Lipid Nanoparticle Raw Materials Market Size, Share, Growth and Forecast (2026 - 2036)
Lipid Nanoparticle Raw Materials Market is segmented by Application (Therapeutics, Research), Product (Kits, Reagents, Other Raw Materials), Disease Indication (Cancer, Infectious Diseases, Blood Diseases, Others), End-use (Pharmaceutical & Biotechnology Companies, Academic & Research Institutes, Others), and Region. Forecast for 2026 to 2036.
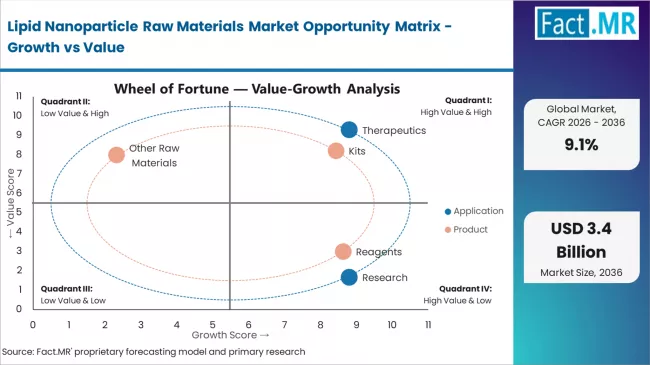
Fact.MR opines the lipid nanoparticle raw materials market was valued at USD 1.32 billion in 2025. Sales are expected to reach USD 1.44 billion in 2026 and USD 3.44 billion by 2036. Kits is poised to lead by Product with 54.5% share in 2026 and Therapeutics is estimated to command by Application in 2026.
Lipid Nanoparticle Raw Materials Market Size, Market Forecast and Outlook By Fact.MR
The lipid nanoparticle raw materials market was valued at USD 1.32 billion in 2025, projected to reach USD 1.44 billion in 2026, and is forecast to expand to USD 3.44 billion by 2036 at a 9.1% CAGR. Expanding mRNA therapeutic and vaccine development pipelines are generating sustained procurement demand for ionizable lipids, PEGylated lipids, helper lipids, and cholesterol formulations that constitute the encapsulation matrix of lipid nanoparticle delivery systems. Drug developers that previously managed lipid sourcing on a per-project basis now face clinical-stage pipeline volumes that require formalized supply agreements with qualified lipid manufacturers.

Summary of Lipid Nanoparticle Raw Materials
- Market Overview
- The lipid nanoparticle raw materials market is valued at USD 1.32 billion in 2025 and is projected to reach USD 3.44 billion by 2036.
- The industry is expected to grow at a 9.1% CAGR from 2026 to 2036, creating an incremental opportunity of USD 2.00 billion.
- The market is a purity-critical, IP-constrained specialty biochemical category where ionizable lipid synthesis capability, GMP compliance, and pharmaceutical supply chain qualification define competitive positioning.
- Demand and Growth Drivers
- Demand is rising as mRNA therapeutic and vaccine development pipelines expand beyond infectious disease into oncology, rare disease, and gene editing applications requiring lipid nanoparticle encapsulation.
- Clinical-stage manufacturing scale-up is generating procurement volume increases that force drug developers to formalize long-term lipid supply agreements with GMP-qualified manufacturers.
- Government pandemic preparedness programs are creating strategic lipid raw material stockpiling demand in the United States, Japan, and European Union member states.
- Among key countries, China leads at 10.0% CAGR, followed by the United States, Japan, and South Korea, each scaling lipid procurement for expanding LNP therapeutic pipelines.
- Product and Segment View
- The market covers ionizable lipids, PEGylated lipids, helper lipids (DSPC, DOPE), and cholesterol formulations used as encapsulation matrix components in lipid nanoparticle drug delivery systems.
- Kits lead by Product with 54.5% share in 2026, serving preclinical and early-stage clinical researchers requiring pre-formulated lipid component sets for LNP assembly and screening.
- Therapeutics leads by Application with 61.4% share in 2026, driven by the expanding pipeline of mRNA-based drugs, gene therapies, and siRNA therapeutics advancing through clinical development.
- Cancer leads by Disease Indication with 37.9% share in 2026, anchored by oncology mRNA vaccine and therapeutic programs requiring patient-specific or tumor-targeted LNP formulations.
- Scope includes ionizable, PEGylated, and helper lipid raw materials and formulation kits, while excluding finished lipid nanoparticle drug products, mRNA active pharmaceutical ingredients, and non-lipid delivery system excipients.
- Geography and Competitive Outlook
- China and the United States are the primary growth centers, with China leading on domestic mRNA pipeline expansion and the United States maintaining the largest global portfolio of LNP-based clinical candidates.
- Japan and South Korea sustain demand through oncology mRNA programs, pandemic preparedness stockpiling, and gene therapy development requiring GMP-grade lipid raw materials.
- Competition is shaped by ionizable lipid synthesis IP, GMP manufacturing scale, and pharmaceutical customer qualification, with key players including Merck KGaA, Echelon Biosciences, BroadPharm, Avanti Polar Lipids, Tebubio, Creative Biolabs, CordenPharma International, Biopharma PEG Scientific Inc., NOF America Corporation, and Polysciences Inc.
- Analyst Opinion at Fact.MR
- Shambhu Nath Jha , Principal Consultant for Life Sciences, opines: 'In my analysis, I have observed that the supply chain bottleneck for lipid nanoparticle therapeutics has migrated from mRNA manufacturing to lipid raw material qualification. Drug developers advancing LNP-based candidates through Phase II and Phase III trials face lipid supplier audit timelines that extend clinical program schedules when dual-sourcing qualification has not been initiated during preclinical stages. Pharmaceutical procurement teams that delay GMP lipid supply agreement execution risk pipeline delays that compound across every subsequent clinical and regulatory milestone.'
- Strategic Implications / Executive Takeaways
- Lipid manufacturers must invest in GMP-grade ionizable lipid synthesis capacity and regulatory filing support capabilities to qualify for pharmaceutical supply agreements tied to late-stage clinical programs.
- Drug developers should initiate dual-source lipid supplier qualification during preclinical development to avoid single-source dependency risks that delay clinical and commercial manufacturing timelines.
- Research procurement teams must validate lipid kit lot-to-lot consistency specifications to ensure screening results generated with commercial-grade materials remain transferable to GMP manufacturing.
Lipid Nanoparticle Raw Materials Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.44 billion |
| Industry Value (2036) | USD 3.44 billion |
| CAGR (2026 to 2036) | 9.1% |
Source: Fact.MR, 2026
The USD 2.00 billion incremental opportunity over the forecast period reflects the broadening of lipid nanoparticle applications from infectious disease vaccines into oncology, rare disease, and gene editing therapeutic programs. Pharmaceutical companies scaling from preclinical to commercial manufacturing volumes encounter lipid purity and batch consistency requirements that compress the qualified supplier base. Ionizable lipid synthesis complexity and intellectual property constraints around proprietary lipid structures create supply concentration risks that drug developers mitigate through dual-sourcing qualification programs.
All monitored geographies confirm expanding lipid raw material procurement trajectories. China sets the pace at a 10.0% CAGR, propelled by domestic mRNA vaccine and therapeutic development programs scaling through clinical trials. The United States maintains strong growth supported by the largest global pipeline of LNP-based therapeutic candidates in clinical development. Japan sustains demand through oncology-focused mRNA therapeutic programs and government-funded pandemic preparedness lipid stockpiling initiatives. South Korea advances through biosimilar and gene therapy development programs requiring GMP-grade lipid raw materials.
Why is the Lipid Nanoparticle Raw Materials Market Experiencing Rapid Growth?
The lipid nanoparticle raw materials market grows by enabling pharmaceutical companies, biotechnology firms, and research institutes to access high-quality delivery system components that support therapeutic innovation while meeting industry demand for effective drug delivery platforms. Pharmaceutical and biotechnology companies face mounting pressure to develop advanced therapeutics with proven efficacy and safety profiles, with lipid nanoparticle formulations typically providing enhanced cellular uptake and targeted delivery capabilities that improve therapeutic outcomes, making these materials essential for competitive product positioning in gene therapy and mRNA therapeutics categories. The pharmaceutical industry's need for versatile delivery systems and formulation compatibility creates demand for diverse lipid types that can provide superior encapsulation efficiency, maintain stability across different storage conditions, and ensure regulatory compliance without compromising drug potency or safety standards.
Government initiatives promoting advanced therapeutic development and biotechnology innovation drive adoption in mRNA vaccines, gene therapy applications, and targeted cancer treatments, where lipid nanoparticle technology has a direct impact on therapeutic efficacy and patient outcomes. The pharmaceutical industry's growing focus on personalized medicine and targeted drug delivery further expands market opportunities, with clinical research demonstrating measurable preference shifts toward lipid-based delivery systems, RNA therapeutics development, and reduced conventional small molecule dependence. However, supply chain complexity during specialized lipid sourcing and the technical requirements for pharmaceutical-grade manufacturing and analytical validation may limit accessibility among smaller biotechnology companies and developing regions with limited infrastructure for advanced lipid nanoparticle production and quality control systems.
Segmental Analysis
The market is segmented by application, product, disease indication, end-use, and region. By application, the market is divided into therapeutics and research. Based on product, the market is categorized into kits, reagents, and other raw materials. By disease indication, the market includes cancer, infectious diseases, blood diseases, and others. Based on end-use, the market covers pharmaceutical & biotechnology companies, academic & research institutes, and others. Regionally, the market is divided into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa.
What Makes Therapeutics the Dominant Application Segment in the Lipid Nanoparticle Raw Materials Market?
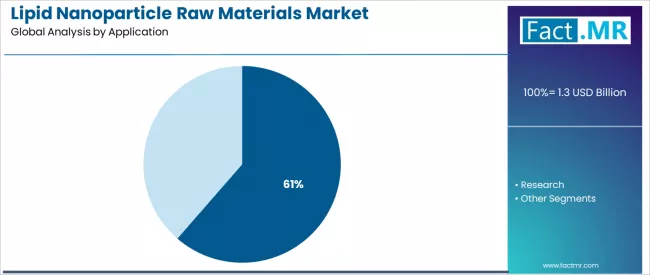
The therapeutics segment represents the dominant force in the lipid nanoparticle raw materials market, capturing approximately 61.4% of total market share in 2025. This established application category encompasses solutions featuring pharmaceutical-grade materials for commercial drug manufacturing operations, including mRNA vaccines and gene therapy products that enable large-scale production and consistent quality standards across all therapeutic manufacturing applications.
The therapeutics segment's market leadership stems from its commercial viability, with products capable of meeting stringent regulatory requirements while maintaining high purity standards and operational reliability across all pharmaceutical manufacturing environments. Within the therapeutics segment, mRNA vaccine applications account for substantial share, driven by successful COVID-19 vaccine deployment and expanding pipeline development.
The research segment maintains a substantial 38.6% market share, serving academic institutions and early-stage biotechnology companies who require high-quality materials for preclinical studies and formulation development applications. These applications offer critical innovation pathways for pharmaceutical discovery while providing sufficient versatility capabilities to meet diverse research protocols and experimental design requirements. The research segment demonstrates steady growth momentum, driven by increasing investment in novel therapeutic modalities and academic-industry collaborations for advanced drug delivery system development.
Key advantages driving the therapeutics segment include:
- Advanced manufacturing infrastructure with integrated pharmaceutical supply networks that reduce procurement complexity and ensure consistent material availability
- High-scale production capabilities allowing commercial manufacturing across different therapeutic categories without significant supply constraints
- Proven regulatory pathway, delivering compliant materials while maintaining quality standards required for commercial pharmaceutical production
- Broad therapeutic application enabling straightforward technology transfer and market adoption across multiple disease indications
How do Kits Lead the Product Segment?

Kits dominate the product segment with approximately 54.5% market share in 2025, reflecting the convenience and standardization benefits supporting lipid nanoparticle formulation across pharmaceutical development and research laboratories worldwide. The kits segment's market leadership is reinforced by comprehensive formulation solutions, consistent component quality, and reduced technical complexity for end-users implementing lipid nanoparticle technology across drug development and manufacturing applications. Within the kits segment, pre-formulated lipid mixtures account for significant adoption, driven by workflow efficiency and reproducibility advantages.
The reagents segment represents the second-largest product category, capturing 36.4% market share through individual lipid components and specialized materials for custom formulation development. This segment benefits from growing pharmaceutical manufacturer preference for flexible formulation approaches that meet specific therapeutic requirements, intellectual property strategies, and proprietary delivery system protocols in competitive drug development markets. Reagents demonstrate strong adoption among established pharmaceutical companies with in-house formulation expertise.
The other raw materials segment holds 9.1% market share, serving specialized applications requiring excipients, stabilizers, and custom synthesis services. This segment serves niche requirements and emerging technology platforms exploring novel lipid structures and formulation approaches.
Key market dynamics supporting product segment growth include:
- Kits expansion driven by ease of use and standardization benefits, requiring minimal formulation expertise for research applications
- Reagents adoption trends require high-quality, customizable components for proprietary formulation development and manufacturing optimization
- Integration of technical support services enabling successful technology adoption and formulation troubleshooting
- Growing emphasis on product diversity driving demand for comprehensive, application-specific material solutions
What Drives Cancer's Leadership in Disease Indication?
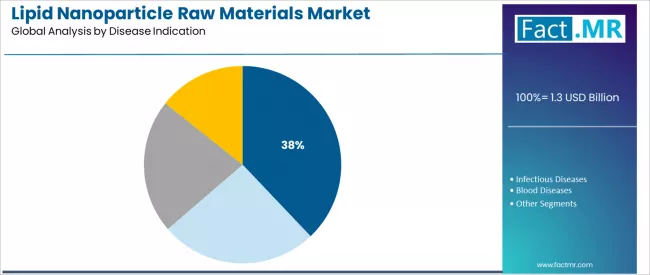
The cancer segment leads the disease indication category with approximately 37.9% market share in 2025, driven by extensive clinical pipeline development and growing investment in targeted oncology therapeutics utilizing lipid nanoparticle delivery systems. Cancer applications benefit from lipid nanoparticle capabilities for tumor-targeted delivery and reduced systemic toxicity, making them preferred platforms for siRNA therapeutics, mRNA cancer vaccines, and gene editing approaches addressing solid tumors and hematologic malignancies. Within the cancer segment, solid tumor applications account for dominant subsegment share, serving pharmaceutical companies focused on precision oncology development.
The infectious diseases segment maintains 30.3% market share, serving mRNA vaccine applications and antiviral therapeutic development. This segment demonstrates strong growth momentum driven by pandemic preparedness initiatives and expanding vaccine pipeline development for emerging infectious diseases beyond COVID-19.
The blood diseases segment holds 18.9% market share, serving gene therapy applications for inherited blood disorders including hemophilia and sickle cell disease. This segment benefits from regulatory approval successes and expanding clinical development programs.
The others segment represents 12.9% market share, encompassing cardiovascular diseases, rare diseases, and metabolic disorders requiring advanced delivery system approaches.
Key factors supporting disease indication segment development:
- Cancer leadership through extensive therapeutic pipeline and significant pharmaceutical investment
- Infectious diseases momentum reflecting vaccine development priorities and pandemic preparedness strategies
- Blood diseases growth driven by gene therapy commercialization and clinical validation success
- Disease diversification enabling comprehensive therapeutic coverage and expanding addressable markets
How do Pharmaceutical & Biotechnology Companies Lead End-Use Applications?
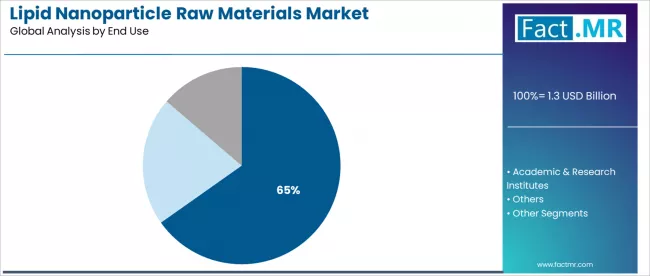
The pharmaceutical & biotechnology companies segment dominates end-use applications with approximately 65.2% market share in 2025, reflecting the critical role of commercial therapeutic development and manufacturing operations across global pharmaceutical industry.
The pharmaceutical & biotechnology companies segment's market leadership is reinforced by large-scale manufacturing requirements, commercial product portfolios, and rising therapeutic pipeline demands for lipid-based delivery systems in drug development programs across developed and emerging markets. Within the pharmaceutical & biotechnology companies segment, large pharmaceutical manufacturers account for highest procurement volume, driven by commercial-scale mRNA vaccine production and gene therapy manufacturing.
The academic & research institutes segment represents 26.5% market share, capturing growth through fundamental research programs and early-stage therapeutic development initiatives. This segment benefits from growing government research funding and industry-academic collaborations that advance lipid nanoparticle technology and explore novel therapeutic applications. The academic & research institutes segment demonstrates steady growth momentum driven by expanding biotechnology research programs and translational medicine initiatives.
The others segment holds 8.3% market share, serving contract research organizations and diagnostic companies utilizing lipid nanoparticle technology for specialized applications.
Key market characteristics across end-use segments:
- Pharmaceutical & biotechnology companies leadership through commercial manufacturing scale and established development pipelines
- Academic & research institutes innovation driving fundamental technology advancement and novel application exploration
- Contract manufacturing growth reflecting pharmaceutical outsourcing trends and specialized production capabilities
- Cross-segment collaboration enabling technology transfer and accelerated therapeutic development timelines
What are the Drivers, Restraints, and Key Trends of the Lipid Nanoparticle Raw Materials Market?
The market is driven by three concrete demand factors tied to therapeutic innovation and pharmaceutical advancement. First, rapid expansion of mRNA therapeutics and vaccines creates growing demand for high-quality lipid components, with global mRNA drug pipeline expanding significantly following COVID-19 vaccine success, requiring comprehensive raw material supply infrastructure. Second, increasing investment in gene therapy and RNA interference therapeutics drives adoption of lipid nanoparticle delivery systems, with pharmaceutical companies prioritizing targeted delivery capabilities and improved therapeutic efficacy for genetic diseases and cancer treatments. Third, technological advancements in ionizable lipid design and formulation science enable more effective delivery systems that enhance cellular uptake while improving safety profiles and therapeutic windows.
Market restraints include high raw material costs for specialized lipid synthesis that can deter smaller biotechnology companies from advancing therapeutic programs, particularly in developing regions where access to pharmaceutical-grade materials and technical expertise remains limited. Regulatory complexity and pharmaceutical manufacturing requirements pose another significant challenge, as lipid nanoparticle materials demand extensive characterization and validation studies, potentially causing increased development costs and timeline delays for therapeutic programs. Technical formulation challenges and stability optimization requirements across different therapeutic modalities create additional market barriers for broad technology adoption, demanding ongoing investment in formulation development and analytical characterization programs.
Key trends indicate accelerated adoption in China and other Asia Pacific markets, where expanding biotechnology sectors and government innovation incentives drive comprehensive lipid nanoparticle technology adoption. Technology integration trends toward next-generation ionizable lipids with improved delivery efficiency, reduced immunogenicity profiles, and enhanced targeting capabilities enable proactive therapeutic development approaches that optimize efficacy and minimize adverse effects. However, the market thesis could face disruption if significant advances in alternative delivery technologies or major changes in regulatory pathways reduce dependence on lipid-based nanoparticle platforms.
Analysis of the Lipid Nanoparticle Raw Materials Market by Key Countries
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| China | 10.0% |
| UK | 9.0% |
| Germany | 8.6% |
| USA | 8.5% |
| France | 8.2% |
| Canada | 8.1% |
| Japan | 7.8% |

The lipid nanoparticle raw materials market is expanding rapidly, with China leading at a 10.0% CAGR through 2036, driven by rapid vaccine production capacity expansion, high R&D growth investment, and government support for biotechnology innovation. UK follows at 9.0%, supported by active nanotechnology research programs and government grant funding for advanced therapeutics.
The USA records 8.5%, reflecting strong pharmaceutical R&D investment and mRNA therapeutics adoption across major biotechnology hubs. Germany posts 8.6%, anchored by established biotech manufacturing infrastructure and pharmaceutical industry expertise. France grows at 8.2%, with focus on infectious disease therapeutics development and academic research programs.
Canada advances at 8.1%, emphasizing government incentives and biotech sector expansion, while Japan grows steadily at 7.8%, focusing on regulatory support for advanced therapies and precision medicine initiatives.
China Leads Global Market Expansion
China demonstrates the strongest growth potential in the lipid nanoparticle raw materials market with a CAGR of 10.0% through 2036. The country's leadership position stems from rapid vaccine production capacity expansion, high R&D growth investment, and government innovation support enabling mainstream lipid nanoparticle technology adoption. Growth is concentrated in major biotechnology centers, including Beijing, Shanghai, Shenzhen, and Guangzhou, where pharmaceutical companies and research institutes are implementing lipid-based delivery systems for therapeutic development and vaccine manufacturing.
Distribution channels through pharmaceutical suppliers, contract manufacturing organizations, and research procurement platforms expand deployment across drug development programs and commercial production facilities. The country's expanding biotechnology sector provides comprehensive policy support for advanced therapeutics development, including mRNA vaccine manufacturing and gene therapy programs.
Key market factors:
- Pharmaceutical demand concentrated in major biotechnology hubs with comprehensive government innovation programs
- Vaccine manufacturing capacity expansion through domestic production initiatives and technology transfer agreements
- Comprehensive biotechnology ecosystem, including established pharmaceutical companies with proven manufacturing capabilities
- Technology integration featuring advanced lipid synthesis platforms, formulation development systems, and analytical characterization technologies
UK Emerges as Innovation Hub
In London, Cambridge, Oxford, and Manchester, the adoption of lipid nanoparticle technology is accelerating across pharmaceutical development programs and academic research initiatives, driven by active nanotechnology research and government grant funding. The market demonstrates strong growth momentum with a CAGR of 9.0% through 2036, linked to research excellence, academic-industry collaborations, and growing biotechnology investment in advanced drug delivery systems. British researchers and pharmaceutical companies are implementing lipid nanoparticle platforms for cancer immunotherapy, rare disease treatments, and vaccine development to enhance therapeutic outcomes while meeting regulatory requirements and commercial viability standards. The country's strong academic research infrastructure creates persistent demand for innovative delivery technologies, while increasing emphasis on translational medicine drives adoption of lipid nanoparticle systems from research laboratories to clinical development.
Key development areas:
- Pharmaceutical and academic sectors leading lipid nanoparticle technology adoption with comprehensive research programs
- Government funding providing integrated support for nanotechnology research and therapeutic development
- Technology partnerships between academic institutions and pharmaceutical companies are expanding market capabilities
- Integration of regulatory science expertise and clinical development pathways
USA Demonstrates Market Leadership

The USA market expansion is driven by diverse pharmaceutical demand across major biotechnology centers including Boston, San Francisco, San Diego, and Research Triangle, where strong R&D investment and mRNA therapeutics adoption support lipid nanoparticle raw materials consumption. The country demonstrates solid growth potential with a CAGR of 8.5% through 2036, supported by strong pharmaceutical R&D investment, established biotechnology infrastructure, and expanding therapeutic pipelines across mRNA vaccines and gene therapy applications. American pharmaceutical companies face competitive dynamics requiring continuous innovation and strategic positioning in advanced therapeutics categories. However, growing clinical validation of lipid-based delivery systems and expanding therapeutic applications create compelling business cases for raw material adoption, particularly in major biotechnology hubs where established pharmaceutical companies and emerging biotech firms have direct impact on market development.
Market characteristics:
- Pharmaceutical and biotechnology sectors showing consistent growth with expanding mRNA therapeutics and gene therapy pipelines
- Regional concentration trends focused on established biotechnology clusters with comprehensive infrastructure
- Strategic partnerships between lipid suppliers and pharmaceutical manufacturers driving market penetration
- Growing emphasis on commercial manufacturing capabilities and regulatory compliance requirements
Germany Shows Manufacturing Excellence
Germany's market expansion benefits from established biotech manufacturing infrastructure, with Frankfurt, Munich, Hamburg, and Berlin leading pharmaceutical development and production capabilities. The country shows strong growth potential with a CAGR of 8.6% through 2036, driven by established biotech manufacturing hub status, pharmaceutical industry expertise, and comprehensive quality control systems for advanced therapeutics production. German manufacturers leverage advanced pharmaceutical infrastructure and regulatory expertise, providing foundation for lipid nanoparticle material production and quality assurance. Growing investment in mRNA therapeutics and gene therapy development creates expanding opportunities for lipid raw material adoption in commercial manufacturing and clinical development applications.
Market characteristics:
- Established pharmaceutical manufacturing providing production expertise and quality systems
- Biotech infrastructure supporting commercial-scale therapeutic manufacturing capabilities
- Advanced regulatory framework enabling efficient product development and approval processes
- Investment in manufacturing capacity for next-generation therapeutic modalities
France Emphasizes Infectious Disease Focus
The French market demonstrates steady development with a CAGR of 8.2% through 2036, driven by focus on infectious disease therapeutics development and academic research programs advancing lipid nanoparticle technology. Major cities including Paris, Lyon, Toulouse, and Marseille showcase active pharmaceutical development in vaccine technologies and antiviral therapeutics where lipid-based delivery systems support therapeutic innovation. French pharmaceutical companies and research institutes prioritize infectious disease applications and preventive medicine approaches, creating opportunities for lipid nanoparticle platform adoption. Distribution channels through pharmaceutical suppliers and academic procurement systems support market expansion across diverse research and development segments.
Leading market segments:
- Infectious disease research driving lipid nanoparticle technology adoption
- Academic institutions supporting fundamental research and translational medicine programs
- Strategic collaborations between pharmaceutical companies and public health organizations
- Focus on vaccine development and antiviral therapeutic applications
Canada Leverages Government Support
Canada's lipid nanoparticle raw materials market benefits from government incentives and biotech sector expansion, with Toronto, Montreal, and Vancouver leading pharmaceutical development activities. The market shows steady growth momentum with a CAGR of 8.1% through 2036, driven by government innovation incentives, expanding biotechnology sector, and growing pharmaceutical investment in advanced therapeutics. Canadian biotechnology companies are developing therapeutic programs utilizing lipid nanoparticle platforms for gene therapy and rare disease treatments. Government funding programs and tax incentives provide foundation for biotechnology innovation, while academic research excellence accelerates technology development and clinical translation.
Key market characteristics:
- Government incentives supporting biotechnology innovation and pharmaceutical development
- Academic research excellence providing technology foundation and talent development
- Emerging biotechnology companies driving therapeutic pipeline development
- Strategic focus on specialized therapeutic areas and precision medicine approaches
Japan Emphasizes Regulatory Excellence

Japan's lipid nanoparticle raw materials market demonstrates mature pharmaceutical landscape with a CAGR of 7.8% through 2036, driven by regulatory support for advanced therapies and precision medicine initiatives. Major pharmaceutical centers including Tokyo, Osaka, and Kobe showcase established drug development programs integrating lipid-based delivery technologies. The country's emphasis on regulatory science and pharmaceutical quality standards creates requirements for high-purity lipid materials that support comprehensive therapeutic development programs. Strategic partnerships between domestic pharmaceutical companies and international technology providers enable access to advanced lipid nanoparticle platforms and formulation expertise.
Market development factors:
- Regulatory framework supporting advanced therapy development and approval
- Established pharmaceutical industry providing manufacturing expertise and quality systems
- Precision medicine focus driving targeted therapeutic development
- International collaborations expanding technology access and development capabilities
Europe Market Split by Country
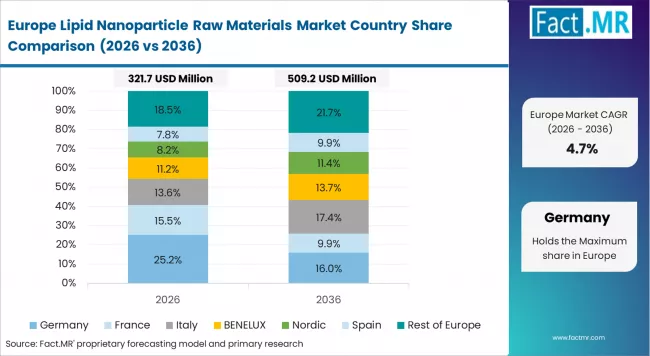
The lipid nanoparticle raw materials market in Europe is projected to grow from USD 380.0 million in 2026 to USD 890.0 million by 2036, registering a CAGR of 8.9% over the forecast period. Germany is expected to maintain its leadership position with a 29.5% market share in 2026, increasing to 30.2% by 2036, supported by its advanced pharmaceutical manufacturing infrastructure, comprehensive biotechnology research networks, and strong regulatory expertise across major commercial centers.
UK follows with a 26.0% share in 2026, projected to reach 26.5% by 2036, driven by comprehensive academic research programs in major universities implementing advanced lipid nanoparticle technology and pharmaceutical partnerships. France holds a 21.0% share in 2026, expected to reach 21.2% by 2036 through the ongoing development of infectious disease therapeutics and vaccine research programs. Italy commands a 11.5% share, projected to maintain 11.0% by 2036, while Spain accounts for 7.0% in 2026, anticipated to hold 6.8% by 2036. The Rest of Europe region is projected to decrease slightly from 5.0% to 4.3% by 2036, attributed to concentrated growth in major Western European pharmaceutical markets with established biotechnology infrastructure and comprehensive therapeutic development programs.
Competitive Landscape of the Lipid Nanoparticle Raw Materials Market
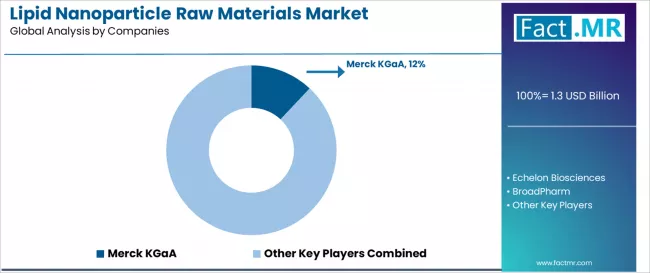
The lipid nanoparticle raw materials market features approximately 25-30 meaningful players with moderate concentration, where the top three companies control roughly 30-35% of global market share through established product portfolios and extensive pharmaceutical industry relationships. Competition centers on material purity, technical support, and supply reliability rather than price competition alone.
Market leaders include Merck KGaA, Echelon Biosciences, and BroadPharm, which maintain competitive advantages through comprehensive lipid component portfolios, global distribution networks, and deep expertise in the pharmaceutical raw materials and drug delivery sectors, creating high customer loyalty among pharmaceutical manufacturers and research institutions.
These companies leverage established pharmaceutical partnerships and ongoing product innovation services to defend market positions while expanding into adjacent therapeutic applications and emerging delivery technologies. Merck KGaA commands approximately 12.0% market share through strategic pharmaceutical partnerships, recent production facility expansion in Germany, and comprehensive product portfolio covering multiple lipid categories.
Challengers encompass Avanti Polar Lipids and Tebubio, which compete through specialized lipid synthesis capabilities and strong technical support presence in key pharmaceutical markets. Avanti Polar Lipids has strengthened its market position through the launch of new ionizable lipid products for mRNA therapeutics in 2025, demonstrating commitment to emerging therapeutic modalities.
Material specialists, including Creative Biolabs, CordenPharma International, and Biopharma PEG Scientific Inc., focus on specific product categories or manufacturing services, offering differentiated capabilities in custom synthesis, cGMP manufacturing, and specialized PEGylated lipid formulations. Creative Biolabs has enhanced its market presence through strategic collaboration with academic institutes for novel LNP research in 2025, expanding its innovation pipeline.
Regional players and emerging specialty chemical companies create competitive pressure through innovative lipid structures and rapid development capabilities, particularly in high-growth markets including China and other Asia Pacific regions, where local presence provides advantages in pharmaceutical customer service and regulatory understanding.
Market dynamics favor companies that combine advanced lipid synthesis technologies with comprehensive technical support services that address the complete development cycle from formulation optimization through commercial manufacturing and regulatory documentation. Strategic collaborations between lipid suppliers and pharmaceutical companies accelerate therapeutic development, while vertical integration initiatives enable cost optimization and quality control across synthesis operations.
Global Lipid Nanoparticle Raw Materials Market - Stakeholder Contribution Framework
Lipid nanoparticle raw materials represent critical enabling components that allow pharmaceutical companies, biotechnology firms, and research institutes to develop advanced therapeutics and delivery systems without substantial in-house synthesis infrastructure, typically providing encapsulation efficiency rates of 80-95% for nucleic acid therapeutics while enabling targeted delivery and enhanced bioavailability. With the market projected to grow from USD 1.32 billion in 2025 to USD 3.15 billion by 2035 at an 9.1% CAGR, these materials offer compelling advantages - superior delivery efficiency, proven therapeutic success, and versatile application capabilities - making them essential for mRNA therapeutics (expanding segment), gene therapy development (growing adoption), and diverse pharmaceutical applications seeking validated delivery platforms. Scaling market penetration and technology capabilities requires coordinated action across regulatory agencies, industry standards organizations, pharmaceutical manufacturers, raw material suppliers, and research institutions.
How Governments Could Spur Local Production and Adoption?
- Innovation Development Programs: Include lipid nanoparticle manufacturing capabilities in national biotechnology initiatives, providing targeted funding for synthesis facilities in pharmaceutical regions and supporting local specialty chemical companies through innovation grants and development support.
- Tax Policy & R&D Support: Implement accelerated depreciation schedules for pharmaceutical-grade synthesis equipment, provide tax incentives for companies investing in advanced lipid production technologies, and establish favorable research accounting standards that encourage domestic production over imported alternatives.
- Regulatory Framework Development: Create streamlined material approval processes across pharmaceutical development and manufacturing applications, establish clear quality standards for lipid nanoparticle components, and develop international harmonization protocols that facilitate cross-border pharmaceutical trade.
- Skills Development & Training: Fund vocational programs for pharmaceutical chemists, lipid synthesis specialists, and analytical development professionals. Invest in technology transfer initiatives that bridge academic research with commercial lipid production and pharmaceutical manufacturing systems.
How Industry Bodies Could Support Market Development?
- Quality Standards & Certification: Define standardized performance metrics for lipid nanoparticle materials across therapeutic development and manufacturing applications, establish universal purity and characterization protocols, and create certification programs for material quality that pharmaceutical companies can rely on.
- Market Education & Best Practices: Lead messaging that demonstrates lipid nanoparticle advantages, emphasizing proven therapeutic success, delivery efficiency, and superior safety profiles compared to alternative delivery technologies.
- Technology Integration Standards: Develop interoperability standards for lipid formulations, comprehensive analytical methodology guidelines, and manufacturing technology platforms, ensuring consistent quality across different production environments and pharmaceutical requirements.
How Manufacturers and Technology Players Could Strengthen the Ecosystem?
- Advanced Synthesis Development: Develop next-generation ionizable lipids with enhanced delivery capabilities, improved safety profiles, and structure-specific targeting features that enhance therapeutic functionality while improving production economics.
- Technical Support Platforms: Provide comprehensive formulation support that integrates analytical characterization, stability testing, regulatory documentation, and manufacturing optimization, enabling customers to maximize therapeutic development success and regulatory approval rates.
- Supply Chain & Partnership Networks: Offer flexible collaboration programs for pharmaceutical companies and research institutions, including custom synthesis options, technical consultation services, and long-term supply agreements that ensure lipid nanoparticle material availability aligns with therapeutic development timelines.
Key Players in the Lipid Nanoparticle Raw Materials Market
- Merck KGaA
- Echelon Biosciences
- BroadPharm
- Avanti Polar Lipids
- Tebubio
- Creative Biolabs
- CordenPharma International
- Biopharma PEG Scientific Inc.
- NOF America Corporation
- Polysciences, Inc.
Bibliography
- United States Food and Drug Administration. (2024). Guidance for industry: Lipid nanoparticle-based drug products, chemistry, manufacturing, and controls. FDA.
- European Medicines Agency. (2024). Reflection paper on the development of mRNA medicines: Quality considerations for lipid nanoparticle components. EMA.
- National Institutes of Health. (2024). ClinicalTrials.gov: Active LNP-based therapeutic and vaccine clinical trials summary. NIH.
- Pharmaceuticals and Medical Devices Agency, Japan. (2024). Guideline on quality aspects of mRNA vaccines and therapeutics. PMDA.
- World Health Organization. (2024). mRNA vaccine technology transfer programme: Lipid raw material specification guidance. WHO.
This bibliography is provided for reader reference. The full Fact.MR report contains the complete reference list with primary research documentation.
This Report Addresses
- Market sizing and quantitative forecast metrics detailing lipid nanoparticle raw material expenditure across therapeutic and research applications through 2036.
- Segmentation analysis mapping procurement patterns across ionizable lipids, PEGylated lipids, helper lipids, and formulation kits by disease indication and end-use category.
- Regional deployment intelligence comparing mRNA pipeline-driven growth in China and the United States against pandemic preparedness stockpiling dynamics in Japan and the European Union.
- Regulatory and quality assessment analyzing how GMP manufacturing standards and pharmaceutical supply chain qualification requirements shape lipid supplier selection.
- Competitive posture evaluation tracking ionizable lipid IP portfolios, GMP synthesis capacity, and pharmaceutical customer audit qualification status.
- Capital project strategic guidance defining the synthesis scale-up and quality system specifications required for GMP-grade lipid manufacturing facility investments.
- Supply chain analysis identifying ionizable lipid synthesis complexity bottlenecks and IP licensing constraints that restrict qualified supplier availability.
- Custom data delivery formats encompassing interactive dashboards, raw Excel datasets, and comprehensive PDF narrative reports.
Lipid Nanoparticle Raw Materials Market Definition
Lipid nanoparticle raw materials refer to the specialty biochemical components, including ionizable lipids, PEGylated lipids, helper lipids, and cholesterol, that constitute the encapsulation matrix of lipid nanoparticle drug delivery systems used to transport mRNA, siRNA, and gene editing payloads into target cells. These materials are supplied as individual reagents, pre-formulated kits, and bulk GMP-grade intermediates. Primary purchasers include pharmaceutical and biotechnology companies developing LNP-based therapeutics, academic research institutions conducting delivery system studies, and contract development and manufacturing organizations.
Lipid Nanoparticle Raw Materials Market Inclusions
Market scope includes ionizable cationic lipids, PEGylated lipid conjugates, helper phospholipids (DSPC, DOPE), cholesterol, and pre-assembled formulation kits. Coverage extends to research-grade and GMP-grade materials across therapeutic and research applications. Regional and segment-level market sizing is provided across the 2026 to 2036 forecast period.
Lipid Nanoparticle Raw Materials Market Exclusions
Finished lipid nanoparticle drug products containing encapsulated active pharmaceutical ingredients are excluded. mRNA, siRNA, and DNA payloads are not covered. Non-lipid delivery system excipients such as polymer nanoparticles and viral vectors fall outside the analytical scope.
Lipid Nanoparticle Raw Materials Market Research Methodology
- Primary Research: Analysts engaged with pharmaceutical lipid procurement directors, LNP formulation scientists, and lipid manufacturing quality managers to map supply qualification timelines and procurement volume projections.
- Desk Research: Data collection phases aggregated clinical trial registry databases for LNP-based therapeutic candidates, GMP manufacturing facility disclosures, and government pandemic preparedness lipid stockpiling program documentation.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of lipid raw material procurement volumes across pharmaceutical and research customers, applying pipeline advancement probabilities to project future clinical and commercial demand.
- Data Validation and Update Cycle: Projections are tested against publicly reported specialty biochemical division revenues and cross-referenced with clinical trial databases tracking LNP-based therapeutic candidate progression.
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 1.44 billion to USD 3.44 billion, at a CAGR of 9.1% |
| Market Definition | Lipid Nanoparticle Raw Materials encompasses ionizable lipids, PEGylated lipids, helper lipids, cholesterol, and formulation kits used as encapsulation matrix components in lipid nanoparticle delivery systems for mRNA, siRNA, and gene editing therapeutics. |
| Application Segmentation | Therapeutics, Research |
| Product Segmentation | Kits, Reagents, Other Raw Materials |
| Disease Indication Segmentation | Cancer, Infectious Diseases, Blood Diseases, Others |
| End-use Segmentation | Pharmaceutical & Biotechnology Companies, Academic & Research Institutes, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, United States, Japan, South Korea, Germany, United Kingdom, and 30 plus countries |
| Key Companies Profiled | Merck KGaA, Echelon Biosciences, BroadPharm, Avanti Polar Lipids, Tebubio, Creative Biolabs, CordenPharma International, Biopharma PEG Scientific Inc., NOF America Corporation, Polysciences Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with global LNP-based therapeutic pipeline candidate counts and projecting lipid raw material consumption by clinical stage advancement and commercial manufacturing volumes. |
Lipid Nanoparticle Raw Materials Market by Segments
-
Application :
- Therapeutics
- Research
-
Product :
- Kits
- Reagents
- Other Raw Materials
-
Disease Indication :
- Cancer
- Infectious Diseases
- Blood Diseases
- Others
-
End-use :
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
- Others
-
Region :
- North America
- USA
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Nordic
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
How large is the demand for Lipid Nanoparticle Raw Materials in the global market in 2026?
Demand for Lipid Nanoparticle Raw Materials in the global market is estimated to be valued at USD 1.44 billion in 2026.
What will be the market size of Lipid Nanoparticle Raw Materials in the global market by 2036?
Market size for Lipid Nanoparticle Raw Materials is projected to reach USD 3.44 billion by 2036.
What is the expected demand growth for Lipid Nanoparticle Raw Materials in the global market between 2026 and 2036?
Demand for Lipid Nanoparticle Raw Materials is expected to grow at a CAGR of 9.1% between 2026 and 2036.
Which Product is poised to lead global sales by 2026?
Kits account for 54.5% share in 2026 as preclinical and early-stage clinical researchers rely on pre-formulated lipid component sets for efficient LNP assembly and screening workflows.
How significant is the role of Therapeutics in driving Lipid Nanoparticle Raw Materials adoption in 2026?
Therapeutics represents 61.4% of application share as the expanding global pipeline of mRNA drugs, gene therapies, and siRNA therapeutics sustains lipid raw material procurement volumes.
What is driving demand in China?
Domestic mRNA vaccine and therapeutic development programs scaling through clinical trials and government-supported biopharmaceutical manufacturing capacity expansion fuel demand in China at a 10.0% CAGR.
What is Lipid Nanoparticle Raw Materials and what is it mainly used for?
Lipid Nanoparticle Raw Materials are specialty biochemical components that form the encapsulation matrix of LNP drug delivery systems. Pharmaceutical companies primarily use them to manufacture mRNA, siRNA, and gene therapy products.
What is included in the scope of this report?
The market covers ionizable lipids, PEGylated lipids, helper phospholipids, cholesterol, and pre-assembled formulation kits in research-grade and GMP-grade formats.
What is excluded from the scope of this report?
Finished LNP drug products, mRNA and siRNA payloads, and non-lipid delivery system excipients such as polymer nanoparticles and viral vectors are excluded.
How does Fact.MR build and validate the forecast?
Forecasting models apply a bottom-up methodology starting with global LNP therapeutic pipeline counts and cross-validate projections against specialty biochemical division revenues and clinical trial databases.
What does zero reliance on speculative third-party market research mean here?
Primary interviews, verified clinical trial registries, and official regulatory guidance documents are used exclusively instead of unverified syndicated estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Therapeutics
- Research
- Therapeutics
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Kits
- Reagents
- Other Raw Materials
- Kits
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Disease Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease Indication, 2026 to 2036
- Cancer
- Infectious Diseases
- Blood Diseases
- Others
- Cancer
- Y to o to Y Growth Trend Analysis By Disease Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Disease Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-use, 2026 to 2036
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutes
- Others
- Pharmaceutical & Biotechnology Companies
- Y to o to Y Growth Trend Analysis By End-use, 2021 to 2025
- Absolute $ Opportunity Analysis By End-use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Application
- By Product
- By Disease Indication
- By End-use
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Product
- By Disease Indication
- By End-use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Application
- By Product
- By Disease Indication
- By End-use
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Product
- By Disease Indication
- By End-use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Application
- By Product
- By Disease Indication
- By End-use
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Product
- By Disease Indication
- By End-use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Application
- By Product
- By Disease Indication
- By End-use
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Product
- By Disease Indication
- By End-use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Application
- By Product
- By Disease Indication
- By End-use
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Product
- By Disease Indication
- By End-use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Application
- By Product
- By Disease Indication
- By End-use
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Product
- By Disease Indication
- By End-use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Application
- By Product
- By Disease Indication
- By End-use
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Product
- By Disease Indication
- By End-use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Product
- By Disease Indication
- By End-use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By Product
- By Disease Indication
- By End-use
- Competition Analysis
- Competition Deep Dive
- Merck KGaA
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Echelon Biosciences
- BroadPharm
- Avanti Polar Lipids
- Tebubio
- Creative Biolabs
- CordenPharma International
- Biopharma PEG Scientific Inc.
- NOF America Corporation
- Polysciences, Inc.
- Merck KGaA
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End-use, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End-use, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by End-use, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by End-use, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by End-use, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by End-use, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by End-use, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by End-use, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Application
- Figure 6: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Product
- Figure 9: Global Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Disease Indication, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Disease Indication
- Figure 12: Global Market Value Share and BPS Analysis by End-use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End-use, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by End-use
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Product
- Figure 32: North America Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Disease Indication, 2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Disease Indication
- Figure 35: North America Market Value Share and BPS Analysis by End-use, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End-use, 2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by End-use
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by Application
- Figure 42: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 44: Latin America Market Attractiveness Analysis by Product
- Figure 45: Latin America Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Disease Indication, 2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Disease Indication
- Figure 48: Latin America Market Value Share and BPS Analysis by End-use, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by End-use, 2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by End-use
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 54: Western Europe Market Attractiveness Analysis by Application
- Figure 55: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 57: Western Europe Market Attractiveness Analysis by Product
- Figure 58: Western Europe Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Disease Indication, 2026 to 2036
- Figure 60: Western Europe Market Attractiveness Analysis by Disease Indication
- Figure 61: Western Europe Market Value Share and BPS Analysis by End-use, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by End-use, 2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by End-use
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Application
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Product
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Disease Indication, 2026 to 2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Disease Indication
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by End-use, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by End-use, 2026 to 2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by End-use
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 80: East Asia Market Attractiveness Analysis by Application
- Figure 81: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 83: East Asia Market Attractiveness Analysis by Product
- Figure 84: East Asia Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Disease Indication, 2026 to 2036
- Figure 86: East Asia Market Attractiveness Analysis by Disease Indication
- Figure 87: East Asia Market Value Share and BPS Analysis by End-use, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by End-use, 2026 to 2036
- Figure 89: East Asia Market Attractiveness Analysis by End-use
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Disease Indication, 2026 to 2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Disease Indication
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by End-use, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by End-use, 2026 to 2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by End-use
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Disease Indication, 2026 to 2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Disease Indication
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by End-use, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by End-use, 2026 to 2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by End-use
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis