Blow Fill Seal Technology Market Size, Share, Growth and Forecast (2026 - 2036)
Blow Fill Seal Technology Market is segmented by Product Type (Bottles, Ampoules, Vials, Prefilled Syringes & Injectable), Material Type (PE, PP, Others), End-Use (Pharmaceuticals, Food & Beverages, Cosmetics & Personal Care, Others), and Region. Forecast for 2026 to 2036.
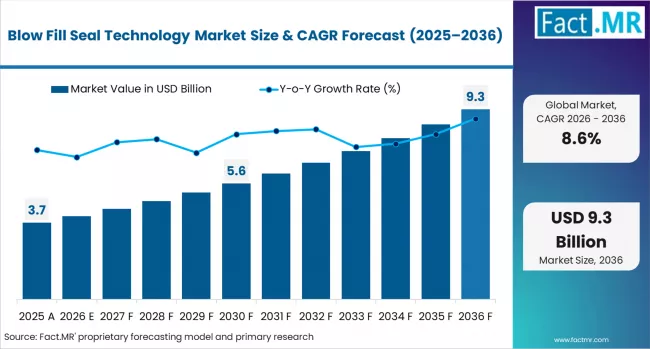
Fact.MR opines the blow fill seal technology market was valued at USD 3.74 billion in 2025. Sales are expected to reach USD 4.06 billion in 2026 and USD 9.27 billion by 2036. Bottles is poised to lead by Product Type with 45.0% share in 2026.
Blow Fill Seal Technology Market Size, Market Forecast and Outlook By Fact.MR
The blow fill seal technology market was valued at USD 3.74 billion in 2025, projected to reach USD 4.06 billion in 2026, and is forecast to expand to USD 9.27 billion by 2036 at a 8.6% CAGR. Expanding pharmaceutical unit-dose packaging requirements and growing demand for preservative-free ophthalmic and respiratory drug delivery formats are accelerating procurement of blow fill seal (BFS) manufacturing systems across global pharmaceutical packaging supply chains. Contract manufacturing organisations specifying BFS production lines for sterile liquid fill operations face tightening regulatory qualification timelines as health authorities mandate validated aseptic processing documentation for each container format.
Summary of Blow Fill Seal Technology Market
- Blow Fill Seal Technology Market Definition
- Blow fill seal technology is an aseptic packaging process that forms, fills, and seals plastic containers in a single machine cycle, used for preservative-free pharmaceutical unit-dose packaging including ophthalmic solutions, injectables, and respiratory therapy products.
- Demand Drivers in the Market
- Pharmaceutical manufacturers converting preservative-free ophthalmic product lines from multi-dose glass bottles to BFS unit-dose ampoules that eliminate the microbial contamination risk associated with repeated patient dispensing.
- Contract manufacturing organisations investing in validated BFS production lines to capture the growing outsourced demand for sterile injectable and biologic drug packaging that requires documented aseptic process qualification.
- Respiratory therapy producers specifying BFS unit-dose containers for nebuliser solution packaging that must meet precise fill volume tolerances and tamper-evident seal integrity requirements under national pharmacopoeia standards.
- Key Segments Analyzed in the Fact.MR Report
- Bottles product type: 45.0% share in 2026.
- PE material type: 62.0% share in 2026.
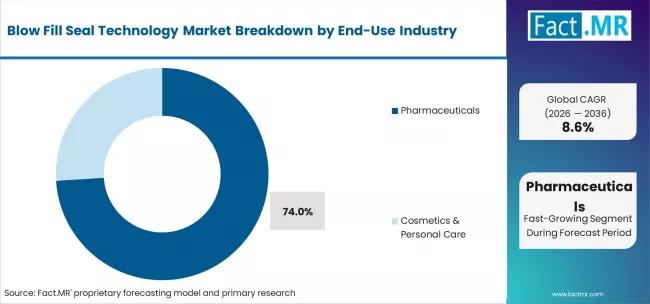
- Pharmaceuticals end-use: 74.0% share in 2026.
- China: 8.7% compound growth during 2026 to 2036.
- Analyst Opinion at Fact.MR
- The blow fill seal technology market is entering an accelerated growth phase as pharmaceutical manufacturers migrate preservative-free ophthalmic and respiratory products from glass to plastic unit-dose formats that reduce contamination risk and improve patient compliance. Contract manufacturers lacking validated BFS production capacity face exclusion from the growing pool of outsourced sterile packaging contracts that represent the fastest-expanding segment of pharmaceutical CDMO revenues. Pharmaceutical companies that delay BFS format conversion risk competitive displacement as regulatory authorities increasingly favour unit-dose preservative-free presentations in updated ophthalmic and respiratory therapy monographs. Investing in multi-format BFS machine configurations that accommodate ampoule, bottle, and prefilled syringe production represents the clearest capacity utilisation pathway for contract packaging operators over the forecast decade.
- Strategic Implications / Executive Takeaways
- Contract manufacturers must invest in multi-format BFS machine platforms to qualify for the growing diversity of pharmaceutical unit-dose packaging contracts spanning ophthalmic, injectable, and respiratory therapy applications.
- Pharmaceutical companies should evaluate BFS format conversion economics for preservative-free product lines where unit-dose packaging eliminates the regulatory burden of multi-dose antimicrobial effectiveness testing.
- Resin suppliers must secure pharmaceutical-grade polyethylene qualification with established BFS machine builders to capture the growing share of validated material supply contracts linked to specific container format approvals.
Blow Fill Seal Technology Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 4.06 billion |
| Industry Value (2036) | USD 9.27 billion |
| CAGR (2026 to 2036) | 8.6% |
The absolute dollar opportunity between 2026 and 2036 amounts to approximately USD 5.21 billion. This expansion captures the structural migration from glass vials and pre-filled syringes toward plastic BFS containers for unit-dose ophthalmic, injectable, and respiratory therapy applications. Resin cost volatility in pharmaceutical-grade polyethylene supply chains, combined with the capital intensity of validated BFS machine installation and qualification, is constraining new entrant capacity while supporting pricing stability for established contract manufacturing operators.
All major consuming regions reflect accelerated deployment parameters. China sets the pace with a 8.7% CAGR, followed by India at 8.2%. South Korea registers a 7.9% rate. USA registers a 7.5% rate. France registers a 7.3% rate. Germany registers a 7.1% rate. ANZ registers a 6.9% rate. Japan registers a 6.8% rate. Italy registers a 6.5% rate. UK expands at a 6.4% trajectory.
Blow Fill Seal Technology Industry Demand Analysis and Impact
The stakeholders of the blow fill seal technology value chain play a crucial role in deciding its growth pattern. The producers and the manufacturers are the pillars of this society, and they are the ones who put the product together and manufacture the final product. They are the driving force behind innovation because they invest in research and development, constantly improving the quality of the product and refining the process of production.
The regulatory standards also directly affect the manufacturers, who must meet the standards of safety, quality, and the environment before they become industry-acceptable. Large-scale production without compromising cost-effectiveness remains a thorny issue, and manufacturers tend to use strategic alliances to overcome production bottlenecks.
Technology vendors are the core part of the revolution of the BFS industry, bringing forth new solutions that enhance manufacturing processes and product performance. These shareholders will likely drive industry growth with automation, AI, and analytics innovation, enabling manufacturers to achieve more productivity at lower costs.
Technology vendors are, however, forced by evolving industry demands to upgrade continuously to stay competitive. They are responsible for ensuring the industry is in sync with evolving customer requirements and technology expectations, developing a culture of continuous change and digital transformation.
Investors are also one of the most important factors for driving the inflow of capital to the industry, investing in new projects, developing manufacturing capacity, and fueling mergers and acquisitions. Their influence is that they can force capital investment into growth initiatives and emerging trends such as sustainability and smart manufacturing.
Investors are conservative, nonetheless, seeking to hedge against risks of industry volatility, regulatory change, and potential competition from emerging players. Investors utilize industry analysis to inform their potential investments, monitoring closely the level of performance and potential return on investment.
Regulators have a powerful level of control over business in the industry by making and enforcing industry guidelines and regulations. Regulators protect consumers, the environment, and fair conduct in the industry. As harsh as these actions are on businesses attempting to cut back, there is also the potential for growth in spaces that align with regulatory strategies, such as green technology and sustainability. The regulators vs. manufacturers dynamic is often one of ensuring compliance with effective operations.
End-users form the final chain of the value chain, setting industry demand by their tastes and buying power. In this dynamism, the industry keeps changing, and end-users increasingly are setting direction for demand in technology-better, sustainable, and affordable products. End-users drive firms to innovate and adjust to their demands, hence making this a dynamic setting where product portfolios must continue to change. Strategic benefit is found in the ability to forecast such evolving tastes and create products that suit target industries.
Throughout the value chain, tensions and interdependencies exist, most prominently between producers, regulators, and technology providers, all of whom must cooperate if they are to meet the demands of the end-users in the context of the regulatory environment. Opportunities for disruption and cooperation arise where these groups converge, as with next-generation technology development, green manufacturing processes, and moves into new geographies.
Blow Fill Seal Technology Industry Analysis by Top Investment Segments
The blow fill seal technology market is segmented by product type into bottles, ampoules, vials, and prefilled syringes & injectable. Material types include PE, PP, and others. The end-use applications span pharmaceuticals, food and beverages, cosmetics & personal care, and others like chemicals. Geographically, the industry covers North America, Latin America, Europe, Asia Pacific, and the Middle East & Africa (MEA).
By Product Type
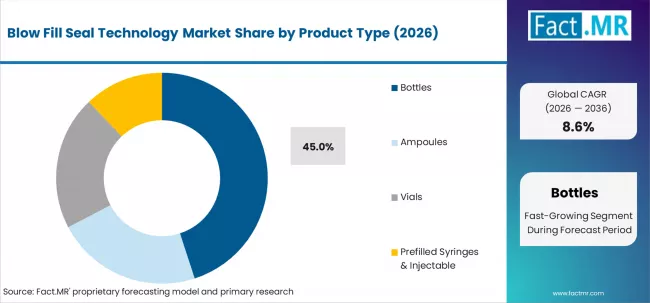
According to the Fact.MR analysis, prefilled syringes & injectables will be the most profitable product type segment. Led by consistent innovation in device technology and enhanced safety aspects, prefilled syringes and injectables will witness aggressive growth, projected at a CAGR of approximately 8.2% during the forecast period, when compared to other product segments.
The growth in prevalence of the application of self-administered, user-comfortable injectable solutions for chronic disease treatment and biologic therapies is driving prefilled syringe usage. Such products benefit from the merits of accurate dose, eliminating the risk of contamination, and enhancing patient compliance, especially with therapy such as insulin therapy and biologics.
By Material Type
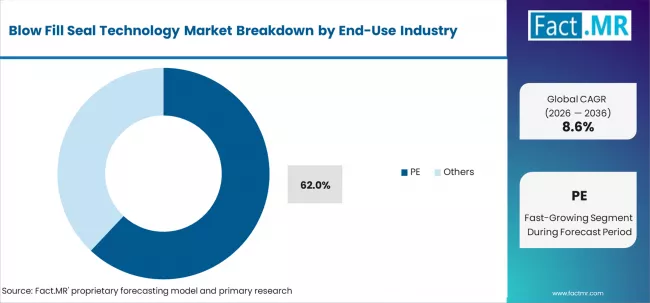
Based on the material, HDPE is set to remain the most profitable segment between 2026 and 2036. The transition towards eco-friendly packaging practices, along with the multidisciplinary use of HDPE in a variety of applications, will propel its industry share. The segment is poised to expand at an estimated CAGR of 7.6% during the study period.
HDPE is chemical-resistant, strong, and easy to handle, hence the best material for pharma packaging. With improving safety standards of products and packaging sustainability requirements, HDPE is a cost-effective but adaptable choice.
By End-use
Of the end-use applications, biologics will most likely be the most profitable segment. As healthcare systems persist in valuing biologic therapy for various conditions, including autoimmune diseases and cancer, the biologics segment is expected to expand at a rate of 9.1% CAGR during the study period, which is higher than the general industry growth rate.
Biologics, including gene therapies, monoclonal antibodies, and vaccines, are leading-edge medicine today, with patient demand spurred by advances in personalized medicine and immunotherapies. Research and development spending rates and high levels of innovation in delivery and therapeutics propel the biologics sector.
Key Strategies of Blow Fill Seal Technology Manufacturers, Suppliers, and Distributors
With the current industry conditions, firms are working on product innovation and alliances aggressively to stay ahead. To meet greater customer demands for high-performance and green products, most companies are focusing on R&D for future-generation products. This includes the incorporation of green material, enhancing the product to make it efficient, and automation to reduce the cost of production.
They are also forming alliances with other makers and tech providers to create new products and expand product offerings together. Geographic expansion, particularly into expansion industries, is the other primary strategy, allowing makers to gain access to new customers and diversify revenues.
Investors target companies that showcase high growth potential and innovation as a measure of response to technology revolutions and regulatory changes. Others are particularly keen on companies embracing sustainability and digitalization, which have become inevitable for prosperity. Investors also want to hedge the risk through geographical and sector diversification of their interests. Strategic investment of capital in startups focused on disruptive technologies such as AI or eco-technologies is also gaining momentum.
Regulators, on the other hand, are interested in making compliance tighter and establishing sound industry standards to ensure safety and sustainability. They lean more towards green technology and stricter environmental controls. Stakeholder regulatory lobbying is thus an important strategy, where players actively engage with legislators to structure the regulatory framework in a way that favors innovation as well as compliance. This lobbying has a tendency to be applied to pricing models as well, where all stakeholders demand environmentally friendly and sustainable pricing models.
For buyers, particularly those in pharma and other industries, reliability and efficiency of the product are given utmost importance. Such buyers are now placing more and more demands on suppliers to provide customized solutions and better after-sales support, thus spurring suppliers to prioritize customer-centric models as their foremost priority.
Tech providers are answering these demands by providing custom-made software and analytics for a better user experience as well as operational efficiency. This includes offering more flexible pricing plans, subscription models, and data-driven information to customers, which are now considered value-added services.
Blow Fill Seal Technology Industry Analysis Across Key Countries
.webp)
United States
Fact.MR estimates the blow fill seal technology market in the U.S. to exhibit outstanding growth of 7.5% CAGR during 2026 to 2036. The U.S. is the world's technology powerhouse, pharmaceuticals, and biologics leader, with ongoing investment in research and development. Health technology advancements coupled with positive regulatory conditions are likely to propel growth, especially for injectable self-injectables and biologics.
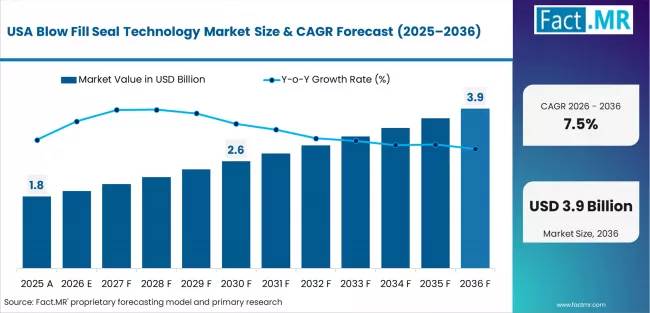
Additionally, the growth of chronic conditions and an aging population both require long-term demand for such solutions. Dynamics of the American industry also considerably depend upon reforms in healthcare policies, for instance, on shifting reimbursement arrangements. The inclination toward more tailored approaches to medicine, like precision therapeutics, will continue to encourage the generation of novel solutions in the course of the next few years.
India
Fact.MR has predicted that the landscape in India will be growing at a CAGR of 8.2% from 2026 to 2036. The healthcare and pharmaceutical sector in India has been growing extremely on the heels of increasing healthcare penetration, increasing incomes, and greater awareness regarding preventive healthcare.
The generic production capability of India remains more dominant on the international platform. With healthcare systems changing, India will be a key player in formulating biologics and injectables. Complicated regulatory landscapes, infrastructure deficits, and price sensitivity continue to be issues, and ongoing investment in healthcare delivery and packaging infrastructure will be required to match the growing demand for effective treatments.
China
The sector in China is expected to see growth at a CAGR of 8.7% between 2026 and 2036, according to Fact.MR estimations. China is among the most rapidly growing and largest healthcare industries in the world, driven by aggressive demand for new therapeutics, biologics, and injectables. China is shifting its attention away from generics and towards new biologic development and innovation adoption.
Government policies for enhancing increased access to healthcare, enhancing insurance coverage, and facilitating technological innovation will continue to be driving factors. The bigger population base, along with the aging population, will also drive higher demand for customized and high-precision therapies, giving the industry good opportunities.
United Kingdom
The blow fill seal technology market in the United Kingdom will grow at a CAGR of 6.4% between 2026 and 2036 as projected by Fact.MR. The UK healthcare industry is controlled by the National Health Service (NHS), which has a strong hold on therapy access and innovation. The application of biologics, particularly in long-term diseases like cancer and autoimmune diseases, is growing at an exponential rate.
New technologies in drug delivery, like prefilled syringes and injectables, are increasingly in demand with the advantage of convenience and less chance of drug error. There could be some regulatory problems based on Brexit that would affect the speed of industry penetration by the new technology, but overall, growth is robust.
Germany
The sector in Germany is expected to witness growth at a CAGR of 7.1% over the period from 2026 to 2036, according to Fact.MR. The country possesses a strong regulatory and healthcare infrastructure and also remains the prime pharma industry of Europe. Healthcare in the country is changing to increasingly patient-focused care in the form of biologics and targeted therapies, further increasing growth.
Germany's focus on the expansion and availability of new forms of delivery, like prefilled syringes, will lead to the expansion of the industry. Germany's research and development intensity for injectables and biologics places it as the industry leader within Europe.
South Korea
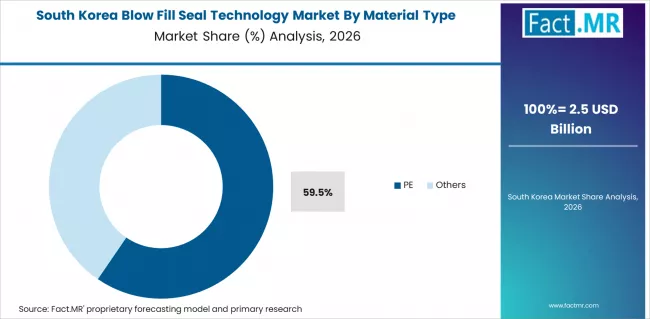
Fact.MR forecasts that the landscape in South Korea will expand at a CAGR of 7.9% during the period 2026 to 2036. South Korean healthcare infrastructure is characterized by technological development and advancement, making it a high-line industry for novel therapies and drug delivery mechanisms.
Growing demand for efficient healthcare therapies is being fueled by the aging population of the country and the rising incidence of chronic diseases. South Korea's biopharma and biotech sectors will be supported by rising research spending, notably in precision medicine and biologics. Furthermore, import reduction and manufacturing-based government policies will fuel these sectors.
Japan
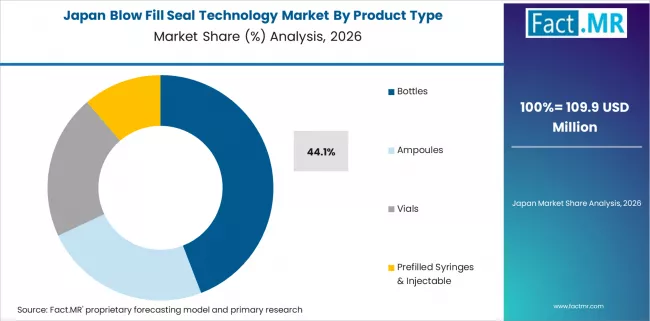
The industry in Japan is anticipated to rise at a 6.8% CAGR from 2026 to 2036, as per Fact.MR. Japan's pharma sector is mature and established, and biologics and precision medicine are highly prioritized by the nation in R&D. The demand for new therapeutics like biologics and injectables is being fueled by aging and the increasing incidence of lifestyle diseases.
Japanese medicine is also rapidly adopting next-generation drug delivery technologies that are patient-friendly and directed toward the convenience of administration according to patient demands, particularly for managing chronic illness. Government policy in facilitating the health delivery process will also keep driving sector expansion.
France
Fact.MR anticipates that the sector in France is expected to register a CAGR of 7.3% from 2026 to 2036 in France. France is one of the largest healthcare industries across Europe, and public as well as private healthcare systems there very high value on the availability of patients to new treatment options.
Increasing need for biologics, coupled with sophisticated drug delivery systems like injectable systems and prefilled syringes, is expected to drive high growth rates over the next couple of years. Also, France has a well-entrenched health system and a strong regulatory system, enabling the commercialization of new drugs and resulting in uniform growth of the industry.
Italy
According to Fact.MR, the landscape in Italy will experience a 6.5% CAGR growth from 2026 to 2036. The pharmaceutical industry of Italy is growing more competitive with the launch of biologics, targeted drugs, and next-generation drug delivery systems by pharmaceutical companies.
Following the experience of having an aging population with a high proportion, Italy is facing rising demands for healthcare and, consequently, for cost-effective and innovative treatment methods. Further, as the country's healthcare system is choosing more sustainable policies and procedures, the use of high-grade injectable products and biologics will rise. Its strong pharma R&D activities further strengthen Italy's leadership position in the European industry.
Australia-New Zealand
Fact.MR projects that the industry in Australia and New Zealand will witness growth at 6.9% CAGR from 2026 to 2036. Both countries have very well-developed healthcare systems that will continue to expand as they continue to adopt more sophisticated drug delivery systems and biologic treatments.
Australia’s high spending habits on health and New Zealand’s strong public health systems, as well as patient care, will lead to continued demand for innovative solutions, particularly in oncology, autoimmune diseases, and chronic diseases. Continued commitment to healthcare infrastructure and a license given for new treatment will facilitate the development in the region.
Leading Blow Fill Seal Technology Companies and their Industry Share
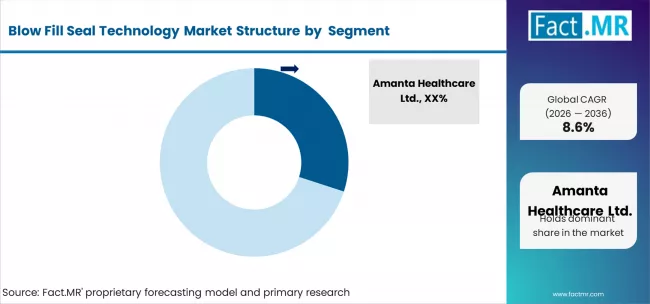
In 2024, Amanta Healthcare Ltd. (25-30% industry share) stayed ahead with its strong portfolio of prefilled syringes and injectables, catering to the growing demand for biologics and complex drug delivery systems. Unither Pharmaceuticals (20-25%) is expanding its global presence, especially in Europe and North America, with its innovative solutions of sterile fill-finish services, including prefilled syringes and vials. Rommelag Kunststoff-Maschinen Vertriebsgesellschaft mbH (15-20%) is also enhancing its blow fill seal technology, focusing on pharma and healthcare industries, to position itself as the aseptic packaging solutions leader.
Catalent, Inc. (10-15%) consolidates its industry leadership through strategic acquisitions and an emerging global manufacturing presence, particularly in biologics and drug delivery systems, including prefilled syringes. Curida AS (8-12%) is capitalizing on the growing demand for contract development and manufacturing services targeting prefilled syringes and biologics with a focus on sustainability. Unipharma LLC (5-8%) is growing its industry standing, particularly in North America, with its expertise in vial packaging of injectable solutions.
Weiler Engineering, Inc. (3-7%) is expanding in the North American industry with state-of-the-art injection molding and packaging solutions tailored to the pharmaceutical and healthcare industries. Its focus on cost-effective and high-quality drug delivery devices is creating its growing industry share.
| Company Name | Estimated Industry Share (%) |
|---|---|
| Amanta Healthcare Ltd. | 25-30% |
| Unither Pharmaceuticals | 20-25% |
| Rommelag Kunststoff-Maschinen Vertriebsgesellschaft mbH | 15-20% |
| Catalent, Inc. | 10-15% |
| Curida AS | 8-12% |
| Unipharma LLC | 5-8% |
| Weiler Engineering, Inc. | 3-7% |
Key Success Factors Driving the Industry
Innovation, collaboration, and sustainability are the key drivers of business success. Though it is high-level solutions that companies seek, companies that invest in product innovation to address emerging customer requirements and optimize operational efficiencies will succeed.
Most prominently, sustainable products and green practices are now significant as customers and regulators increasingly value sustainability. Those companies that can successfully integrate these factors into their product or service lines are poised to capture industry share and strengthen their position.
Strategic alliances and partnering are equally as core to marketplace triumph. Partnering with technology suppliers enables manufacturers to leverage complementary capabilities to facilitate product improvement and the creation of new industries.
Furthermore, partnering for entry into high-growth emerging industries and geographic expansion partnering to enter such industries is being increasingly recognized as companies go global to diversify and access opportunities for expansion internationally. These partnerships, coupled with a strong focus on operating effectiveness and inexpensive manufacturing, are essential to propelling growth and long-term success.
Bibliography
- 1. European Medicines Agency. (2024). Annex 1: Manufacture of sterile medicinal products, revised guidelines. EMA.
- 2. United States Food and Drug Administration. (2024). Guidance for industry: Sterile drug products produced by aseptic processing. FDA.
- 3. International Organization for Standardization. (2024). ISO 15378: Primary packaging materials for medicinal products, quality management system requirements. ISO.
- 4. Parenteral Drug Association. (2024). Technical report: Blow-fill-seal technology, aseptic process validation. PDA.
- 5. Organisation for Economic Co-operation and Development. (2024). Pharmaceutical packaging technology market assessment. OECD.
- 6. Catalent, Inc. (2024). Annual report 2023: Biologics and specialty drug delivery segment review. Catalent.
This bibliography is provided for reader reference. The full Fact.MR report contains the complete reference list with primary research documentation.
This Report Addresses
- Market sizing and quantitative forecast metrics detailing blow fill seal technology consumption across major end-use industries through 2036.
- Segmentation analysis mapping adoption velocity across product type, material type, end-use categories and evaluating structural demand shifts.
- Regional deployment intelligence comparing consumption patterns across Asia Pacific, Europe, North America, and other regions.
- Regulatory compliance assessment analysing how material safety directives and environmental standards influence procurement specifications.
- Competitive positioning evaluation tracking market share distribution, vertical integration advantages, and buyer leverage dynamics among leading producers.
- Capital project strategic guidance defining procurement specifications and supply qualification requirements for major industrial consumers.
- Supply chain risk analysis identifying feedstock concentration, logistics constraints, and capacity utilisation bottlenecks.
- Custom data delivery formats encompassing interactive dashboards, raw Excel datasets, and comprehensive PDF narrative reports.
Blow Fill Seal Technology Market Definition
Blow fill seal technology is an automated aseptic packaging process in which a plastic container is formed from extruded polymer resin, filled with liquid product, and hermetically sealed in a single continuous machine cycle under controlled cleanroom conditions. This technology produces unit-dose ampoules, bottles, vials, and prefilled syringes from polyethylene or polypropylene resins for pharmaceutical, food and beverage, and cosmetic applications. Primary pharmaceutical end uses include preservative-free ophthalmic solutions, sterile injectable preparations, respiratory therapy unit doses, and wound care solutions.
Blow Fill Seal Technology Market Inclusions
Market scope covers global and regional blow fill seal technology market volumes, forecast from 2026 to 2036. Segment breakdowns include product type (bottles, ampoules, vials, prefilled syringes and injectables), material type (PE including LDPE and HDPE, PP), and end use (pharmaceuticals including ophthalmic, injectable, biologics, wound care, respiratory therapy, food and beverages, cosmetics and personal care). Regional pricing trends and technology adoption curve analysis are incorporated.
Blow Fill Seal Technology Market Exclusions
The scope excludes conventional glass vial filling lines, pre-formed plastic container filling equipment, and downstream pharmaceutical distribution services. Active pharmaceutical ingredient manufacturing and drug substance formulation development fall outside analytical parameters. Standalone plastic resin market sizing is not included unless directly linked to BFS container production consumption metrics.
Blow Fill Seal Technology Market Research Methodology
- Primary Research: Analysts conducted structured interviews with procurement directors, production managers, and specification engineers across major blow fill seal technology consuming industries in 30 countries to validate adoption timelines and volume commitments.
- Desk Research: Data collection aggregated regulatory filings, trade association production statistics, company annual reports, and published pricing indices relevant to the blow fill seal technology supply chain.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of production capacity data and consumption volumes, applying region-specific demand curves to project future adoption trajectories.
- Data Validation and Update Cycle: Projections undergo cross-validation against publicly reported financial guidance from leading producers and quarterly trade data published by national statistical agencies.
Scope of the Report
| Quantitative Units | USD 4.06 billion to USD 9.27 billion, at a CAGR of 8.6% |
|---|---|
| Market Definition | Blow fill seal technology is an aseptic packaging process that forms, fills, and seals plastic containers in a single machine cycle, used for preservative-free pharmaceutical unit-dose packaging including ophthalmic solutions, injectables, and respiratory therapy products. |
| Segmentation | Product Type: Bottles, Ampoules, Vials, Prefilled Syringes & Injectable; Material Type: PE, PP, Others; End-Use: Pharmaceuticals, Food & Beverages, Cosmetics & Personal Care, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, South Korea, USA, Germany, Japan, France, Italy, UK, ANZ, India, and 40 plus countries |
| Key Companies Profiled | Amanta Healthcare Ltd., Unither Pharmaceuticals, Rommelag Kunststoff-Maschinen Vertriebsgesellschaft mbH, Catalent, Inc., Curida AS, Unipharma LLC, Weiler Engineering, Inc., Schott AG, West Pharmaceutical Services, Biocorp Production, Stevanato Group, SMC Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid modelling combining bottom-up production capacity analysis with top-down demand consumption patterns, cross-validated against primary research and public financial disclosures. |
Segmentation
-
By Product Type :
- Bottles
- 0.2-100ml
- 100-500ml
- Above 500ml
- Ampoules
- 0.1-10ml
- 10-100ml
- Vials
- 0.1-10ml
- 10-50ml
- Prefilled Syringes & Injectable
- Bottles
-
By Material Type :
- PE
- LDPE
- HDPE
- PP
- Others (EVOH, etc.)
- PE
-
By End-Use :
- Pharmaceuticals
- Ophthalmic
- Injectable
- Biologics
- Wound Care
- Respiratory Therapy
- Food & Beverages
- Cosmetics & Personal Care
- Others (Chemicals, etc.)
- Pharmaceuticals
-
By Region :
- North America
- Latin America
- Europe
- East Asia
- South Asia & Oceania
- Middle East & Africa
- Frequently Asked Questions -
How large is the demand for Blow Fill Seal Technology Market globally in 2026?
Demand for blow fill seal technology in the global market is estimated to be valued at USD 4.06 billion in 2026.
What will be the market size of Blow Fill Seal Technology Market by 2036?
Market size for blow fill seal technology is projected to reach USD 9.27 billion by 2036.
What is the expected demand growth for Blow Fill Seal Technology Market between 2026 and 2036?
Demand for blow fill seal technology is expected to grow at a CAGR of 8.6% between 2026 and 2036.
Which Product Type is poised to lead global sales by 2026?
Bottles accounts for 45.0% share in 2026 within the product type category.
What is the China growth outlook in this report?
China is projected to grow at a CAGR of 8.7% during 2026 to 2036.
How significant is the role of PE in the material type category?
PE represents 62.0% of segment share in the material type category as of 2026.
What is Blow Fill Seal Technology and what is it mainly used for?
Blow fill seal technology is an aseptic packaging process that forms, fills, and seals plastic containers in a single machine cycle, used for preservative-free pharmaceutical unit-dose packaging including ophthalmic solutions, injectables, and respiratory therapy products.
How does Fact.MR build and validate the Blow Fill Seal Technology Market forecast?
Forecasting models apply a hybrid methodology combining bottom-up production capacity data with top-down consumption benchmarks, cross-validated against quarterly financial disclosures from leading producers and national trade statistics.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Bottles
- 0.2-100ml
- 100-500ml
- Above 500ml
- Ampoules
- 0.1-10ml
- 10-100ml
- Vials
- 0.1-10ml
- 10-50ml
- Prefilled Syringes & Injectable
- Bottles
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material Type, 2026 to 2036
- PE
- LDPE
- HDPE
- PP
- Others
- PE
- Y to o to Y Growth Trend Analysis By Material Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Material Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-Use, 2026 to 2036
- Pharmaceuticals
- Ophthalmic
- Injectable
- Biologics
- Wound Care
- Respiratory Therapy
- Food & Beverages
- Cosmetics & Personal Care
- Others
- Pharmaceuticals
- Y to o to Y Growth Trend Analysis By End-Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End-Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Material Type
- By End-Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End-Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Material Type
- By End-Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End-Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Material Type
- By End-Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End-Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Material Type
- By End-Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End-Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Material Type
- By End-Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End-Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Material Type
- By End-Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End-Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Material Type
- By End-Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End-Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End-Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Material Type
- By End-Use
- Competition Analysis
- Competition Deep Dive
- Amanta Healthcare Ltd.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Value (USD Million)her Pharmaceuticals
- Rommelag Kunststoff-Maschinen Vertriebsgesellschaft mbH
- Catalent, Inc.
- Curida AS
- Unipharma LLC
- Weiler Engineering, Inc.
- Schott AG
- West Pharmaceutical Services
- Biocorp Production
- Stevanato Group
- SMC Ltd.
- Groninger & Co. GmbH
- Ismeca Semiconductor
- Pharmapack Co. Ltd.
- Automatic Liquid Packaging Solutions LLC
- Asept Pak, Inc.
- NuPharm
- BioConnection
- Plastikon
- Amanta Healthcare Ltd.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End-Use, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End-Use, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End-Use, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End-Use, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End-Use, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End-Use, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End-Use, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End-Use, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Material Type, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Material Type
- Figure 9: Global Market Value Share and BPS Analysis by End-Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End-Use, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End-Use
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Material Type, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Material Type
- Figure 29: North America Market Value Share and BPS Analysis by End-Use, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End-Use, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End-Use
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Material Type, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Material Type
- Figure 39: Latin America Market Value Share and BPS Analysis by End-Use, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End-Use, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End-Use
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Material Type, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Material Type
- Figure 49: Western Europe Market Value Share and BPS Analysis by End-Use, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End-Use, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End-Use
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Material Type, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Material Type
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End-Use, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End-Use, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End-Use
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Material Type, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Material Type
- Figure 69: East Asia Market Value Share and BPS Analysis by End-Use, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End-Use, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End-Use
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Material Type, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Material Type
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End-Use, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End-Use, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End-Use
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Material Type, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Material Type
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End-Use, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End-Use, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End-Use
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis