Healthcare Regulatory Affairs Outsourcing Market Size, Share, Growth and Forecast (2026 - 2036)
Healthcare Regulatory Affairs Outsourcing Market is segmented by Service (Regulatory Consulting, Legal Representation, Regulatory Writing and Publishing, Product Registration and Clinical Trial Applications, and Others), End-User (Pharmaceutical and Biotechnology Companies, Medical Device Companies, Food and Beverage Companies), and Region. Forecast for 2026 to 2036.
Fact MR opines that the healthcare regulatory affairs outsourcing market was valued at USD 4.3 billion in 2025 and is expected to increase to USD 4.5 billion in 2026 and USD 6.6 billion by 2036, growing at a 6.7% CAGR. Regulatory consulting is anticipated to account for 27.4% of the service market, while pharmaceutical and biotechnology companies are expected to make up 55% of the end-user segment.
Segmental Analysis
Healthcare Regulatory Affairs Outsourcing Market Analysis by Service
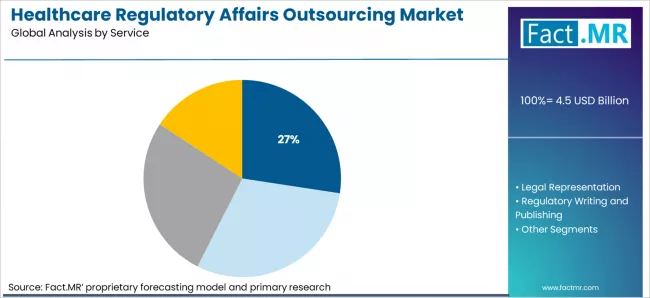
- Market Overview: Regulatory consulting is expected to capture 27.4% of the healthcare regulatory affairs outsourcing market by 2026. Regulatory consulting services provide companies with expertise on navigating complex regulatory frameworks, ensuring that their products comply with local, national, and international regulations.
- Demand Drivers:
- Navigating Complex Regulations: As healthcare companies increasingly expand their operations globally, the demand for regulatory consulting services grows. These services help companies understand and comply with ever-evolving regulations in different markets, ensuring smooth product approvals and market access.
- Expanding Product Portfolios: With healthcare companies continuously developing new drugs, devices, and therapies, regulatory consulting is essential for ensuring that these innovations meet the regulatory requirements of multiple regions.
- Compliance and Risk Management: Regulatory consultants provide strategic guidance on managing compliance risks and ensuring that businesses avoid costly fines, delays, or rejections of product approvals. This risk mitigation factor drives the demand for these services in the industry.
Healthcare Regulatory Affairs Outsourcing Market Analysis by End-User
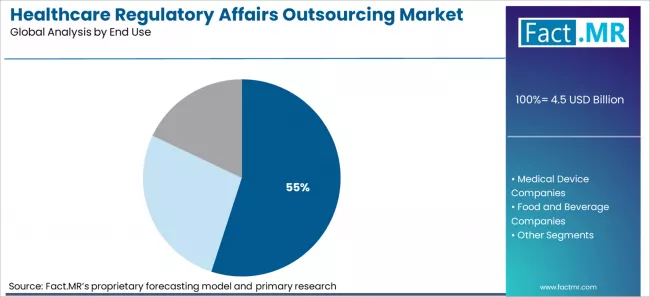
- Market Overview: Pharmaceutical and biotechnology companies are projected to hold 55% of the market share by 2026. Regulatory affairs outsourcing is crucial for these companies as they face complex regulatory requirements in drug development, clinical trials, and product approvals across multiple jurisdictions.
- Demand Drivers:
- Regulatory Complexity in Drug Development: Pharmaceutical and biotechnology companies require specialized regulatory expertise to navigate the complex and diverse regulations involved in drug development, clinical trials, and market authorization. Outsourcing regulatory affairs ensures these companies have access to the latest knowledge and insights on regulatory changes.
- Global Expansion of Biotech and Pharma: As pharmaceutical and biotechnology companies expand their operations globally, the need for regulatory outsourcing increases. These companies must manage a range of regulations across various regions, making outsourcing an efficient and cost-effective solution.
- Increased Focus on Innovation and Speed to Market: As the biopharmaceutical sector grows, companies are focused on bringing new drugs and therapies to market quickly. Outsourcing regulatory affairs enables these companies to streamline their processes and reduce time-to-market while ensuring compliance with regulatory standards.
Healthcare Regulatory Affairs Outsourcing Market Drivers, Restraints, and Opportunities
FMR analysts observe that the healthcare regulatory affairs outsourcing market is a growing compliance‑driven segment rooted in the increasing complexity of regulatory requirements across drugs, biologics, medical devices, and digital health products. Historically, many healthcare companies managed regulatory submissions and compliance in‑house; as global regulatory frameworks proliferated and requirements (e.g., unique device identification, clinical evaluation reports, evolving pharmacovigilance) became more demanding, structural demand emerged for specialized outsourced services. The 2026 market valuation reflects this shift, with rising demand tied to global product approvals, post‑market surveillance obligations, and cross‑border regulatory harmonization efforts.
While many firms still maintain internal regulatory teams, complex submissions, diverse regulatory environments, and the need for up‑to‑date compliance expertise are driving growth in outsourced solutions. These services carry higher per‑engagement value and are expanding as companies seek to mitigate risk and shorten time‑to‑market. The market exists at its current size because the pace of regulatory change outstrips many internal teams’ capacity, and outsourcing remains a cost‑effective way to navigate multifaceted regulatory landscapes.
- Compliance Complexity Demand: Companies outsource to navigate diverse global regulatory frameworks and meet requirements that internal teams struggle to manage alone.
- Regulatory Standards Pressure: Standards such as unique device identification and pharmacovigilance mandates push firms toward specialized outsourcing to meet stringent compliance criteria.
- Regional Regulatory Dynamics: In North America and Europe, stringent regulatory enforcement and frequent updates drive higher uptake of outsourced regulatory affairs services compared with regions where frameworks are less rigorous.
Regional Analysis
The market analysis covers key global regions, including East Asia, South Asia, Western Europe, North America, and China. It is segmented geographically, with specific market dynamics for each region. The full report provides a detailed market attractiveness analysis.
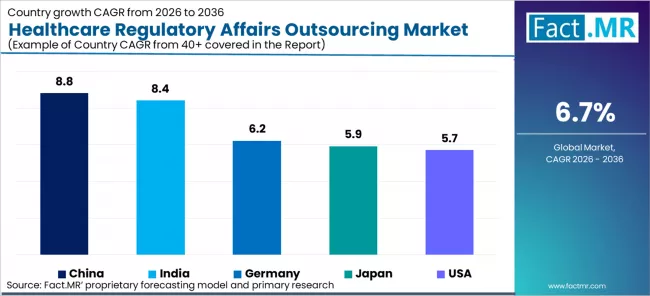
| Country | CAGR (2026-2036) |
|---|---|
| China | 8.8% |
| India | 8.4% |
| Germany | 6.2% |
| Japan | 5.9% |
| USA | 5.7% |
Source: Fact MR (FMR) analysis, based on proprietary forecasting model and primary research.
East Asia:
In East Asia, China and Japan are leading the healthcare regulatory affairs outsourcing market, driven by the growing pharmaceutical industry, increasing outsourcing trends, and regulatory complexity in these countries.
- China: Demand for healthcare regulatory affairs outsourcing in China is projected to rise at 8.8% CAGR through 2036. China’s growing pharmaceutical and biotech industries and increasing regulatory demands contribute to the market’s rapid growth.
- Japan: Demand for healthcare regulatory affairs outsourcing in Japan is projected to rise at 5.9% CAGR through 2036. Japan’s strong healthcare system and complex regulatory requirements continue to drive demand for outsourcing services in the healthcare regulatory affairs sector.
South Asia:
In South Asia, India is a prominent player in the healthcare regulatory affairs outsourcing market, driven by its robust outsourcing capabilities and growing pharmaceutical and healthcare industries.
- India: Demand for healthcare regulatory affairs outsourcing in India is projected to rise at 8.4% CAGR through 2036. India’s established outsourcing industry, coupled with growing regulatory complexities in the healthcare sector, continues to fuel market growth.
Western Europe:
In Western Europe, Germany is a key market for healthcare regulatory affairs outsourcing, supported by its large pharmaceutical industry and regulatory environment.
- Germany: Demand for healthcare regulatory affairs outsourcing in Germany is projected to rise at 6.2% CAGR through 2036. Germany’s strong healthcare and pharmaceutical sectors and the increasing need for regulatory compliance continue to drive the demand for outsourcing services.
North America:
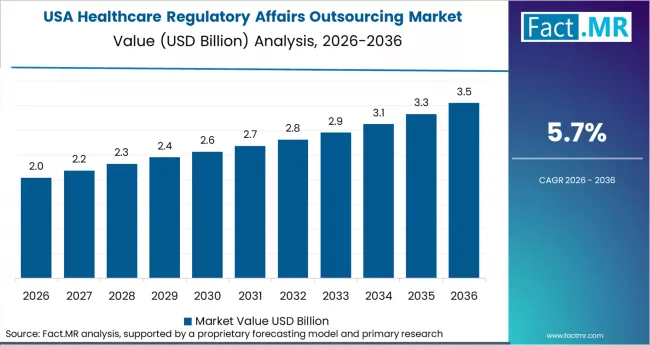
In North America, the USA remains the dominant market for healthcare regulatory affairs outsourcing, fueled by the rising complexity of regulatory processes in the healthcare and pharmaceutical industries.
- USA: Demand for healthcare regulatory affairs outsourcing in the U.S. is projected to rise at 5.7% CAGR through 2036. The USA’s advanced healthcare and pharmaceutical industries and the increasing need for regulatory expertise contribute to steady market growth.
Fact MR's analysis of the Healthcare Regulatory Affairs Outsourcing Market in East Asia, South Asia, Western Europe, North America, and China consists of country-wise assessments that include China, India, Germany, Japan, and the USA. Readers can find detailed trends, regulatory updates, and company-specific investments shaping the market’s growth in these countries.
How Are Key Players Competing in the Healthcare Regulatory Affairs Outsourcing Market?
In the healthcare regulatory affairs outsourcing market, IQVIA Holdings Inc. stands out with its extensive data analytics capabilities, offering regulatory affairs outsourcing alongside its deep insights into healthcare trends. Charles River Laboratories and Syneos Health are competitive with their integrated services, combining regulatory expertise with clinical trial management. Laboratory Corporation of America and Medpace leverage their strong clinical trial backgrounds to offer regulatory solutions tailored for pharmaceutical and biotech companies. ICON plc and PAREXEL International provide specialized regulatory and compliance services globally, capitalizing on their established reputations. Meanwhile, Thermo Fisher Scientific and WuXi AppTec emphasize end-to-end services, facilitating faster market entry through comprehensive regulatory support. Promedica International and Accell Clinical Research focus on niche markets, providing personalized regulatory solutions to enhance product development and compliance for emerging pharmaceutical companies.
Recent Industry Developments
- FDA QMSR Mandatory Compliance: The FDA’s Quality Management System Regulation (QMSR) became effective on February 2, 2026, officially incorporating ISO 13485:2016 and requiring manufacturers to open previously exempt internal quality audits to federal inspectors.
- EUDAMED Mandatory Registration Deadline: The European Commission confirmed that mandatory registration for actors and devices in the EUDAMED database will take effect on May 26, 2026, following the formal functionality notice published in late 2025.
- Parexel and Weave Bio AI Partnership: On September 30, 2025, Parexel announced a strategic partnership with Weave Bio to integrate AI-native automation into regulatory workflows, specifically to accelerate Investigational New Drug (IND) authoring.
Key Players of the Healthcare Regulatory Affairs Outsourcing Market
- IQVIA Holdings Inc.
- Accell Clinical Research, LLC
- Charles River Laboratories International, Inc.
- Syneos Health, Inc.
- Laboratory Corporation of America Holdings
- ICON plc
- Medpace Holdings, Inc.
- PAREXEL International Corporation
- Thermo Fisher Scientific Inc.
- Promedica International, Inc.
- WuXi AppTec Co., Ltd.
Report Scope
| Metric | Value |
|---|---|
| Quantitative Units | USD 4.5 billion (2026) to USD 6.6 billion (2036), at a CAGR of 6.7% |
| Market Definition | The healthcare regulatory affairs outsourcing market includes services related to regulatory affairs for the healthcare industry, such as consulting, legal representation, and clinical trial applications. |
| By Service | Regulatory Consulting, Legal Representation, Regulatory Writing and Publishing, Product Registration and Clinical Trial Applications, Others |
| By End-User | Pharmaceutical and Biotechnology Companies, Medical Device Companies, Food and Beverage Companies |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia, and 40+ countries |
| Key Companies Profiled | IQVIA Holdings Inc., Accell Clinical Research, LLC, Charles River Laboratories International, Inc., Syneos Health, Inc., Laboratory Corporation of America Holdings, ICON plc, Medpace Holdings, Inc., PAREXEL International Corporation, Thermo Fisher Scientific Inc., Promedica International, Inc., WuXi AppTec Co., Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with manufacturers and supported by trade data benchmarking and market research. |
Bibliographies
- U.S. Food and Drug Administration. (2026, February 2). Quality Management System Regulation (QMSR): Final rule aligning 21 CFR Part 820 with ISO 13485:2016. U.S. Food and Drug Administration.
- European Commission. (2025). Commission Implementing Regulation confirming the mandatory use of the European Database on Medical Devices (EUDAMED) from 26 May 2026. European Commission.
- World Health Organization. (2023). WHO global benchmarking tool for evaluation of national regulatory systems of medical products. World Health Organization.
- Regulatory Affairs Professionals Society. (2024). Global regulatory trends impacting pharmaceutical and medical device development. Regulatory Affairs Professionals Society.
- IQVIA Holdings Inc. (2024). Regulatory and compliance services for life sciences organizations. IQVIA Holdings Inc.
- Syneos Health, Inc. (2024). Regulatory and clinical development outsourcing trends in global healthcare. Syneos Health, Inc.
- Parexel International Corporation. (2025, September 30). Parexel announces AI partnership with Weave Bio to enhance regulatory workflow automation [Press release]. Parexel International Corporation.
This report addresses:
- Market intelligence enabling comprehensive assessment of leading countries and service segments across the Healthcare Regulatory Affairs Outsourcing Market globally.
- Market volume (sales units) estimates and 10-year revenue forecasts from 2026 to 2036, validated through manufacturer shipment data, channel partner surveys, and country-level demand modeling.
- Growth opportunity mapping across service classes (Regulatory Consulting, Legal Representation, Regulatory Writing and Publishing, Product Registration and Clinical Trial Applications, Others), end-user sectors (Pharmaceutical and Biotechnology Companies, Medical Device Companies, Food and Beverage Companies), and regions.
- Segment and regional revenue forecasts by service class, end-user sector, and geography across 40+ countries.
- Competition strategy analysis covering dealer network structure, recent developments, product portfolio, USPs, and market share analysis.
- Product and regulatory compliance tracking aligned with regulatory guidelines and clinical trial approval standards.
- Regulatory impact analysis addressing global healthcare compliance, market access regulations, and evolving standards for pharmaceutical and biotechnology products.
- Report delivery in PDF, Excel, PowerPoint, and interactive dashboard formats for executive, procurement, and operational planning use.
Healthcare Regulatory Affairs Outsourcing Market Key Segments
-
Service :
- Regulatory Consulting
- Legal Representation
- Regulatory Writing and Publishing
- Product Registration and Clinical Trial Applications
- Others
-
End-Use Industry :
- Pharmaceutical and Biotechnology Companies
- Medical Device Companies
- Food and Beverage Companies
-
Region :
- North America
- USA
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East and Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Rest of Middle East and Africa
- Other Regions
- Oceania
- Central Asia
- Other Markets
- North America
- Frequently Asked Questions -
How large is the demand for Healthcare Regulatory Affairs Outsourcing in the global market in 2026?
Demand for Healthcare Regulatory Affairs Outsourcing in the global market is estimated to be valued at USD 4.5 billion in 2026.
What will be the market size of Healthcare Regulatory Affairs Outsourcing in the global market by 2036?
The market size for Healthcare Regulatory Affairs Outsourcing is projected to reach USD 6.6 billion by 2036.
What is the expected demand growth for Healthcare Regulatory Affairs Outsourcing in the global market between 2026 and 2036?
Demand for Healthcare Regulatory Affairs Outsourcing in the global market is expected to grow at a CAGR of 6.7% between 2026 and 2036.
Which end-user is expected to dominate the market?
Pharmaceutical and Biotechnology Companies are expected to dominate the market, accounting for 55% of the market share in 2026, due to their growing need for regulatory expertise and outsourcing to navigate complex global regulatory requirements.
Which region is expected to show the highest growth rate for Healthcare Regulatory Affairs Outsourcing?
China is projected to show the highest regional CAGR at 8.8% during the forecast period, driven by the country's expanding pharmaceutical and biotechnology industries and increasing demand for regulatory affairs expertise.
How significant is the growth outlook for India in this market?
India is expected to grow at a CAGR of 8.4%, reflecting the increasing outsourcing of regulatory affairs by pharmaceutical and biotech companies in the country, leveraging cost advantages and a growing regulatory landscape.
What is the growth outlook for Germany in the Healthcare Regulatory Affairs Outsourcing market?
Germany is expected to grow at a CAGR of 6.2%, supported by the country's strong pharmaceutical and biotechnology sectors, with companies increasingly relying on outsourcing to navigate the European regulatory environment.
What is the growth forecast for Japan in the Healthcare Regulatory Affairs Outsourcing market?
Japan is expected to grow at a CAGR of 5.9%, driven by the country's advanced pharmaceutical industry and growing demand for regulatory support as companies expand their global presence.
What is the growth forecast for the United States in the Healthcare Regulatory Affairs Outsourcing market?
The United States is anticipated to grow at a CAGR of 5.7%, supported by the country’s established pharmaceutical and biotechnology industries, and an increasing reliance on outsourcing to comply with complex global regulations.
Which company is identified as a leading player in the Healthcare Regulatory Affairs Outsourcing market?
IQVIA Holdings Inc. is recognized as a leading player in this market, providing comprehensive regulatory affairs outsourcing services to pharmaceutical and biotechnology companies globally.
What is Healthcare Regulatory Affairs Outsourcing?
Healthcare Regulatory Affairs Outsourcing refers to the practice of pharmaceutical, biotechnology, and medical device companies outsourcing regulatory affairs tasks such as compliance management, product registration, and approval processes to third-party service providers with expertise in regulatory matters.
What does the Healthcare Regulatory Affairs Outsourcing market include in this report?
The market scope includes outsourcing services provided to pharmaceutical and biotechnology companies, particularly focusing on regulatory affairs management, product approvals, compliance, and related services across various regions.
How is the market forecast developed in this report?
The forecast is developed using historical data on pharmaceutical and biotechnology industry growth, trends in regulatory affairs outsourcing, and insights from key industry players involved in providing outsourcing services to healthcare companies.
What is meant by the Healthcare Regulatory Affairs Outsourcing market in this report?
The market refers to the global production, trade, and consumption of outsourcing services for regulatory affairs in the healthcare sector, particularly for pharmaceutical and biotechnology companies seeking to navigate complex and evolving regulatory environments.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service , 2026 to 2036
- Regulatory Consulting
- Legal Representation
- Regulatory Writing and Publishing
- Product Registration and Clinical Trial Applications
- Regulatory Consulting
- Y to o to Y Growth Trend Analysis By Service , 2021 to 2025
- Absolute $ Opportunity Analysis By Service , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical and Biotechnology Companies
- Medical Device Companies
- Food and Beverage Companies
- Pharmaceutical and Biotechnology Companies
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Service
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service
- By End Use
- Competition Analysis
- Competition Deep Dive
- IQVIA Holdings Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Accell Clinical Research, LLC
- Charles River Laboratories International, Inc.
- Syneos Health, Inc.
- Laboratory Corporation of America Holdings
- ICON plc
- Medpace Holdings, Inc.
- PAREXEL International Corporation
- Thermo Fisher Scientific Inc.
- Promedica International, Inc.
- WuXi AppTec Co., Ltd.
- IQVIA Holdings Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region,2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service,2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use,2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country,2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Service,2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End Use,2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country,2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Service,2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End Use,2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country,2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Service,2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End Use,2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country,2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Service,2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End Use,2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country,2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Service,2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End Use,2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country,2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Service,2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End Use,2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country,2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Service,2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End Use,2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Service,2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Service,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Service
- Figure 6: Global Market Value Share and BPS Analysis by End Use,2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by End Use,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region,2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Service,2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Service,2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Service
- Figure 23: North America Market Value Share and BPS Analysis by End Use,2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by End Use,2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by End Use
- Figure 26: Latin America Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Service,2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Service,2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Service
- Figure 30: Latin America Market Value Share and BPS Analysis by End Use,2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by End Use,2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by End Use
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Service,2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Service,2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Service
- Figure 37: Western Europe Market Value Share and BPS Analysis by End Use,2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by End Use,2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by End Use
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Service,2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Service,2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Service
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End Use,2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by End Use,2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 47: East Asia Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Service,2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Service,2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Service
- Figure 51: East Asia Market Value Share and BPS Analysis by End Use,2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by End Use,2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by End Use
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Service,2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Service,2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Service
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End Use,2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by End Use,2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country,2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Service,2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Service,2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Service
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End Use,2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by End Use,2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis