Stent Graft Balloon Catheter Market Size, Share, Growth and Forecast (2026 - 2036)
Stent Graft Balloon Catheter Market is segmented by Product (Stent Graft Balloon Catheters, Semi-Compliant Balloon Catheters, Non-Compliant Balloon Catheters, Drug-Coated Balloon Catheters), End Use (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Academic & Research Institutes), and Region, with forecasts covering the period from 2026 to 2036.
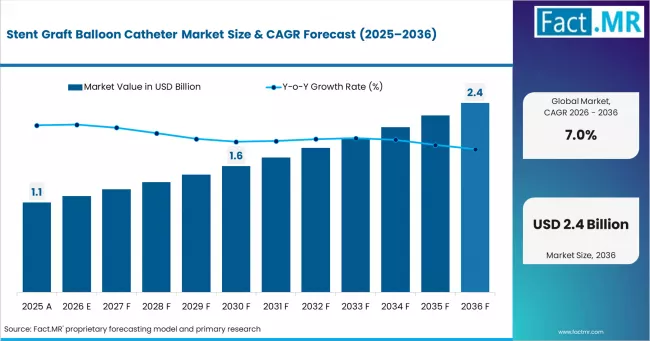
According to Fact.MR estimates, the global stent graft balloon catheter market market was valued at USD 1.1 billion in 2025. The market is projected to reach USD 1.2 billion in 2026 and is expected to grow to USD 2.4 billion by 2036, expanding at a CAGR of 7.0%. Stent Graft Balloon Catheters is anticipated to account for 54.0% of the product segment in 2026, while Stent Graft Balloon Catheters is expected to remain the leading application with around 54.0% share.
Stent Graft Balloon Catheter Market Analysis and Forecast by Fact.MR
The global market for stent graft balloon catheter is estimated to be around USD 1.2 billion in 2026 and has been forecast to expand at a CAGR of 7.0% to end up at USD 2.4 billion by 2036. The Stent Graft Balloon Catheter segment is led by Stent Graft Balloon Catheters, which is anticipated to account for 54.0% of the product segment in 2026, while Stent Graft Balloon Catheters is expected to remain the leading application with around 54.0% share. The market was valued at USD 1.1 billion in 2025 and is poised for steady growth driven by evolving industry dynamics and regional demand patterns.
Summary of Stent Graft Balloon Catheter Market
- Market Snapshot
- In 2025, the global Stent Graft Balloon Catheter Market was valued at approximately USD 1.1 billion.
- The market is estimated to reach USD 1.2 billion in 2026 and is projected to attain USD 2.4 billion by 2036
- The stent graft balloon catheter market is likely to expand at a CAGR of 7.0% during the forecast period.
- The market is anticipated to create an absolute dollar opportunity of USD 1.2 billion between 2026 and 2036.
- Stent Graft Balloon Catheters account for 54.0% of market share in 2026.
- China (9.5%) and India (8.8%) are the key growth markets during the forecast period
- Demand and Growth Drivers
- Growing adoption of endovascular aortic repair (EVAR) and thoracic endovascular repair (TEVAR) is increasing demand for stent graft balloon catheters used in graft deployment and post-dilation.
- Rising prevalence of aortic aneurysms and peripheral vascular disease is expanding the patient population requiring endovascular intervention.
- Catheterization and hybrid operating room infrastructure expansion in Asia Pacific is broadening access to complex endovascular procedures.
- Product and Segment View
- Stent graft balloon catheters account for 54% of the product segment in 2026, serving as essential devices for endovascular graft deployment and vessel preparation.
- Balloon expansion technology holds 51% in 2026, reflecting the standard approach for stent-graft deployment and post-dilation in endovascular procedures.
- Hospitals hold 63% of end-use demand in 2026, with vascular surgery and interventional radiology departments as the primary procedure settings.
- Geography and Competitive Outlook
- China leads at 9.5% CAGR, driven by expanding endovascular procedure volumes and vascular surgery infrastructure development.
- North America and Europe remain the largest markets with established EVAR/TEVAR procedure bases.
- Medtronic holds 28% market share through its integrated endovascular stent-graft and catheter delivery systems.
- Analyst Opinion
- The stent graft balloon catheter market is closely tied to endovascular aortic repair procedure volumes, which are growing as the technique displaces open surgical repair across a broader patient population. Device demand is procedure-linked, with each EVAR/TEVAR case requiring balloon catheters for graft deployment and molding. Growth is concentrated in Asia Pacific where vascular surgery infrastructure is scaling rapidly. The competitive advantage accrues to companies offering integrated stent-graft delivery systems where the balloon catheter is a component of a proprietary platform, creating switching costs and procedure-level lock-in. Drug-coated balloon variants represent an emerging opportunity for combining vessel preparation with anti-restenotic therapy.
Why is the Stent Graft Balloon Catheter Market Growing?
- Endovascular aortic repair adoption is expanding as the technique displaces open surgery across a broader range of aortic pathologies and patient demographics.
- Peripheral vascular disease prevalence growth is increasing interventional procedure volumes requiring balloon catheter-assisted stent-graft deployment.
- Hybrid operating room construction in emerging markets is enabling complex endovascular procedures previously limited to specialized centers.
The stent graft balloon catheter market is a direct derivative of endovascular procedure volume growth. Each EVAR, TEVAR, or peripheral stent-graft procedure requires balloon catheters for device deployment, vessel preparation, and post-dilation, creating a procedurally linked demand model.
The shift from open surgical to endovascular repair continues to expand the addressable patient population. Advances in stent-graft design, including fenestrated and branched configurations, are extending endovascular treatment to more complex anatomies, with each case requiring specialized balloon catheter support.
Segment-wise Analysis of the Stent Graft Balloon Catheter Market
- Stent graft balloon catheters lead at 54% share in 2026, serving endovascular graft deployment and post-dilation.
- Balloon expansion technology holds 51% in 2026, with inflation-based graft deployment as the standard technique.
- Sterile single-use kits account for 67% of packaging in 2026, meeting regulatory sterilization and traceability requirements.
The market is segmented by product type (stent graft balloon, semi-compliant balloon, non-compliant balloon, drug-coated balloon catheters), end use (hospitals, ambulatory surgical centers, specialty clinics, academic and research institutes), technology (balloon expansion, drug-coated balloon, hybrid stent-graft systems), and packaging (sterile single-use kits, medical pouch, bulk hospital packaging).
Which Product Segment Leads?
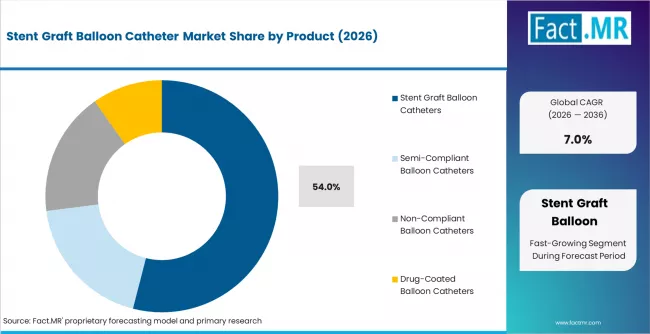
Stent graft balloon catheters hold 54% of the product segment in 2026. Their leadership reflects the essential role of balloon inflation in endovascular graft deployment and vessel molding. Non-compliant and semi-compliant variants serve post-dilation and vessel preparation roles, while drug-coated balloon catheters are gaining share as pharmacological therapy integration advances.
Which Technology Drives Largest Demand?
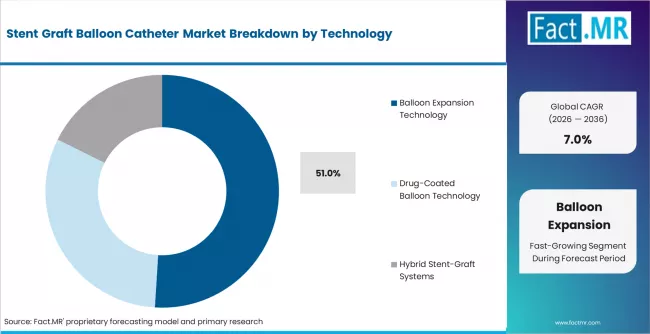
Balloon expansion technology accounts for 51% in 2026. Inflation-based stent-graft deployment remains the standard approach in EVAR and TEVAR procedures. Drug-coated balloon technology is the fastest-growing sub-segment, combining vessel preparation with localized drug delivery to reduce restenosis in peripheral applications.
Key Growth Drivers, Constraints, and Market Scope
- Endovascular procedure volume growth creates procedurally linked device demand.
- Expanding treatment indications for complex aortic anatomies increase per-case device utilization.
- High device costs and reimbursement limitations constrain adoption in price-sensitive markets.
The stent graft balloon catheter market is shaped by vascular disease epidemiology, endovascular procedure adoption, device technology development, and reimbursement frameworks.
Growth Drivers
Demand is shaped by EVAR/TEVAR procedure volume growth, expanding endovascular treatment indications, and hybrid OR infrastructure development in emerging markets. Aging population demographics increase vascular disease prevalence and procedure candidacy.
Constraints
High device costs limit adoption in emerging markets without adequate reimbursement. Procedural complexity restricts use to specialized centers with trained vascular surgeons. Regulatory approval timelines for new device designs delay market entry.
Opportunities
Drug-coated balloon catheters for peripheral applications combine mechanical and pharmacological therapy. Complex aortic repair requiring fenestrated/branched grafts increases per-case balloon utilization. Asian market infrastructure expansion creates the largest procedural volume growth opportunity.
Regional Outlook Across Key Markets
.webp)
- China leads at 9.5% CAGR, driven by vascular surgery infrastructure expansion and growing aortic disease prevalence.
- India follows at 8.8% CAGR, reflecting expanding endovascular procedure capacity.
- Germany records 8.1% CAGR, supported by established vascular surgery volumes and complex case capabilities.
- USA grows at 6.7% CAGR, with mature procedure volumes and device technology advancement.
Country CAGR Table
| Country | CAGR (%) |
|---|---|
| China | 9.5% |
| India | 8.8% |
| Germany | 8.1% |
| Brazil | 7.4% |
| USA | 6.7% |
| UK | 6.0% |
| Japan | 5.3% |
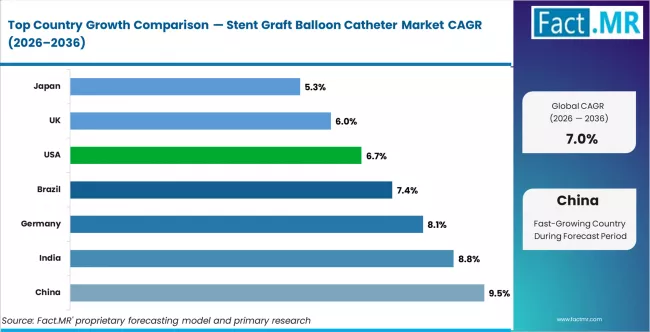
Regional demand reflects vascular disease prevalence, endovascular procedure adoption, hospital infrastructure maturity, and reimbursement support for advanced vascular devices.
Market Outlook in China
The China stent graft balloon catheter market is projected to grow at a CAGR of 9.5% through 2036. Demand is driven by rapid vascular surgery infrastructure expansion, increasing aortic disease prevalence, and growing adoption of EVAR/TEVAR techniques.
- Vascular surgery infrastructure expansion broadens procedure access.
- Aortic disease prevalence growth increases intervention volumes.
- Local device manufacturers are entering the market with cost-competitive offerings.
Demand Trends in India
The India stent graft balloon catheter market is projected to grow at a CAGR of 8.8% through 2036. Growth reflects expanding endovascular procedure capacity, growing tertiary hospital infrastructure, and increasing vascular disease burden.
- Endovascular capacity expansion drives procedure volume growth.
- Tertiary hospital infrastructure supports complex vascular interventions.
- Growing disease burden increases patient candidacy for intervention.
Germany Market Growth
The Germany stent graft balloon catheter market is projected to grow at a CAGR of 8.1% through 2036. Demand is supported by established vascular surgery centers, complex case expertise, and reimbursement support for advanced endovascular procedures.
- Established vascular centers support high procedure volumes.
- Complex case capabilities drive advanced device utilization.
- Reimbursement frameworks support specialty device procurement.
USA Market Outlook
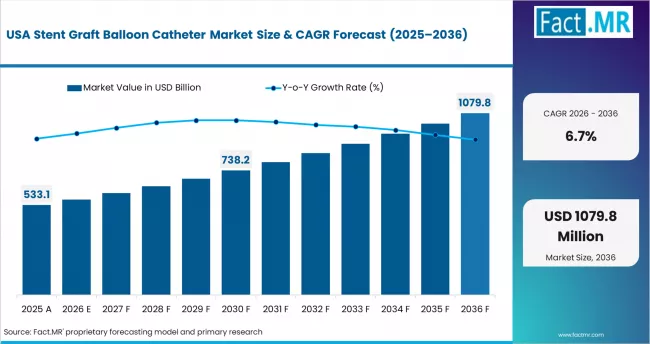
The USA stent graft balloon catheter market is projected to grow at a CAGR of 6.7% through 2036. Demand is shaped by mature EVAR/TEVAR procedure volumes, advancing device technology, and expanding treatment indications for complex anatomies.
- Mature procedure volumes sustain consistent device demand.
- Technology advancement drives device iteration and upgrade cycles.
- Expanding treatment indications increase per-case device utilization.
Competitive Benchmarking and Company Positioning
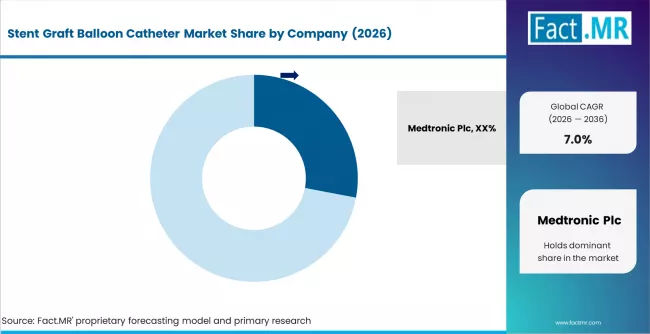
- Medtronic holds 28% market share through its integrated endovascular stent-graft delivery systems spanning aortic and peripheral applications.
- Cook Medical and B. Braun compete through established vascular device portfolios and hospital distribution networks.
- Competition is shaped by stent-graft platform integration, delivery system design, physician training programs, and clinical evidence.
The competitive landscape is shaped by integration between stent-graft platforms and balloon catheter delivery systems. Medtronic leads through its Endurant and Valiant stent-graft systems, where balloon catheters are integral to the deployment mechanism. Cook Medical, B. Braun, and Terumo compete through established vascular device portfolios.
Strategic priorities include developing drug-coated balloon variants, improving catheter crossing profiles and deliverability, expanding hybrid stent-graft system capabilities, and building distribution in emerging market vascular surgery centers.
Competitive Benchmarking
| Company | Platform Integration | Vascular Portfolio | Clinical Evidence | Geographic Reach |
|---|---|---|---|---|
| Medtronic | Full system | Comprehensive | Extensive | Global |
| Cook Medical | Moderate | Broad | Strong | Global |
| B. Braun | Moderate | Focused | Moderate | Europe/Asia |
| Terumo | Moderate | Catheter focused | Moderate | Global |
| Boston Scientific | Growing | Broad | Strong | Global |
Recent Industry Developments
- Medtronic launched an updated balloon delivery system for its aortic stent-graft platform in 2025, featuring improved catheter profile for percutaneous access.
- Cook Medical expanded its peripheral stent-graft balloon catheter line in early 2026, targeting growing femoropopliteal intervention volumes.
- MicroPort received regulatory approval for its stent graft balloon catheter system in China in 2025, targeting domestic vascular surgery market growth.
Leading Companies Shaping the Stent Graft Balloon Catheter Market
-
Major Players
- Medtronic Plc
- B. Braun Melsungen AG
- Cook Medical INC
- Terumo Corporation
- Boston Scientific Corporation
-
Emerging Players
- Cordis Corporation
- MicroPort Scientific Corporation
- Jotech GmbH
- Cardionovum GmbH
- Hexacath
- Abbott Laboratories
- Meril Life Sciences Pvt. Ltd.
Sources and Research References
- Society for Vascular Surgery (SVS). Procedure volume and clinical practice data.
- European Society for Vascular Surgery (ESVS). Clinical guidelines for endovascular repair.
- Medtronic Plc. Annual reports and endovascular portfolio disclosures.
- World Health Organization (WHO). Cardiovascular and vascular disease epidemiology.
- United States Food and Drug Administration (FDA). Medical device regulatory databases.
- Primary interviews with vascular surgeons, interventional radiologists, hospital procurement managers, and device distributors.
This bibliography is provided for reader reference. The full report contains the complete reference list.
Key Questions This Report Addresses
- What is the current and projected size of the stent graft balloon catheter market?
- What CAGR is expected between 2026 and 2036?
- Which product type holds the largest share?
- Which technology drives the highest demand?
- What factors shape demand?
- Which countries show fastest growth?
- How is the competitive landscape structured?
- How does Fact.MR validate the forecast?
Stent Graft Balloon Catheter Market Definition
The stent graft balloon catheter market covers balloon-based catheter devices used in endovascular stent-graft deployment, post-dilation, and vessel preparation during aortic, peripheral, and other vascular repair procedures. The market spans device manufacturing and distribution to hospital vascular surgery and interventional radiology departments.
Stent Graft Balloon Catheter Market Inclusions
The scope covers global and regional market size and forecasts for 2026 to 2036 across product type, end use, technology, packaging, and region. It includes demand analysis based on vascular disease prevalence, EVAR/TEVAR procedure volumes, and endovascular infrastructure development.
Stent Graft Balloon Catheter Market Exclusions
The scope excludes standalone bare-metal and drug-eluting stents without balloon delivery, diagnostic catheters, non-vascular balloon devices, and open surgical graft materials not deployed endovascularly.
Stent Graft Balloon Catheter Market Research Methodology
- Primary research includes interviews with vascular surgeons, interventional radiologists, hospital procurement managers, and device distributors.
- Desk research draws on vascular disease epidemiology, procedure volume statistics, device regulatory databases, and company financial disclosures.
- Market sizing uses a demand-side model validated through procedure volume cross-checks, device utilization rates, and pricing benchmarks.
- Data validation involves reconciliation with medical device registry data and expert panel review.
Scope of Analysis

| Parameter | Details |
|---|---|
| Quantitative Units | USD 1.2 billion to USD 2.4 billion, at a CAGR of 7.0% |
| Market Definition | Covers balloon catheter devices for endovascular stent-graft deployment, post-dilation, and vessel preparation in aortic, peripheral, and other vascular procedures. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, China, India, Germany, Brazil, UK, Japan, and 30+ countries |
| Key Companies | Medtronic, B. Braun, Cook Medical, Terumo, Boston Scientific, Cordis, MicroPort, Jotech, Cardionovum, Hexacath, Abbott, Meril |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid demand-side methodology built on procedure volume data, vascular disease epidemiology, device utilization rates, and primary interviews across vascular surgeons, hospitals, and device distributors. |
Analysis by Product Type, End Use, Technology, Source, Packaging, and Region
-
Stent Graft Balloon Catheter Market Market Segmented by Product:
- Stent Graft Balloon Catheters
- Compliant Balloon Catheters
- Aortic Stent Deployment Balloons
- Compliant Balloon Catheters
- Semi-Compliant Balloon Catheters
- Multi-Purpose Expansion Balloons
- Peripheral Vascular Balloons
- Multi-Purpose Expansion Balloons
- Non-Compliant Balloon Catheters
- High Pressure Deployment Balloons
- Calcified Lesion Expansion Systems
- High Pressure Deployment Balloons
- Drug-Coated Balloon Catheters
- Drug-Eluting Stent Support Balloons
- Anti-Restenosis Balloon Systems
- Drug-Eluting Stent Support Balloons
- Stent Graft Balloon Catheters
-
Stent Graft Balloon Catheter Market Market Segmented by Product:
- Stent Graft Balloon Catheters
- Compliant Balloon Catheters
- Aortic Stent Deployment Balloons
- Compliant Balloon Catheters
- Semi-Compliant Balloon Catheters
- Multi-Purpose Expansion Balloons
- Peripheral Vascular Balloons
- Multi-Purpose Expansion Balloons
- Non-Compliant Balloon Catheters
- High Pressure Deployment Balloons
- Calcified Lesion Expansion Systems
- High Pressure Deployment Balloons
- Drug-Coated Balloon Catheters
- Drug-Eluting Stent Support Balloons
- Anti-Restenosis Balloon Systems
- Drug-Eluting Stent Support Balloons
- Stent Graft Balloon Catheters
-
Stent Graft Balloon Catheter Market Market Segmented by End Use:
- Hospitals
- Cardiology Departments
- Catheterization Laboratories
- Cardiology Departments
- Ambulatory Surgical Centers
- Day Care Vascular Centers
- Minimally Invasive Surgery Units
- Day Care Vascular Centers
- Specialty Clinics
- Cardiovascular Clinics
- Outpatient Interventional Centers
- Cardiovascular Clinics
- Academic & Research Institutes
- Clinical Training Hospitals
- Medical Research Facilities
- Clinical Training Hospitals
- Hospitals
-
Stent Graft Balloon Catheter Market Market Segmented by Technology:
- Balloon Expansion Technology
- Controlled Dilation Systems
- High-Pressure Inflation Mechanisms
- Controlled Dilation Systems
- Drug-Coated Balloon Technology
- Targeted Drug Delivery Systems
- Anti-Restenosis Coating Technology
- Targeted Drug Delivery Systems
- Hybrid Stent-Graft Systems
- Integrated Deployment Platforms
- AI-Assisted Navigation Systems
- Integrated Deployment Platforms
- Balloon Expansion Technology
-
Stent Graft Balloon Catheter Market Market Segmented by Source:
- Medical-Grade Polymers
- Nylon-Based Materials
- Polyurethane & Composite Materials
- Biocompatible Polymer Blends
- Nylon-Based Materials
- Medical-Grade Polymers
-
Stent Graft Balloon Catheter Market Market Segmented by Packaging:
- Sterile Single-Use Kits
- Blister-Sealed Catheter Packs
- Procedure Ready Surgical Kits
- Blister-Sealed Catheter Packs
- Medical Pouch Packaging
- Sterile Barrier Packaging
- Hospital Supply Packs
- Sterile Barrier Packaging
- Bulk Hospital Packaging
- Catheterization Lab Kits
- Institutional Supply Boxes
- Catheterization Lab Kits
- Sterile Single-Use Kits
-
Stent Graft Balloon Catheter Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
What is the market size in 2026?
The global stent graft balloon catheter market is estimated at USD 1.2 billion in 2026.
What will be the market size in 2036?
The market is projected to reach USD 2.4 billion by 2036.
What is the expected CAGR?
The market is expected to grow at a 7.0% CAGR between 2026 and 2036.
Which product type leads?
Stent graft balloon catheters hold 54% of the product segment in 2026.
Which technology accounts for highest demand?
Balloon expansion technology holds 51% in 2026.
Which country is fastest growing?
China is the fastest growing market at 9.45% CAGR through 2036.
What does the market cover?
Balloon catheter devices for endovascular stent-graft deployment, post-dilation, and vessel preparation in aortic, peripheral, and vascular procedures.
How does Fact.MR validate the forecast?
Fact.MR uses a hybrid demand-side methodology validated through procedure data, vascular epidemiology, and primary interviews across 30+ countries.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Stent Graft Balloon Catheters
- Compliant Balloon Catheters
- _Aortic Stent Deployment Balloons
- Semi-Compliant Balloon Catheters
- Multi-Purpose Expansion Balloons
- _Peripheral Vascular Balloons
- Non-Compliant Balloon Catheters
- High Pressure Deployment Balloons
- _Calcified Lesion Expansion Systems
- Drug-Coated Balloon Catheters
- Drug-Eluting Stent Support Balloons
- _Anti-Restenosis Balloon Systems
- Stent Graft Balloon Catheters
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Stent Graft Balloon Catheters
- Compliant Balloon Catheters
- _Aortic Stent Deployment Balloons
- Semi-Compliant Balloon Catheters
- Multi-Purpose Expansion Balloons
- _Peripheral Vascular Balloons
- Non-Compliant Balloon Catheters
- High Pressure Deployment Balloons
- _Calcified Lesion Expansion Systems
- Drug-Coated Balloon Catheters
- Drug-Eluting Stent Support Balloons
- _Anti-Restenosis Balloon Systems
- Stent Graft Balloon Catheters
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Cardiology Departments
- _Catheterization Laboratories
- Ambulatory Surgical Centers
- Day Care Vascular Centers
- _Minimally Invasive Surgery Value (USD Million)s
- Specialty Clinics
- Cardiovascular Clinics
- _Outpatient Interventional Centers
- Academic & Research Institutes
- Clinical Training Hospitals
- _Medical Research Facilities
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Balloon Expansion Technology
- Controlled Dilation Systems
- _High-Pressure Inflation Mechanisms
- Drug-Coated Balloon Technology
- Targeted Drug Delivery Systems
- _Anti-Restenosis Coating Technology
- Hybrid Stent-Graft Systems
- Integrated Deployment Platforms
- _AI-Assisted Navigation Systems
- Balloon Expansion Technology
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Source
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Source, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Source, 2026 to 2036
- Medical-Grade Polymers
- Nylon-Based Materials
- _Polyurethane & Composite Materials
- _Biocompatible Polymer Blends
- Medical-Grade Polymers
- Y to o to Y Growth Trend Analysis By Source, 2021 to 2025
- Absolute $ Opportunity Analysis By Source, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging, 2026 to 2036
- Sterile Single-Use Kits
- Blister-Sealed Catheter Packs
- _Procedure Ready Surgical Kits
- Medical Pouch Packaging
- Sterile Barrier Packaging
- _Hospital Supply Packs
- Bulk Hospital Packaging
- Catheterization Lab Kits
- _Institutional Supply Boxes
- Sterile Single-Use Kits
- Y to o to Y Growth Trend Analysis By Packaging, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Product
- By End Use
- By Technology
- By Source
- By Packaging
- Competition Analysis
- Competition Deep Dive
- Medtronic Plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Cordis Corporation
- B. Braun Melsungen AG
- Cook Medical INC
- Terumo Corporation
- Boston Scientific Corporation
- MicroPort Scientific Corporation
- Jotech GmbH; Cardionovum GmbH
- Hexacath; Abbott Laboratories
- Meril Life Sciences Pvt. Ltd.
- Medtronic Plc
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Packaging, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Product
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Source, 2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Source
- Figure 18: Global Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Packaging, 2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Packaging
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Product
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: North America Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Source, 2026 to 2036
- Figure 46: North America Market Attractiveness Analysis by Source
- Figure 47: North America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Packaging, 2026 to 2036
- Figure 49: North America Market Attractiveness Analysis by Packaging
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Product
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 62: Latin America Market Attractiveness Analysis by Technology
- Figure 63: Latin America Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Source, 2026 to 2036
- Figure 65: Latin America Market Attractiveness Analysis by Source
- Figure 66: Latin America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Packaging, 2026 to 2036
- Figure 68: Latin America Market Attractiveness Analysis by Packaging
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Product
- Figure 76: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 78: Western Europe Market Attractiveness Analysis by End Use
- Figure 79: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 81: Western Europe Market Attractiveness Analysis by Technology
- Figure 82: Western Europe Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Source, 2026 to 2036
- Figure 84: Western Europe Market Attractiveness Analysis by Source
- Figure 85: Western Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Packaging, 2026 to 2036
- Figure 87: Western Europe Market Attractiveness Analysis by Packaging
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Product
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Source, 2026 to 2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Source
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Packaging, 2026 to 2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Packaging
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 113: East Asia Market Attractiveness Analysis by Product
- Figure 114: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 116: East Asia Market Attractiveness Analysis by End Use
- Figure 117: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 119: East Asia Market Attractiveness Analysis by Technology
- Figure 120: East Asia Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Source, 2026 to 2036
- Figure 122: East Asia Market Attractiveness Analysis by Source
- Figure 123: East Asia Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Packaging, 2026 to 2036
- Figure 125: East Asia Market Attractiveness Analysis by Packaging
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Source, 2026 to 2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Source
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging, 2026 to 2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Packaging
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Source, 2026 to 2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Source
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging, 2026 to 2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Packaging
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis