COPD & Asthma Therapeutics Market Size, Share, Growth and Forecast (2025 - 2035)
COPD & Asthma Therapeutics Market Size and Share Forecast Outlook 2025 to 2035
COPD & asthma therapeutics market is projected to grow from USD 100.1 billion in 2025 to USD 245.9 billion by 2035, at a CAGR of 9.4%. Anti-IgE & Anti-IL Monoclonal Antibodies will dominate with a 22.7% market share, while inhalers will lead the device/product type segment with a 46.5% share.
COPD & Asthma Therapeutics Market Forecast and Outlook 2025 to 2035
The global COPD & asthma therapeutics market is projected to grow from USD 100.1 billion in 2025 to approximately USD 245.9 billion by 2035, recording an extraordinary absolute increase of USD 145.8 billion over the forecast period. This translates into a total growth of 145.7%, with the market forecast to expand at a compound annual growth rate (CAGR) of 9.4% between 2025 and 2035.
The overall market size is expected to grow by over 2.5X during this period, supported by exponential demand for advanced biologic therapies, rising adoption of smart inhaler technologies, and growing emphasis on personalized respiratory medicine innovation and digital health integration across global healthcare operations.
Quick Stats on COPD & Asthma Therapeutics Market
- COPD & Asthma Therapeutics Market Value (2025): USD 100.1 billion
- COPD & Asthma Therapeutics Market Forecast Value (2035): USD 245.9 billion
- COPD & Asthma Therapeutics Market Forecast CAGR (2025 to 2035): 9.4%
- Leading Drug Class in COPD & Asthma Therapeutics Market: Anti-IgE & Anti-IL Monoclonal Antibodies (22.7%)
- Leading Device/Product in COPD & Asthma Therapeutics Market: Inhalers (46.5%)
- Key Growth Regions in COPD & Asthma Therapeutics Market: Asia Pacific, North America, and Europe
- Key Players in COPD & Asthma Therapeutics Market: GSK plc, Teva Pharmaceutical, Merck & Co, F. Hoffmann-La Roche, AstraZeneca, Boehringer Ingelheim, Sanofi, Royal Philips, BD, Novartis, Cipla, Chiesi, Covis Pharma, Findair

The COPD & asthma therapeutics market is positioned for substantial expansion, driven by increasing recognition of chronic respiratory disease burden, growing patient populations with enhanced treatment access, and rising adoption of advanced therapeutic modalities across respiratory care practices globally.
The market demonstrates robust fundamentals supported by expanding healthcare infrastructure networks, respiratory specialists' focus on disease management protocols, and rising recognition of innovative biologic therapies as critical healthcare components in achieving enhanced patient outcomes, exacerbation prevention capabilities, and quality of life improvement within modern respiratory care architectures across diverse patient populations.
Market growth is underpinned by technological innovations in drug delivery procedures, particularly advanced inhaler systems and biologic therapy integration, which offer enhanced treatment efficacy, improved patient compliance, and superior compatibility with comprehensive disease management protocols prevalent in contemporary respiratory practices.
Healthcare providers increasingly prioritize therapeutic solutions that deliver optimal balance between clinical effectiveness, safety profiles, and cost-effectiveness while adhering to increasingly stringent regulatory standards and environmental requirements across global pharmaceutical markets.
The convergence of biologics development acceleration in developed regions, generic medication expansion in emerging economies, and specialized digital health infrastructure advancement creates multifaceted growth opportunities for pharmaceutical manufacturers and respiratory care facility operators.
The COPD & asthma therapeutics landscape is experiencing transformative changes as manufacturers develop sophisticated biologic agents including anti-IgE therapies, anti-IL monoclonal antibodies, and innovative combination formulations that enable targeted immune modulation and precision treatment approaches.
These therapeutic advancements are complemented by evolving delivery technologies encompassing smart inhaler systems, advanced nebulization platforms, and innovative digital health integration that significantly improve medication adherence and treatment monitoring outcomes.
The integration of artificial intelligence platforms and remote patient monitoring capabilities further expands therapeutic effectiveness for respiratory disease management, particularly benefiting chronic disease populations and underserved healthcare regions where specialist availability remains limited.
Between 2025 and 2030, the COPD & asthma therapeutics market is projected to expand from USD 100.1 billion to USD 155.3 billion, demonstrating strong foundational growth driven by global respiratory disease awareness expansion, increasing adoption of biologic therapies, and initial deployment of smart inhaler technologies across primary care and specialist respiratory platforms. This growth phase establishes market infrastructure, validates personalized treatment protocols, and creates comprehensive care delivery networks supporting global respiratory healthcare operations.
From 2030 to 2035, the market is forecast to reach USD 245.9 billion, driven by mature biologic therapy penetration, next-generation targeted therapeutics requiring sophisticated clinical expertise, and comprehensive integration of digital health platforms demanding enhanced patient monitoring capabilities. The growing adoption of precision medicine approaches specialized respiratory training initiatives, and universal healthcare coverage expansion will drive demand for comprehensive COPD & asthma therapeutic solutions with enhanced treatment outcomes and seamless respiratory care network integration functionality.
COPD & Asthma Therapeutics Market Key Takeaways
| Metric | Value |
|---|---|
| Estimated Value (2025E) | USD 100.1 billion |
| Forecast Value (2035F) | USD 245.9 billion |
| Forecast CAGR (2025 to 2035) | 9.4% |
Why is the COPD & Asthma Therapeutics Market Growing?
Market expansion is being supported by the exponential increase in chronic respiratory disease prevalence and the corresponding need for sophisticated therapeutic solutions in disease management applications across global healthcare operations. Respiratory care practitioners are increasingly focused on advanced biologic therapies that can improve patient outcomes, reduce exacerbation frequency, and optimize long-term disease control while meeting stringent safety requirements.
The proven efficacy of innovative therapeutic agents in various respiratory disease applications makes them an essential component of comprehensive disease management strategies and patient care programs. The growing emphasis on personalized medicine standards and biologic therapy integration is driving demand for advanced therapeutic solutions that meet stringent efficacy specifications and safety requirements for respiratory applications.
Healthcare providers' preference for reliable, high-performance treatment protocols that can ensure consistent clinical outcomes is creating opportunities for innovative drug formulations and customized combination therapies. The rising influence of environmental sustainability standards and inhaler carbon footprint regulations is also contributing to increased adoption of next-generation respiratory therapeutic solutions across different patient categories and healthcare systems requiring advanced pharmaceutical technology.
Opportunity Pathways - COPD & Asthma Therapeutics Market
The COPD & asthma therapeutics market represents a transformative growth opportunity, expanding from USD 100.1 billion in 2025 to USD 245.9 billion by 2035 at a 9.4% CAGR. As respiratory care practitioners prioritize patient outcome optimization, exacerbation prevention, and treatment excellence in complex disease management environments, COPD & asthma therapeutic solutions have evolved from conventional bronchodilator therapy to an essential healthcare component enabling precise inflammatory control, comprehensive symptom management strategies, and multi-pathway treatment operations across severe asthma platforms and COPD care applications.
The convergence of respiratory disease burden acceleration, increasing biologic therapy penetration, advanced drug delivery technology integration, and stringent quality mandates creates momentum in demand. High-precision monoclonal antibody therapies offering superior disease control, cost-effective combination treatments balancing functionality with economics, and specialized biologics for severe asthma applications will capture market premiums, while geographic expansion into high-growth Asian respiratory care markets and emerging healthcare access ecosystems will drive volume leadership. Healthcare provider emphasis on clinical innovation and patient safety provides structural support.
- Pathway A - Anti-IgE & Anti-IL Monoclonal Antibodies Segment Dominance: Leading with 22.7% market share, biologic antibody applications drive premium demand through targeted immune modulation requiring comprehensive therapeutic capabilities for severe respiratory disease management. Advanced biologic formulations enabling improved exacerbation control, reduced corticosteroid dependence, and enhanced quality of life outcomes command premium pricing from healthcare systems requiring stringent efficacy specifications and safety compliance. Expected revenue pool: USD 22.7-55.8 billion.
- Pathway B - Inhalers Device Leadership: Dominating with 46.5% market share through optimal balance of clinical effectiveness and patient convenience requirements, inhaler device applications serve most respiratory therapy delivery requirements while meeting diverse patient care demands. This device category addresses both maintenance treatment needs and acute symptom management expectations, making it the preferred delivery system for respiratory practitioners and patient populations seeking comprehensive therapeutic capabilities. Opportunity: USD 46.5-114.3 billion.
- Pathway C - Asian Market Acceleration: China (11.1% CAGR) and India (10.6% CAGR) lead global growth through respiratory disease prevalence expansion, healthcare infrastructure development, and therapeutic access capability advancement. Strategic partnerships with local healthcare providers, product localization expertise, and distribution network optimization enable the expansion of respiratory therapeutics in major urban healthcare and emerging rural care hubs. Geographic expansion upside: USD 32.4-68.7 billion.
- Pathway D - Combination Therapy Segment: Combination therapeutic formulations with 19.3% market share serve critical multi-pathway disease management applications requiring synergistic drug interactions for diverse patient populations. Optimized combination protocols supporting multiple mechanism targets, treatment adherence requirements, and proven clinical effectiveness maintain significant volumes from respiratory specialists and primary care facilities. Revenue potential: USD 19.3-47.5 billion.
- Pathway E - Advanced Biologic Development & Smart Inhaler Technologies: Companies investing in next-generation monoclonal antibodies, novel biologics targeting new pathways, and digital inhaler systems gain competitive advantages through breakthrough clinical efficacy and enhanced patient monitoring. Advanced capabilities enabling personalized treatment algorithms and real-time adherence tracking capture premium healthcare partnerships. Technology premium: USD 18.5-42.3 billion.
- Pathway F - Market Access Optimization & Patient Support Excellence: Comprehensive patient assistance programs, strategic payer negotiations, and integrated care delivery systems create competitive differentiation in respiratory markets requiring consistent therapeutic access. Companies offering guaranteed medication availability, adherence support services, and comprehensive disease management programs gain preferred provider status with quality-focused healthcare operators. Access value: USD 15.2-34.1 billion.
- Pathway G - Emerging Applications & Pipeline Development: Beyond traditional COPD & asthma management, respiratory therapeutics in rare lung diseases, pediatric asthma care, and precision phenotype-targeted treatments represent growth opportunities. Companies developing novel mechanisms of action, supporting investigator-initiated research, and expanding into adjacent respiratory disease markets capture incremental demand while diversifying revenue streams. Emerging opportunity: USD 12.3-27.6 billion.
Segmental Analysis
The market is segmented by drug class, device/product type, and region. By drug class, the market is divided into anti-IgE & anti-IL monoclonal antibodies, combination therapy, bronchodilators, corticosteroids, leukotriene modifiers, and others.
Based on device/product type, the market is categorized into inhalers (including dry powder inhalers, metered dose inhalers, and soft mist inhalers), nebulizers, and oxygen therapy devices. Regionally, the market is divided into Asia Pacific, North America, Europe, Latin America, and Middle East & Africa.
How do Anti-IgE & Anti-IL Monoclonal Antibodies Drive Premium Market Positioning?
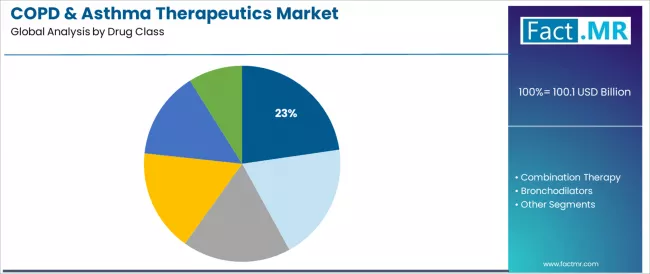
The anti-IgE & anti-IL monoclonal antibodies segment is projected to account for 22.7% of the COPD & asthma therapeutics market in 2025, reaffirming its position as the category's leading drug class specification. Respiratory specialists increasingly recognize the optimal balance of targeted efficacy and transformative clinical outcomes offered by biologic antibody therapies for severe asthma management applications, particularly in type 2 inflammatory phenotypes and difficult-to-control disease presentations.
This drug class addresses both eosinophilic inflammation requirements and IgE-mediated pathway blockade while providing superior exacerbation reduction across diverse severe asthma populations. This segment forms the foundation of severe asthma treatment protocols for biologic-eligible patients and specialty respiratory care, as it represents the most clinically transformative and evidence-based drug category in the respiratory therapeutics industry.
Clinical trial validation standards and extensive real-world effectiveness data continue to strengthen confidence in monoclonal antibody therapies among respiratory specialists and pulmonology professionals. With increasing recognition of biologic therapy impact on hospitalization reduction and oral corticosteroid sparing requirements, anti-IgE & anti-IL monoclonal antibodies align with both current specialty practice standards and precision medicine evolution goals, making them the central premium growth driver of comprehensive severe asthma management strategies across multiple healthcare platforms.
Combination therapy represents 19.3% of drug class applications, reflecting significant clinical utility in optimizing multi-pathway disease control and improving medication adherence through fixed-dose formulations. Bronchodilators account for a substantial portion of prescriptions, driven by first-line COPD management and acute symptom relief requirements.
Corticosteroids represent foundational anti-inflammatory therapy across disease severities, while leukotriene modifiers and other drug classes encompass specialized therapeutic options including phosphodiesterase inhibitors, anticholinergics, and emerging targeted agents requiring distinct mechanism interventions.
Why do Inhalers Command a Dominant Market Share?
Inhalers are projected to represent 46.5% of COPD & asthma therapeutics demand in 2025, underscoring its role as the primary delivery system category driving market adoption and respiratory therapy deployment. Healthcare providers recognize that inhaler device requirements, including portability convenience, direct lung delivery, and comprehensive medication formulation compatibility, provide the largest addressable market that alternative delivery systems cannot match in patient preference and therapeutic versatility.
Inhalers utilized in respiratory care offer enhanced patient autonomy and broad medication compatibility essential for serving maintenance therapy requirements and acute symptom management applications. The segment is supported by the expanding nature of global ambulatory respiratory care development, requiring delivery devices capable of accommodating diverse medication types, varied patient capabilities, and comprehensive therapeutic regimens, and the increasing recognition that advanced inhaler technologies can improve medication deposition and treatment adherence.
Practitioners are increasingly adopting smart inhaler platforms that integrate digital health tracking for optimal adherence monitoring and treatment optimization. As understanding of drug delivery technology capabilities advances and patient-centric care standards become more sophisticated, inhaler-based delivery systems will continue to play a crucial role in comprehensive respiratory disease management differentiation strategies.
Dry powder inhalers, metered dose inhalers, and soft mist inhalers represent distinct inhaler subcategories addressing specific patient populations and medication formulation requirements. Nebulizers represent 18.8% of device applications, reflecting critical utility in pediatric populations, severe exacerbations, and patients unable to coordinate inhaler techniques. Oxygen therapy devices comprise the remaining segment, addressing hypoxemia management in advanced COPD and respiratory failure applications requiring supplemental oxygen delivery.
What drives Combination Therapy Drug Class Prominence?
The combination therapy drug class is projected to account for 19.3% of the COPD & asthma therapeutics market in 2025, establishing its position as a leading therapeutic approach category. Respiratory practitioners increasingly recognize that combination formulations, encompassing fixed-dose combinations of anti-inflammatory and bronchodilator agents, represent highly clinically significant and adherence-optimized therapeutic options requiring synergistic mechanism integration due to disease complexity and treatment goals.
This drug class addresses both inflammation control requirements and bronchodilation needs while delivering superior convenience outcomes across varied patient populations. The segment is supported by the continuing nature of chronic respiratory disease management in both asthma and COPD patient populations, driven by multifactorial disease pathophysiology, adherence optimization requirements, and therapeutic guideline recommendations supporting combination approaches.
Respiratory practitioners are increasingly focusing on triple therapy combinations that enhance treatment comprehensiveness and reduce pill burden while maintaining safety standards. As respiratory care sophistication expands and treatment algorithms evolve, combination therapy approaches will continue to serve a crucial role in ensuring optimal disease control and medication adherence within the global respiratory therapeutics market.
Bronchodilators represent a substantial portion of therapeutic interventions, reflecting foundational treatment across disease severities and stages. Corticosteroids account for essential anti-inflammatory therapy requirements, driven by inflammation control priorities and exacerbation prevention.
Leukotriene modifiers represent alternative anti-inflammatory pathways, addressing specific patient phenotypes, while other drug classes account for specialized mechanisms including emerging biologics, targeted small molecules, and adjunctive therapies requiring distinct pharmacological interventions.
What are the Drivers, Restraints, and Key Trends of the COPD & Asthma Therapeutics Market?
The COPD & asthma therapeutics market is advancing rapidly due to increasing recognition of chronic respiratory disease burden and growing demand for specialized pharmaceutical solutions across the respiratory healthcare sector.
The market faces challenges, including high biologic therapy costs in price-sensitive healthcare systems, patent expiration pressures on blockbuster respiratory medications, and concerns about inhaler environmental impact in sustainability-focused markets. Innovation in biologic mechanisms and advanced drug delivery algorithms continues to influence therapeutic development and market expansion patterns.
Proliferation of Advanced Biologic Therapies and Targeted Mechanisms
The accelerating adoption of sophisticated biologic platforms is enabling the development of more precise respiratory therapeutic applications and treatment protocols that can meet stringent efficacy and safety requirements.
Respiratory specialists demand comprehensive biologic integration for severe disease management, including phenotype-specific targeting capabilities and multi-pathway modulation formulations that are particularly important for achieving superior exacerbation reduction requirements in complex asthma applications.
Advanced biologic technologies provide access to targeted immune pathway intervention that can optimize treatment response strategies and enhance patient quality of life while maintaining safety profiles for diverse respiratory disease populations.
Integration of Digital Health Platforms and Smart Inhaler Capabilities
Modern pharmaceutical organizations are incorporating advanced technologies such as connected inhaler devices, real-time adherence monitoring capabilities, and digital patient engagement interfaces to enhance therapeutic effectiveness and treatment optimization.
These systems improve medication compliance, enable seamless patient-provider communication transitions, and provide better integration between prescribed therapies and actual medication usage throughout the treatment and management experience.
Advanced digital health capabilities also enable personalized treatment adjustments and early identification of disease deterioration or adherence challenges, supporting proactive intervention management and improved respiratory disease control outcomes.
Analysis of the COPD & Asthma Therapeutics Market by Key Countries
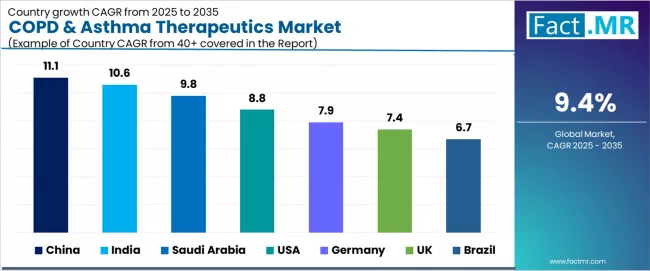
| Country | CAGR (2025-2035) |
|---|---|
| China | 11.1% |
| India | 10.6% |
| Saudi Arabia | 9.8% |
| USA | 8.8% |
| Germany | 7.9% |
| UK | 7.4% |
| Brazil | 6.7% |
The COPD & asthma therapeutics market is experiencing exceptional growth globally, with China leading at an 11.1% CAGR through 2035, driven by extremely high COPD prevalence, expanding healthcare access, and increasing government health initiatives across major urban and industrial centers. India follows at 10.6%, supported by rapid generic medication expansion, rising respiratory disease awareness, and comprehensive digital diagnostic infrastructure initiatives.
Saudi Arabia records 9.8% growth, benefiting from urban pollution challenges and healthcare system digitization programs. USA demonstrates 8.8% growth, emphasizing largest biologics uptake and smart inhaler technology adoption. Germany shows 7.9% growth with strong reimbursement frameworks and pharmaceutical R&D excellence. UK records 7.4% growth, representing focus on low-carbon inhaler initiatives and NHS procurement optimization, while Brazil shows 6.7% growth, representing expanding treatment access and increased disease diagnosis rates.
How does China Demonstrate Exceptional Market Leadership with COPD Prevalence?
The COPD & asthma therapeutics market in China is projected to exhibit exceptional growth with a CAGR of 11.1% through 2035, driven by the world's highest COPD disease burden and increasing recognition of respiratory disease management as an essential component for public health improvement and healthcare system modernization. The country's massive patient population and growing availability of specialized respiratory care infrastructure are creating significant opportunities for therapeutic deployment across both urban tertiary hospitals and emerging rural healthcare segments.
Major international pharmaceutical manufacturers and domestic producers are establishing comprehensive market presence to serve the expanding population of COPD and asthma patients requiring advanced respiratory medications across inhalation therapies, biologic treatments, and combination formulations throughout China's diverse metropolitan and regional healthcare systems. The government's strategic emphasis on healthcare reform advancement and essential medicine access is driving substantial investments in respiratory disease management capabilities and pharmaceutical coverage availability.
This policy support, combined with the country's enormous patient population and growing healthcare insurance penetration, creates a favorable environment for respiratory therapeutics market development. Pharmaceutical manufacturers are increasingly focusing on domestic biologic development capacity to improve treatment access, with advanced respiratory therapies representing a key component in this healthcare capability building.
- Government initiatives supporting universal health coverage expansion and chronic disease management enhancement are driving demand for COPD & asthma therapeutic solutions across hospital and primary care segments
- Healthcare infrastructure expansion and respiratory specialty advancement are supporting appropriate utilization of advanced therapies among practitioners and healthcare facilities nationwide
- Patients and healthcare providers are increasingly recognizing respiratory disease control importance in quality-of-life improvement, creating new treatment demand capabilities
- Rising healthcare expenditure and growing awareness of exacerbation prevention benefits are accelerating respiratory therapeutic adoption across patient categories
What makes India Demonstrate Strong Market Potential with Generic Expansion?
The COPD & asthma therapeutics market in India is expanding at a CAGR of 10.6%, supported by rapid generic medication manufacturing capacity, expanding respiratory disease diagnosis rates, and advancing digital health infrastructure integration across the country's developing urban and rural healthcare corridors. The country's substantial pharmaceutical production capability and increasing sophistication of respiratory diagnostic platforms are driving demand for specialized therapeutic solutions in both asthma and COPD management applications.
International pharmaceutical companies and domestic generic manufacturers are establishing market capacity to serve the growing demand for affordable respiratory medications while supporting the country's position as a leading generic pharmaceutical manufacturing hub. India's pharmaceutical sector continues to benefit from favorable manufacturing policies, expanding healthcare access programs, and growing middle-class healthcare spending.
The country's focus on expanding generic medication availability is driving investments in critical respiratory therapy production including inhaled corticosteroids, bronchodilators, and combination therapies. This development is particularly important for respiratory disease applications, as healthcare systems seek cost-effective treatment sources for medication supply to enhance disease management capabilities and reduce treatment cost barriers.
- Rising respiratory disease awareness and improving domestic pharmaceutical manufacturing capabilities are creating opportunities for advanced generic therapeutic solutions
- Growing healthcare access and respiratory diagnostic modernization are supporting increased deployment of affordable respiratory medications across patient categories
- Expanding pharmaceutical export capacity and emerging quality standards are driving innovative applications of generic respiratory therapeutics in domestic and international applications
- Manufacturing scale advantages are enabling delivery of cost-effective treatment protocols, supporting market growth and improving medication accessibility
How does Saudi Arabia Drive Regional Market Growth with Healthcare Modernization?
The COPD & asthma therapeutics market in Saudi Arabia is projected to exhibit strong growth with a CAGR of 9.8% through 2035, driven by rapid urbanization-related air quality challenges and comprehensive healthcare system digitization initiatives.
The country's position as a leading Gulf economy and increasing healthcare infrastructure investment are creating significant opportunities for respiratory therapeutic integration across both modern hospital systems and emerging specialized respiratory care applications. Pharmaceutical distributors and healthcare providers are leveraging regional development to serve the growing demand for respiratory medications while supporting the country's position as a regional healthcare hub.
The market benefits from substantial government healthcare investment supporting universal coverage expansion, enabling optimized respiratory care deployment and comprehensive therapeutic access. This development is particularly important for respiratory therapeutic applications, as healthcare systems seek advanced treatment solutions that maximize disease control while ensuring quality standards in specialized respiratory care and primary healthcare environments.
Strategic Market Considerations:
- Modern healthcare facilities and specialty respiratory care segments leading growth with focus on treatment quality and comprehensive disease management applications
- Healthcare investment priorities and digital health integration are driving diverse respiratory therapeutic portfolios from conventional bronchodilator treatments to advanced biologic therapy platforms
- Healthcare digitization excellence and regional hub positioning supporting competitive market development in Middle Eastern respiratory care markets
- Air quality concerns and urbanization patterns beginning to influence disease prevalence and therapeutic demand timelines
What drives the USA’s Market Growth with Biologics Leadership?

The USA's advanced pharmaceutical market demonstrates sophisticated COPD & asthma therapeutic deployment with documented effectiveness in specialty respiratory care platforms and primary care applications through integration with cutting-edge biologic therapies and smart inhaler technologies.
The country leverages pharmaceutical innovation leadership and comprehensive healthcare infrastructure to maintain an 8.8% CAGR through 2035. Specialty respiratory centers, including major academic medical centers in metropolitan regions and integrated healthcare systems, showcase advanced therapeutic implementations where sophisticated biologic agents integrate with comprehensive disease management programs and ongoing clinical research to optimize treatment effectiveness and patient outcomes.
Healthcare providers prioritize clinical evidence and innovative therapeutic options in respiratory disease management, creating demand for premium biologic therapies with advanced capabilities, including targeted monoclonal antibodies and integration with comprehensive care coordination systems. The market benefits from established pharmaceutical innovation infrastructure and willingness to invest in breakthrough therapies that provide superior clinical outcomes and compliance with stringent FDA standards and evidence-based medicine expectations.
Strategic Market Considerations:
- Specialty biologic and smart inhaler segments leading growth with focus on advanced therapeutic mechanisms and comprehensive digital health applications
- High innovation adoption rates are driving sophisticated therapeutic portfolios from conventional maintenance treatments to cutting-edge precision medicine platforms
- Pharmaceutical R&D excellence and clinical trial leadership supporting continued innovation in respiratory therapeutic delivery
- Payer coverage frameworks and value-based care models ensuring appropriate biologic access and outcome optimization
How does Germany Maintain Pharmaceutical Excellence with Strong Reimbursement?
Germany's advanced pharmaceutical market demonstrates sophisticated COPD & asthma therapeutic utilization with documented effectiveness in comprehensive disease management platforms and specialty care applications through integration with cutting-edge treatment protocols and evidence-based guidelines.
The country leverages pharmaceutical industry excellence and comprehensive health insurance infrastructure to maintain a 7.9% CAGR through 2035. Respiratory specialty clinics, including major university hospitals in leading medical centers, showcase advanced therapeutic implementations where sophisticated medication regimens integrate with comprehensive patient education programs and ongoing outcomes research to optimize treatment adherence and disease control.
German healthcare providers prioritize evidence-based medicine and long-term treatment outcomes in respiratory disease management, creating demand for high-quality therapeutic solutions with proven efficacy, including advanced inhalation systems and integration with German healthcare quality standards. The market benefits from established pharmaceutical research infrastructure and commitment to comprehensive reimbursement frameworks that ensure patient access to innovative therapies.
Strategic Market Considerations:
- Comprehensive disease management and specialty care segments leading growth with focus on evidence-based protocols and quality outcome applications
- Stringent reimbursement assessment requirements are driving sophisticated therapeutic portfolios from conventional respiratory medications to advanced targeted therapy platforms
- Pharmaceutical research excellence and health technology assessment leadership supporting competitive positioning in European respiratory markets
- Quality standards and outcome-based evaluation emphasizing proven efficacy and comprehensive cost-effectiveness in therapeutic applications
What drives UK’s Market Growth with Environmental Sustainability Focus?
The UK's expanding COPD & asthma therapeutics market demonstrates accelerating therapeutic adoption with a 7.4% CAGR through 2035, driven by environmental sustainability initiatives, low-carbon inhaler development, and comprehensive NHS procurement optimization across major respiratory care regions.
The country's emphasis on healthcare sustainability and clinical effectiveness is creating substantial demand for environmentally conscious respiratory therapeutic solutions across diverse hospital and primary care platforms. UK healthcare providers and pharmaceutical suppliers are increasingly prioritizing respiratory medications that incorporate reduced environmental impact for optimal clinical outcomes and carbon footprint reduction.
Market dynamics focus on sustainable respiratory therapeutic solutions that balance clinical effectiveness with environmental considerations important to UK healthcare policies and climate change commitments. Growing focus on propellant-free inhalers and dry powder formulations creates opportunities for integrated sustainability programs and therapeutic innovation deployment.
Strategic Market Considerations:
- Primary care and respiratory specialty segments demonstrating focused growth with emphasis on sustainable prescribing and comprehensive disease management applications
- Environmental sustainability requirements driving therapeutic preferences toward low-carbon inhaler specifications with reduced greenhouse gas characteristics
- Clinical guideline development supported by NHS sustainability initiatives and pharmaceutical environmental responsibility
- Healthcare provider preferences emphasizing proven clinical efficacy and comprehensive environmental impact assessment in respiratory therapeutic applications
How does Brazil Demonstrate Latin American Market Expansion?
The COPD & asthma therapeutics market in Brazil is projected to exhibit moderate growth with a CAGR of 6.7% through 2035, driven by expanding universal healthcare coverage and improving respiratory disease diagnosis rates. The country's position as the largest Latin American pharmaceutical market and growing healthcare access programs are creating opportunities for respiratory therapeutic integration across both public health system and private healthcare applications.
Brazilian pharmaceutical distributors and healthcare providers are leveraging regional market leadership to serve the demand for respiratory medications while supporting the country's position as a major pharmaceutical economy. The market benefits from established generic pharmaceutical infrastructure supporting affordable medication access, enabling optimized respiratory therapy deployment and comprehensive treatment availability. This development is particularly important for respiratory therapeutic applications, as healthcare systems seek cost-effective medication solutions that maximize disease control while ensuring treatment accessibility in diverse socioeconomic healthcare environments.
Strategic Market Considerations:
- Public health system and primary care segments leading growth with focus on treatment access expansion and diagnosis improvement applications
- Healthcare access economics and universal coverage expansion are driving diverse respiratory therapeutic portfolios from essential bronchodilator treatments to combination therapy platforms
- Generic pharmaceutical expertise and regional distribution capabilities supporting competitive positioning in Latin American respiratory markets
- Healthcare access initiatives and disease awareness programs beginning to influence treatment utilization patterns and market growth trajectories
Europe Market Split by Country
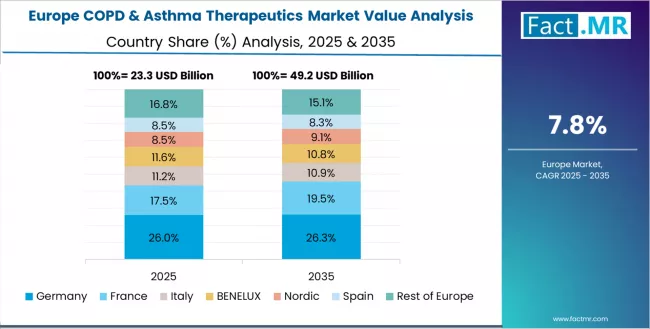
The COPD & asthma therapeutics market in Europe is projected to grow from USD 24.8 billion in 2025 to USD 48.3 billion by 2035, registering a CAGR of 6.9% over the forecast period. Germany is expected to maintain its leadership position with a 28.4% market share in 2025, rising to 29.8% by 2035, supported by its dominant pharmaceutical industry infrastructure, comprehensive health insurance coverage capabilities, and advanced respiratory care excellence throughout major university hospitals and primary care networks.
The UK follows with a 23.6% share in 2025, projected to reach 22.9% by 2035, driven by NHS-based universal healthcare delivery, pharmaceutical procurement sophistication, and environmental sustainability emphasis serving both domestic and European markets. France holds a 19.7% share in 2025, expected to increase to 20.1% by 2035, supported by comprehensive healthcare system infrastructure and respiratory specialty capabilities.
Italy commands a 13.8% share in 2025, projected to reach 13.2% by 2035, while Spain accounts for 10.2% in 2025, expected to reach 9.7% by 2035. The Rest of Europe region, including Nordic countries with advanced healthcare quality standards, Eastern European emerging pharmaceutical markets, and smaller Western European respiratory care centers, is anticipated to hold 4.3% in 2025, declining slightly to 4.3% by 2035, attributed to market consolidation toward larger core markets with established pharmaceutical distribution infrastructure and respiratory care expertise.
Competitive Landscape of the COPD & Asthma Therapeutics Market
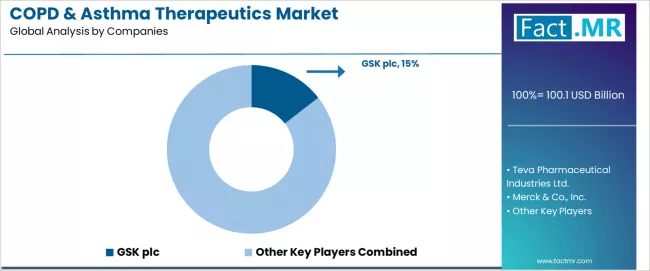
The COPD & asthma therapeutics market is characterized by intense competition among established pharmaceutical giants, specialized respiratory medicine companies, and comprehensive biopharmaceutical organizations focused on delivering high-quality, effective, and accessible respiratory therapeutic solutions.
Companies are investing in clinical research programs, advanced biologic development, strategic market access partnerships, and comprehensive patient support initiatives to deliver effective, safe, and accessible respiratory medications that meet stringent regulatory standards and healthcare provider expectations. Product innovation, patent portfolio management, and market access optimization strategies are central to strengthening therapeutic portfolios and market presence.
GSK plc leads the market with a 14.5% market share, offering comprehensive COPD & asthma therapeutic solutions with a focus on respiratory disease expertise and advanced inhaler technology capabilities for diverse patient applications. Teva Pharmaceutical provides specialized generic respiratory platforms with emphasis on affordable medication access and comprehensive primary care support across global markets.
Merck & Co focuses on innovative therapeutic development and comprehensive respiratory disease research serving international healthcare communities. F. Hoffmann-La Roche delivers biologic therapy excellence with strong monoclonal antibody development capabilities and severe asthma treatment integration.
AstraZeneca operates with a focus on respiratory disease franchise leadership and comprehensive inhaler technology innovation for specialty and primary care applications. Boehringer Ingelheim provides COPD therapeutic expertise emphasizing long-acting bronchodilator development and comprehensive disease management solutions. Sanofi specializes in biologic therapy innovation and severe asthma treatment with emphasis on targeted mechanism development.
Royal Philips delivers respiratory care device solutions to enhance therapeutic delivery and provide comprehensive patient monitoring. BD, Novartis, Cipla, Chiesi, Covis Pharma, and Findair focus on specialized delivery devices, generic medication access, and regional therapeutic solutions, emphasizing product quality and comprehensive respiratory care protocols through dedicated pharmaceutical strategies.
Key Players in the COPD & Asthma Therapeutics Market
- GSK plc
- Teva Pharmaceutical Industries Ltd.
- Merck & Co., Inc.
- F. Hoffmann-La Roche Ltd
- AstraZeneca plc
- Boehringer Ingelheim International GmbH
- Sanofi S.A.
- Royal Philips (Koninklijke Philips N.V.)
- Becton, Dickinson and Company (BD)
- Novartis AG
- Cipla Limited
- Chiesi Farmaceutici S.p.A.
- Covis Pharma Group
- FindAir Sp. z o.o.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD 100.1 Billion |
| Drug Class | Anti-IgE & Anti-IL Monoclonal Antibodies, Combination Therapy, Bronchodilators, Corticosteroids, Leukotriene Modifiers, Others |
| Device/Product Type | Inhalers (Dry Powder Inhalers, Metered Dose Inhalers, Soft Mist Inhalers), Nebulizers, Oxygen Therapy Devices |
| Regions Covered | Asia Pacific, North America, Europe, Latin America, Middle East & Africa |
| Countries Covered | USA, Germany, UK, Japan, India, China, Brazil, Saudi Arabia and 40+ countries |
| Key Companies Profiled | GSK plc, Teva Pharmaceutical, Merck & Co, F. Hoffmann-La Roche, AstraZeneca, Boehringer Ingelheim, Sanofi, Royal Philips, BD, Novartis, Cipla, Chiesi, Covis Pharma, Findair |
| Additional Attributes | Dollar sales by drug class, device/product type, regional demand trends, competitive landscape, healthcare provider preferences for specific therapeutic agents, integration with comprehensive disease management systems, innovations in biologic mechanism development, drug delivery technology advancement, and patient outcome optimization capabilities |
COPD & Asthma Therapeutics Market by Segments
-
Drug Class :
- Anti-IgE & Anti-IL Monoclonal Antibodies
- Combination Therapy
- Bronchodilators
- Corticosteroids
- Leukotriene Modifiers
- Others
-
Device/Product Type :
- Inhalers
- Dry Powder Inhalers
- Metered Dose Inhalers
- Soft Mist Inhalers
- Nebulizers
- Oxygen Therapy Devices
- Inhalers
-
Region :
-
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
-
North America
- United States
- Canada
- Mexico
-
Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
-
Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
-
Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Countries
- Rest of Middle East & Africa
-
- Frequently Asked Questions -
How big is the COPD & asthma therapeutics market in 2025?
The global COPD & asthma therapeutics market is estimated to be valued at USD 100.1 billion in 2025.
What will be the size of COPD & asthma therapeutics market in 2035?
The market size for the COPD & asthma therapeutics market is projected to reach USD 245.9 billion by 2035.
How much will be the COPD & asthma therapeutics market growth between 2025 and 2035?
The COPD & asthma therapeutics market is expected to grow at a 9.4% CAGR between 2025 and 2035.
What are the key product types in the COPD & asthma therapeutics market?
The key product types in COPD & asthma therapeutics market are anti-ige & anti-il monoclonal antibodies, combination therapy, bronchodilators, corticosteroids, leukotriene modifiers and others.
Which device/product type segment to contribute significant share in the COPD & asthma therapeutics market in 2025?
In terms of device/product type, inhalers segment to command 46.5% share in the COPD & asthma therapeutics market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Drug Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Class, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Class, 2025 to 2035
- Anti-IgE & Anti-IL Monoclonal Antibodies
- Combination Therapy
- Bronchodilators
- Corticosteroids
- Leukotriene Modifiers
- Others
- Y to o to Y Growth Trend Analysis By Drug Class, 2020 to 2024
- Absolute $ Opportunity Analysis By Drug Class, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Device/Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Device/Product Type, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Device/Product Type, 2025 to 2035
- Inhalers
- Nebulizers
- Oxygen Therapy Devices
- Y to o to Y Growth Trend Analysis By Device/Product Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Device/Product Type, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Drug Class
- By Device/Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Device/Product Type
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Drug Class
- By Device/Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Device/Product Type
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Drug Class
- By Device/Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Device/Product Type
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Drug Class
- By Device/Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Device/Product Type
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Drug Class
- By Device/Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Device/Product Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Drug Class
- By Device/Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Device/Product Type
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Drug Class
- By Device/Product Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Class
- By Device/Product Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Class
- By Device/Product Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Drug Class
- By Device/Product Type
- Competition Analysis
- Competition Deep Dive
- GSK plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Teva Pharmaceutical Industries Ltd.
- Merck & Co., Inc.
- F. Hoffmann-La Roche Ltd
- AstraZeneca plc
- Boehringer Ingelheim International GmbH
- Sanofi S.A.
- Royal Philips (Koninklijke Philips N.V.)
- Becton, Dickinson and Company (BD)
- Novartis AG
- Cipla Limited
- Chiesi Farmaceutici S.p.A.
- Covis Pharma Group
- FindAir Sp. z o.o.
- GSK plc
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Global Market Value (USD Million) Forecast by Drug Class, 2020 to 2035
- Table 3: Global Market Value (USD Million) Forecast by Device/Product Type, 2020 to 2035
- Table 4: North America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: North America Market Value (USD Million) Forecast by Drug Class, 2020 to 2035
- Table 6: North America Market Value (USD Million) Forecast by Device/Product Type, 2020 to 2035
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 8: Latin America Market Value (USD Million) Forecast by Drug Class, 2020 to 2035
- Table 9: Latin America Market Value (USD Million) Forecast by Device/Product Type, 2020 to 2035
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 11: Western Europe Market Value (USD Million) Forecast by Drug Class, 2020 to 2035
- Table 12: Western Europe Market Value (USD Million) Forecast by Device/Product Type, 2020 to 2035
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Drug Class, 2020 to 2035
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Device/Product Type, 2020 to 2035
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 17: East Asia Market Value (USD Million) Forecast by Drug Class, 2020 to 2035
- Table 18: East Asia Market Value (USD Million) Forecast by Device/Product Type, 2020 to 2035
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Drug Class, 2020 to 2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Device/Product Type, 2020 to 2035
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Drug Class, 2020 to 2035
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Device/Product Type, 2020 to 2035
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020 to 2035
- Figure 3: Global Market Value Share and BPS Analysis by Drug Class, 2025 and 2035
- Figure 4: Global Market Y to o to Y Growth Comparison by Drug Class, 2025 to 2035
- Figure 5: Global Market Attractiveness Analysis by Drug Class
- Figure 6: Global Market Value Share and BPS Analysis by Device/Product Type, 2025 and 2035
- Figure 7: Global Market Y to o to Y Growth Comparison by Device/Product Type, 2025 to 2035
- Figure 8: Global Market Attractiveness Analysis by Device/Product Type
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2025 to 2035
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2025 to 2035
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: North America Market Value Share and BPS Analysis by Drug Class, 2025 and 2035
- Figure 21: North America Market Y to o to Y Growth Comparison by Drug Class, 2025 to 2035
- Figure 22: North America Market Attractiveness Analysis by Drug Class
- Figure 23: North America Market Value Share and BPS Analysis by Device/Product Type, 2025 and 2035
- Figure 24: North America Market Y to o to Y Growth Comparison by Device/Product Type, 2025 to 2035
- Figure 25: North America Market Attractiveness Analysis by Device/Product Type
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 27: Latin America Market Value Share and BPS Analysis by Drug Class, 2025 and 2035
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Drug Class, 2025 to 2035
- Figure 29: Latin America Market Attractiveness Analysis by Drug Class
- Figure 30: Latin America Market Value Share and BPS Analysis by Device/Product Type, 2025 and 2035
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Device/Product Type, 2025 to 2035
- Figure 32: Latin America Market Attractiveness Analysis by Device/Product Type
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 34: Western Europe Market Value Share and BPS Analysis by Drug Class, 2025 and 2035
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Drug Class, 2025 to 2035
- Figure 36: Western Europe Market Attractiveness Analysis by Drug Class
- Figure 37: Western Europe Market Value Share and BPS Analysis by Device/Product Type, 2025 and 2035
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Device/Product Type, 2025 to 2035
- Figure 39: Western Europe Market Attractiveness Analysis by Device/Product Type
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Drug Class, 2025 and 2035
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Drug Class, 2025 to 2035
- Figure 43: Eastern Europe Market Attractiveness Analysis by Drug Class
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Device/Product Type, 2025 and 2035
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Device/Product Type, 2025 to 2035
- Figure 46: Eastern Europe Market Attractiveness Analysis by Device/Product Type
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 48: East Asia Market Value Share and BPS Analysis by Drug Class, 2025 and 2035
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Drug Class, 2025 to 2035
- Figure 50: East Asia Market Attractiveness Analysis by Drug Class
- Figure 51: East Asia Market Value Share and BPS Analysis by Device/Product Type, 2025 and 2035
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Device/Product Type, 2025 to 2035
- Figure 53: East Asia Market Attractiveness Analysis by Device/Product Type
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Drug Class, 2025 and 2035
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Drug Class, 2025 to 2035
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Drug Class
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Device/Product Type, 2025 and 2035
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Device/Product Type, 2025 to 2035
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Device/Product Type
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Drug Class, 2025 and 2035
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Drug Class, 2025 to 2035
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Drug Class
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Device/Product Type, 2025 and 2035
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Device/Product Type, 2025 to 2035
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Device/Product Type
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis