Healthcare Contract Manufacturing Market Size, Share, Growth and Forecast (2026 - 2036)
The Healthcare Contract Manufacturing Market is segmented by Service Type (Pharmaceutical Contract Manufacturing and Medical Device Manufacturing), Business Line (API Manufacturing, Finished Dosage Formulation, and Biologics & Advanced Therapies), End Use (Pharmaceutical & Biotech Companies and Medical Device OEMs) and Region. Forecast for 2026 to 2036.
Fact.MR indicates that the healthcare contract manufacturing market is at a structural inflection point. Outsourcing is no longer driven primarily by cost arbitrage. The focus is shifting toward capability-led partnerships. Pharma companies are prioritising advanced manufacturing capabilities. Demand is rising for biologics, cell, and gene therapy production. Technical expertise and regulatory readiness are becoming critical selection criteria.
Healthcare Contract Manufacturing Market Forecast and Outlook By Fact.MR
In 2025, the healthcare contract manufacturing market was valued at USD 240 billion. Based on Fact.MR analysis, demand for healthcare contract manufacturing services is estimated to grow to USD 262 billion in 2026 and USD 646 billion by 2036. FACT.MR projects a CAGR of 9.4% during the forecast period.
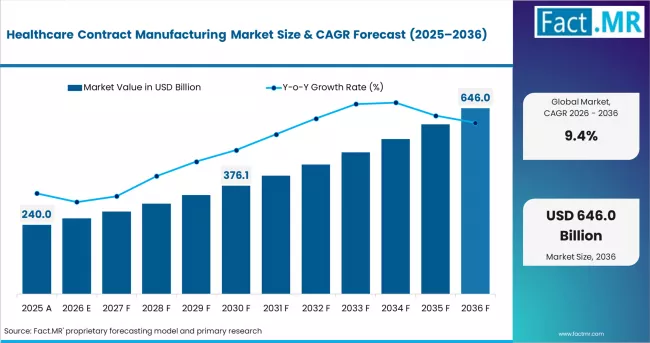
| Metric | Value |
|---|---|
| Estimated Value in 2026 | USD 262 billion |
| Forecast Value in 2036 | USD 646 billion |
| Forecast CAGR (2026 to 2036) | 9.4% |
Summary of Healthcare Contract Manufacturing Market
- Market Definition
- Market covers outsourced pharmaceutical and medical device production. It includes API manufacturing, biologics, finished dosage, and device assembly for global pharma and biotech clients.
- Demand Drivers
- Biologics Outsourcing Growth: Global pharma sponsors are outsourcing mammalian cell culture and mRNA manufacturing. Internal capacity is insufficient for accelerating pipeline volumes.
- API Supply Chain Restructuring: Regulatory pressure to reduce Asia-Pacific API dependency is driving near-shore and domestic outsourcing. US and European CDMOs are benefiting from reshoring mandates.
- Advanced Therapy Expansion: Cell and gene therapy pipelines are generating outsourcing demand that proprietary manufacturers cannot internally absorb. Specialist CDMOs are capturing programme contracts early.
- Key Segments Analyzed
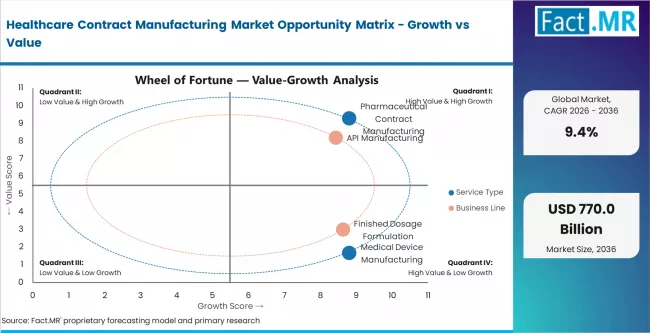
- By Service Type: Pharmaceutical contract manufacturing leads with 75% share in 2026. It dominates due to complex drug pipelines and outsourcing by large pharma.
- By Business Line: API manufacturing leads with 65% share in 2026. Biologics and advanced therapies represent the fastest-growing sub-segment.
- By End Use: Pharmaceutical and biotech companies account for 72% share in 2026. They drive volume through large-molecule and specialty drug outsourcing.
- Analyst Opinion at FACT.MR
- Shambhu Nath Jha, Principal Consultant at Fact.MR, opines, CXOs will gain access to granular, segment-level insights across the market. Detailed data on biologics outsourcing growth highlights where demand is accelerating. It shows how capacity is shifting toward complex, high-value manufacturing.
- Strategic Implications
- Invest in biologics fill-finish and cell and gene therapy manufacturing infrastructure. This captures the fastest-growing outsourcing segment across US and European sponsor networks.
- Build dual-shore API manufacturing capacity across India and a US or EU site. This addresses regulatory mandates and sponsor risk diversification requirements simultaneously.
- Establish long-term supply agreements with mid-sized biotech sponsors in early clinical stages. This secures commercial manufacturing rights ahead of competitive CDMO consolidation.
- Methodology
- Market sizing combines CDMO revenues, sponsor outsourcing rates by drug class, and contract value benchmarks. Validated using FDA facility registrations and EMA inspection data.
- Incorporates WHO and national health authority manufacturing capacity data. Includes regulatory approval databases for biologics and advanced therapies.
- Forecasts account for pipeline conversion rates and patent expiry-driven outsourcing shifts. Supported by expert interviews with CDMO executives and pharma procurement leaders.
Between 2026 and 2036, the market is projected to generate an absolute opportunity of USD 384 billion. Growth is driven by rising outsourcing of biologics manufacturing, accelerating complex molecule pipelines, and pharma companies divesting internal production assets. Cost pressure and speed-to-market requirements are sustaining CDMO demand across both established and emerging geographies.
Outsourcing decisions are increasingly contract-structured. Large pharmaceutical sponsors are locking in multi-year agreements with CDMOs offering end-to-end capabilities. Single-source manufacturing partnerships for high-value biologics and cell and gene therapy programmes are becoming common, particularly across North American and European sponsor networks.
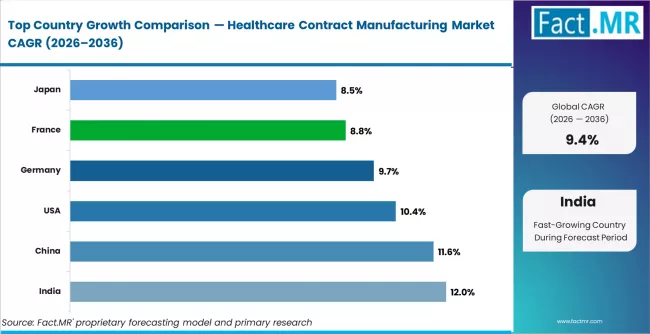
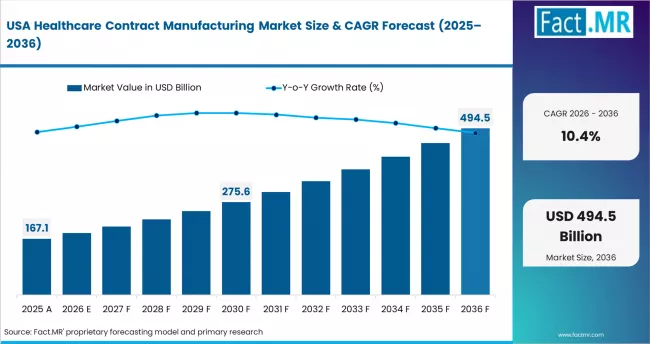
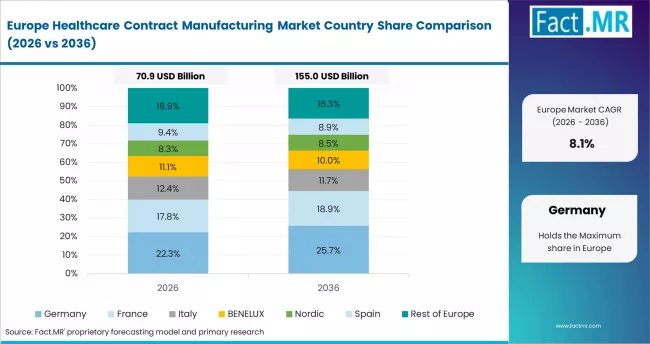
Regionally, India is projected to record the fastest CAGR of 12.0% through 2036, driven by API manufacturing scale-up and biologics investment under government production-linked incentive schemes. China follows at 11.6%, led by capacity expansion among large CDMOs and biopharma outsourcing from global sponsors. The United States grows at 10.4%, supported by reshoring of API supply chains and advanced therapy manufacturing demand. Germany records 9.7%, anchored by precision formulation and biologics fill-finish capabilities. France grows at 8.8%, driven by public health manufacturing mandates and CDMO investment. Japan follows at 8.5%, shaped by domestic pharma outsourcing and regulatory modernisation.
Segmental Analysis
Healthcare Contract Manufacturing Market Analysis by Service Type
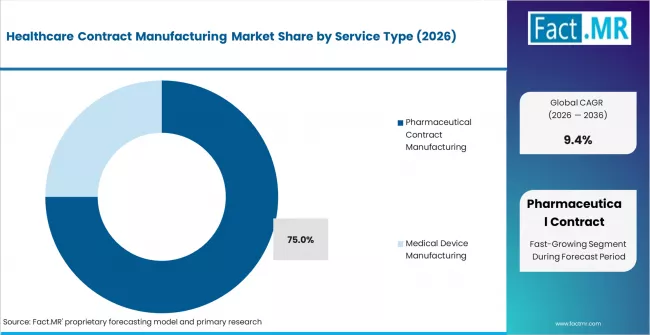
Based on FACT.MR analysis, pharmaceutical contract manufacturing holds 75% share in 2026. It leads because large-molecule and specialty drug pipelines require specialist production assets. Outsourcing rates among top-20 pharma companies have increased steadily. Biologics and advanced therapies are redefining service scope. Medical device manufacturing is smaller but growing faster in absolute rate terms.
- Lonza Biologics Capacity: Lonza Group announced a CHF 800 million investment in its Visp biologics campus in Q2 2024. The expansion adds mammalian cell culture and mRNA manufacturing capacity. This targets large pharma and biotech sponsors with commercial-stage biologics programmes [4].
- Samsung Biologics Expansion: Samsung Biologics commenced operations at its Plant 4 in Songdo, South Korea in 2023 and broke ground on Plant 5 in 2024. Combined capacity exceeds 784,000 litres of bioreactor volume, targeting global biosimilar and innovator biologic sponsors [5].
- Medical Device Outsourcing Trend: Jabil Inc. reported 14% growth in its healthcare segment revenue in FY2024, driven by outsourced medical device assembly and combination product manufacturing. Growth is concentrated in minimally invasive devices and connected health platforms [6].
Healthcare Contract Manufacturing Market Analysis by Business Line
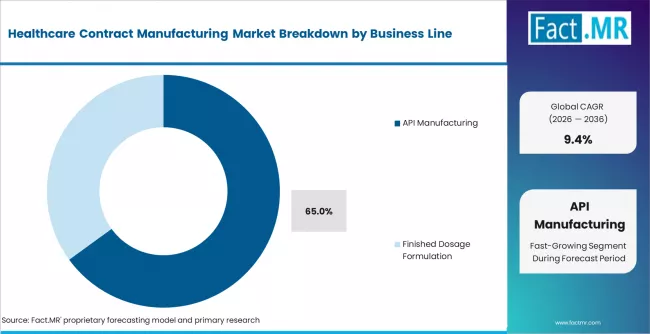
Based on FACT.MR analysis, API manufacturing leads with 65% share in 2026. It dominates because small-molecule APIs remain the largest volume category globally. Biologics and advanced therapies are the fastest-growing sub-segment. Finished dosage formulation is a stable mid-tier segment. Reshoring and near-shoring dynamics are restructuring API geography.
- WuXi Biologics API Expansion: WuXi Biologics expanded its biologics API manufacturing at its Wuxi facility in Q3 2024. The site added two 15,000-litre bioreactor trains to support global clinical and commercial biologics contracts. Programme capacity is linked to 580+ active client projects [7].
- Thermo Fisher mRNA Platform: Thermo Fisher Scientific launched its Genoptix mRNA manufacturing platform in 2024. It targets clinical-stage biotech sponsors. The platform integrates lipid nanoparticle formulation and fill-finish. Commercial agreements with three biotech companies were signed in H1 2024 [8].
- Finished Dosage Formulation Trend: Recipharm AB reported 8% growth in finished dosage formulation revenue in 2024. Growth is driven by complex formulation projects including inhalation, parenteral, and transdermal dosage forms. Partnerships with mid-sized European pharma sponsors underpinned volume growth [9].
Healthcare Contract Manufacturing Market Analysis by End Use
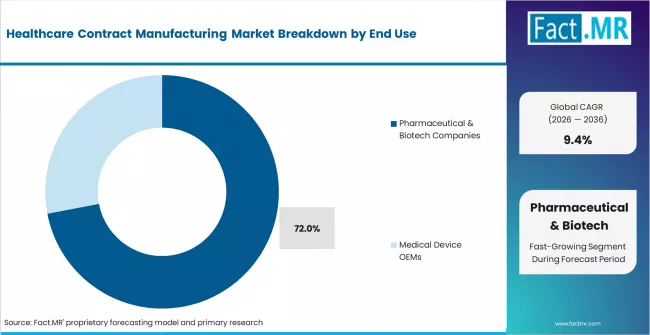
Based on FACT.MR analysis, pharmaceutical and biotech companies account for 72% share in 2026. They drive the market through biologics programmes, specialty molecule pipelines, and biosimilar development projects. Medical device OEMs represent a smaller but faster-growing end-use segment. Outsourcing decisions by pharma clients are programme-level, making contract values large and switching costs high.
- Boehringer Ingelheim CDMO Expansion: Boehringer Ingelheim Biopharmaceuticals GmbH expanded its CDMO services for mammalian-derived biologics in 2024. The company added fill-finish capacity at its Vienna site and extended its bioreactor capacity in Ingelheim. Agreements with three oncology biotech clients were disclosed in Q3 2024.
- Biotech Outsourcing Growth: Catalent Inc. reported in its FY2024 results that biotech clients accounted for over 65% of new biologics manufacturing contracts. Cell and gene therapy programme wins increased 18% year-on-year. FDA approval of client products drove commercial fill-finish volume growth.
- Medical Device OEM Trend: Jabil Healthcare expanded its medical device contract manufacturing footprint in Malaysia and Mexico in 2024. The expansion targets US and European OEM clients seeking lower-cost assembly combined with regulatory-compliant quality systems. ISO 13485 certified capacity was added at both sites.
Drivers, Restraints, and Opportunities
FACT.MR analysis indicates that the healthcare contract manufacturing market is in an expansionary cycle driven by structural outsourcing rather than cyclical demand. The market has grown consistently as pharmaceutical pipelines have shifted toward biologics and specialty molecules that require specialised manufacturing infrastructure.
API manufacturing remains the largest business line by revenue, but biologics and advanced therapies are the primary growth engine.
- FDA DSCSA Implementation: The US Drug Supply Chain Security Act, fully operational since November 2023, is increasing compliance costs for non-integrated manufacturers. CDMOs with serialisation-ready infrastructure are gaining preferred supplier status from major pharma sponsors in 2024 and 2025 [1].
- India PLI Scheme for Pharmaceuticals: India's Production Linked Incentive scheme, with USD 2 billion allocated for API and formulation manufacturing, is scaling CDMO capacity. Divi's Laboratories and Aurobindo Pharma expanded API production in 2024 under this programme, reducing unit costs and improving global competitiveness [2].
- EU Pharmaceutical Strategy Reshoring: The European Medicines Agency's Critical Medicine Alliance identified 200+ shortage-prone APIs in 2024. This is driving reshoring contracts to European CDMOs. Lonza expanded its Visp site in Switzerland, and Recipharm added capacity in Sweden to meet EMA resilience requirements [3].
Regional Analysis
The healthcare contract manufacturing market is assessed across North America, Europe, Asia Pacific, Latin America, and Middle East and Africa, covering 40+ countries with distinct CDMO ecosystem maturity, regulatory frameworks, and outsourcing demand profiles. The full report offers market attractiveness analysis by region and country.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 12.0% (fastest) |
| China | 11.6% |
| United States | 10.4% |
| Germany | 9.7% |
| France | 8.8% |
| Japan | 8.5% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
Asia Pacific Healthcare Contract Manufacturing Market Analysis
Asia Pacific is the production volume backbone of global healthcare contract manufacturing. India and China together account for the majority of global API supply. Both countries are upgrading to biologics and advanced formulation manufacturing. South Korea's Samsung Biologics has positioned the country as a premium biologics CDMO hub for global sponsors.
- China: Demand for healthcare contract manufacturing in China is projected to rise at 11.6% CAGR through 2036. China's CDMO sector is expanding rapidly despite geopolitical headwinds. WuXi Biologics reported 580+ active client projects in its 2024 annual results, with capacity expansion at its Wuxi and Hangzhou sites. Samsung Biologics' Plant 5 construction began in 2024, while WuXi AppTec expanded small-molecule API capacity by 20% in Chengdu.
- India: India is projected to rise at 12.0% CAGR through 2036, the fastest among covered countries. India's PLI scheme for pharmaceuticals, with a USD 2 billion allocation, is directing investment into domestic API and formulation manufacturing. Divi's Laboratories expanded its API complex in Andhra Pradesh in 2024, targeting regulated market exports. Aurobindo Pharma commissioned a new biologics unit in FY2024.
- Japan: Japan is projected to rise at 8.5% CAGR through 2036. Japan's CDMO market is being stimulated by domestic pharma outsourcing and regulatory modernisation under the Pharmaceuticals and Medical Devices Act (PMDA) revisions of 2024. Fujifilm Diosynth Biotechnologies expanded its biologics manufacturing in Japan in 2024, focusing on mammalian cell culture.
FACT.MR covers China, India, Japan, South Korea, ASEAN, and Australia and New Zealand. It includes PLI scheme data, biosimilar manufacturing trends, and sponsor outsourcing profiles.
North America Healthcare Contract Manufacturing Market Analysis
North America is the global revenue epicentre for healthcare contract manufacturing. FDA-regulated biologics and advanced therapy programmes generate the highest contract values. US-based CDMOs hold technology and compliance leadership. Catalent Inc. and Thermo Fisher Scientific anchor the competitive landscape through broad service portfolios and multi-site manufacturing networks.
- United States: Healthcare contract manufacturing in the United States is projected to rise at 10.4% CAGR through 2036. The US CDMO market is being reshaped by two concurrent forces: advanced therapy outsourcing growth and API supply chain reshoring. The FDA approved 55 novel drugs in 2023 and 50 in 2024, each generating commercial manufacturing outsourcing demand. Catalent expanded its biologics fill-finish capacity in Bloomington, Indiana in Q1 2024.
FACT.MR's analysis of the healthcare contract manufacturing market in North America consists of country-wise assessment that includes the United States, Canada, and Mexico. Readers can find API reshoring trends, biologics outsourcing contract data, and advanced therapy manufacturing investment profiles.
Europe Healthcare Contract Manufacturing Market Analysis
Europe is a compliance-driven CDMO market shaped by EMA approval standards and the EU Pharmaceutical Strategy. Reshoring of critical APIs and biologics manufacturing is a policy priority. Germany and France host large integrated CDMOs. Lonza and Recipharm maintain dominant positions through multi-site networks and broad technology platforms.
- Germany: Growth for healthcare contract manufacturing in Germany is projected to rise at 9.7% CAGR through 2036. Germany hosts several of Europe's highest-capacity biologics CDMOs. Boehringer Ingelheim Biopharmaceuticals expanded its Vienna and Ingelheim biologics sites in 2024, adding fill-finish and bioreactor capacity for oncology and immunology programmes. The German government's IPCEI Health initiative supported biomanufacturing investment in 2024 and 2025.
- France: Demand for healthcare contract manufacturing in France is projected to rise at 8.8% CAGR through 2036. France's public health manufacturing mandate, reinforced by the 2024 National Medicines Resilience Strategy, is directing state contracts to domestic CDMOs. Recipharm expanded its French formulation and sterile manufacturing sites in 2024.
FACT.MR covers Germany, the UK, France, Italy, Spain, Nordic countries, and BENELUX. It includes EMA approval data, reshoring contract trends, and regulatory investment analysis.
Competitive Aligners for Market Players
The healthcare contract manufacturing market is slightly concentrated at the top tier. The five largest CDMOs, including Lonza, Catalent, Samsung Biologics, WuXi Biologics, and Thermo Fisher Scientific, collectively account for approximately 35% of global revenues. The remaining share is fragmented across hundreds of regional and specialist operators. The primary competitive variable is manufacturing technology breadth, specifically the ability to serve biologics, advanced therapies, and small-molecule programmes from a single CDMO partner.
Structural advantages are held by CDMOs with multi-site networks, regulatory approval portfolios across FDA, EMA, and PMDA jurisdictions, and vertically integrated supply chains. Lonza's captive raw material capabilities and Samsung Biologics' scale economics provide cost-per-litre advantages that smaller operators cannot replicate without significant capital. Long-term master service agreements with top-20 pharma companies further entrench incumbents by reducing re-tendering frequency.
Buyer concentration is high. The top 10 pharma and biotech sponsors account for a disproportionate share of CDMO revenue. This creates leverage in pricing negotiations and forces CDMOs to offer volume-linked discounts on multi-programme contracts. However, advanced therapy and rare disease programmes are so technically specialised that pricing power shifts toward CDMOs with unique capabilities, improving margins in those niches.
Key Players
- Lonza Group
- Catalent Inc.
- Samsung Biologics
- WuXi Biologics
- Thermo Fisher Scientific
- Recipharm AB
- Boehringer Ingelheim Biopharmaceuticals GmbH
- Jabil Inc.
Bibliography
- [1] U.S. Food and Drug Administration. (2024). Novel Drug Approvals for 2024 and Drug Supply Chain Security Act Implementation Progress Report: Serialisation Compliance and Track-and-Trace System Deployment Across Licensed Contract Manufacturing Facilities. fda.gov/drugs/development-approval-process-drugs
- [2] Ministry of Chemicals and Fertilizers, Government of India. (2024). Production Linked Incentive Scheme for Pharmaceuticals: Annual Implementation Report 2023-24 Covering API Manufacturing Expansion, Eligible Investment Disbursements, and Export-Oriented CDMO Capacity under Scheme Guidelines. pharmaceuticals.gov.in
- [3] European Medicines Agency. (2024). Critical Medicine Alliance Report 2024: API Shortage-Prone Medicines List, Reshoring Policy Recommendations, and Manufacturing Resilience Investment Frameworks for EU Member State Health Authorities. ema.europa.eu/en/human-regulatory-overview/medicines-shortages
- [4] Lonza Group AG. (Q2 2024). Investor Relations Press Release: CHF 800 Million Visp Campus Biologics Expansion Announcement Covering Mammalian Cell Culture Capacity Addition and mRNA Manufacturing Infrastructure for Pharmaceutical Sponsor Partnerships. lonza.com/investors
- [5] Samsung Biologics Co. Ltd. (2024). Annual Report 2024 and Plant 5 Groundbreaking Press Release: Biologics Contract Manufacturing Capacity Expansion in Songdo, South Korea Targeting Global Biosimilar and Originator Biologic Sponsor Programmes. samsungbiologics.com/investors
- [6] Jabil Inc. (FY2024). Annual Report and Healthcare Segment Results: Medical Device Contract Manufacturing Revenue Growth, ISO 13485 Certified Facility Expansion in Malaysia and Mexico, and Connected Health Device Assembly Programme Wins. jabil.com/investors
- [7] WuXi Biologics Cayman Inc. (2024). Annual Results 2024 and Capacity Expansion Update: Active Client Programme Count, Wuxi and Hangzhou Bioreactor Addition, and Biologics API Manufacturing Scale-Up Details for Global Pharmaceutical Clients. wuxibiologics.com/investors
- [8] Thermo Fisher Scientific Inc. (2024). Press Release: Genoptix mRNA Manufacturing Platform Launch and Commercial Manufacturing Agreement Announcements with Clinical-Stage Biotech Partners in H1 2024. thermofisher.com/global/en/home/news-gallery
- [9] Recipharm AB. (2024). Annual Report 2024: Finished Dosage Formulation Revenue Growth, French and Swedish Site Expansion Investments, and European Pharma Client Contract Portfolio Update Including EUROAPI Collaboration Details. recipharm.com/investors
This Report Addresses
- Strategic intelligence on healthcare contract manufacturing demand across pharmaceutical outsourcing, biologics manufacturing, API production, and medical device assembly globally.
- Market forecast from USD 262 billion in 2026 to USD 646 billion by 2036, at a CAGR of 9.4%.
- Opportunity mapping across India PLI-driven API and biologics expansion, China CDMO capacity growth, US advanced therapy outsourcing, EU reshoring mandates, and Japan PMDA-aligned programme development.
- Segment analysis by service type (pharmaceutical contract manufacturing, medical device manufacturing), business line (API, finished dosage, biologics), and end use (pharma and biotech companies, device OEMs) across global and regional markets.
- Regional outlook covering Asia Pacific API and biologics manufacturing scale-up, North America advanced therapy programme outsourcing, European reshoring-driven CDMO investment, and emerging market capacity expansion.
- Competitive landscape of Lonza Group, Catalent Inc., Samsung Biologics, WuXi Biologics, Thermo Fisher Scientific, Recipharm AB, Boehringer Ingelheim, and Jabil Inc., covering manufacturing technology breadth, regulatory approval portfolios, and client programme depth.
- Regulatory analysis covering US DSCSA and BIOSECURE Act implications, India PLI pharmaceutical scheme, EU Pharmaceutical Strategy reshoring policy, EMA critical medicine shortage response, and Japan PMDA manufacturing modernisation.
- Report delivered in PDF, Excel, and presentation formats supported by validated CDMO revenue data, FDA and EMA regulatory benchmarks, and industry expert inputs.
Healthcare Contract Manufacturing Market Definition
The healthcare contract manufacturing market covers outsourced production services for pharmaceutical and medical device companies. It includes API synthesis, finished dosage formulation, biologics manufacturing, and medical device assembly.
Healthcare Contract Manufacturing Market Inclusions
Covers global and regional forecasts from 2026 to 2036. Segmentation includes service type, business line, and end use. Includes pharmaceutical contract manufacturing and medical device manufacturing. Business lines span API manufacturing, finished dosage formulation, and biologics and advanced therapies.
Healthcare Contract Manufacturing Market Exclusions
Excludes in-house manufacturing by vertically integrated pharmaceutical companies. Excludes raw material and excipient supply. Omits downstream retail pharmaceutical distribution. Excludes clinical research and regulatory consulting services unless bundled with manufacturing contracts.
Healthcare Contract Manufacturing Market Research Methodology
- Primary Research:Includes interviews with CDMO executives, procurement heads at pharma sponsors, and biologics manufacturing specialists. Covers quality and regulatory affairs managers across contract sites globally.
- Desk Research:Includes analysis of company filings from Lonza Group, Catalent Inc., and Samsung Biologics. Incorporates FDA, EMA, and WHO manufacturing capacity and approval data.
- Market-Sizing and Forecasting:Uses a hybrid approach. Combines CDMO revenue data, outsourcing rate estimates by drug class, and average contract values. Covers pharmaceutical and medical device manufacturing separately.
- Data Validation and Update Cycle:Validated using sponsor procurement disclosures and CDMO annual reports. Includes FDA facility registration data and EMA manufacturing inspection records. Updated through expert interviews.
Scope of Report
| Attribute | Details |
|---|---|
| Quantitative Units | USD 262 billion (2026) to USD 646 billion (2036), at a CAGR of 9.4% |
| Market Definition | Outsourced pharmaceutical and medical device production services, including API manufacturing, finished dosage formulation, biologics and advanced therapy manufacturing, and medical device assembly for global pharma, biotech, and device OEM clients. |
| Service Type Segmentation | Pharmaceutical Contract Manufacturing, Medical Device Manufacturing |
| Business Line Segmentation | API Manufacturing, Finished Dosage Formulation, Biologics & Advanced Therapies |
| End Use Segmentation | Pharmaceutical & Biotech Companies, Medical Device OEMs |
| Application Coverage | Biologics outsourcing for mammalian cell culture and mRNA manufacturing, API supply chain restructuring for small-molecule drugs, finished dosage formulation for oral solids and injectables, and medical device contract assembly for combination and connected health products. |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Canada, Mexico, Germany, France, UK, Italy, Spain, Nordic, BENELUX, China, India, Japan, South Korea, ASEAN, Australia and New Zealand, Brazil, Argentina, Saudi Arabia, GCC, Turkey, South Africa, Rest of MEA |
| Key Companies Profiled | Lonza Group, Catalent Inc., Samsung Biologics, WuXi Biologics, Thermo Fisher Scientific, Recipharm AB, Boehringer Ingelheim Biopharmaceuticals GmbH, Jabil Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using CDMO revenue data, sponsor outsourcing rates by drug class, average contract values, FDA facility registration data, EMA inspection records, and primary interviews with CDMO executives and pharma procurement specialists. |
Healthcare Contract Manufacturing Market by Segments
-
By Service Type:
- Pharmaceutical Contract Manufacturing
- Medical Device Manufacturing
-
By Business Line:
- API Manufacturing
- Finished Dosage Formulation
- Biologics & Advanced Therapies
-
By End Use:
- Pharmaceutical & Biotech Companies
- Medical Device OEMs
-
By Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Western Europe
- Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia and New Zealand
- Latin America
- Brazil
- Argentina
- Mexico
- Rest of Latin America
- Middle East and Africa
- Saudi Arabia
- GCC Countries
- Turkey
- South Africa
- Rest of MEA
- North America
- Frequently Asked Questions -
How large is the global healthcare contract manufacturing market in 2025?
The global healthcare contract manufacturing market was valued at USD 240 billion in 2025.
What will the market size be in 2026?
Based on Fact.MR analysis, demand for healthcare contract manufacturing services is estimated to grow to USD 262 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 646 billion by 2036, generating USD 384 billion in absolute dollar opportunity over the forecast period.
What is the expected CAGR from 2026 to 2036?
FACT.MR projects a CAGR of 9.4% for the global healthcare contract manufacturing market during the 2026 to 2036 forecast period.
Which service type is poised to lead the market?
Pharmaceutical contract manufacturing leads with approximately 75% share in 2026, driven by large-molecule biologics pipelines, specialty drug outsourcing, and the cost advantages of CDMO-based production versus in-house manufacturing.
Which business line shows the fastest growth?
Biologics and advanced therapies represent the fastest-growing business line, as cell and gene therapy programmes, mRNA platforms, and biosimilar manufacturing require specialised CDMO infrastructure not available in-house at most sponsors.
Which country shows the fastest growth in the market?
India leads at 12.0% CAGR through 2036, driven by PLI scheme investments, expanding API manufacturing scale, and growing biologics outsourcing demand from US and European sponsors.
What are the primary services covered?
Healthcare contract manufacturing covers API synthesis, finished dosage formulation including oral and injectable forms, biologics manufacturing including cell culture and mRNA, advanced therapy manufacturing including cell and gene therapies, and medical device contract assembly.
What drives demand in this market?
Demand is driven by pharma companies outsourcing biologics manufacturing, API supply chain reshoring mandates in the US and Europe, advanced therapy programme growth, cost-reduction outsourcing of small-molecule formulation, and regulatory compliance requirements that favour experienced CDMO partners.
Who are the key players in the market?
Major players include Lonza Group, Catalent Inc., Samsung Biologics, WuXi Biologics, Thermo Fisher Scientific, Recipharm AB, Boehringer Ingelheim Biopharmaceuticals GmbH, and Jabil Inc.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Component
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Component , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Component , 2026 to 2036
- Software
- Services
- Software
- Y to o to Y Growth Trend Analysis By Component , 2021 to 2025
- Absolute $ Opportunity Analysis By Component , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- Cloud-Based
- On-Premise
- Cloud-Based
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Component
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Component
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Component
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Component
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Component
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Component
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Component
- By Deployment
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Component
- By Deployment
- Competition Analysis
- Competition Deep Dive
- Autodesk Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Dassault Systèmes SE
- Siemens AG
- PTC Inc.
- Altair Engineering Inc.
- nTopology Inc.
- ANSYS Inc.
- Autodesk Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Component,2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Component,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Component
- Figure 6: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Deployment
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Component,2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Component,2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Component
- Figure 23: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Deployment
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Component,2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Component,2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Component
- Figure 30: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Deployment
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Component,2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Component,2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Component
- Figure 37: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Deployment
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Component,2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Component,2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Component
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Component,2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Component,2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Component
- Figure 51: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Deployment
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Component,2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Component,2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Component
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Component,2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Component,2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Component
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis