Bioinductive Implant Market Size, Share, Growth and Forecast (2026 - 2036)
Bioinductive Implant Market Size and Share Forecast Outlook 2026 to 2036
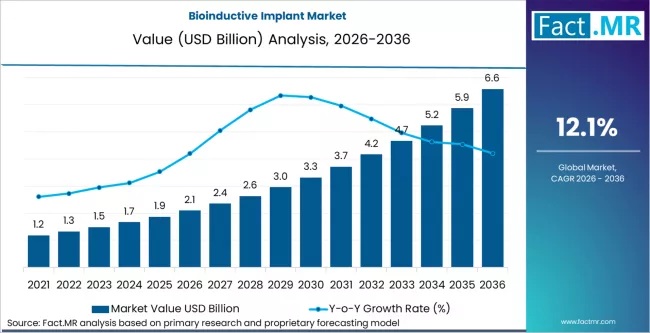
Bioinductive implant market is projected to grow from USD 2.1 billion in 2026 to USD 6.6 billion by 2036, at a CAGR of 12.1%. Collagen bioinductive patches will dominate with a 32.0% market share, while rotator cuff repair augmentation will lead the application or procedure segment with a 40.0% share.
Bioinductive Implant Market Forecast and Outlook By Fact.MR
- The bioinductive implant market records USD 2.1 billion in 2026 and advances to USD 6.6 billion by 2036 at a 12.1% CAGR.
Summary of BioinductiveImplant Market
- Market Snapshot
- Global bioinductive implant market revenue stood at USD 2.1 billion in 2026 and is forecast to reach USD 6.6 billion by 2036.
- At a 12.1% CAGR from 2026 to 2036, this market is set to expand ~3.1x in value, adding USD 4.5 billion in absolute opportunity.
- Growth is being driven by increasing incidence of sports injuries, rising adoption of biologic repair technologies, and growing preference for minimally invasiveorthopedicprocedures.
- Bioinductiveimplants are increasingly adopted to enhance tissue regeneration and healing whilemaintainingstructural support, surgical efficiency, and improved patient outcomes.
- Rising investments in regenerative medicine,orthopedicinnovation, and biologics-based treatment approaches are reinforcing market expansion globally.
- Demand and Growth Drivers
- Increasing prevalence of tendon, ligament, and rotator cuff injuries worldwide
- Rising adoption of biologic augmentation technologies inorthopedicsurgery
- Growing demand for minimally invasive and tissue-preserving treatment options
- Expansion of sports medicine and regenerative healthcare practices
- Need for solutions that provide:
- Enhanced tissue healing and regeneration
- Improved surgical outcomes and recovery rates
- Reduced re-tear and revision surgery risks
- Better long-term functional performance
- Product and Segment View
- Collagen bioinductive patchese mergeas the leading implant type in 2026.
- Key product categories include:
- Collagenbioinductivepatches
- ECM scaffolds (xenograft or allograft)
- Syntheticbioinductivescaffolds
- Bioactive-coated repair adjuncts
- Fixation and delivery accessories
- Geography and Competitive Outlook
- Asia Pacificemergesas the leading growth region for thebioinductiveimplant market.
- Key growth markets and CAGR: India 15.6%, Brazil 14.8%, China 13.9%, USA 10.3%, Germany 6.6%, France 5.0%, UK 3.4%
- Market expansion is closely tied to:
- Increasing adoption of biologic repair solutions inorthopedicprocedures
- Rising investments in sports medicine and regenerative therapies
- Growing demand for advanced tissue healing technologies and improved patient outcomes
- Key companies active in this market include Smith & Nephew, Arthrex, Zimmer Biomet, Stryker, and Integra LifeSciences.
BioinductiveImplant Market — At a Glance
| Attribute | Details |
|---|---|
| Market Value 2026 | USD 2.1 billion |
| Market Value 2036 | USD 6.6 billion |
| Absolute Dollar Opportunity 2026–2036 | USD 4.5 billion |
| Total Growth 2026–2036 | 214.3% |
| CAGR 2026–2036 | 12.1% |
| Growth Multiple | ~3.1x |
| Key Demand Theme | Growing adoption of biologic tissue regeneration andorthopedicrepair augmentation technologies |
| Leading Implant Type (2026) | Collagenbioinductivepatches |
| Leading Application (2026) | Rotator cuff repair augmentation |
| Key Growth Region | Asia Pacific |
| Country CAGRs | India 15.6%, Brazil 14.8%, China 13.9%, USA 10.3%, Germany 6.6%, France 5.0%, UK 3.4% |
| Top Companies | Smith & Nephew, Arthrex, Zimmer Biomet, Stryker, Integra LifeSciences |
| Segmentation by Implant Type | Collagenbioinductivepatches, ECM scaffolds (xenograft or allograft), Syntheticbioinductivescaffolds, Bioactive-coated repair adjuncts,Fixationand delivery accessories |
| Segmentation by Application or Procedure | Rotator cuff repair augmentation, Tendon repair (Achilles or hand), Ligament repair and reconstruction adjuncts,Meniscusor cartilage repair support,Othersports medicine procedures |
| Segmentation by End User / Sales Channel | Hospitals, Ambulatory SurgeryCenters(ASCs),Orthopedicspecialty clinics, Distributor channels,GPOor tender procurement |
| Segmentation by Region | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
The development path reflects steady adoption from earlier levels at USD 1.2 billion, USD 1.5 billion, USD 1.9 billion, USD 2.4 billion, and USD 3.0 billion before the current base was established. Use is concentrated in procedures where tissue quality limits direct repair and where surgeons seek reinforcement that integrates into native structures over time. Product selection places weight on handling properties, fixation behavior, and predictable resorption profiles, as these factors affect operating time and post procedure follow up. Once a system is validated within a surgical protocol, change occurs infrequently due to training, documentation, and outcome tracking requirements. Volume growth follows expansion of indications and wider inclusion in standard repair pathways rather than short cycle product turnover.
Category
| Category | Segments |
|---|---|
| Implant Type | Collagen bioinductive patches; ECM scaffolds (xenograft or allograft); Synthetic bioinductive scaffolds; Bioactive-coated repair adjuncts; Fixation and delivery accessories |
| Application or Procedure | Rotator cuff repair augmentation; Tendon repair (Achilles or hand); Ligament repair and reconstruction adjuncts; Meniscus or cartilage repair support; Other sports medicine procedures |
| End User or Sales Channel | Hospitals; Ambulatory surgery centers (ASCs); Orthopedic specialty clinics; Distributor channels; GPO or tender procurement |
| Region | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
Segmental Analysis
Why Do Collagen Bioinductive Patches Anchor Most Implant Selection Decisions?
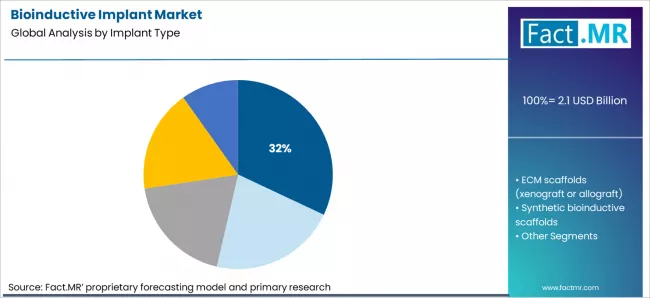
Use patterns in the bioinductive implant market place collagen bioinductive patches at 32.0% of implant type demand. Surgeons treat these patches as a predictable way to add biological stimulus without altering core repair mechanics. Operating room workflows accommodate patch placement within existing procedure steps, which keeps case time within familiar limits. Inventory teams prefer this category since sizes, handling methods, and storage rules stay consistent across suppliers. Training requirements also remain limited, with technique steps fitting standard arthroscopic or open repair routines. Clinical documentation frequently references this format as a baseline option when tissue quality appears compromised. ECM scaffolds, synthetic systems, and coated adjuncts remain active choices. Case planning still starts most often from patch based solutions as the default reference.
Why Does Rotator Cuff Augmentation Dominate the Clinical Use Mix?
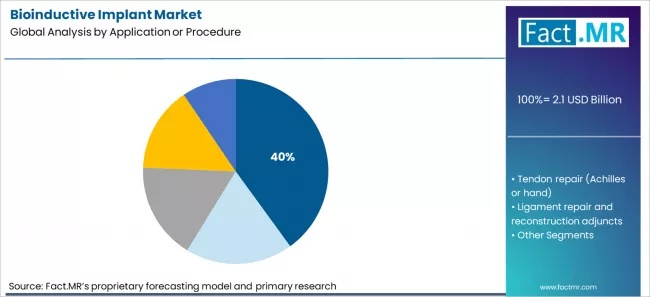
Procedure distribution in the bioinductive implant market shows rotator cuff repair augmentation at 40.0% of application volume. This area generates a high number of cases that involve partial healing risk and variable tendon quality. Surgeons follow patients through long imaging and functional assessment cycles, which keeps attention focused on reinforcement strategies. Operating schedules in sports medicine centers contain a steady flow of shoulder repairs across age groups and activity levels. Implant use in this setting fits into established coding, documentation, and follow up routines. Tendon, ligament, meniscus, and cartilage procedures also use bioinductive systems. The scale and frequency of cuff repairs keep this indication at the center of implant utilization planning.
What are the Drivers, Restraints, and Key Trends of the Bioinductive Implant Market?
- Drivers: Growing incidence of orthopedic and soft tissue injuries, rising demand for biologically active solutions that support tissue regeneration, and increased adoption of minimally invasive surgical techniques are driving use of bioinductive implants in musculoskeletal care.
- Restraint: High cost of bioinductive implant procedures, reimbursement variability across regions, and cautious clinician uptake due to evidence requirements limit rapid market expansion in some healthcare settings.
- Trend 1: Development of implants with tailored bioactive coatings and matrices designed to enhance cellular response and tissue integration.
- Trend 2: Expansion of indications beyond tendon repair into broader applications such as cartilage and spinal tissue support.
Analysis of the Bioinductive Implant Market by Key Country
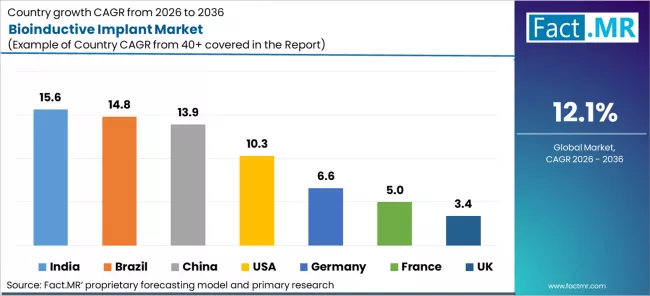
| Country | CAGR 2026 to 2036 |
|---|---|
| USA | 10.3% |
| Brazil | 14.8% |
| China | 13.9% |
| India | 15.6% |
| Germany | 6.6% |
| France | 5.0% |
| UK | 3.4% |
The report covers an in-depth analysis of 30+ countries; top-performing countries are highlighted below.
How Are Orthopedic Procedure Standardization Programs in USA Shaping Use of Bioinductive Implants?
In USA, this market advances at close to 10.3% per year through 2036. Adoption follows procedure standardization inside sports medicine and shoulder repair pathways rather than surgeon by surgeon experimentation. Hospital systems evaluate these implants through outcomes committees focused on revision rates and recovery timelines. Initial use concentrates in rotator cuff and tendon repair cases. Purchasing decisions sit with value analysis teams rather than individual departments. Rollouts usually begin in high volume centers. Case volumes increase when protocols include bioinductive options as default steps. Training schedules influence expansion speed. Continued use depends on measured reductions in retear rates and follow up imaging findings rather than on marketing exposure or short term surgeon preference.
What Is Driving Faster Expansion of Bioinductive Implants in Brazil?
Brazil records growth near 14.8% per year through 2036. Demand rises from private hospital groups seeking to differentiate orthopedic services. Many facilities still rely on conventional repair methods, which creates room for procedural upgrades. Adoption often starts inside high throughput sports injury centers. Purchasing decisions balance clinical benefit against implant cost. Training support plays a large role in early uptake. Rollout speed depends on surgeon acceptance and hospital investment cycles. Case volumes grow as more surgeons receive certification. Continued use depends on recovery outcomes and patient throughput rather than on promotional activity. Market expansion remains tied to private hospital investment rather than to broad public system adoption.
What Is Supporting Broader Use of Bioinductive Implants in China?
China shows expansion close to 13.9% per year through 2036. Hospital modernization programs and rising surgical volumes shape most adoption patterns. Initial use concentrates in large urban orthopedic centers. Procurement decisions align with multiyear equipment and implant upgrade plans. Surgeon training programs influence how quickly procedures spread. Integration into treatment guidelines determines long term volume. Rollouts often proceed in waves tied to hospital investment phases. Case numbers increase when procedures move beyond flagship departments. Local distribution and service support influence supplier selection. Market development remains linked to institutional upgrade cycles rather than to individual surgeon driven trial usage.
How Are Multi Site Hospital Networks in India Changing Use of Bioinductive Implants?
In India, this market progresses at roughly 15.6% per year through 2036. Growth is driven by hospital chains seeking standardized repair outcomes across locations. High sports injury and trauma caseloads support adoption. Initial use appears in tertiary care centers before reaching secondary facilities. Purchasing decisions weigh cost, training needs, and patient affordability. Surgeon education programs shape rollout pace. Case volumes expand as protocols become standardized across networks. Continued use depends on recovery metrics and patient satisfaction rather than on novelty appeal. Market growth follows network expansion and service line development rather than isolated adoption by individual clinics.
What Is Defining Adoption of Bioinductive Implants in Germany?
Germany shows growth near 6.6% per year through 2036. Adoption follows structured evaluation pathways and clinical evidence review. Hospitals emphasize guideline alignment and reimbursement clarity before scaling use. Initial cases concentrate in specialist orthopedic centers. Procurement committees require documented outcome improvements. Rollouts proceed cautiously across departments. Training certification remains a prerequisite for wider use. Case volume increases after formal inclusion in treatment protocols. Supplier choice depends on compliance support and service reliability. Market development stays tied to formal assessment cycles rather than to rapid expansion based on early adopter enthusiasm.
How Are Reimbursement and Evaluation Frameworks in France Influencing Use of Bioinductive Implants?
France records expansion near 5.0% per year through 2036. Adoption centers on careful assessment of clinical benefit within established reimbursement structures. Initial use appears in selected reference centers. Procurement decisions emphasize cost effectiveness and evidence strength. Integration into standard care pathways takes time. Rollouts remain gradual and department focused. Case volumes grow after inclusion in approved procedure lists. Supplier competition centers on clinical data support and training programs. Continued usage depends on audit outcomes and budget reviews. Market development remains incremental rather than rapid.
What Is Shaping Demand for Bioinductive Implants in UK?
In UK, growth holds near 3.4% per year through 2036. Adoption is influenced by centralized evaluation and funding approval processes. Use often begins through pilot programs inside selected trusts. Procurement decisions emphasize value for money and evidence based outcomes. Rollout speed depends on approval pathways rather than clinical interest alone. Case volumes increase slowly as programs extend across sites. Training availability affects utilization rates. Supplier selection favors proven references and service support. Continued use depends on alignment with pathway level outcome targets rather than on individual surgeon preference.
How Do Surgeon Preference and Procedure Pathways Shape Competition in the Bioinductive Implant Market?
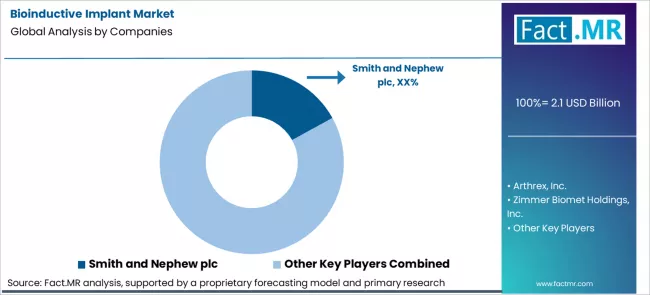
Case adoption in the bioinductive implant market is decided inside operating rooms, not purchasing offices. Surgeons first set indications, fixation methods, and post procedure protocols before any brand is considered. Smith and Nephew, Arthrex, Zimmer Biomet, Stryker, and Integra LifeSciences compete through portfolio fit across rotator cuff, sports medicine, and soft tissue repair workflows. Training programs, instrument compatibility, and published outcomes influence inclusion in hospital preference cards.
Committees review suppliers using handling consistency, revision rates, and documentation support rather than unit price. Once a system is accepted, usage follows surgeon volume and case mix. Supplier movement appears mainly after guideline updates, changes in surgeon leadership, or platform consolidation decisions.
Key Players in Bioinductive Implant Market
- Smith & Nephew plc
- Arthrex, Inc.
- Zimmer Biomet Holdings, Inc.
- Stryker Corporation
- Integra LifeSciences Holdings Corporation
References
- U.S. Food and Drug Administration. (2023). Use of International Standard ISO 10993-1, “Biological evaluation of medical devices - Part 1: Evaluation and testing within a risk management process”. U.S. Department of Health and Human Services.
- U.S. Food and Drug Administration. (2024). Class II special controls guidance document: Surgical meshes and soft tissue reinforcement implants. U.S. Department of Health and Human Services.
- U.S. Food and Drug Administration. (2023). Technical considerations for medical devices that contain materials derived from animal sources. U.S. Department of Health and Human Services.
- European Commission, Medical Device Coordination Group. (2023). Clinical evaluation guidance under Regulation (EU) 2017/745 (MDR). European Commission.
- International Organization for Standardization. (2023). ISO 10993-1: Biological evaluation of medical devices - Part 1: Evaluation and testing within a risk management process. ISO.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Implant Type | Collagen bioinductive patches, ECM scaffolds (xenograft or allograft), synthetic bioinductive scaffolds, bioactive-coated repair adjuncts, fixation and delivery accessories |
| Application or Procedure | Rotator cuff repair augmentation, tendon repair (Achilles or hand), ligament repair and reconstruction adjuncts, meniscus or cartilage repair support, other sports medicine procedures |
| End User or Sales Channel | Hospitals, ambulatory surgery centers (ASCs), orthopedic specialty clinics, distributor channels, GPO or tender procurement |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordics, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Smith & Nephew, Arthrex, Zimmer Biomet, Stryker, Integra LifeSciences |
| Additional Attributes | Dollar sales by implant type, application, and end user; handling behavior, fixation method, and resorption profile; procedure pathway adoption, surgeon training requirements, and rollout by hospital networks |
Bioinductive Implant Market Segmentation
-
Implant Type :
- Collagen bioinductive patches
- ECM scaffolds (xenograft or allograft)
- Synthetic bioinductive scaffolds
- Bioactive-coated repair adjuncts
- Fixation & delivery accessories
-
Application or Procedure :
- Rotator cuff repair augmentation
- Tendon repair (Achilles or hand)
- Ligament repair & reconstruction adjuncts
- Meniscus or cartilage repair support
- Other sports medicine procedures
-
End User or Sales Channel :
- Hospitals
- Ambulatory surgery centers (ASCs)
- Orthopedic specialty clinics
- Distributor channels
- GPO or tender procurement
-
Region :
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
- Asia Pacific
- Frequently Asked Questions -
How big is the bioinductive implant market in 2026?
The global bioinductive implant market is estimated to be valued at USD 2.1 billion in 2026.
What will be the size of bioinductive implant market in 2036?
The market size for the bioinductive implant market is projected to reach USD 6.6 billion by 2036.
How much will be the bioinductive implant market growth between 2026 and 2036?
The bioinductive implant market is expected to grow at a 12.1% CAGR between 2026 and 2036.
What are the key product types in the bioinductive implant market?
The key product types in bioinductive implant market are collagen bioinductive patches, ecm scaffolds (xenograft or allograft), synthetic bioinductive scaffolds, bioactive-coated repair adjuncts and fixation and delivery accessories.
Which application or procedure segment to contribute significant share in the bioinductive implant market in 2026?
In terms of application or procedure, rotator cuff repair augmentation segment to command 40.0% share in the bioinductive implant market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Implant Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Implant Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Implant Type, 2026 to 2036
- Collagen bioinductive patches
- ECM scaffolds (xenograft or allograft)
- Synthetic bioinductive scaffolds
- Bioactive-coated repair adjuncts
- Fixation and delivery accessories
- Collagen bioinductive patches
- Y to o to Y Growth Trend Analysis By Implant Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Implant Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application or Procedure
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application or Procedure, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application or Procedure, 2026 to 2036
- Rotator cuff repair augmentation
- Tendon repair (Achilles or hand)
- Ligament repair and reconstruction adjuncts
- Meniscus or cartilage repair support
- Other sports medicine procedures
- Rotator cuff repair augmentation
- Y to o to Y Growth Trend Analysis By Application or Procedure, 2021 to 2025
- Absolute $ Opportunity Analysis By Application or Procedure, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User or Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User or Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User or Sales Channel, 2026 to 2036
- Hospitals
- Ambulatory surgery centers (ASCs)
- Orthopedic specialty clinics
- Distributor channels
- GPO or tender procurement
- Hospitals
- Y to o to Y Growth Trend Analysis By End User or Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By End User or Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Implant Type
- By Application or Procedure
- By End User or Sales Channel
- Competition Analysis
- Competition Deep Dive
- Smith and Nephew plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Arthrex, Inc.
- Zimmer Biomet Holdings, Inc.
- Stryker Corporation
- Integra LifeSciences Holdings Corporation
- Smith and Nephew plc
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application or Procedure, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User or Sales Channel, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application or Procedure, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User or Sales Channel, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application or Procedure, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User or Sales Channel, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application or Procedure, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User or Sales Channel, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application or Procedure, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User or Sales Channel, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application or Procedure, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User or Sales Channel, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application or Procedure, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User or Sales Channel, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Implant Type, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application or Procedure, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User or Sales Channel, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Implant Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Implant Type
- Figure 6: Global Market Value Share and BPS Analysis by Application or Procedure, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Application or Procedure, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application or Procedure
- Figure 9: Global Market Value Share and BPS Analysis by End User or Sales Channel, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by End User or Sales Channel, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User or Sales Channel
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Implant Type, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Implant Type
- Figure 26: North America Market Value Share and BPS Analysis by Application or Procedure, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Application or Procedure, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application or Procedure
- Figure 29: North America Market Value Share and BPS Analysis by End User or Sales Channel, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by End User or Sales Channel, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User or Sales Channel
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Implant Type, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Implant Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Application or Procedure, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Application or Procedure, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Application or Procedure
- Figure 39: Latin America Market Value Share and BPS Analysis by End User or Sales Channel, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End User or Sales Channel, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User or Sales Channel
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Implant Type, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Implant Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application or Procedure, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Application or Procedure, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application or Procedure
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User or Sales Channel, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End User or Sales Channel, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User or Sales Channel
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Implant Type, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Implant Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application or Procedure, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Application or Procedure, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application or Procedure
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User or Sales Channel, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End User or Sales Channel, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User or Sales Channel
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Implant Type, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Implant Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Application or Procedure, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Application or Procedure, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Application or Procedure
- Figure 69: East Asia Market Value Share and BPS Analysis by End User or Sales Channel, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End User or Sales Channel, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User or Sales Channel
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Implant Type, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Implant Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application or Procedure, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Application or Procedure, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application or Procedure
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User or Sales Channel, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End User or Sales Channel, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User or Sales Channel
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Implant Type, 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Implant Type, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Implant Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application or Procedure, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Application or Procedure, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application or Procedure
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User or Sales Channel, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End User or Sales Channel, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User or Sales Channel
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis