Continuous Cuff Pressure Monitoring Devices Market Size, Share, Growth and Forecast (2026 - 2036)
The Continuous Cuff Pressure Monitoring Devices Market is segmented by Product Type (Continuous Cuff Pressure Monitors and Manual Cuff Manometers), Technology (Electronic Automated Systems and Pneumatic/Mechanical Systems), Application (ICU Ventilation, Surgical Anesthesia, and Emergency Care), and Region. Forecast for 2026 to 2036.
FACT.MR analysis indicates the market is undergoing a protocol-driven shift. Demand is consolidating around automated continuous monitoring systems as VAP prevention mandates tighten across ICUs. Hospitals are replacing manual manometers with electronic systems that integrate directly into ventilator and bedside monitoring platforms. Procedural care segments remain secondary but are growing as operating room safety standards evolve.
Continuous Cuff Pressure Monitoring Devices Market Forecast and Outlook By Fact.MR
In 2025, the continuous cuff pressure monitoring devices market was valued at USD 0.9 billion. Based on Fact.MR analysis, demand for continuous cuff pressure monitoring devices is estimated to grow to USD 1.0 billion in 2026 and USD 1.9 billion by 2036. FACT.MR projects a CAGR of 7.5% during the forecast period.
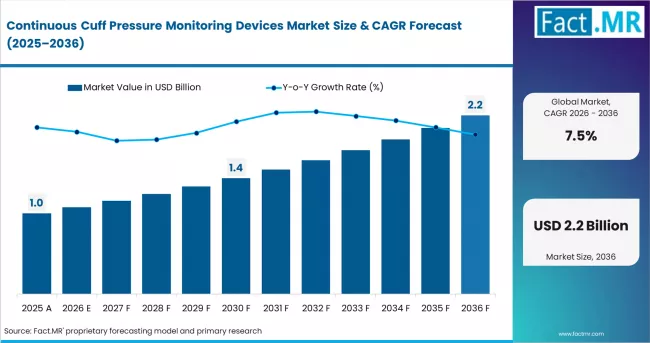
| Metric | Value |
|---|---|
| Estimated Value in 2026 | USD 1.0 billion |
| Forecast Value in 2036 | USD 1.9 billion |
| Forecast CAGR (2026 to 2036) | 7.5% |
Summary of Continuous Cuff Pressure Monitoring Devices Market
- Market Definition
- The market includes automated continuous and manual cuff pressure monitoring devices used for endotracheal tube management across ICU, surgical, and emergency care settings.
- Demand Drivers
- U.S. ICU networks require continuous monitoring systems compliant with VAP prevention protocols under CMS quality reporting frameworks.
- China is expanding ICU bed capacity with integrated monitoring systems driving institutional procurement.
- Germany is adopting ICU automation standards, increasing demand for electronic cuff management devices.
- Key Segments Analyzed
- By Product Type: Continuous cuff pressure monitors hold approximately 58% share in 2026 due to VAP prevention requirements.
- By Technology: Electronic automated systems hold approximately 60% share in 2026, driven by ventilator integration.
- By Application: ICU ventilation holds approximately 55% share in 2026, supported by continuous intubation monitoring mandates.
- By Geography: India leads at 8.2% CAGR, supported by critical care infrastructure investment.
- Analyst Opinion at FACT.MR
- Shambhu Nath Jha, Principal Consultant at Fact.MR, opines, 'CXOs will find that automated cuff monitoring systems represent the compliance-driven upgrade path as VAP benchmarks harden across global ICU accreditation frameworks.'
- Strategic Implications
- Invest in automated electronic cuff monitors with documented VAP compliance outputs for ICU procurement programmes.
- Develop cost-optimised continuous monitoring devices for emerging markets in India and China.
- Build partnerships with ventilator OEMs to integrate cuff pressure monitoring within ICU platform ecosystems.
- Methodology
- Market sizing uses device shipment volumes and ASP, validated with 2024 to 2025 procurement data.
- Analysis includes FDA 510(k) clearances, NHS data, CMS quality reporting, and company disclosures.
- Forecasts consider ICU capacity expansion, VAP prevention mandates, and hospital automation investment trends.
The market is expected to generate USD 0.9 billion in incremental revenue over the forecast period. Growth is steady rather than transformational, supported by consistent ICU infrastructure investment and mandatory VAP prevention protocols. Expansion is constrained by high device unit costs and budget limitations in public hospital systems across emerging markets.
Demand is shifting from periodic manual cuff checks toward real-time automated monitoring embedded in ICU ventilation workflows. Hospitals facing VAP audit requirements are accelerating procurement of continuous systems. Regulatory pressure from patient safety bodies is compelling institutions to standardise on automated devices with documented compliance outputs.
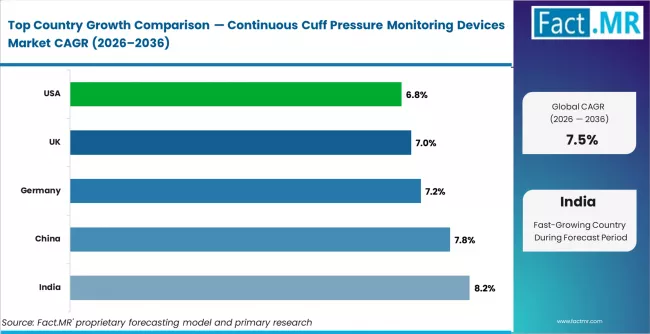
India leads with an estimated CAGR of 8.2% through 2036, supported by rapid critical care infrastructure expansion. China follows at 7.8%, driven by growing ICU bed capacity and respiratory disease burden. Germany records 7.2%, supported by high ICU automation adoption. The United Kingdom grows at 7.0%, driven by NHS patient safety programmes. The United States records 6.8%, supported by advanced VAP prevention protocols in established ICU networks.
Segmental Analysis
Continuous Cuff Pressure Monitoring Devices Market Analysis by Product Type
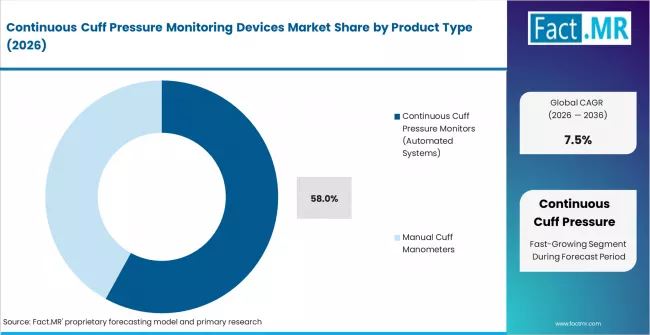
Based on FACT.MR's continuous cuff pressure monitoring devices market report, continuous cuff pressure monitors are estimated to hold approximately 58% share in 2026. They dominate due to mandated VAP prevention workflows in ICU settings. Automated systems eliminate reliance on manual checks and provide documented pressure logs for clinical audit compliance.
- Medtronic Product Expansion: Medtronic expanded its Nellcor portfolio with updated cuff pressure management accessories in 2024. The expansion targeted U.S. and European ICU procurement. Devices were certified under updated EU MDR requirements, strengthening institutional sales. [4]
- Dräger ICU Integration: Dräger launched an updated Evita ventilator series in 2024 with integrated cuff pressure monitoring functionality. The system links cuff management directly to ventilator data streams. This reduces standalone device procurement for Dräger ventilator users. [5]
- Manual Manometer Decline Trend: NHS England procurement data from 2024 shows a shift toward automated continuous monitoring in critical care units. Manual manometer procurement fell as a share of total cuff device spend. NHS Trusts under CQC inspection cited automated monitoring as a patient safety standard. [6]
Continuous Cuff Pressure Monitoring Devices Market Analysis by Technology
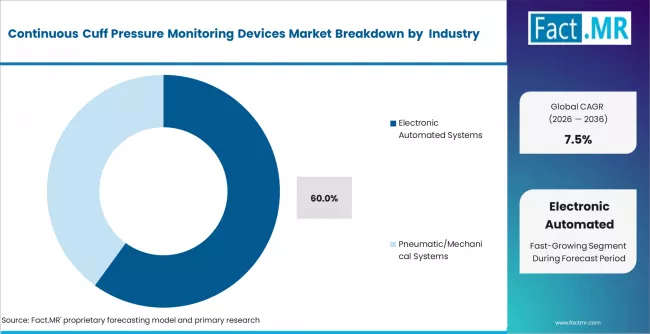
Based on FACT.MR's report, electronic automated systems hold approximately 60% share in 2026. Their dominance reflects demand for ventilator-integrated monitoring and real-time alarm capability. Pneumatic systems remain in use in portable and emergency settings where power independence is required.
- Hamilton Medical Integration: Hamilton Medical advanced ventilator-integrated cuff management in its HAMILTON-C6 platform in 2024. The system automates cuff inflation and deflation cycles based on continuous pressure feedback. ICU customers in Germany and Switzerland adopted the platform for clinical workflow integration. [7]
- Vyaire Medical Technology Update: Vyaire Medical updated its AirLife cuff pressure line in 2025 with digital readout capability for operating room use. The update addressed demand from anesthesia departments requiring precise sub-30 cmH2O pressure management. [8]
- Pneumatic System Persistence: Emergency medical services in the United Kingdom continue to use pneumatic cuff manometers for pre-hospital airway management. NHS Ambulance Service procurement data from 2024 confirms ongoing manual device supply contracts. Portability and battery independence sustain demand in this segment. [9]
Drivers, Restraints, and Opportunities
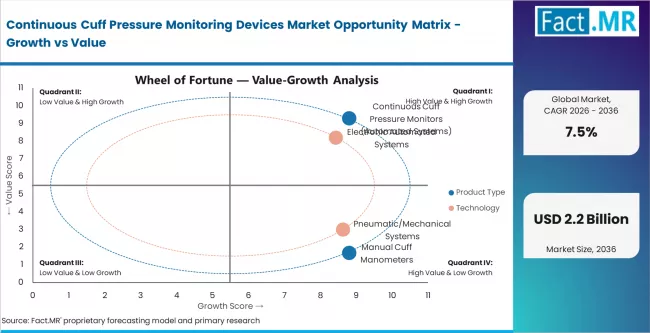
FACT.MR analysts observe the market is mature in developed economies and transitioning in Asia Pacific and Latin America. Demand is concentrated in ICU ventilation applications where continuous monitoring is becoming a standard-of-care requirement. Growth is structurally linked to ICU bed expansion, VAP prevention protocol adoption, and hospital accreditation requirements.
Manual manometers remain in use in cost-sensitive and lower-acuity settings but are declining as a share of procurement in high-income markets. Automated continuous systems are growing faster in volume and value. Higher unit cost of electronic devices is partially offset by reduced nursing intervention time and measurable VAP outcome improvement.
- VAP Prevention Mandates: The U.S. Centers for Medicare and Medicaid Services (CMS) includes VAP rates in hospital quality reporting under the Hospital Inpatient Quality Reporting programme. Hospitals with elevated VAP rates face reimbursement adjustments. This creates direct procurement incentive for continuous cuff monitoring devices over manual alternatives. [1]
- ICU Capacity Expansion in Asia: China added over 70,000 ICU beds between 2020 and 2024 under the National Health Commission's critical care capacity programme. India's ICMR guidelines updated in 2024 require cuff pressure monitoring in mechanically ventilated patients. Both expansions are driving volume procurement of cuff management devices. [2]
EU Medical Device Regulation Compliance: The EU MDR framework, enforced from 2021 with transition deadlines in 2024 and 2025, requires updated clinical evidence and post-market surveillance for active monitoring devices. Medtronic and Dräger submitted updated MDR dossiers in 2024, reinforcing their EU market positions while raising barriers for smaller entrants. [3]
Regional Analysis
The continuous cuff pressure monitoring devices market is assessed across Asia Pacific, North America, Europe, Latin America, and Middle East and Africa, covering 40+ countries with demand shaped by ICU infrastructure maturity, VAP prevention protocol adoption, ventilator integration capabilities, and hospital accreditation frameworks. The full report provides market attractiveness analysis by region and country.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 8.2% |
| China | 7.8% |
| Germany | 7.2% |
| United Kingdom | 7.0% |
| United States | 6.8% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
Asia Pacific Continuous Cuff Pressure Monitoring Devices Market Analysis
Asia Pacific is the fastest-growing region. Growth is supported by ICU capacity expansion and rising respiratory disease burden. China and India drive volume procurement. South Korea and Japan lead in hospital automation adoption. Domestic and multinational suppliers are scaling supply chains to meet public hospital tenders.
- China: ICU expansion and respiratory disease volume drive demand. Demand is projected at 7.8% CAGR through 2036. The National Health Commission added over 70,000 ICU beds between 2020 and 2024. Central government procurement guidelines from 2024 require cuff pressure management devices in tertiary hospital ICUs. Domestic producers including Mindray are increasing cuff monitoring integration in their ventilator platforms. Import substitution policies are supporting local device procurement while international brands retain premium positioning.
- India: Rapid critical care infrastructure build-out accelerates procurement. Demand is projected at 8.2% CAGR through 2036. ICMR updated mechanical ventilation guidelines in 2024 requiring cuff pressure monitoring in intubated patients. The National Health Mission is funding ICU upgrades in tier 2 and tier 3 hospitals. This creates new institutional buyer segments. Medtronic and local distributors are expanding sales networks across government hospital procurement channels.
North America Continuous Cuff Pressure Monitoring Devices Market Analysis
North America is the largest revenue market. Demand is driven by mandatory VAP benchmarking and established ICU automation investment. Medtronic, GE Healthcare, and Smiths Medical dominate procurement. Long-term supply contracts and GPO frameworks define buying patterns.
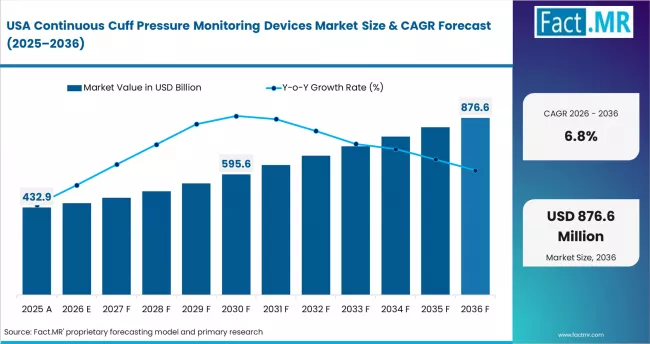
- United States: VAP protocol compliance and advanced ICU infrastructure define market leadership. Demand is projected at 6.8% CAGR through 2036. CMS hospital quality reporting penalises elevated VAP rates under the Hospital-Acquired Condition Reduction Programme. This sustains procurement of automated continuous monitoring devices. GE Healthcare expanded CARESCAPE platform capabilities in Q3 2024 to include cuff pressure alerts. Medtronic's Nellcor product line received updated 510(k) clearances in 2024 for continuous airway management accessories. GPO contract renewals from 2025 are sustaining institutional supply relationships.
Europe Continuous Cuff Pressure Monitoring Devices Market Analysis
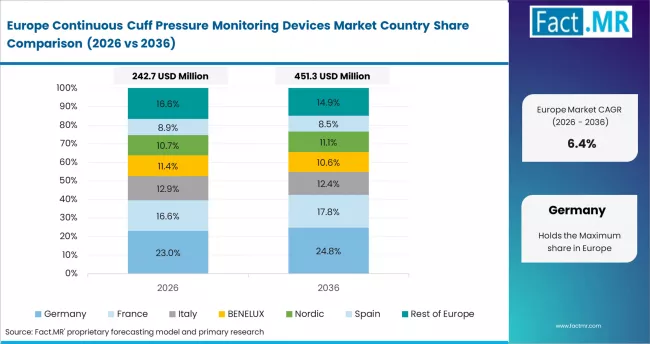
Europe is a regulation-driven and clinically structured market. EU MDR compliance frameworks define procurement eligibility. Germany leads in ICU automation. Dräger, Hamilton Medical, and Smiths Medical hold dominant supply positions.
- Germany: ICU automation and ventilator integration leadership drive consistent demand. Demand is projected at 7.2% CAGR through 2036. Dräger's Evita series updated with cuff monitoring integration in 2024 for German hospital systems. Hospital procurement committees under the German Hospital Federation standards require documented cuff management in mechanically ventilated ICU patients. Hamilton Medical's HAMILTON-C6 adoption expanded across Baden-Württemberg hospital networks in 2024. EU MDR compliance requirements strengthened established suppliers while raising barriers for new entrants.
- United Kingdom: NHS patient safety frameworks and CQC inspection criteria shape procurement. Demand is projected at 7.0% CAGR through 2036. NHS England issued updated critical care guidance in 2024 recommending continuous cuff pressure monitoring in ventilated ICU patients. NHS procurement data shows growth in automated cuff device spend relative to manual manometers. Smiths Medical Portex line retained NHS supply contract renewals in 2024. CQC inspections citing VAP prevention compliance are accelerating device standardisation across NHS Trusts.
FACT.MR's analysis of the continuous cuff pressure monitoring devices market in Europe consists of country-wise assessment covering Germany, the United Kingdom, France, Italy, Spain, and Rest of Europe. Readers can find VAP protocol adoption trends, MDR compliance impacts, and hospital procurement forecasts.
Competitive Aligners for Market Players
Competitive intensity in the continuous cuff pressure monitoring devices market is shaped by platform integration, regulatory compliance, and ICU procurement positioning. A small group of established medical device companies’ controls most of the market. A second tier of regional suppliers competes mainly on price in cost-sensitive hospitals. Competitive advantage depends on ventilator integration, VAP prevention documentation, and compliance with EU MDR and FDA 510(k) standards.
Medtronic, Dräger, and Hamilton Medical sit at the center of ICU procurement. Their strength comes from fully integrated ventilator and monitoring systems. Cuff pressure monitoring is embedded into broader critical care workflows. This reduces the need for standalone devices. These companies also benefit from group purchasing organization contracts in North America and long-term supply agreements in Europe. Once installed, switching vendors becomes difficult for hospitals. GE Healthcare and Smiths Medical compete through complementary offerings. GE Healthcare integrates cuff monitoring into its CARESCAPE platform. This supports centralized patient monitoring across ICU environments. Smith’s Medical focuses on anesthesia and procedural care through its Portex product line. It serves operating rooms and perioperative settings.
Hospital buyers are increasingly consolidating vendors. Large health systems prefer fewer suppliers with broader portfolios. This simplifies procurement and improves system compatibility. As a result, integrated platform providers gain pricing power and contract stability. Standalone device manufacturers face growing pressure. Without integration capabilities or strong clinical data, they are often limited to niche or lower-margin segments.
Key Players
- Medtronic
- Smiths Medical
- Vyaire Medical
- Hamilton Medical
- Dräger
- GE Healthcare
Bibliography
- [1] Centers for Medicare and Medicaid Services (CMS). Hospital Inpatient Quality Reporting Programme: ventilator-associated events and HAC reduction programme measures; 2024. Available from: cms.gov/medicare/quality/initiatives/hospital-quality-initiative
- [2] National Health Commission of China. Critical care capacity expansion programme: ICU bed addition targets and procurement requirements for mechanically ventilated patients; 2024. Available from: nhc.gov.cn
- [3] European Commission. EU Medical Device Regulation (MDR) 2017/745 post-market surveillance and clinical evidence requirements for active monitoring devices; 2024. Available from: ec.europa.eu/health/md_sector/new_regulations
- [4] Medtronic. Annual Report and Nellcor product line expansion disclosure: updated cuff pressure management accessories for ICU and EU MDR compliance certification; 2024. Available from: medtronic.com/investor-relations
- [5] Dräger. Product launch announcement: Evita ventilator series update with integrated cuff pressure monitoring functionality for ICU applications; 2024. Available from: draeger.com/en/products/ventilators
- [6] NHS England. Critical care procurement and patient safety reporting: shift from manual to automated cuff pressure monitoring in mechanically ventilated patients; 2024. Available from: england.nhs.uk/critical-care
- [7] Hamilton Medical. HAMILTON-C6 ventilator product update: automated cuff inflation and deflation cycles with continuous pressure feedback for ICU deployment; 2024. Available from: hamilton-medical.com/products
- [8] Vyaire Medical. AirLife product line update: digital readout cuff pressure monitors for operating room and anesthesia applications; 2025. Available from: vyaire.com/products/airlife
- [9] NHS Ambulance Service. National procurement contract data: pneumatic cuff manometer supply for pre-hospital emergency airway management; 2024. Available from: england.nhs.uk/ambulances
This Report Addresses
- Strategic insights on demand across India, China, Germany, the United Kingdom, and the United States. Covers ICU ventilation, surgical anesthesia, and emergency care procurement trends.
- Market forecast from USD 1.0 billion in 2026 to USD 1.9 billion by 2036, at 7.5% CAGR.
- Opportunity mapping across India ICU infrastructure build-out, China hospital expansion, U.S. VAP compliance procurement, and Germany ICU automation demand.
- Segment analysis by product type, technology, and application. Covers continuous monitors, electronic systems, and key clinical use cases.
- Regional outlook covering Asia Pacific capacity-driven growth, North America compliance-led procurement, and Europe regulation-driven standardisation.
- Competitive analysis of Medtronic, Dräger, Hamilton Medical, GE Healthcare, Smiths Medical, and Vyaire Medical.
- Technology tracking on ventilator integration, electronic cuff pressure systems, VAP documentation compliance, and EU MDR certification.
- Report delivery in PDF, Excel, and presentation formats with validated data and industry inputs.
Continuous Cuff Pressure Monitoring Devices Market Definition
The market covers devices used to continuously measure and maintain endotracheal tube cuff pressure within safe clinical ranges.
Continuous Cuff Pressure Monitoring Devices Market Inclusions
Covers global and regional forecasts from 2026 to 2036 by product type and application. Includes electronic automated and pneumatic systems. Covers ICU ventilation, surgical anesthesia, and emergency care use across hospital and CDMO channels.
Continuous Cuff Pressure Monitoring Devices Market Exclusions
Excludes endotracheal tubes and ventilator systems. Omits general airway management accessories and non-cuff pressure monitoring equipment. Excludes standalone bedside monitoring platforms not dedicated to cuff pressure.
Continuous Cuff Pressure Monitoring Devices Market Research Methodology
- Primary Research: Interviews with ICU device procurement managers, respiratory care specialists, and hospital biomedical engineers across key markets.
- Desk Research: Uses NHS procurement data, FDA 510(k) clearances, CMS VAP quality reporting, Medtronic and Dräger public filings, and clinical guidelines from 2024 to 2025.
- Market Sizing and Forecasting: Based on device shipment volumes and ASP by product category. Includes public hospital procurement and private ICU demand benchmarks.
- Data Validation and Update Cycle: Validated using manufacturer revenues and hospital procurement records. Cross-checked with clinical trial and regulatory databases. Updated regularly.
Scope of Report

| Attribute | Details |
|---|---|
| Quantitative Units | USD 1.0 billion (2026) to USD 1.9 million (2036), at a CAGR of 7.5% |
| Market Definition | Electronic and mechanical devices that measure and maintain ETT or tracheostomy tube cuff pressure in therapeutic ranges, supplied in automated, semi-automated, and manual formats for ICU, home care, ambulatory, and long-term care applications globally. |
| Product Type Segmentation | Continuous Cuff Pressure Monitors (Automated Systems), Manual Cuff Manometers |
| Technology Segmentation | Electronic Automated Systems, Pneumatic/Mechanical Systems |
| Application Segmentation | ICU Ventilation, Surgical Anesthesia, Emergency Care |
| Application Coverage | ICU VAP prevention protocol compliance, anaesthesia airway management, tracheostomy home care cuff management, ambulatory respiratory care, and long-term ventilation airway safety. |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Canada, Mexico, Germany, UK, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, ASEAN, Australia and New Zealand, Brazil, Argentina, Chile, Saudi Arabia, GCC, Turkey, South Africa, Rest of MEA |
| Key Companies Profiled | Smiths Medical, Nellcor (Medtronic), Tracoe Medical, Endocore Medical, IMD Medical, VBM Medizintechnik, Weinmann Emergency, Intersurgical, Flexicare Medical, Bard Medical (BD) |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using device manufacturer revenues, ICU procurement volumes by product type, average selling prices by automation grade, hospital device purchasing benchmarks, and primary interviews with ICU procurement specialists and critical care clinicians. |
Continuous Cuff Pressure Monitoring Devices Market By Segment
-
By Product Type:
- Continuous Cuff Pressure Monitors (Automated Systems)
- Manual Cuff Manometers
-
By Technology:
- Electronic Automated Systems
- Pneumatic/Mechanical Systems
-
By Application:
- ICU Ventilation
- Surgical Anesthesia
- Emergency Care
-
By Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Rest of Middle East and Africa
- North America
- Frequently Asked Questions -
How large is the continuous cuff pressure monitoring devices market in 2025?
The market was valued at USD 0.9 billion in 2025.
What will the market size be in 2026?
The market is estimated to reach USD 1.0 billion in 2026
What is the projected market size by 2036?
The market is projected to reach USD 1.9 billion by 2036.
What is the expected CAGR of the continuous cuff pressure monitoring devices market?
The market is expected to grow at a CAGR of 7.5% from 2026 to 2036.
What is the absolute dollar opportunity in this market?
The market is expected to create an absolute dollar opportunity of USD 0.9 billion between 2026 and 2036.
Which product type segment leads the market?
Continuous cuff pressure monitors lead with approximately 58% share in 2026.
Which technology segment is poised to lead?
Electronic automated systems lead with approximately 60% share in 2026.
Which application segment holds the largest share?
ICU ventilation holds approximately 55% share in 2026, driven by continuous intubation monitoring mandates.
Which country shows the fastest growth?
India leads with a CAGR of 8.2% through 2036.
What are continuous cuff pressure monitoring devices used for?
They are used to continuously measure and maintain endotracheal tube cuff pressure within safe ranges during mechanical ventilation, surgical anesthesia, and emergency airway management.
What is driving market growth?
Growth is driven by VAP prevention protocol mandates, ICU infrastructure expansion in Asia Pacific, and integration of cuff monitoring into ventilator platforms.
Which region leads market growth?
Asia Pacific leads due to rapid ICU capacity build-out in China and India and increasing adoption of patient safety monitoring standards.
Who are the key market players?
Key players include Medtronic, Dräger, Hamilton Medical, GE Healthcare, Smiths Medical, and Vyaire Medical.
What challenges does the market face?
Challenges include high device unit costs limiting adoption in budget-constrained public hospitals and complexity of EU MDR compliance for smaller device suppliers.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Continuous Cuff Pressure Monitors (Automated Systems)
- Manual Cuff Manometers
- Continuous Cuff Pressure Monitors (Automated Systems)
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Electronic Automated Systems
- Pneumatic/Mechanical Systems
- Electronic Automated Systems
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Technology
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Technology
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Technology
- Competition Analysis
- Competition Deep Dive
- Medtronic
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Smiths Medical
- Vyaire Medical
- Hamilton Medical
- Dräger
- GE Healthcare
- Medtronic
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Technology
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Technology
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Technology
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Technology
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Technology
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis