Checkweighers with Rejection Systems for Pharma Blister Lines Market Size, Share, Growth and Forecast (2026 - 2036)
The Checkweighers with Rejection Systems for Pharma Blister Lines Market is segmented by Rejection Type (Air Blast Rejection, Pusher Rejection, Drop Rejection, and Diverter Rejection), Application (Tablet Blister Packs, Capsule Blister Packs, Combination Packs, and Medical Device Blisters), Speed Capacity (Up to 200 Packs per Minute, 200 to 400 Packs per Minute, and Above 400 Packs per Minute), and Region. Forecast for 2026 to 2036.
Fact MR analysts observe the checkweighers with rejection systems for pharma blister lines market expanding from USD 268.4 million in 2025 to USD 628.9 million by 2036 at a 7.9% CAGR. Tablet blister packs represent 41% share supported by high volume packaging demand, while up to 200 packs per minute holds 36% reflecting standardized production speeds.
Checkweighers with Rejection Systems for Pharma Blister Lines Market Forecast and Outlook By Fact.MR
Valuation of the checkweighers with rejection systems for pharma blister lines market stood at USD 268.4 million in 2025. According to Fact MR, demand for automated checkweighing and rejection systems for pharmaceutical blister packaging is expected to reach USD 294.7 million in 2026 and USD 628.9 million by 2036. A CAGR of 7.9% is projected for the forecast timeline.
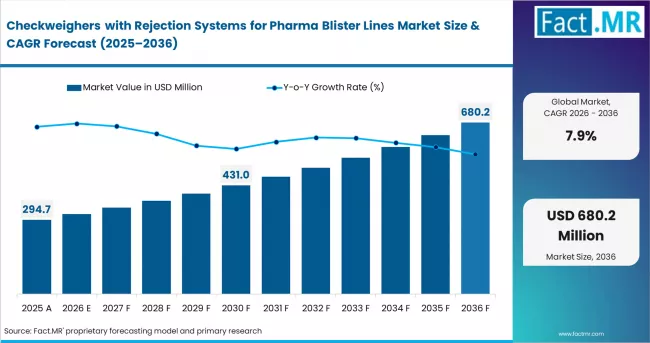
Checkweighers with Rejection Systems for Pharma Blister Lines Market
| Metric | Details |
|---|---|
| Industry Size (2026E) | USD 294.7 million |
| Industry Value (2036F) | USD 628.9 million |
| CAGR (2026 to 2036) | 7.9% |
Summary of the Checkweighers with Rejection Systems for Pharma Blister Lines Market
- Market Definition
- The market includes automated in line weighing systems integrated with rejection mechanisms designed to verify blister pack weight accuracy and remove non compliant pharmaceutical packages during high speed packaging operations.
- Demand Drivers
- Increasing pharmaceutical serialization compliance requirements supporting automated weight verification across blister packaging lines.
- Rising need for dosage accuracy validation ensuring consistent tablet count within sealed blister cavities.
- Growing deployment of integrated rejection systems supporting removal of underweight or incomplete blister packs.
- Expansion of high throughput packaging environments requiring continuous inspection capability across production workflows.
- Increasing regulatory enforcement linked to cGMP packaging control standards requiring validated inspection equipment.
- Rising adoption of precision load cell technologies supporting stable weight measurement accuracy across high speed conveyor systems.
- Key Segments Analyzed
- Application: Tablet blister packs lead with 41% share supported by high volume solid dosage packaging demand.
- Speed Capacity: Up to 200 packs per minute holds 36% share reflecting standardized pharmaceutical packaging throughput configurations.
- Rejection Technology: Air blast and pusher rejection systems support rapid removal of non conforming blister packs.
- Inspection Role: Dynamic checkweighing supports real time verification of dosage completeness across automated packaging lines.
- Geography: Asia Pacific and Europe maintain strong deployment due to pharmaceutical manufacturing capacity expansion.
- Analyst Opinion at Fact MR
- Shambhu Nath Jha, Principal Consultant, Fact MR, opines, 'In this updated edition of the Checkweighers with Rejection Systems for Pharma Blister Lines Market report, automated weight inspection technologies remain critical for maintaining pharmaceutical packaging accuracy and regulatory compliance across high speed solid dosage manufacturing environments through 2036.'
- Strategic Implications or Executive Takeaways
- Strengthen precision weighing capability supporting consistent detection of missing tablet conditions.
- Invest in integrated rejection mechanisms enabling removal of defective blister packs without disrupting packaging flow.
- Improve compatibility with serialization and electronic batch record systems supporting regulatory audit requirements.
- Expand deployment across moderate speed packaging lines requiring stable inspection accuracy performance.
- Focus on validation ready inspection equipment aligned with pharmaceutical quality control frameworks.
- Enhance synchronization capability supporting real time response across continuous blister packaging operations.
- Methodology
- Primary interviews conducted with pharmaceutical packaging equipment manufacturers and inspection system integrators.
- Benchmarked against pharmaceutical production output influencing demand for automated packaging verification systems.
- Evaluated installation trends across high speed blister packaging environments requiring dosage accuracy validation.
- Hybrid modeling applied combining top down pharmaceutical packaging automation demand assessment with bottom up installation benchmarking.
- Validation conducted using supplier level inspection equipment deployment indicators across regulated pharmaceutical production facilities.
- Peer review applied using Fact MR analytical frameworks linking packaging compliance requirements with automated inspection system demand patterns.
A CAGR of 7.9% indicates moderate to transformational expansion supported by pharmaceutical compliance requirements for weight accuracy and defect rejection in blister packaging lines. Variability persists due to capital expenditure cycles and validation timelines, while adoption remains sustained by serialization integrity and quality assurance mandates.
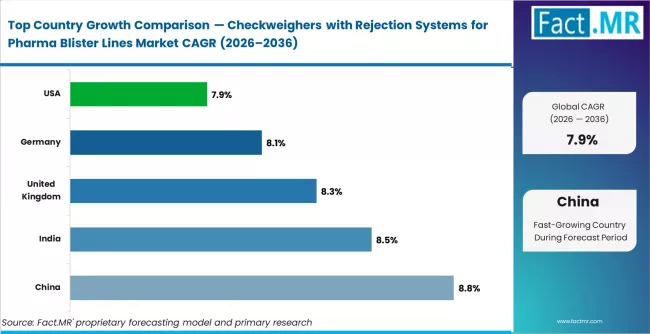
China leads with a projected CAGR of 8.8%, supported by expansion of automated pharmaceutical packaging lines requiring in-line weight verification systems. India follows with a CAGR of 8.5%, driven by increasing integration of quality control automation across drug packaging production environments. The United Kingdom records a CAGR of 8.3%, reflecting steady adoption of automated inspection and rejection systems across regulated pharmaceutical manufacturing processes. Germany shows a CAGR of 8.1%, supported by consistent implementation of precision weighing technologies across high-speed blister packaging operations. The United States records the slowest growth at 7.9%, reflecting a relatively mature packaging automation environment tied to replacement demand within existing pharmaceutical inspection infrastructures.
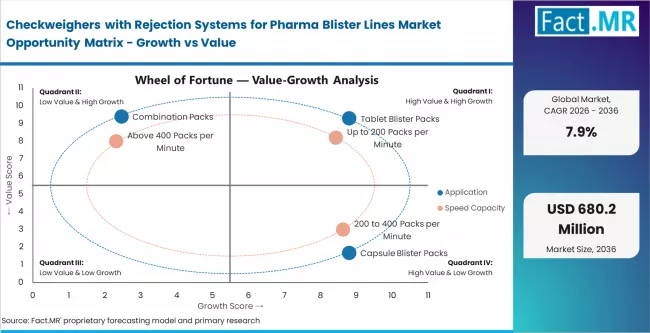
Segmental Analysis
Checkweighers with Rejection Systems for Pharma Blister Lines Market Analysis by Application
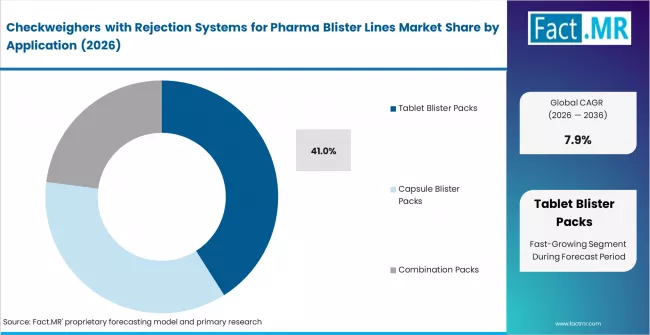
- Market Overview: Based on Fact MR assessment, tablet blister packs are projected to account for 41% share of the checkweighers with rejection systems for pharma blister lines market in 2026. In line weight verification systems are integrated across blister packaging lines requiring precise measurement of filled cavities to detect missing or broken tablets. Automated rejection mechanisms remove non conforming blister packs based on defined weight tolerance thresholds aligned with pharmaceutical packaging quality control requirements. Equipment configuration supports continuous monitoring across high speed packaging workflows requiring controlled dosage verification performance aligned with regulatory compliance parameters governing pharmaceutical product consistency.
- Demand Drivers:
- Dosage Verification Requirements: Checkweighers support identification of underfilled blister packs enabling removal of defective units across pharmaceutical packaging lines.
- Quality Control Parameters: Automated rejection systems support compliance with defined packaging weight tolerance standards across tablet blister production workflows.
- Process Reliability Needs: Continuous weight monitoring supports detection of packaging inconsistencies across high throughput pharmaceutical manufacturing operations.
Checkweighers with Rejection Systems for Pharma Blister Lines Market Analysis by Speed Capacity
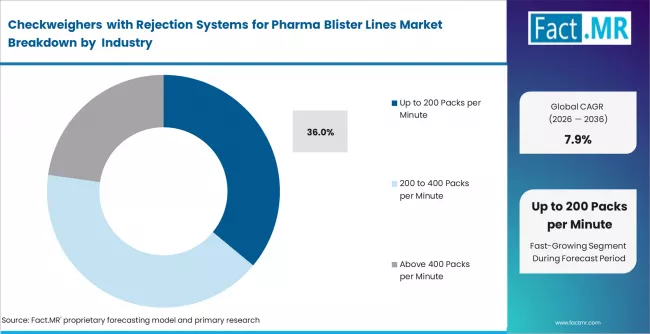
- Market Overview: Speed capacity up to 200 packs per minute is estimated to hold 36% share of the checkweighers with rejection systems for pharma blister lines market in 2026, supported by requirement for inline inspection systems capable of operating within moderate to high speed packaging environments. Equipment design supports stable weighing accuracy across continuous conveyor movement requiring controlled synchronization with blister sealing and cutting processes. Performance configuration supports consistent rejection response timing aligned with defined production throughput parameters applied across pharmaceutical blister packaging operations requiring reliable inspection efficiency.
- Demand Drivers:
- Inspection Throughput Requirements: Checkweighers support weight verification across continuous packaging flow enabling stable performance at moderate production speeds.
- Synchronization Parameters: Inspection equipment supports integration with conveyor systems requiring coordinated rejection timing across packaging line operations.
- Accuracy Stability Needs: Controlled weighing mechanisms support consistent detection of weight deviation across high frequency blister pack production workflows.
Key Dynamics
Checkweighers with Rejection Systems for Pharma Blister Lines Market Drivers, Restraints, and Opportunities
FMR analysts observe that historical demand originates from pharmaceutical packaging validation processes where batch-level weight verification ensured dosage consistency and prevented incomplete blister packs from entering distribution channels. The current market size reflects a structurally stable quality assurance segment shaped by regulatory enforcement requiring automated in-line verification of tablet count, fill accuracy, and packaging integrity. Structural reality indicates a mature but steadily expanding market because high-speed blister packaging lines require continuous inspection capability to maintain compliance with strict drug product quality specifications and minimize recall exposure associated with packaging deviations.
The present structural shift reflects declining reliance on manual sampling inspection as high-speed checkweighers integrated with automatic rejection systems enable real-time identification of underfilled or overfilled blister packs. Higher capital cost per inspection line is offset by reduced product giveaway, lower batch rejection rates, and improved compliance documentation aligned with electronic batch record systems. Value growth remains supported by serialization expansion and higher throughput packaging lines requiring continuous validation of packaging accuracy across diverse dosage formats.
- cGMP Packaging Control: FDA 21 CFR Part 211 packaging and process control requirements sustain demand for automated weight verification ensuring batch uniformity and product integrity.
- Zero-Error Packaging: Integrated rejection systems remove incomplete blister packs at high line speeds, reducing recall risk linked to missing tablets and dosage inconsistencies.
- Europe Solid Dose Base: Germany, Italy, and Switzerland maintain strong adoption due to dense concentration of high-speed blister packaging facilities supporting global generic drug supply chains.
Regional Analysis
The checkweighers with rejection systems for pharma blister lines market is assessed across North America, Europe, and Asia Pacific, segmented by country-level demand in pharmaceutical packaging weight verification, automated rejection handling, serialization-compliant inspection, and production line quality assurance systems. Regional demand reflects tightening drug packaging accuracy standards and adoption of integrated inspection technologies. The full report offers market attractiveness analysis.
.webp)
| Country | CAGR (2026–2036) |
|---|---|
| China | 8.8% |
| India | 8.5% |
| United Kingdom | 8.3% |
| Germany | 8.1% |
| United States | 7.9% |
Source: Fact MR analysis, based on proprietary forecasting model and primary research
Asia Pacific
Asia Pacific functions as the pharmaceutical packaging line automation hub, supported by expansion of solid dosage manufacturing capacity and increased adoption of integrated inspection systems. Mettler Toledo International Inc. strengthens precision checkweighing technology capability. Ishida Co., Ltd. expands high-speed weight inspection systems. Anritsu Corporation supports pharmaceutical packaging compliance solutions.
- China: China is projected to record 8.8% CAGR in checkweighers with rejection systems for pharma blister lines through 2036. Drug serialization compliance update (National Medical Products Administration, February 2024) supports automated packaging inspection adoption. Anritsu Corporation expanded high-precision checkweighing integration capability (June 2023).
- India: Installation of checkweighers with rejection systems for pharma blister lines in India is forecast to grow at 8.5% CAGR through 2036. Central Drugs Standard Control Organization packaging validation guideline update (January 2024) supports automated weight verification deployment. Mettler Toledo expanded pharmaceutical inspection engineering capability (May 2023).
Europe
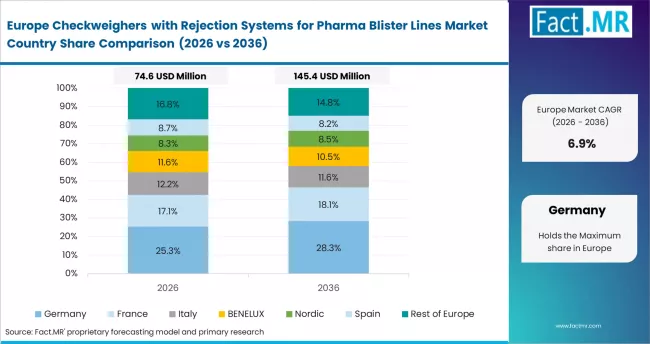
Europe operates as the pharmaceutical packaging accuracy compliance center, supported by strict medicinal product verification frameworks and structured quality assurance standards. Syntegon Technology GmbH strengthens integrated inspection platform capability. Körber AG expands pharmaceutical packaging automation software. Mettler Toledo International Inc. supports high-precision weighing technologies.
- United Kingdom: Utilization of checkweighers with rejection systems for pharma blister lines in United Kingdom is expected to expand at 8.3% CAGR through 2036. Medicines and Healthcare products Regulatory Agency packaging verification update (March 2024) supports automated inspection adoption. Körber AG expanded pharmaceutical packaging inspection software capability (July 2023).
- Germany: Germany is anticipated to observe 8.1% CAGR in checkweighers with rejection systems for pharma blister lines through 2036. EU Falsified Medicines Directive serialization compliance update (January 2024) supports high-precision packaging verification technologies. Syntegon Technology GmbH expanded integrated inspection system engineering capability (April 2023).
North America
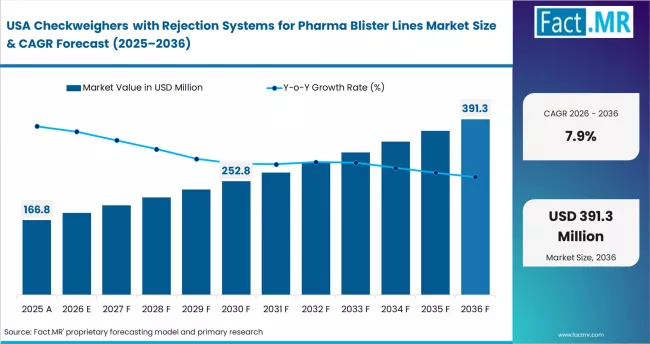
North America represents the pharmaceutical quality control technology deployment environment, supported by adoption of automated inspection and rejection handling systems across high-speed packaging lines. Thermo Fisher Scientific Inc. strengthens pharmaceutical weighing accuracy capability. Mettler Toledo International Inc. expands high-speed checkweighing systems. Antares Vision Group supports integrated packaging verification technologies.
- United States: The United States is forecast to witness 7.9% CAGR in checkweighers with rejection systems for pharma blister lines through 2036. U.S. Food and Drug Administration data integrity guidance update (April 2024) supports automated packaging verification deployment. Thermo Fisher Scientific Inc. expanded pharmaceutical inspection instrumentation capability (August 2023).
Fact MR's analysis of checkweighers with rejection systems for pharma blister lines market in global regions consists of country-wise assessment that includes China, India, United Kingdom, Germany, and United States. Readers can find pharmaceutical packaging inspection trends, automated rejection system developments, regulatory compliance signals, and competitive positioning across key markets.
Competitive Landscape
Competitive Structure and Buyer Dynamics in the Checkweighers with Rejection Systems for Pharma Blister Lines Market
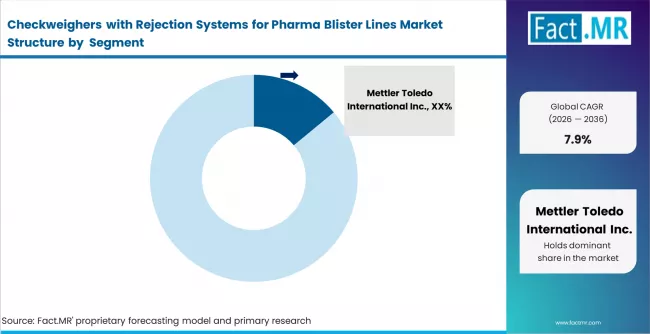
The competitive structure of the Checkweighers with Rejection Systems for Pharma Blister Lines Market is moderately concentrated, with precision measurement equipment manufacturers controlling a significant share of pharmaceutical packaging inspection solutions. Companies such as Mettler Toledo International Inc., WIPOTEC OCS GmbH, Ishida Co. Ltd., Anritsu Corporation, Minebea Intec GmbH, Thermo Fisher Scientific Inc., Bizerba SE & Co. KG, Loma Systems Ltd., Multivac Group, and Yamato Scale Co. Ltd. maintain strong positions through established weighing technologies and automated rejection mechanisms designed for high speed packaging lines. Competition is primarily influenced by weighing accuracy, rejection precision, validation capability, and compliance with pharmaceutical quality standards.
Several companies maintain structural advantages through proprietary load cell technologies and established relationships with pharmaceutical packaging equipment manufacturers. Firms such as Mettler Toledo International Inc. and Minebea Intec GmbH benefit from strong expertise in precision weighing systems and global service infrastructure supporting validation requirements. WIPOTEC OCS GmbH and Anritsu Corporation maintain advantages through high speed inspection technologies integrated with packaging automation systems. Pharmaceutical manufacturers typically adopt multi vendor inspection strategies to reduce dependence on a single equipment provider and ensure compliance reliability. Procurement decisions evaluate suppliers based on measurement precision, system validation support, and long term operational stability, moderating supplier pricing leverage across pharmaceutical packaging environments.
Key Players of the Checkweighers with Rejection Systems for Pharma Blister Lines Market
- Mettler Toledo International Inc.
- WIPOTEC OCS GmbH
- Ishida Co. Ltd.
- Anritsu Corporation
- Thermo Fisher Scientific Inc.
- Minebea Intec GmbH
- Bizerba SE & Co. KG
- Loma Systems Ltd.
- Multivac Group
- Yamato Scale Co. Ltd.
Bibliographies
- [1] National Medical Products Administration. (2024, February). Drug serialization compliance guidelines for pharmaceutical packaging lines. Government of China.
- [2] Central Drugs Standard Control Organization. (2024, January). Packaging validation guidelines for automated pharmaceutical blister lines. Government of India.
- [3] Medicines and Healthcare products Regulatory Agency. (2024, March). Packaging verification standards update for MHRA compliance. UK Government.
- [4] European Medicines Agency. (2024, January). Falsified medicines directive serialization compliance update for high speed packaging verification. European Medicines Agency.
- [5] U.S. Food and Drug Administration. (2024, April). Data integrity guidance for automated pharmaceutical packaging inspection systems 21 CFR Part 11 compliance. U.S. Department of Health and Human Services.
- [6] Anritsu Corporation. (2023, June). High precision checkweighing integration for pharmaceutical blister packaging lines. Anritsu Corporation.
- [7] Mettler Toledo International Inc. (2023, May). Pharmaceutical inspection engineering expansion for automated blister pack weight verification. Mettler Toledo International Inc.
This Report Addresses
- Market size forecasts for 2026 to 2036 based on adoption of automated weight verification systems across pharmaceutical blister packaging lines.
- Opportunity mapping across air blast, pusher, drop, and diverter rejection mechanisms supporting high precision defect removal workflows.
- Segment and regional forecasts covering tablet blister packs, capsule blister packs, combination packs, and medical device blister packaging applications.
- Competition benchmarking based on weighing accuracy tolerance capability, rejection response timing precision, and integration compatibility with high speed blister packaging equipment.
- Regulatory assessment covering cGMP packaging controls and dosage verification requirements influencing deployment of automated checkweighing systems.
- Report delivery in PDF, Excel, PPT, and dashboard formats supporting pharmaceutical packaging engineers, quality assurance specialists, and inspection equipment integrators.
- Operational risk analysis covering calibration drift, rejection synchronization timing variability, vibration sensitivity in conveyor weighing systems, and validation complexity across regulated pharmaceutical packaging workflows.
Checkweighers with Rejection Systems for Pharma Blister Lines Market Definition
Checkweighers with Rejection Systems for Pharma Blister Lines Market Definition Paragraph
The Checkweighers with Rejection Systems for Pharma Blister Lines Market includes automated in line weighing systems integrated with rejection mechanisms that verify the weight accuracy of blister packs and automatically remove packages with missing tablets, incorrect counts, or deviations from specified limits during pharmaceutical packaging operations.
Checkweighers with Rejection Systems for Pharma Blister Lines Market Inclusions
The report includes global and regional market size estimates, forecast analysis, and segmentation by checkweigher type, rejection mechanism, inspection accuracy level, integration architecture, application stage, end use industry, pricing structure, and compatibility with pharmaceutical packaging automation systems.
Checkweighers with Rejection Systems for Pharma Blister Lines Market Exclusions
The scope excludes standalone blister inspection vision systems without weight verification, manual weighing equipment, packaging machinery not equipped with automated rejection functionality, and pharmaceutical production equipment unrelated to packaging quality control.
Checkweighers with Rejection Systems for Pharma Blister Lines Research Methodology
- Primary Research: Interviews were conducted with inspection equipment manufacturers, pharmaceutical packaging line integrators, quality assurance specialists, and automation technology providers.
- Desk Research: Public sources included pharmaceutical packaging standards, company technical documentation, regulatory guidelines, and technical literature on dynamic weighing and rejection systems.
- Market-Sizing and Forecasting: A hybrid model combining top-down pharmaceutical packaging automation demand evaluation and bottom-up analysis of checkweigher installation across blister packaging lines was applied.
- Data Validation and Update Cycle: Outputs were validated through cross comparison of supplier data, expert consultation, and periodic monitoring of pharmaceutical quality inspection technology adoption trends.
Report Scope

| Metric | Value |
| Quantitative Units | USD 294.7 million (2026) to USD 628.9 million (2036), at a CAGR of 7.9% |
| Market Definition | The checkweighers with rejection systems for pharma blister lines market includes automated weight inspection equipment integrated with rejection mechanisms to identify underweight or overweight blister packs and remove non-compliant units from high-speed pharmaceutical packaging lines. |
| Rejection Type Segmentation | Air blast rejection, Pusher rejection, Drop rejection, Diverter rejection |
| Application Segmentation | Tablet blister packs, Capsule blister packs, Combination packs, Medical device blisters |
| Speed Capacity Segmentation | Up to 200 packs per minute, 200 to 400 packs per minute, Above 400 packs per minute |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, France, United Kingdom, Italy, Spain, Netherlands, Switzerland, China, Japan, South Korea, India, Singapore, Australia, Brazil, Mexico, United Arab Emirates, and 40+ countries |
| Key Companies Profiled | Mettler Toledo International Inc., WIPOTEC OCS GmbH, Ishida Co. Ltd., Anritsu Corporation, Thermo Fisher Scientific Inc., Minebea Intec GmbH, Bizerba SE & Co. KG, Loma Systems Ltd., Multivac Group, Yamato Scale Co. Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market estimation based on pharmaceutical packaging automation adoption rates, high-speed inspection equipment demand benchmarking, regulatory compliance requirements for dosage accuracy, packaging line efficiency optimization trends, and validation through primary interviews with pharmaceutical equipment manufacturers, packaging system integrators, and quality assurance specialists. |
Checkweighers with Rejection Systems for Pharma Blister Lines Market Key Segments
-
Rejection Type:
- Air Blast Rejection
- Pusher Rejection
- Drop Rejection
- Diverter Rejection
-
Application:
- Tablet Blister Packs
- Capsule Blister Packs
- Combination Packs
- Medical Device Blisters
-
Speed Capacity:
- Up to 200 Packs per Minute
- 200 to 400 Packs per Minute
- Above 400 Packs per Minute
-
Region
- North America
- USA
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East and Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Rest of Middle East and Africa
- Other Regions
- Oceania
- Central Asia
- Other Markets
- North America
- Frequently Asked Questions -
How large is the demand for Checkweighers with Rejection Systems for Pharma Blister Lines in the global market in 2026?
Demand for checkweighers with rejection systems for pharma blister lines in the global market is estimated to be valued at USD 294.7 million in 2026.
What will be the market size of Checkweighers with Rejection Systems for Pharma Blister Lines in the global market by 2036?
Market size for checkweighers with rejection systems for pharma blister lines is projected to reach USD 628.9 million by 2036.
What is the expected demand growth for Checkweighers with Rejection Systems for Pharma Blister Lines in the global market between 2026 and 2036?
Demand for checkweighers with rejection systems for pharma blister lines is expected to grow at a CAGR of 7.9% between 2026 and 2036.
Which company is identified as a leading provider in the Checkweighers with Rejection Systems for Pharma Blister Lines market?
Mettler Toledo International Inc. is identified as a leading participant due to its precision weighing technology portfolio and pharmaceutical inspection system capabilities.
Which application segment is projected to dominate checkweigher adoption by 2026?
Tablet blister packs are expected to account for approximately 41% of total market share in 2026 due to high volume pharmaceutical packaging requirements.
Why are checkweighers with rejection systems widely used in pharmaceutical blister packaging lines?
Checkweighers enable verification of product weight accuracy and automated removal of defective packages from high speed production lines.
What is driving demand for checkweighers for pharma blister lines in China?
Expansion of pharmaceutical packaging automation and increasing implementation of quality control technologies are supporting market growth.
What is the growth outlook for the Checkweighers with Rejection Systems for Pharma Blister Lines market in China?
China is projected to expand at a CAGR of 8.8% during 2026 to 2036 supported by pharmaceutical packaging inspection demand.
Why is India an important market for pharmaceutical weight inspection systems?
Growth in pharmaceutical manufacturing capacity and increasing adoption of automated packaging verification technologies contribute to steady demand.
What is the growth outlook for the Checkweighers with Rejection Systems for Pharma Blister Lines market in India?
India is projected to grow at a CAGR of 8.5% between 2026 and 2036 supported by packaging quality control demand.
How is demand for pharma checkweighers evolving in the United Kingdom pharmaceutical sector?
Demand is supported by implementation of automated inspection systems and integration of precision weighing technologies.
What is the growth outlook for the Checkweighers with Rejection Systems for Pharma Blister Lines market in the United Kingdom?
The United Kingdom is projected to expand at a CAGR of 8.3% during 2026 to 2036 supported by pharmaceutical inspection demand.
What is the growth outlook for the Checkweighers with Rejection Systems for Pharma Blister Lines market in Germany?
Germany is projected to grow at a CAGR of 8.1% between 2026 and 2036 supported by pharmaceutical packaging automation demand.
How is the United States positioned in the Checkweighers with Rejection Systems for Pharma Blister Lines market?
The United States demonstrates steady demand supported by integration of automated inspection technologies in pharmaceutical production facilities.
What is the growth outlook for the Checkweighers with Rejection Systems for Pharma Blister Lines market in the United States?
The United States is projected to expand at a CAGR of 7.9% during 2026 to 2036 supported by packaging quality verification demand.
What are checkweighers with rejection systems and what are they mainly used for in pharmaceutical blister lines?
Checkweighers with rejection systems are automated inspection devices used to measure product weight and remove non conforming blister packages from production lines.
What does the Checkweighers with Rejection Systems for Pharma Blister Lines market include in this report?
The market includes precision weighing systems, automated rejection mechanisms, inspection sensors, and pharmaceutical packaging verification technologies.
What applications are included in the scope of the Checkweighers with Rejection Systems for Pharma Blister Lines market?
Scope covers tablet blister packaging inspection, capsule packaging verification, automated weight checking processes, pharmaceutical quality control systems, and packaging compliance monitoring.
What is excluded from the scope of the Checkweighers with Rejection Systems for Pharma Blister Lines market report?
General industrial weighing equipment not configured for pharmaceutical packaging inspection applications are excluded unless integrated within automated checkweighing systems.
What does market forecast mean in the Checkweighers with Rejection Systems for Pharma Blister Lines market report?
Market forecast represents a structured projection based on pharmaceutical packaging automation trends and adoption of inspection technologies.
How is the Checkweighers with Rejection Systems for Pharma Blister Lines market forecast developed in this report?
Forecast modeling is based on evaluation of pharmaceutical production activity, packaging automation demand patterns, and supplier technology deployment indicators.
What does primary validation indicate in the Checkweighers with Rejection Systems for Pharma Blister Lines market analysis?
Primary validation involves assessment of inspection technology adoption indicators, pharmaceutical packaging production data, and supplier level system deployment trends supporting forecast assumptions.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application , 2026 to 2036
- API Intermediate
- Research
- API Intermediate
- Y to o to Y Growth Trend Analysis By Application , 2021 to 2025
- Absolute $ Opportunity Analysis By Application , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Purity
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Purity, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Purity, 2026 to 2036
- ≥ 99%
- < 99%
- ≥ 99%
- Y to o to Y Growth Trend Analysis By Purity, 2021 to 2025
- Absolute $ Opportunity Analysis By Purity, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Application
- By Purity
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Purity
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Application
- By Purity
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Purity
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Application
- By Purity
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Purity
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Application
- By Purity
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Purity
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Application
- By Purity
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Purity
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Application
- By Purity
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Purity
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Application
- By Purity
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Purity
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Purity
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By Purity
- Competition Analysis
- Competition Deep Dive
- Teva Pharmaceutical Industries Ltd.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Dr. Reddy’s Laboratories Ltd.
- Lupin Limited
- Sandoz Group AG
- Aurobindo Pharma Limited
- Sun Pharmaceutical Industries Limited
- Cipla Limited
- Hetero Drugs Limited
- Zhejiang Huahai Pharmaceutical Co., Ltd.
- Amneal Pharmaceuticals, Inc.
- Torrent Pharmaceuticals Limited
- Alkem Laboratories Limited
- Zydus Lifesciences Limited
- Jubilant Pharmova Limited
- Apotex Inc.
- Teva Pharmaceutical Industries Ltd.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Purity, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Application
- Figure 6: Global Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Purity
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Application
- Figure 23: North America Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Purity
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Application
- Figure 30: Latin America Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Purity
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Application
- Figure 37: Western Europe Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Purity
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Application
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Purity
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Application
- Figure 51: East Asia Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Purity
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Purity
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Purity, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Purity
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis