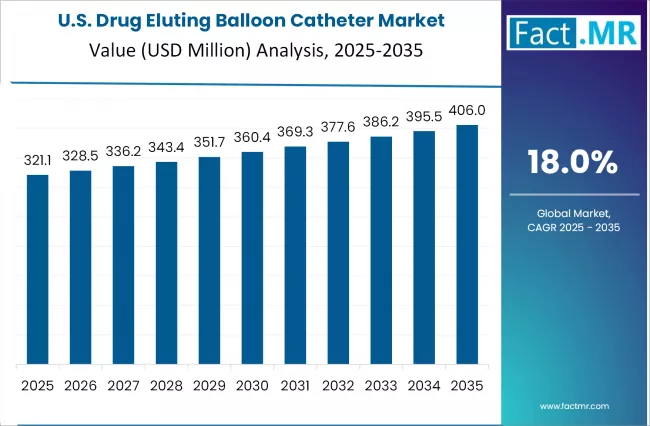
Technological progress in catheter design and drug formulations has made treatments more effective and reliable. Notable product launches such as BrosMed’s VaSecure Paclitaxel-coated PTA Balloon Catheter, featuring a protective sheath and micro-needle crystal coating, highlight ongoing innovation. These advances improve drug retention and transfer efficiency, addressing unmet needs in restenosis and small vessel disease while broadening therapeutic options.
- Rising cardiovascular disease burden increases treatment adoption
- Innovations like micro-needle coating enhance drug retention and outcomes
- Minimally invasive preference fuels faster uptake of advanced devices
Increasing Healthcare Investment Initiatives to Encourage Drug Eluting Balloon Catheter Demand in China
China is witnessing rapid uptake of drug eluting balloon catheters due to a rising cardiovascular disease burden and expanding hospital infrastructure. A clear shift toward minimally invasive solutions has aligned with government healthcare spending and regulatory approvals. Clinical adoption is accelerating as physicians favor technologies with strong safety and efficacy records.
A pivotal development came in 2025 with NMPA approval of MicroPort’s Firelimus Rapamycin Drug-Eluting Balloon Catheter, which treats primary coronary bifurcation lesions.
Firelimus uses an excipient-free microcrystalline coating for controlled drug release, reducing inflammation risks and improving safety. The broad sizing range also allows it to address varied patient anatomies, reinforcing MicroPort’s positioning in stent-free interventions.
- Strong healthcare investments accelerate procedural adoption
- NMPA approvals boost confidence in domestic innovations
- Excipient-free rapamycin devices reduce inflammation and thrombosis risks
Increase in Geriatric Demographics to Elevate Drug Eluting Balloon Catheter Insertion Procedures in Japan
Japan’s drug eluting balloon catheter market is shaped by its rapidly aging population, which faces high rates of coronary and peripheral artery disease. This demographic factor is creating sustained demand for effective interventional treatments. Minimally invasive procedures are favored due to quicker recovery and lower complication rates, further encouraging use.
Advances in balloon technology, including low-profile designs and optimized drug release systems, enhance physician efficiency and broaden access to complex lesions.
Thes catheters are increasingly delivering antiproliferative drugs such as paclitaxel directly to arterial walls, lowering restenosis rates while improving patient safety. Efforts to improve catheter flexibility and biocompatibility also contribute to wider acceptance across clinical settings.
- Aging population drives demand for advanced vascular solutions
- Low-profile balloon designs improve procedural access to complex lesions
- Drug delivery technologies enhance restenosis prevention and safety
Category-wise Analysis
The drug eluting balloon catheter market demonstrates differentiated growth across disease indications and care delivery settings. Peripheral vascular applications continue to dominate due to early clinical adoption, while ambulatory surgical centers are emerging as the fastest-growing end-use environment.
Peripheral Vascular Indications Driving Adoption
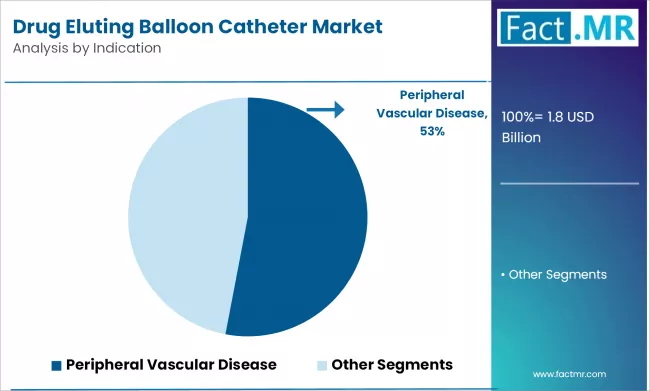
Peripheral drug eluting balloons hold the largest market share, supported by strong evidence in femoropopliteal and below-the-knee interventions. These anatomical regions often resist stenting due to vessel flexion, external compression, and calcification, making balloons the preferred choice. Trials such as SELUTION SFA Japan, with an 87.9 percent patency rate at 12 months, underscore the ability of drug-coated balloons to sustain durable outcomes in complex lesions.
The installed base remains weighted toward peripheral usage since paclitaxel devices were first introduced in this segment, creating clinician familiarity and procedural confidence.
However, coronary applications are gaining traction as evidence expands in de-novo and small-vessel disease. With reimbursement barriers expected to ease, coronary procedures could narrow the revenue gap, though peripheral demand will remain resilient over the forecast horizon.
- Peripheral vascular disease remains the largest treatment segment
- Clinical data validates efficacy in femoropopliteal and BTK lesions
- Coronary pipeline expected to close the revenue gap gradually
Ambulatory Surgical Centers Leading End Use Expansion
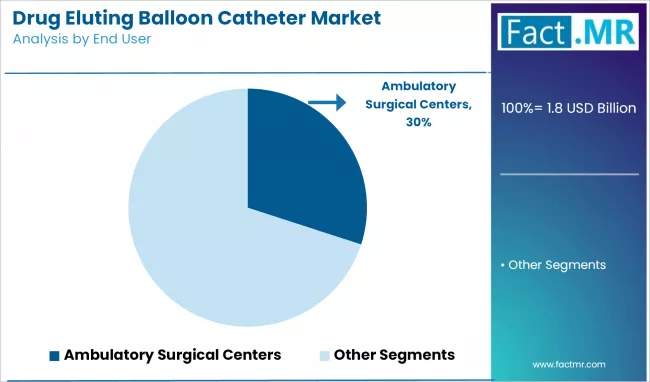
Ambulatory surgical centers are becoming the fastest-growing channel for drug eluting balloon catheter procedures. These centers benefit from lower overhead costs, streamlined workflows, and growing payer support for outpatient cardiovascular care. The shift is reinforced by same-day discharge protocols, which are more feasible with balloons since no permanent implants are left behind, reducing monitoring requirements.
Clinical evidence adds confidence: a Medicare study in 2025 reported similar 30-day outcomes for percutaneous coronary interventions in ASCs compared to hospital outpatient departments. By 2023, ASCs already accounted for 1.8 percent of procedures, marking consistent expansion.
Financially, ASCs sustain better margins despite lower reimbursement, as operational costs remain significantly lower than hospitals. Drug eluting balloons are well-suited to this setting, accelerating the migration of interventional cardiology toward cost-efficient outpatient models.
- ASCs register the highest growth rate through 2035
- Same-day discharge protocols favor balloon catheter adoption
- Cost efficiencies strengthen ASC competitiveness against hospitals
Competitive Analysis
The drug eluting balloon market exhibits moderate consolidation. Medtronic, Boston Scientific, and Koninklijke Philips N.V. leverage integrated cardiovascular portfolios, strong distributor ties, and deep data sets to anchor share. Boston Scientific’s TransPac technology underpins the AGENT coronary balloon, reinforcing its coronary franchise leadership.
Cordis’ USD 1.1 billion buyout of MedAlliance introduces MicroReservoir sirolimus delivery and signals renewed competitive intensity from mid-sized strategic buyers. Teleflex’s 2025 acquisition of BIOTRONIK’s vascular assets expands its peripheral toolkit, highlighting ongoing inorganic expansion as a route to differentiation.
Emerging firms including Concept Medical, Cardionovum, and iVascular focus on next-generation excipient chemistries and dual-drug payloads aimed at restenosis-prone segments. Intellectual-property depth around coating polymers increasingly dictates partnering and licensing negotiations, underscoring R&D’s central role in shaping future drug eluting balloon market share contests.
Key Players in the Market
- Cardionovum GmbH
- Cook Medical Inc.
- Cordis Corporation
- Abbott Laboratories
- Meril Life Sciences Pvt Ltd
- Jotech GmbH
- MicroPort Scientific Corporation
- Terumo Corporation
- Boston Scientific Corporation
Recent Developments
- In May 2025, Cordis launched the SELUTION Global Coronary Registry, which is a prospective study designed to enroll up to 10,000 patients worldwide. This registry is one of the largest of its kind for coronary drug-eluting balloons and will track real-world outcomes of the SELUTION SLR sirolimus drug-eluting balloon for up to five years.
- The registry aims to complement ongoing randomized trials and enhance clinical evidence for the technology. The SELUTION SLR balloon is notable for sustained sirolimus release without a permanent implant, preserving future treatment options.
- In February 2025, MicroPort obtained Chinese regulatory approval for its Firelimus rapamycin drug-eluting balloon, targeting primary coronary bifurcation lesions. This marks an important expansion in the availability of drug-coated balloon options in China.
- In March 2024, Boston Scientific received US FDA clearance for its AGENT drug-coated balloon, designed to treat coronary in-stent restenosis, expanding treatment options for this condition.
Segmentation of Drug Eluting Balloon Catheter Market
-
By Indication :
- Coronary Artery Disease
- Peripheral Vascular Disease
-
By End User :
- Hospitals
- Ambulatory Surgical Centers
- Cardiac Catheterization Laboratories
-
By Raw Material :
- Polyurethane
- Nylon
- Others
-
By Region :
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
- Frequently Asked Questions -
What is the global drug eluting balloon catheter market size in 2025?
The drug eluting balloon catheter market is valued at USD 1.8 billion in 2025.
Who are the major players operating in the drug eluting balloon catheter market?
Prominent players in the market include Cardionovum GmbH, Cook Medical INC, Cordis Corporation, Abbott Laboratories, Meril Life Sciences Pvt Ltd, and Other.
What is the estimated valuation of the drug eluting balloon catheter market by 2035?
The market is expected to reach a valuation of USD 9.4 billion by 2035.
At what CAGR is the drug eluting balloon catheter market slated to grow during the study period?
The growth rate of the drug eluting balloon catheter market is 18.0% from 2025-2035.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Bn) & Units Analysis, 2020-2024
- Current and Future Market Size Value (USD Bn) & Units Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020-2024 and Forecast 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Bn) & Units Analysis By Indication, 2020-2024
- Current and Future Market Size Value (USD Bn) & Units Analysis and Forecast By Indication, 2025-2035
- Coronary Artery Disease
- Peripheral Vascular Disease
- Y-o-Y Growth Trend Analysis By Indication, 2020-2024
- Absolute $ Opportunity Analysis By Indication, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Bn) & Units Analysis By End User, 2020-2024
- Current and Future Market Size Value (USD Bn) & Units Analysis and Forecast By End User, 2025-2035
- Hospitals
- Ambulatory Surgical Centers
- Cardiac Catheterization Laboratories
- Y-o-Y Growth Trend Analysis By End User, 2020-2024
- Absolute $ Opportunity Analysis By End User, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Raw Material
- Introduction / Key Findings
- Historical Market Size Value (USD Bn) & Units Analysis By Raw Material, 2020-2024
- Current and Future Market Size Value (USD Bn) & Units Analysis and Forecast By Raw Material, 2025-2035
- Polyurethane
- Nylon
- Others
- Y-o-Y Growth Trend Analysis By Raw Material, 2020-2024
- Absolute $ Opportunity Analysis By Raw Material, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Bn) & Units Analysis By Region, 2020-2024
- Current Market Size Value (USD Bn) & Units Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Bn) & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Bn) & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- U.S.
- Canada
- Mexico
- By Indication
- By End User
- By Raw Material
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- By Raw Material
- Key Takeaways
- Latin America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Bn) & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Bn) & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Indication
- By End User
- By Raw Material
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- By Raw Material
- Key Takeaways
- Western Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Bn) & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Bn) & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- U.K.
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Europe
- By Indication
- By End User
- By Raw Material
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- By Raw Material
- Key Takeaways
- Eastern Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Bn) & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Bn) & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltics
- Rest of Eastern Europe
- By Indication
- By End User
- By Raw Material
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- By Raw Material
- Key Takeaways
- East Asia Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Bn) & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Bn) & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Indication
- By End User
- By Raw Material
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- By Raw Material
- Key Takeaways
- South Asia & Pacific Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Bn) & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Bn) & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Res of South Asia & Pacific
- By Indication
- By End User
- By Raw Material
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- By Raw Material
- Key Takeaways
- Middle East & Africa Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Bn) & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Bn) & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Indication
- By End User
- By Raw Material
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By End User
- By Raw Material
- Key Takeaways
- Key Countries Market Analysis
- U.S.
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- U.K.
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Nordic
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- BENELUX
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Balkan & Baltics
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Indication
- By End User
- By Raw Material
- U.S.
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Indication
- By End User
- By Raw Material
- Competition Analysis
- Competition Deep Dive
- Cardionovum GmbH
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Cook Medical INC
- Cordis Corporation
- Abbott Laboratories
- Meril Life Sciences Pvt Ltd
- Jotech GmbH
- MicroPort Scientific Corporation
- Terumo Corporation
- Boston Scientific Corporation
- Cardionovum GmbH
- Assumptions & Acronyms Used
- Research Methodology
- Competition Deep Dive
List Of Table
- Table 1: Global Market Value (USD Bn) Forecast by Region, 2020 to 2035
- Table 2: Global Market Units Forecast by Region, 2020 to 2035
- Table 3: Global Market Value (USD Bn) Forecast by Indication, 2020 to 2035
- Table 4: Global Market Units Forecast by Indication, 2020 to 2035
- Table 5: Global Market Value (USD Bn) Forecast by End User, 2020 to 2035
- Table 6: Global Market Units Forecast by End User, 2020 to 2035
- Table 7: Global Market Value (USD Bn) Forecast by Raw Material, 2020 to 2035
- Table 8: Global Market Units Forecast by Raw Material, 2020 to 2035
- Table 9: North America Market Value (USD Bn) Forecast by Country, 2020 to 2035
- Table 10: North America Market Units Forecast by Country, 2020 to 2035
- Table 11: North America Market Value (USD Bn) Forecast by Indication, 2020 to 2035
- Table 12: North America Market Units Forecast by Indication, 2020 to 2035
- Table 13: North America Market Value (USD Bn) Forecast by End User, 2020 to 2035
- Table 14: North America Market Units Forecast by End User, 2020 to 2035
- Table 15: North America Market Value (USD Bn) Forecast by Raw Material, 2020 to 2035
- Table 16: North America Market Units Forecast by Raw Material, 2020 to 2035
- Table 17: Latin America Market Value (USD Bn) Forecast by Country, 2020 to 2035
- Table 18: Latin America Market Units Forecast by Country, 2020 to 2035
- Table 19: Latin America Market Value (USD Bn) Forecast by Indication, 2020 to 2035
- Table 20: Latin America Market Units Forecast by Indication, 2020 to 2035
- Table 21: Latin America Market Value (USD Bn) Forecast by End User, 2020 to 2035
- Table 22: Latin America Market Units Forecast by End User, 2020 to 2035
- Table 23: Latin America Market Value (USD Bn) Forecast by Raw Material, 2020 to 2035
- Table 24: Latin America Market Units Forecast by Raw Material, 2020 to 2035
- Table 25: Western Europe Market Value (USD Bn) Forecast by Country, 2020 to 2035
- Table 26: Western Europe Market Units Forecast by Country, 2020 to 2035
- Table 27: Western Europe Market Value (USD Bn) Forecast by Indication, 2020 to 2035
- Table 28: Western Europe Market Units Forecast by Indication, 2020 to 2035
- Table 29: Western Europe Market Value (USD Bn) Forecast by End User, 2020 to 2035
- Table 30: Western Europe Market Units Forecast by End User, 2020 to 2035
- Table 31: Western Europe Market Value (USD Bn) Forecast by Raw Material, 2020 to 2035
- Table 32: Western Europe Market Units Forecast by Raw Material, 2020 to 2035
- Table 33: Eastern Europe Market Value (USD Bn) Forecast by Country, 2020 to 2035
- Table 34: Eastern Europe Market Units Forecast by Country, 2020 to 2035
- Table 35: Eastern Europe Market Value (USD Bn) Forecast by Indication, 2020 to 2035
- Table 36: Eastern Europe Market Units Forecast by Indication, 2020 to 2035
- Table 37: Eastern Europe Market Value (USD Bn) Forecast by End User, 2020 to 2035
- Table 38: Eastern Europe Market Units Forecast by End User, 2020 to 2035
- Table 39: Eastern Europe Market Value (USD Bn) Forecast by Raw Material, 2020 to 2035
- Table 40: Eastern Europe Market Units Forecast by Raw Material, 2020 to 2035
- Table 41: East Asia Market Value (USD Bn) Forecast by Country, 2020 to 2035
- Table 42: East Asia Market Units Forecast by Country, 2020 to 2035
- Table 43: East Asia Market Value (USD Bn) Forecast by Indication, 2020 to 2035
- Table 44: East Asia Market Units Forecast by Indication, 2020 to 2035
- Table 45: East Asia Market Value (USD Bn) Forecast by End User, 2020 to 2035
- Table 46: East Asia Market Units Forecast by End User, 2020 to 2035
- Table 47: East Asia Market Value (USD Bn) Forecast by Raw Material, 2020 to 2035
- Table 48: East Asia Market Units Forecast by Raw Material, 2020 to 2035
- Table 49: South Asia & Pacific Market Value (USD Bn) Forecast by Country, 2020 to 2035
- Table 50: South Asia & Pacific Market Units Forecast by Country, 2020 to 2035
- Table 51: South Asia & Pacific Market Value (USD Bn) Forecast by Indication, 2020 to 2035
- Table 52: South Asia & Pacific Market Units Forecast by Indication, 2020 to 2035
- Table 53: South Asia & Pacific Market Value (USD Bn) Forecast by End User, 2020 to 2035
- Table 54: South Asia & Pacific Market Units Forecast by End User, 2020 to 2035
- Table 55: South Asia & Pacific Market Value (USD Bn) Forecast by Raw Material, 2020 to 2035
- Table 56: South Asia & Pacific Market Units Forecast by Raw Material, 2020 to 2035
- Table 57: Middle East & Africa Market Value (USD Bn) Forecast by Country, 2020 to 2035
- Table 58: Middle East & Africa Market Units Forecast by Country, 2020 to 2035
- Table 59: Middle East & Africa Market Value (USD Bn) Forecast by Indication, 2020 to 2035
- Table 60: Middle East & Africa Market Units Forecast by Indication, 2020 to 2035
- Table 61: Middle East & Africa Market Value (USD Bn) Forecast by End User, 2020 to 2035
- Table 62: Middle East & Africa Market Units Forecast by End User, 2020 to 2035
- Table 63: Middle East & Africa Market Value (USD Bn) Forecast by Raw Material, 2020 to 2035
- Table 64: Middle East & Africa Market Units Forecast by Raw Material, 2020 to 2035
List Of Figures
- Figure 1: Global Market Units Forecast 2020 to 2035
- Figure 2: Global Market Pricing Analysis
- Figure 3: Global Market Value (USD Bn) Forecast 2020 to 2035
- Figure 4: Global Market Value Share and BPS Analysis by Indication, 2025 and 2035
- Figure 5: Global Market Y-o-Y Growth Comparison by Indication, 2025 to 2035
- Figure 6: Global Market Attractiveness Analysis by Indication
- Figure 7: Global Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 8: Global Market Y-o-Y Growth Comparison by End User, 2025 to 2035
- Figure 9: Global Market Attractiveness Analysis by End User
- Figure 10: Global Market Value Share and BPS Analysis by Raw Material, 2025 and 2035
- Figure 11: Global Market Y-o-Y Growth Comparison by Raw Material, 2025 to 2035
- Figure 12: Global Market Attractiveness Analysis by Raw Material
- Figure 13: Global Market Value (USD Bn) Share and BPS Analysis by Region, 2025 and 2035
- Figure 14: Global Market Y-o-Y Growth Comparison by Region, 2025 to 2035
- Figure 15: Global Market Attractiveness Analysis by Region
- Figure 16: North America Market Incremental $ Opportunity, 2025 to 2035
- Figure 17: Latin America Market Incremental $ Opportunity, 2025 to 2035
- Figure 18: Western Europe Market Incremental $ Opportunity, 2025 to 2035
- Figure 19: Eastern Europe Market Incremental $ Opportunity, 2025 to 2035
- Figure 20: East Asia Market Incremental $ Opportunity, 2025 to 2035
- Figure 21: South Asia & Pacific Market Incremental $ Opportunity, 2025 to 2035
- Figure 22: Middle East & Africa Market Incremental $ Opportunity, 2025 to 2035
- Figure 23: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 24: North America Market Value Share and BPS Analysis by Indication, 2025 and 2035
- Figure 25: North America Market Y-o-Y Growth Comparison by Indication, 2025 to 2035
- Figure 26: North America Market Attractiveness Analysis by Indication
- Figure 27: North America Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 28: North America Market Y-o-Y Growth Comparison by End User, 2025 to 2035
- Figure 29: North America Market Attractiveness Analysis by End User
- Figure 30: North America Market Value Share and BPS Analysis by Raw Material, 2025 and 2035
- Figure 31: North America Market Y-o-Y Growth Comparison by Raw Material, 2025 to 2035
- Figure 32: North America Market Attractiveness Analysis by Raw Material
- Figure 33: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 34: Latin America Market Value Share and BPS Analysis by Indication, 2025 and 2035
- Figure 35: Latin America Market Y-o-Y Growth Comparison by Indication, 2025 to 2035
- Figure 36: Latin America Market Attractiveness Analysis by Indication
- Figure 37: Latin America Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 38: Latin America Market Y-o-Y Growth Comparison by End User, 2025 to 2035
- Figure 39: Latin America Market Attractiveness Analysis by End User
- Figure 40: Latin America Market Value Share and BPS Analysis by Raw Material, 2025 and 2035
- Figure 41: Latin America Market Y-o-Y Growth Comparison by Raw Material, 2025 to 2035
- Figure 42: Latin America Market Attractiveness Analysis by Raw Material
- Figure 43: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 44: Western Europe Market Value Share and BPS Analysis by Indication, 2025 and 2035
- Figure 45: Western Europe Market Y-o-Y Growth Comparison by Indication, 2025 to 2035
- Figure 46: Western Europe Market Attractiveness Analysis by Indication
- Figure 47: Western Europe Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 48: Western Europe Market Y-o-Y Growth Comparison by End User, 2025 to 2035
- Figure 49: Western Europe Market Attractiveness Analysis by End User
- Figure 50: Western Europe Market Value Share and BPS Analysis by Raw Material, 2025 and 2035
- Figure 51: Western Europe Market Y-o-Y Growth Comparison by Raw Material, 2025 to 2035
- Figure 52: Western Europe Market Attractiveness Analysis by Raw Material
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 54: Eastern Europe Market Value Share and BPS Analysis by Indication, 2025 and 2035
- Figure 55: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2025 to 2035
- Figure 56: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 57: Eastern Europe Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 58: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2025 to 2035
- Figure 59: Eastern Europe Market Attractiveness Analysis by End User
- Figure 60: Eastern Europe Market Value Share and BPS Analysis by Raw Material, 2025 and 2035
- Figure 61: Eastern Europe Market Y-o-Y Growth Comparison by Raw Material, 2025 to 2035
- Figure 62: Eastern Europe Market Attractiveness Analysis by Raw Material
- Figure 63: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 64: East Asia Market Value Share and BPS Analysis by Indication, 2025 and 2035
- Figure 65: East Asia Market Y-o-Y Growth Comparison by Indication, 2025 to 2035
- Figure 66: East Asia Market Attractiveness Analysis by Indication
- Figure 67: East Asia Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 68: East Asia Market Y-o-Y Growth Comparison by End User, 2025 to 2035
- Figure 69: East Asia Market Attractiveness Analysis by End User
- Figure 70: East Asia Market Value Share and BPS Analysis by Raw Material, 2025 and 2035
- Figure 71: East Asia Market Y-o-Y Growth Comparison by Raw Material, 2025 to 2035
- Figure 72: East Asia Market Attractiveness Analysis by Raw Material
- Figure 73: South Asia & Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 74: South Asia & Pacific Market Value Share and BPS Analysis by Indication, 2025 and 2035
- Figure 75: South Asia & Pacific Market Y-o-Y Growth Comparison by Indication, 2025 to 2035
- Figure 76: South Asia & Pacific Market Attractiveness Analysis by Indication
- Figure 77: South Asia & Pacific Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 78: South Asia & Pacific Market Y-o-Y Growth Comparison by End User, 2025 to 2035
- Figure 79: South Asia & Pacific Market Attractiveness Analysis by End User
- Figure 80: South Asia & Pacific Market Value Share and BPS Analysis by Raw Material, 2025 and 2035
- Figure 81: South Asia & Pacific Market Y-o-Y Growth Comparison by Raw Material, 2025 to 2035
- Figure 82: South Asia & Pacific Market Attractiveness Analysis by Raw Material
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 84: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2025 and 2035
- Figure 85: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2025 to 2035
- Figure 86: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 87: Middle East & Africa Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 88: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2025 to 2035
- Figure 89: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 90: Middle East & Africa Market Value Share and BPS Analysis by Raw Material, 2025 and 2035
- Figure 91: Middle East & Africa Market Y-o-Y Growth Comparison by Raw Material, 2025 to 2035
- Figure 92: Middle East & Africa Market Attractiveness Analysis by Raw Material
- Figure 93: Global Market - Tier Structure Analysis
- Figure 94: Global Market - Company Share Analysis