Venous Stents Market Size, Share, Growth and Forecast (2026 - 2036)
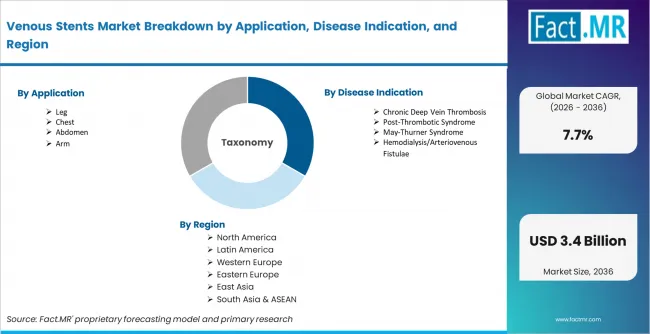
The Venous Stents Market is segmented by Application (Leg, Chest, Abdomen, Arm), Disease Indication (Chronic Deep Vein Thrombosis, Post-Thrombotic Syndrome, May-Thurner Syndrome, Hemodialysis/Arteriovenous Fistulae), Technology (Iliac Vein Stent Technologies, Wallstent Technologies), Product Type (Self-Expanding Venous Stents, Balloon-Expandable Venous Stents, Hybrid Venous Stents), End User (Hospitals, Ambulatory Surgical Centers, Specialty Vascular Clinics), and Region. Forecast for 2026 to 2036.
Fact.MR analysis indicates that venous stent demand is moving from adapted vascular implants toward dedicated venous systems. Chronic deep vein thrombosis is expected to lead the disease indication during the forecast period. Hospitals are projected to lead because complex venous interventions need imaging support and specialist oversight.
Venous Stents Market Size, Market Forecast and Outlook by Fact.MR
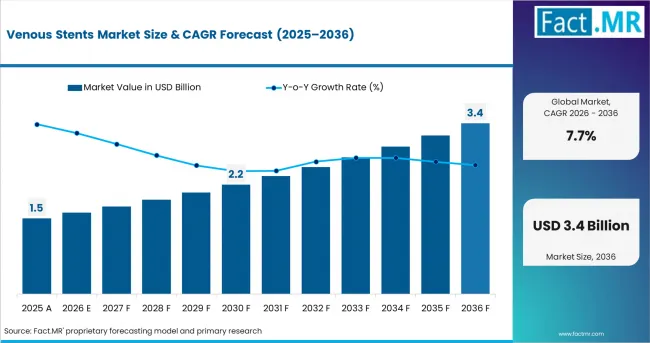
- The venous stents market was valued at USD 1.4 billion in 2025. Demand is set to reach USD 1.5 billion in 2026 at a CAGR of 7.7% during the forecast period.
- Sustained treatment demand takes the valuation to USD 3.1 billion through 2036 as vascular specialists treat chronic venous obstruction with dedicated implant systems.
- Vascular specialists are being pushed to choose between older adapted stent use and purpose-built venous systems that better match iliofemoral anatomy. The FDA approved the Duo Venous Stent System for narrowed or reduced blood flow in iliofemoral veins. The device includes nitinol stents and a delivery catheter, which confirms the shift toward venous-specific designs. [1]
Summary of venous stents market
- Market Definition
- Venous stents are implantable devices used to reopen or support veins affected by obstruction or compression. Scope is defined by venous placement, implant design, disease indication, delivery system, and use in chronic venous treatment.
- Demand Drivers in the Market
- Vascular surgeons need dedicated venous stents for iliofemoral obstruction and chronic outflow restriction.
- Interventional radiologists need reliable venous implants when imaging confirms vessel narrowing.
- Hospital value committees require clearer evidence before wider venous stent adoption.
- Key Segments Analyzed
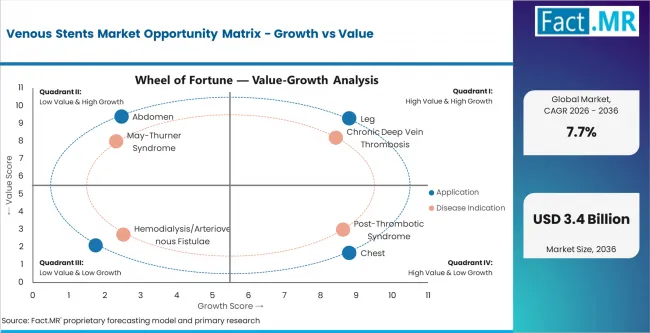
- Chronic Deep Vein Thrombosis: Chronic deep vein thrombosis is expected to lead by disease indication because persistent obstruction creates recurring treatment need. This segment is projected to account for 42.0% share of venous stent demand by disease indication in 2026.
- Iliac Vein Stent Technologies: Iliac vein stent technologies are projected to account for 56.0% share of venous stent technology demand in 2026 because iliofemoral obstruction remains a major treatment area.
- Self-Expanding Venous Stents: Venous anatomy needs sustained outward support after placement. Self-expanding venous stents are estimated to capture 68.0% share of venous stent demand by product type in 2026.
- Hospitals: Hospitals are likely to remain the main procedural base for venous stenting. They are projected to represent 61.0% share of venous stent demand by end user in 2026 because complex procedures require imaging and specialist backup.
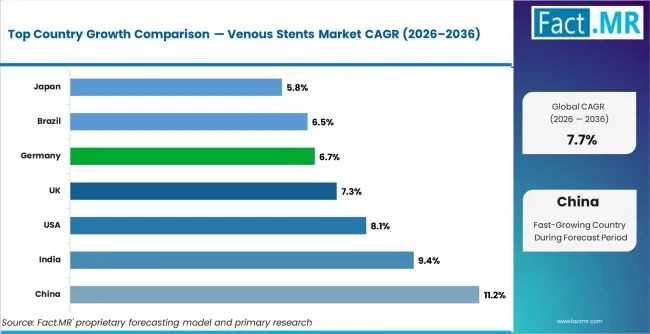
- Geography: China is forecast to grow at 11.2% CAGR in venous stent demand from 2026 to 2036, supported by expanding vascular intervention access.
- Analyst Opinion at Fact.MR
- Shambhu Nath Jha, Senior Analyst at Fact.MR, notes, “Venous stents adoption depends on patient selection and follow-up discipline. Device design matters, but clinical confidence depends on whether physicians can confirm the lesion and size the implant correctly. Patency monitoring strengthens supplier relevance across the full venous care pathway.”
- Strategic Implications
- Venous stent suppliers must support training around case selection and post-procedure surveillance.
- Hospitals need defined pathways for imaging, sizing, and follow-up before routine adoption expands.
- Device makers should focus on venous-specific performance rather than broad vascular positioning.
- Methodology
- Fact.MR interviews vascular surgeons and interventional radiologists. Hospital sourcing managers, device suppliers, and cath lab administrators are covered separately.
- Research reviews FDA approvals and product portfolios. Clinical trial references, company disclosures, and venous disease treatment guidance are reviewed separately.
- Estimates use venous procedure demand and device adoption cycles. Regulatory approvals and hospital intervention capacity guide th4e final model.
- Forecasts are validated through supplier checks and clinician feedback. Hospital adoption signals and application-level triangulation support final validation.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.5 billion |
| Industry Value (2036) | USD 3.1 billion |
| CAGR (2026 to 2036) | 7.7% |
Demand changes as venous stenting enters structured chronic venous disease care beyond selective rescue procedure. Hospitals support this shift through imaging and case selection. Procedure planning and follow up surveillance make venous stents part of long term venous care.
China is projected to record 11.2% CAGR in venous stent demand from 2026 to 2036 as vascular intervention capacity expands. India is likely to grow at 9.4% CAGR through hospital expansion and wider specialist access. The United States is forecast to post 8.1% CAGR as chronic venous disease treatment gains stronger clinical structure. The UK is projected to grow at 7.3% CAGR, Germany at 6.7% CAGR, Brazil at 6.5% CAGR, and Japan at 5.8% CAGR. Country variation depends on specialist access and hospital readiness.
Segmental Analysis
Venous Stents Market Analysis by Application
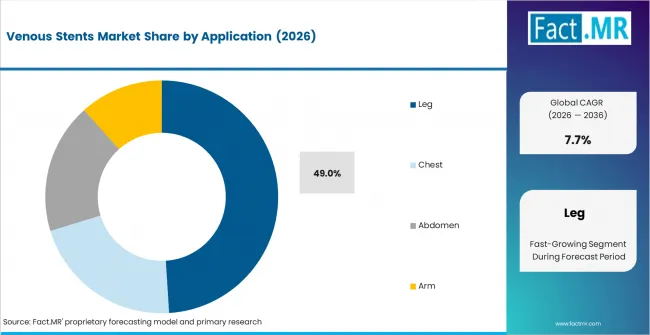
Leg applications are projected to account for 49.0% share of venous stent demand by application in 2026 because lower-limb obstruction creates visible symptoms and strong treatment need. Iliofemoral venous obstruction lead to swelling and walking pain, which increases clinical attention. Chest and arm applications remain more selective because they depend on narrower anatomical indications.
- Clinical visibility: Leg procedures receive earlier attention because swelling and walking limits are clear to patients and physicians.
- Selective placement: Chest and abdominal cases require closer anatomical review because vessel location changes access route and device choice. Arm cases need separate assessment.
- Treatment accuracy: Imaging before the procedure helps physicians judge lesion length and vessel diameter before stent deployment. Landing zone review improves placement accuracy.
Venous Stents Market Analysis by Disease Indication
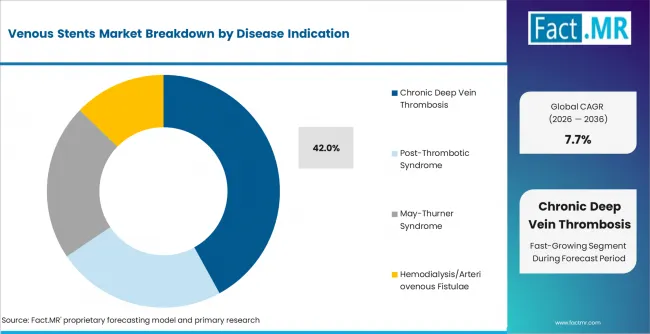
Chronic deep vein thrombosis leads by disease indication because obstruction can persist after the initial clot event. CDC guidance updated in 2025 states that venous thromboembolism includes deep vein thrombosis and pulmonary embolism. [2] Post-thrombotic syndrome remains important when prior thrombosis causes long-term venous damage. Chronic deep vein thrombosis is projected to account for 42.0% share of venous stent demand by disease indication in 2026.
- Chronic obstruction: Chronic deep vein thrombosis accounts for a major share of treatment demand when post-thrombotic symptoms persist after clot management.
- Compression cases: May-Thurner syndrome supports stenting in confirmed iliac vein compression cases where outflow restriction remains clinically relevant.
- Follow-up need: Long-term monitoring remains central because restenosis, stent patency and symptom recurrence influence durable outcomes.
Venous Stents Market Analysis by Technology
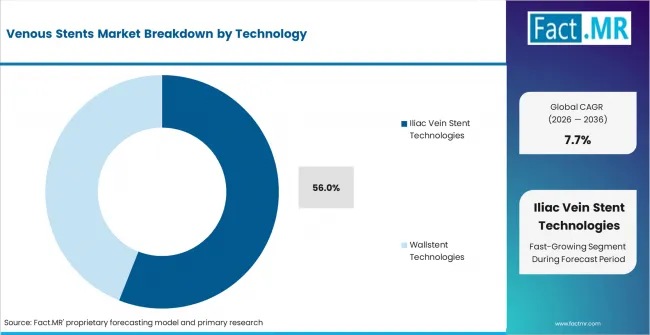
Iliac vein stent technologies lead because iliac obstruction creates a broad clinical base for venous intervention. Dedicated iliac systems are used when physicians need compression resistance and flexibility across curved venous anatomy. Iliac vein stent technologies are projected to account for 56.0% share of venous stent technology demand in 2026. Wallstent technologies remain relevant because many clinicians have experience with their use in iliofemoral obstruction.
- Iliac focus: Iliac vein technologies lead because iliofemoral obstruction remains a core treatment area for symptomatic venous outflow disease.
- Established use: Wallstent technologies retain demand in complex venous cases where physician familiarity and long clinical use support continued selection.
- Device choice: Venous anatomy guides stent flexibility, radial strength and compression resistance requirements during product selection.
Venous Stents Market Analysis by Product Type
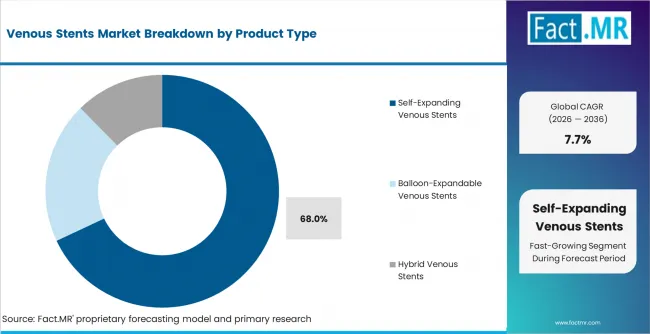
Venous vessels need devices that can expand against compression and maintain lumen support after placement. Self-expanding venous stents are expected to account for 68.0% share of venous stent demand by product type in 2026. Balloon-expandable stents remain selective because venous movement can make flexibility important.
- Self expansion need: Self expanding systems suit venous pathways because iliac and femoral veins can narrow during posture change and hip flexion. External compression adds further placement pressure.
- Material role: Nitinol supports flexibility and shape recovery. Steady radial force helps the device adapt to curved venous anatomy after deployment.
- Placement discipline: Correct sizing helps improve wall apposition because undersizing can reduce support. Oversizing may increase vessel irritation during follow up.
Venous Stents Market Analysis by End User
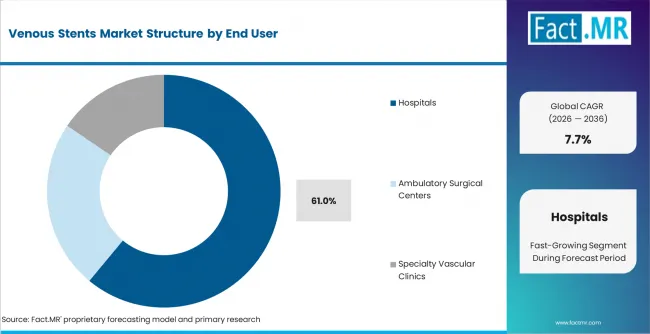
Hospitals remain central because patient selection can involve ultrasound and intravascular imaging. Ambulatory surgical centers gain in standardized cases as protocols improve. Hospitals are projected to account for 61.0% share of venous stent demand by end user in 2026 because complex venous procedures devices need imaging support and specialist availability. Specialty vascular clinics support evaluation and follow-up, though implant procedures often need stronger clinical backup.
- Hospital infrastructure: Hospitals lead because venous interventions need fluoroscopy and ultrasound support. Recovery monitoring after stent placement strengthens hospital use.
- Specialist control: Vascular surgeons and interventional radiologists guide device selection based on lesion site and access route. Vessel diameter review supports correct sizing.
- Care continuity: Follow up protocols influence long term stent performance by tracking patency and restenosis risk. Symptom recurrence review supports ongoing care.
Venous Stents Market Drivers, Restraints, and Opportunities
Chronic venous obstruction and post-thrombotic complications are the main demand anchors for venous stents. CDC data updated in 2025 states that up to 900,000 people in the United States are affected by venous thromboembolism each year. Dedicated venous stent approvals support clinical confidence because newer systems are designed for venous anatomy.
Strict product approval and evidence expectations remain the main restraints. Implantable venous devices must satisfy regulatory review and hospital value analysis. The 2025 ESVM guideline document provides recommendations on interventional treatment of venous thromboembolism, showing the role of structured clinical evaluation in treatment decisions. [3] Smaller suppliers may face slower adoption when they cannot support training or follow-up needs.
Opportunities in the Venous Stents Market
- Dedicated venous design: Suppliers can expand through stents built for venous compression and vessel movement. Long lesion coverage is important in iliac and femoral vein procedures.
- Clinical training: Physician education can improve patient selection and sizing discipline. Procedure consistency depends on anatomy review and accurate lesion assessment.
- Hospital access: Large vascular centers support demand through imaging systems and specialist physicians. Follow up capacity supports diagnosis and long term patency monitoring.
Regional Analysis
.webp)
Based on regional analysis, the venous stents market is segmented into North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, and Middle East and Africa across 30 plus countries.
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 11.2% |
| India | 9.4% |
| United States | 8.1% |
| UK | 7.3% |
| Germany | 6.7% |
| Brazil | 6.5% |
| Japan | 5.8% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research.
East Asia Venous Stents Market Analysis
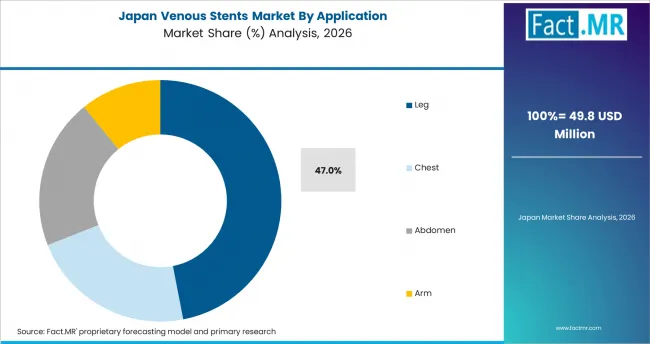
East Asia shows strong growth because large hospitals are adding advanced vascular intervention capacity. China creates the largest regional opportunity through expanding specialty care and higher diagnosis of chronic venous disorders. Japan has a more established hospital base, where adoption depends on clinical protocols and device review. Fact.MR analysis indicates that suppliers in this region compete through training depth and access to major vascular centers.
- China: China is projected to record 11.2% CAGR in venous stent demand from 2026 to 2036 as vascular intervention access expands. Large urban hospitals lead adoption as they have the imaging base needed for complex venous procedures. Specialist training is important because venous stenting requires accurate lesion assessment.
- Japan: Age-related vascular care needs support treatment demand in Japan. Venous stent demand in Japan is expected to post 5.8% CAGR over the assessment period. Established hospital review processes keep adoption measured. Suppliers need strong clinical evidence and reliable local support to build physician confidence.
South Asia and Pacific Venous Stents Market Analysis
South Asia and Pacific benefits from private hospital expansion and rising access to interventional radiology. India leads regional growth because larger hospitals are adding advanced vascular procedures. Price sensitivity remains important, though clinical need is rising where chronic venous obstruction is diagnosed earlier.
- India: Large private hospitals are expanding vascular care capacity, which supports wider use of venous implants. Venous stent demand in India is anticipated to rise at 9.4% CAGR through 2036. Affordability and physician training will influence the adoption curve. Suppliers with strong distributor coverage can improve access beyond top metro hospitals.
North America Venous Stents Market Analysis
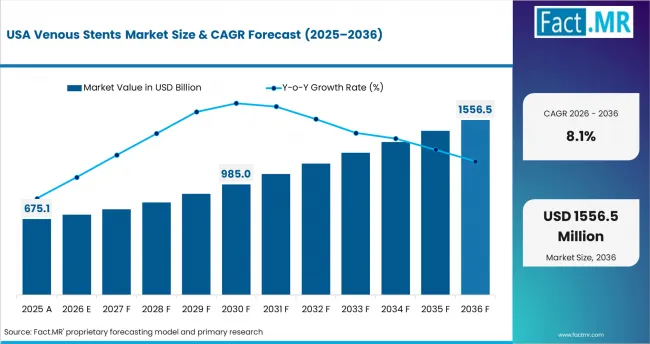
North America remains a major demand base because venous disease treatment is supported by specialist access and regulated device pathways. The United States has strong demand for chronic venous obstruction treatment and advanced interventional care. Philips launched the Duo Venous Stent System in 2024 for symptomatic venous outflow obstruction after FDA approval. [4]
- United States: Specialist-led treatment pathways support venous stent use in the United States. CDC data keeps venous thromboembolism visible in clinical practice. The United States is forecast to post 8.1% CAGR in venous stent demand from 2026 to 2036. Hospitals will continue to evaluate devices through evidence and follow-up reliability.
Western Europe Venous Stents Market Analysis
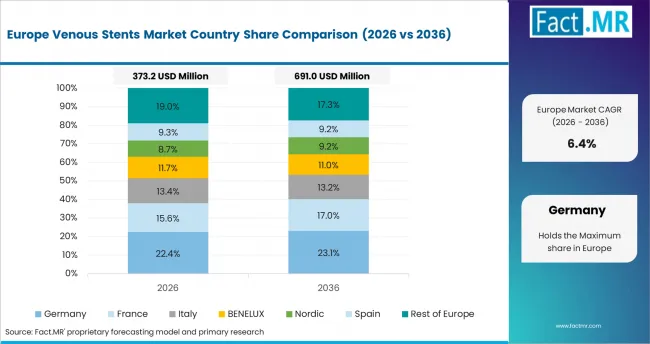
Western Europe demand is supported by advanced vascular care and structured hospital evaluation. Germany leads regional demand because vascular care standards and specialist access remain strong. The UK supports adoption through hospital-based venous disease treatment and structured referral pathways. Supplier selection remains selective because implantable devices require evidence, training, and service support.
- Germany: Germany is projected to reach at a 6.7% CAGR over the study period. Vascular specialists use venous implants where imaging confirms obstruction and symptoms justify intervention. Hospitals are likely to favor suppliers with strong documentation and clinical support.
- UK: Specialist referral pathways and hospital capacity shape procedure volumes in the UK. The UK is expected to expand at 7.3% CAGR in venous stent demand through 2036. Adoption improves when clinicians link diagnosis, procedure planning, and follow-up under one care pathway.
Latin America Venous Stents Market Analysis
Latin America demand is concentrated in major hospitals and private vascular centers. Brazil leads regional demand because it has a wide hospital base and growing access to advanced interventions. Cost sensitivity remains relevant, though specialist-led procedures create demand for proven devices. Distributor availability and training support will shape supplier position outside leading cities.
- Brazil: Urban hospitals support most venous stent adoption in Brazil as complex venous procedures need trained physicians and imaging access. Brazil is projected to record 6.5% CAGR in venous stent demand from 2026 to 2036. Suppliers with local service partners can build stronger repeat demand.
Competitive Aligners for Market Players
The venous stents market is moderately concentrated across global vascular device companies and specialized venous implant suppliers. Becton, Dickinson and Company, Boston Scientific, Cook Medical, Gore Medical, Philips, and OptiMed compete through device design, clinical evidence, physician familiarity, and service depth. Buyers evaluate venous stents through safety, sizing reliability, compression resistance, and long-term outcome confidence.
Competitive advantage depends on matching stent behavior with venous anatomy. FDA approval of the Duo Venous Stent System and Gore’s 2026 FDA approval for the VIABAHN FORTEGRA Venous Stent show that dedicated venous devices are becoming more central to supplier strategy. Gore states that the FORTEGRA venous stent is indicated for deep venous disease in the IVC, iliac, and iliofemoral veins. [5]
Portfolio depth is becoming more important as hospitals evaluate venous treatment pathways. Stryker completed its acquisition of Inari Medical in 2025, giving it entry into the peripheral vascular devices segment. Suppliers that combine device reliability, training support, and evidence generation are better placed through 2036. Low-cost products may gain in selected accounts, but high-risk procedures will favor proven venous platforms. [6]
Key Players in Venous Stents Market
- Becton, Dickinson and Company
- Boston Scientific Corporation
- Cook Medical LLC
- Gore Medical
- Jotec GmbH
- OptiMed Medizinische Instrumente GmbH
- Philips
- Medtronic plc
- Stryker Corporation
- Cordis
Venous Stents Market Analysis by Segments
-
By Application:
- Leg
- Chest
- Abdomen
- Arm
-
By Disease Indication:
- Chronic Deep Vein Thrombosis
- Post-Thrombotic Syndrome
- May-Thurner Syndrome
- Hemodialysis/Arteriovenous Fistulae
-
By Technology:
- Iliac Vein Stent Technologies
- Wallstent Technologies
-
By Product Type:
- Self-Expanding Venous Stents
- Balloon-Expandable Venous Stents
- Hybrid Venous Stents
-
By End User:
- Hospitals
- Ambulatory Surgical Centers
- Specialty Vascular Clinics
-
Region:
- North America
- United States
- Canada
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia Pacific
- China
- Japan
- South Korea
- Taiwan
- Singapore
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- GCC Countries
- South Africa
- Israel
- North America
Bibliography
- [1]. U.S. Food and Drug Administration. (2024). Duo Venous Stent System, P230021. FDA.
- [2]. Centers for Disease Control and Prevention. (2025). Data and statistics on venous thromboembolism. CDC.
- [3]. European Society of Vascular Medicine. (2025). 2025 ESVM Guidelines on interventional treatment of venous thromboembolism. PubMed.
- [4] Philips. (2024). Philips launches Duo Venous Stent System for treatment of symptomatic venous outflow obstruction. Philips.
- [5]. W. L. Gore & Associates. (2026). GORE gains FDA approval for first deep venous stent indicated for the IVC and iliofemoral veins. Gore Medical.
- [6]. Stryker Corporation. (2025). Stryker completes acquisition of Inari Medical, Inc., providing entry into the peripheral vascular segment. Stryker.
This Report Addresses
- Strategic intelligence on venous stent demand across chronic deep vein thrombosis, post-thrombotic syndrome, May-Thurner syndrome, and iliofemoral venous obstruction globally.
- Growth opportunity mapping across China vascular intervention growth, India hospital expansion, United States dedicated device adoption, Germany specialist care, and Japan measured replacement demand.
- Segment analysis by application, disease indication, technology, product type, and end user.
- Regional outlook covering East Asia specialist expansion, South Asia hospital access, North America FDA-approved devices, Western Europe structured vascular care, and Latin America private hospital demand.
- Competitive analysis of BD, Boston Scientific, Cook Medical, Gore Medical, Philips, Medtronic, Stryker, and OptiMed.
- Product assessment covering self-expanding venous stents, balloon-expandable venous stents, hybrid stents, iliac vein technologies, and Wallstent technologies.
- Report delivered through PDF, Excel datasets, and presentation formats, supported by primary interviews, regulatory review, supplier portfolio analysis, hospital buyer checks, and clinician validation.
Venous Stents Market Definition
The venous stents market covers implantable stent systems used to restore blood flow in narrowed or obstructed veins. These devices are used mainly in deep venous pathways such as iliofemoral veins and iliac veins. The market differs from arterial stents because venous applications require compression resistance and flexibility suited to low-pressure vessels.
Venous Stents Market Inclusions
The scope includes self-expanding venous stents, balloon-expandable venous stents, hybrid venous stents, iliac vein stent technologies, Wallstent technologies, and delivery systems sold with venous stent implants. Devices used for chronic deep vein thrombosis, post-thrombotic syndrome, May-Thurner syndrome, and hemodialysis-linked venous obstruction are included when venous stenting is the core treatment function.
Venous Stents Market Exclusions
The scope excludes coronary stents, peripheral arterial stents used only in arteries, thrombectomy-only devices, balloons sold without stents, general vascular catheters, and drug therapies for venous disease. Temporary tools are excluded because they do not remain implanted. Arterial products are excluded because they do not address venous anatomy as the primary use.
Venous Stents Market Research Methodology
Primary Research:
Interviews with vascular surgeons, interventional radiologists, cath lab directors, hospital value analysis committees, and venous device suppliers.
Desk Research:
Review of FDA approvals, company product pages, venous device instructions, clinical literature and vascular care guidelines.
Market-Sizing and Forecasting:
Forecasting uses venous procedure demand, device approval activity, replacement of adapted stents and specialist-led treatment pathways.
Data Validation and Update Cycle:
Forecasts are validated through supplier checks and clinician inputs. Hospital sourcing interviews and venous device approval trends are reviewed separately.
Scope of the Report
| Attribute | Details |
|---|---|
| Quantitative Units | USD 1.5 billion in 2026 to USD 3.1 billion by 2036, at a CAGR of 7.7% |
| Market Definition | Implantable stent systems used to restore blood flow in narrowed or obstructed veins. |
| Application | Leg, Chest, Abdomen, Arm |
| Disease Indication | Chronic Deep Vein Thrombosis, Post-Thrombotic Syndrome, May-Thurner Syndrome, Hemodialysis/Arteriovenous Fistulae |
| Technology | Iliac Vein Stent Technologies, Wallstent Technologies |
| Product Type | Self-Expanding Venous Stents, Balloon-Expandable Venous Stents, Hybrid Venous Stents |
| End User | Hospitals, Ambulatory Surgical Centers, Specialty Vascular Clinics |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | China, India, United States, UK, Germany, Brazil, Japan, and 30 plus countries |
| Key Companies Profiled | Becton, Dickinson and Company, Boston Scientific Corporation, Cook Medical LLC, Gore Medical, Jotec GmbH, OptiMed Medizinische Instrumente GmbH, Philips, Medtronic plc, Stryker Corporation, Cordis |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using venous procedure demand, device approvals, hospital adoption, and primary interviews with vascular care stakeholders. |
- Frequently Asked Questions -
How large is the venous stents market in 2025?
The venous stents market was valued at USD 1.4 billion in 2025.
What is the expected value in 2026?
Demand is estimated to reach USD 1.5 billion in 2026.
What is the forecast value by 2036?
The venous stents market is forecast to reach USD 3.1 billion by 2036.
What CAGR is projected for 2026 to 2036?
Fact.MR projects a CAGR of 7.7% for venous stent demand from 2026 to 2036.
Which application leads venous stent demand?
Leg applications lead with 49.0% share of venous stent demand by application in 2026.
Which disease indication accounts for the highest share?
Chronic deep vein thrombosis leads with 42.0% share of venous stent demand by disease indication in 2026.
Which technology accounts for the highest share?
Iliac vein stent technologies lead with 56.0% share of venous stent technology demand in 2026.
Which product type leads the market?
Self-expanding venous stents lead with 68.0% share of venous stent demand by product type in 2026.
Which end user accounts for the highest share?
Hospitals lead with 61.0% share of venous stent demand by end user in 2026.
Which country shows the fastest growth?
China is projected to grow at 11.2% CAGR in venous stent demand from 2026 to 2036.
Why do leg applications lead demand?
Leg applications lead because lower-limb venous obstruction often affects mobility and daily function.
Why is chronic deep vein thrombosis a major indication?
Chronic deep vein thrombosis creates treatment need when obstruction persists after the initial clot event.
Why are iliac vein stent technologies widely used?
Iliac vein stent technologies are widely used because iliofemoral obstruction is a core venous treatment area.
Why do self-expanding venous stents lead product demand?
Self-expanding venous stents lead because venous pathways need sustained outward support and flexibility.
Why do hospitals lead end-user demand?
Hospitals lead because venous stenting requires imaging, procedure support, and specialist follow-up.
What is the main restraint in this market?
The main restraint is strict clinical evidence and approval requirements for implantable venous devices.
What supports demand in China?
Hospital vascular intervention capacity supports venous stent demand in China.
What supports demand in India?
Private hospital expansion and wider specialist access support venous stent demand in India.
What supports demand in the United States?
Dedicated venous device approvals and structured chronic venous care support demand in the United States.
Why is physician training important?
Physician training supports correct lesion assessment, sizing, placement, and follow-up.
What defines competition in this market?
Competition depends on venous-specific design, clinical evidence, service support, and physician confidence.
Who are the leading companies?
Leading companies include BD, Boston Scientific, Cook Medical, Gore Medical, Philips, Medtronic, Stryker, and OptiMed.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application , 2026 to 2036
- Leg
- Chest
- Abdomen
- Arm
- Leg
- Y to o to Y Growth Trend Analysis By Application , 2021 to 2025
- Absolute $ Opportunity Analysis By Application , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Disease Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease Indication, 2026 to 2036
- Chronic Deep Vein Thrombosis
- Post-Thrombotic Syndrome
- May-Thurner Syndrome
- Hemodialysis/Arteriovenous Fistulae
- Chronic Deep Vein Thrombosis
- Y to o to Y Growth Trend Analysis By Disease Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Disease Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Iliac Vein Stent Technologies
- Wallstent Technologies
- Iliac Vein Stent Technologies
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Self-Expanding Venous Stents
- Balloon-Expandable Venous Stents
- Hybrid Venous Stents
- Self-Expanding Venous Stents
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Ambulatory Surgical Centers
- Specialty Vascular Clinics
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By Disease Indication
- By Technology
- By Product Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Becton, Dickinson and Company
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Boston Scientific Corporation
- Cook Medical LLC
- Gore Medical
- Jotec GmbH
- OptiMed Medizinische Instrumente GmbH
- Philips
- Medtronic plc
- Stryker Corporation
- Cordis
- Becton, Dickinson and Company
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Application ,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Application
- Figure 6: Global Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Disease Indication
- Figure 9: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Technology
- Figure 12: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Product Type
- Figure 15: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by End User
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 22: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Application ,2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Application
- Figure 32: North America Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Disease Indication
- Figure 35: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Technology
- Figure 38: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by Product Type
- Figure 41: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by End User
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Application ,2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Application
- Figure 48: Latin America Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by Disease Indication
- Figure 51: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Technology
- Figure 54: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Product Type
- Figure 57: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by End User
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Application ,2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by Application
- Figure 64: Western Europe Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 66: Western Europe Market Attractiveness Analysis by Disease Indication
- Figure 67: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 69: Western Europe Market Attractiveness Analysis by Technology
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by End User
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Application ,2026 to 2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Application
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Disease Indication
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End User
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Application ,2026 to 2036
- Figure 95: East Asia Market Attractiveness Analysis by Application
- Figure 96: East Asia Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 98: East Asia Market Attractiveness Analysis by Disease Indication
- Figure 99: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 101: East Asia Market Attractiveness Analysis by Technology
- Figure 102: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 104: East Asia Market Attractiveness Analysis by Product Type
- Figure 105: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 107: East Asia Market Attractiveness Analysis by End User
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Application ,2026 to 2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Disease Indication
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Application ,2026 to 2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Disease Indication
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type,2026 to 2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End User,2026 to 2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis