Peptide Therapeutics CDMO Market Size, Share, Growth and Forecast (2026 - 2036)
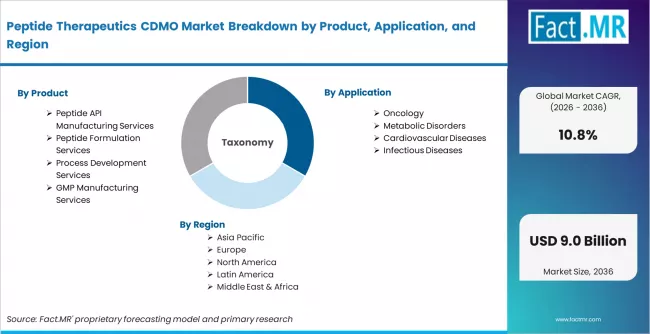
Peptide Therapeutics CDMO Market is segmented by Product (Peptide API Manufacturing Services, Peptide Formulation Services, Process Development Services, GMP Manufacturing Services), Application (Oncology, Metabolic Disorders, Cardiovascular Diseases, Infectious Diseases), End Use (Pharmaceutical & Biotechnology Companies, Contract Development & Manufacturing Organizations (CDMOs), Academic & Research Institutes, Contract Research Organizations (CROs)), and Region, with forecasts covering the period from 2026 to 2036.
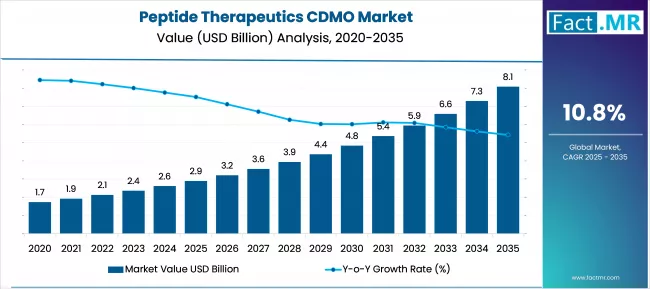
According to Fact.MR estimates, the global peptide therapeutics cdmo market market was valued at USD 2.9 billion in 2025. The market is projected to reach USD 3.2 billion in 2026 and is expected to grow to USD 9.0 billion by 2036, expanding at a CAGR of 10.8%. Peptide API Manufacturing Services is anticipated to account for 42.0% of the product segment in 2026, while Oncology is expected to remain the leading application with around 34.0% share.
Peptide Therapeutics CDMO Market Analysis and Forecast by Fact.MR
According to Fact.MR estimates, the global peptide therapeutics cdmo market market was valued at USD 2.9 billion in 2025. The market is projected to reach USD 3.2 billion in 2026 and is expected to grow to USD 9.0 billion by 2036, expanding at a CAGR of 10.8%. Peptide API Manufacturing Services is anticipated to account for 42.0% of the product segment in 2026, while Oncology is expected to remain the leading application with around 34.0% share.
Summary of Peptide Therapeutics CDMO Market
- Market Snapshot
- The Peptide Therapeutics CDMO market was valued at USD 2.9 billion in 2025.
- By 2036, the Peptide Therapeutics CDMO market is expected to be worth USD 8.96 billion.
- From 2026 to 2036, the market is projected to expand at a CAGR of 10.8%.
- The market is projected to create an incremental opportunity of USD 5.75 billion between 2026 and 2036.
- In 2026, Peptide API Manufacturing Services are expected to account for 42% of the service segment, driven by surging demand for GLP-1 class peptides, increased complexity of long-chain peptides, and sponsor preference for outsourced solid-phase peptide synthesis capacity.
- India (15.8%) and China (14.2%) are two of the fastest-growing markets in the world.
- Demand and Growth Drivers
- Demand is shaped by the unprecedented commercial success of GLP-1 peptide therapeutics, led by semaglutide, tirzepatide, and the next wave including retatrutide, which has created structural capacity shortages across global solid-phase peptide synthesis infrastructure.
- Growth reflects sponsor preference for outsourced peptide manufacturing because SPPS capacity is capital-intensive, requires specialized reactors and purification infrastructure, and benefits from the scale economics that only dedicated CDMOs can achieve.
- Adoption is increasing due to the rising complexity of therapeutic peptides, including longer chain lengths, non-standard amino acids, lipidation and PEGylation chemistry, and complex conjugation steps that favor experienced CDMO partners over in-house manufacturing.
- Product and Segment View
- Peptide API Manufacturing Services lead the service segment at 42% share in 2026, anchored by commercial-scale solid-phase peptide synthesis capacity serving approved and late-stage GLP-1 and metabolic disease peptides.
- Pharmaceutical Companies represent the majority of end-user demand in 2026, reflecting concentrated procurement by large pharma sponsors with approved peptide products requiring high-volume commercial supply, along with biotech sponsors running clinical-stage peptide programs.
- Solid-phase peptide synthesis dominates the technology segment in 2026, with liquid-phase and hybrid synthesis platforms used selectively for specific peptide classes and scale requirements.
- Geography and Competitive Outlook
- Asia Pacific leads growth, with India at a 15.8% CAGR supported by expanding CDMO capacity from Piramal Pharma Solutions, Auro Peptides, and domestic capacity additions, and China at 14.2% anchored by WuXi AppTec and expanding domestic peptide manufacturing.
- Europe contributes strongly through established leaders, with Switzerland at a 10.1% CAGR anchored by Bachem and Lonza, and Germany (10.8%) supported by CordenPharma, Wacker Chemie, and Fresenius Kabi.
- North America grows at a slower pace, with the United States at a 9.6% CAGR, reflecting mature sponsor demand and selective CDMO capacity expansion. Leading players include Bachem, Lonza Group, Polypeptide Group, AmbioPharm, CordenPharma, CPC Scientific, WuXi AppTec, Fresenius Kabi, Piramal Pharma Solutions, and Almac.
- Analyst Opinion
- Peptide therapeutics CDMO has moved from a steady specialty manufacturing niche to one of the most commercially attractive and capacity-constrained segments in biopharma contract manufacturing. The demand surge is driven almost entirely by GLP-1 peptides, where semaglutide, tirzepatide, and the next-wave candidates have created order volumes that substantially exceed historical peptide manufacturing capacity. CDMOs with established SPPS infrastructure are booking multi-year commercial supply contracts at premium economics, and new capacity additions are being absorbed before they come online. The strategic question for sponsors is whether to lock in long-term capacity commitments now or pursue partial in-house manufacturing to reduce supply risk. The strategic question for CDMOs is whether to compete on volume capacity, on complex peptide chemistry capability, or on integrated sterile fill-finish services. Firms combining SPPS scale with complex chemistry depth and integrated fill-finish will set the pricing and service benchmarks over the forecast period.
Why is the Peptide Therapeutics CDMO Market Growing?
- GLP-1 peptide therapeutics have created a structural demand shock for peptide CDMO capacity, with semaglutide, tirzepatide, and retatrutide driving order volumes that substantially exceed pre-2022 global SPPS infrastructure.
- Sponsor outsourcing preferences favor specialized peptide CDMOs because in-house SPPS capacity requires specialized reactors, purification trains, amino acid supply relationships, and dedicated cGMP regulatory infrastructure that most sponsors cannot justify for single-program programs.
- Rising peptide complexity, including longer chains, non-standard amino acids, lipidation and PEGylation chemistry, and complex conjugation steps, is increasing per-kilogram service value and driving sponsor preference for experienced CDMO partners.
GLP-1 demand is the primary demand engine. Semaglutide and tirzepatide sales growth has been unprecedented in biopharma, and both molecules require high-volume peptide API at commercial scale. Retatrutide and other next-wave GLP-1 or GIP-GLP-1-glucagon triagonist candidates in clinical development represent additional demand waves that will continue scaling across the forecast period. Each approved or late-stage GLP-1 program generates multi-year commercial supply contracts at premium economics, and global SPPS capacity has been insufficient to meet demand even after substantial capacity expansion announcements.
Outsourcing economics are the second driver. Building in-house peptide manufacturing requires a multi-hundred-million-dollar investment in specialized SPPS reactors, HPLC purification trains, amino acid and resin supply relationships, sterile fill-finish infrastructure, and cGMP regulatory expertise. For most biopharma sponsors, particularly those with one or two peptide programs in their pipeline, that investment does not generate acceptable returns. Specialized peptide CDMOs spread that infrastructure across multiple sponsor programs, which makes the unit economics work and creates structural preference for outsourced peptide supply.
Chemistry complexity is the third driver. Therapeutic peptides are getting longer, with 30-plus amino acid backbones, complex lipidation patterns, PEG conjugations, and cyclization steps that require experienced manufacturing expertise. Each step of chemistry complexity increases the per-kilogram service value and raises the barrier to in-house manufacturing, which strengthens the CDMO value proposition over the forecast period.
Segment-wise Analysis of Peptide Therapeutics CDMO Market
- Peptide API Manufacturing Services lead the service segment at 42% share in 2026, driven by commercial-scale SPPS capacity serving approved and late-stage GLP-1 and metabolic disease peptides.
- GLP-1 and metabolic disease peptides dominate the therapeutic class segment in 2026, reflecting the unprecedented commercial success of semaglutide and tirzepatide and the depth of the late-stage pipeline.
- Solid-phase peptide synthesis leads the technology segment in 2026, with liquid-phase and hybrid synthesis platforms used selectively for short peptides, specific conjugations, and selected production scales.
The peptide therapeutics CDMO market segments across service type, peptide class, end use, technology, distribution channel, and packaging. Service type coverage includes peptide API manufacturing, process development and scale-up, sterile fill-finish, analytical and quality control, and integrated end-to-end services. Peptide class coverage spans GLP-1 agonists and metabolic disease peptides, oncology peptides, antimicrobial peptides, peptide vaccines, and specialty peptides. Technology coverage includes solid-phase peptide synthesis, liquid-phase peptide synthesis, hybrid synthesis, and chemoenzymatic approaches.
Peptide API Manufacturing Leads the Service Segment
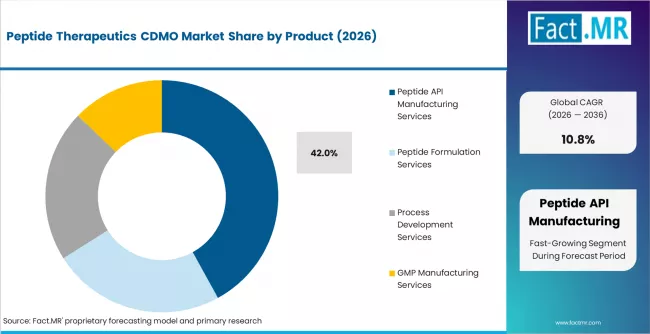
Peptide API manufacturing services account for 42% of service revenue in 2026. The subsegment is anchored by commercial-scale SPPS capacity dedicated to GLP-1 peptides, by process development and scale-up services supporting clinical-stage programs moving toward approval, and by high-complexity manufacturing for lipidated and conjugated peptides. Multi-year commercial supply contracts with GLP-1 sponsors are the single largest revenue pool within the service segment.
Sterile fill-finish services and analytical characterization represent the second and third largest service subsegments. Integrated CDMOs that offer end-to-end services from API manufacturing through fill-finish are capturing premium service economics because they reduce supply chain complexity for sponsors. Analytical services are growing as peptide complexity rises, because longer and more complex peptides require more sophisticated characterization and release testing.
GLP-1 Peptides Dominate the Therapeutic Class Segment
GLP-1 agonists and metabolic disease peptides together represent the largest therapeutic class in 2026. Semaglutide and tirzepatide drive the current commercial demand, with retatrutide and other late-stage candidates providing a pipeline of additional high-volume programs. The therapeutic class is uniquely attractive for CDMOs because individual programs generate very high API volumes over long commercial horizons, which creates predictable capacity utilization and strong economic returns.
Oncology peptides form a second therapeutic class pool, covering peptide-drug conjugates, tumor-targeting peptides, and peptide vaccines in cancer indications. Antimicrobial peptides, cardiovascular peptides, and specialty peptide classes represent smaller but strategically important pools. The therapeutic class mix will continue tilting toward metabolic disease through the forecast period as additional GLP-1 and related programs reach commercial approval.
Key Growth Drivers, Constraints, and Market Scope
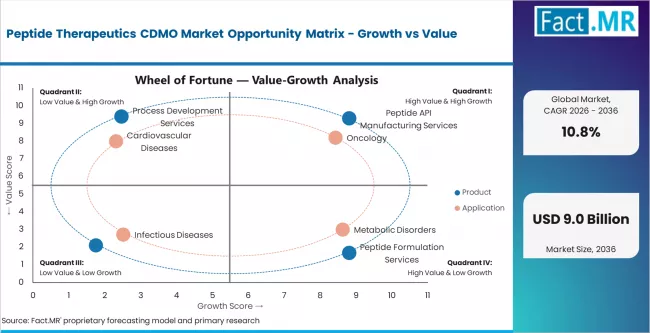
- GLP-1 demand surge, sponsor outsourcing economics, and rising peptide chemistry complexity are creating structural demand growth across the forecast period.
- SPPS capacity constraints, specialty amino acid and resin supply bottlenecks, and skilled peptide chemistry workforce shortages constrain the pace at which supply can match demand.
- Expansion opportunities exist in Asia Pacific, particularly India and China, where capacity additions are absorbing overflow demand from constrained Western CDMO infrastructure.
The peptide therapeutics CDMO market is at a structural inflection point. Demand fundamentals are exceptionally strong, supply is constrained, and capacity expansion plans are being announced and built across major regions. The key variables over the forecast period are the pace of new SPPS capacity coming online, the entry of additional GLP-1 and next-wave candidates into commercial supply, and specialty amino acid and resin supply chain resilience.
Growth Drivers
Three primary forces drive growth. First, GLP-1 peptide therapeutic demand has created a structural capacity shortage that supports premium CDMO economics and drives multi-year capacity expansion programs. Second, sponsor outsourcing economics favor specialized peptide CDMOs because the capital intensity, specialized expertise, and regulatory infrastructure required for in-house SPPS manufacturing cannot be justified by most sponsors running single programs. Third, rising peptide chemistry complexity, including longer chains, non-standard amino acids, lipidation and conjugation steps, increases per-kilogram service value and raises the competitive barrier for new entrants.
Constraints
SPPS capacity is the most significant constraint on near-term growth. Building new peptide manufacturing capacity requires 24 to 36 months of construction and qualification time, which means near-term supply is structurally constrained even with aggressive capacity expansion programs. Specialty amino acid supply, particularly for non-standard amino acids used in complex peptide synthesis, is concentrated among a small number of suppliers and can become a bottleneck when demand surges. Skilled peptide chemistry workforce is in short supply, because peptide synthesis requires specialized training that is not broadly available. Regulatory complexity around GMP peptide manufacturing, particularly for new facility qualifications, extends timelines and adds cost.
Opportunities
The largest near-term opportunity is in commercial-scale GLP-1 API supply, where CDMOs with available SPPS capacity can book long-dated supply contracts at premium economics. A second opportunity is in Asia Pacific capacity expansion, particularly India and China, where lower cost structures and aggressive capacity additions are capturing demand overflow from constrained Western CDMOs. A third opportunity is in complex peptide chemistry, where CDMOs with expertise in lipidation, PEGylation, cyclization, and conjugation can command premium pricing for differentiated services. Integrated service offerings that combine peptide API manufacturing with sterile fill-finish and analytical services capture additional value per program and create customer stickiness.
Regional Outlook Across Key Markets
.webp)
- Asia Pacific leads growth, with India at a 15.8% CAGR anchored by expanding capacity at Piramal Pharma Solutions, Auro Peptides, and domestic CDMO additions, and China (14.2%) supported by WuXi AppTec and domestic peptide capacity programs.
- South Korea (12.9%) and Japan (11.7%) grow steadily, supported by domestic CDMO capacity additions and established biopharma peptide programs.
- Europe contributes strongly through established leaders, with Germany (10.8%), Switzerland (10.1%), and continued Lonza, Bachem, and Polypeptide expansion programs.
- North America grows more slowly, with the United States at a 9.6% CAGR, reflecting mature sponsor demand and selective CDMO capacity expansion.
| Country | CAGR (2026-2036) |
|---|---|
| India | 15.8% |
| China | 14.2% |
| South Korea | 12.9% |
| Japan | 11.7% |
| Germany | 10.8% |
| Switzerland | 10.1% |
| United States | 9.6% |
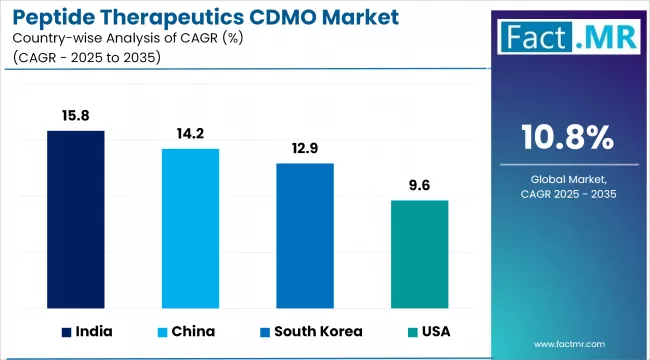
Regional performance reflects both the geography of established peptide CDMO leaders and the geographic pattern of capacity expansion. Asia Pacific leads growth because lower cost structures and aggressive capacity addition programs are capturing demand that cannot be served by constrained Western infrastructure. Europe remains the historical center of peptide manufacturing expertise through Bachem, Polypeptide Group, Lonza, and CordenPharma, but growth rates are slower because the installed base is already large. North America has a substantial installed base at AmbioPharm, Polypeptide, and other firms but grows more slowly than Asia Pacific from a mature baseline.
India
India is the fastest-growing major market, with a 15.8% CAGR through 2036. The country hosts Piramal Pharma Solutions, Auro Peptides, and multiple domestic CDMOs building peptide manufacturing capacity, and is increasingly capturing commercial-scale GLP-1 and complex peptide manufacturing demand. Hyderabad, Mumbai, and Bengaluru are the primary manufacturing centers. Government pharma export promotion programs and competitive labor and construction costs support aggressive capacity expansion.
- Piramal Pharma Solutions, Auro Peptides, and domestic CDMOs are scaling peptide manufacturing capacity aggressively.
- Hyderabad, Mumbai, and Bengaluru are the primary peptide CDMO manufacturing centers.
- Government pharma export promotion, competitive costs, and skilled chemistry workforce support rapid capacity expansion.
China
China records a 14.2% CAGR through 2036. WuXi AppTec anchors domestic peptide CDMO leadership, and additional Chinese CDMOs are building peptide-specific manufacturing capacity to serve domestic and export demand. Shanghai, Suzhou, and Tianjin are the primary manufacturing centers. Domestic biopharma demand for peptide therapeutics is growing rapidly, supporting sustained local CDMO growth alongside export-oriented capacity.
- WuXi AppTec anchors Chinese peptide CDMO leadership with substantial SPPS capacity and integrated services.
- Shanghai, Suzhou, and Tianjin are the primary peptide CDMO manufacturing centers.
- Growing domestic biopharma peptide pipeline demand supports sustained local CDMO growth alongside export manufacturing.
South Korea
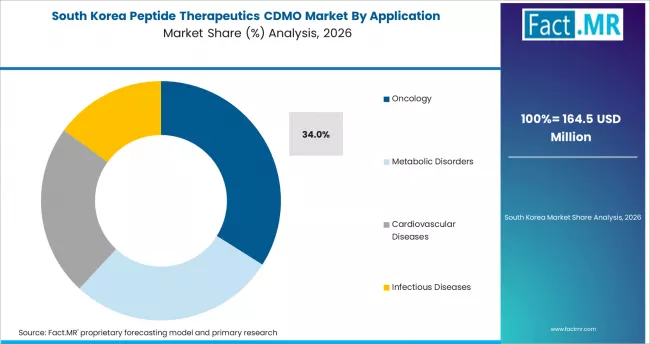
South Korea posts a 12.9% CAGR through 2036. Samsung Biologics and Korean peptide-focused CDMOs are building peptide capacity to serve global biopharma sponsors and a growing domestic biotech pipeline. Government bio-innovation programs direct substantial public funding into CDMO capacity development, and Seoul and Incheon are the primary manufacturing centers.
- Samsung Biologics and peptide-focused Korean CDMOs are scaling capacity to serve global and domestic peptide demand.
- Government bio-innovation programs direct public funding into CDMO capacity development and peptide manufacturing expansion.
- Seoul and Incheon biocluster infrastructure supports peptide CDMO expansion and operations.
Japan
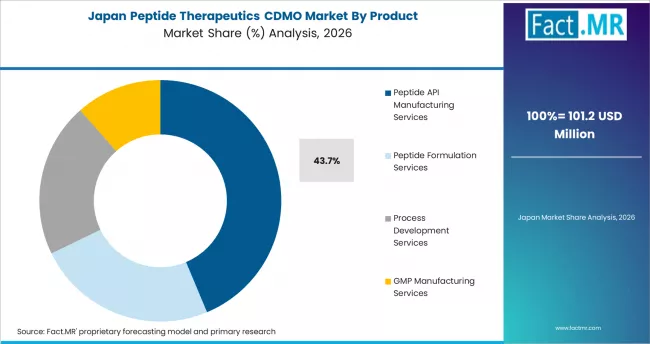
Japan records an 11.7% CAGR through 2036. Established domestic CDMOs serve Japanese pharma sponsors in diabetes, oncology, and cardiovascular peptide programs, and Takeda, Daiichi Sankyo, and Astellas anchor domestic peptide therapeutic pipelines. PMDA regulatory engagement on peptide therapeutics is well-developed, supporting efficient clinical and commercial translation.
- Established Japanese peptide CDMOs serve domestic sponsors across diabetes, oncology, and cardiovascular indications.
- Takeda, Daiichi Sankyo, and Astellas anchor domestic peptide therapeutic pipelines and CDMO demand.
- PMDA regulatory engagement on peptide therapeutics is mature and supports efficient clinical and commercial translation.
Germany
Germany posts a 10.8% CAGR through 2036. CordenPharma and Wacker Chemie anchor German peptide CDMO capability, with additional capacity from Fresenius Kabi and specialty CDMOs serving commercial peptide supply. Frankfurt, Cologne, and Munich biotech and pharma clusters support peptide CDMO operations.
- CordenPharma and Wacker Chemie anchor German peptide CDMO capability with substantial installed SPPS capacity.
- Fresenius Kabi and specialty CDMOs provide additional peptide manufacturing capacity serving commercial supply.
- Frankfurt, Cologne, and Munich biotech and pharma clusters support peptide CDMO operations and workforce.
Switzerland
Switzerland records a 10.1% CAGR through 2036. Bachem anchors global peptide CDMO leadership from its Bubendorf facility, and Lonza's Visp peptide operations provide substantial additional commercial capacity. The country's concentrated pharma and biotech ecosystem, high-quality workforce, and established regulatory infrastructure support premium peptide CDMO economics.
- Bachem anchors global peptide CDMO leadership from its Bubendorf facility with substantial commercial SPPS capacity.
- Lonza's Visp peptide operations provide additional commercial-scale peptide manufacturing capacity.
- Concentrated pharma and biotech ecosystem and skilled workforce support premium peptide CDMO economics.
United States
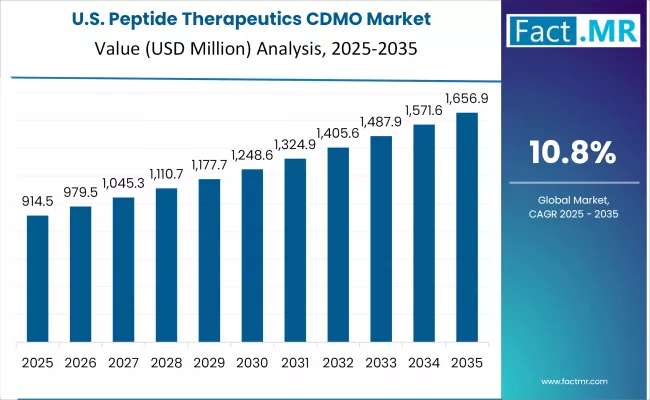
The United States posts a 9.6% CAGR through 2036. AmbioPharm and Polypeptide Laboratories anchor US peptide CDMO capability, with additional capacity from specialty CDMOs serving clinical and commercial supply. FDA regulatory engagement on peptide therapeutics is mature, and the country hosts substantial sponsor pipeline demand for peptide therapeutic programs.
- AmbioPharm and Polypeptide Laboratories anchor US peptide CDMO capability with established SPPS capacity.
- FDA regulatory engagement on peptide therapeutics is mature and supports efficient clinical and commercial translation.
- Substantial US sponsor pipeline demand for peptide therapeutics drives consistent CDMO utilization.
Competitive Benchmarking and Company Positioning
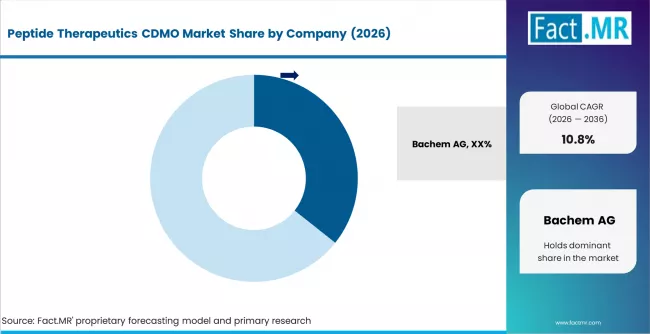
- Bachem, Lonza, and Polypeptide Group collectively represent the historical core of the peptide CDMO category, supported by decades of SPPS expertise, commercial-scale capacity, and established sponsor relationships.
- Asia Pacific CDMOs led by WuXi AppTec and Piramal Pharma Solutions are rapidly gaining share through aggressive capacity expansion and competitive cost structures.
- Specialty and integrated CDMOs including CordenPharma, CPC Scientific, AmbioPharm, Fresenius Kabi, and Almac participate with differentiated capability in complex chemistry, sterile fill-finish, or integrated end-to-end services.
The peptide therapeutics CDMO market features a clear tier structure. Bachem, Lonza, and Polypeptide Group form the leadership tier through decades of SPPS scale expertise and long-term sponsor relationships. WuXi AppTec and major Asia Pacific CDMOs form a rapidly rising second tier through aggressive capacity expansion and competitive cost economics. Specialty CDMOs form a third tier, competing on complex chemistry, sterile fill-finish integration, or regional service. Competitive positions will shift through the forecast period as capacity expansion programs come online and sponsor preferences on supply diversification influence CDMO selection.
Bachem holds estimated 22% share in 2026, anchored by its Bubendorf facility and long-term GLP-1 and peptide therapeutic supply contracts. Lonza, through its Visp peptide operations, holds a strong second position with integrated small-molecule and peptide capacity. Polypeptide Group maintains substantial capacity across multiple global sites. AmbioPharm in the United States is a significant specialty player with deep GLP-1 manufacturing expertise.
CordenPharma, CPC Scientific, Fresenius Kabi, Wacker Chemie, and Almac participate with differentiated service offerings. CordenPharma offers strong integrated small-molecule and peptide services across multiple European facilities. CPC Scientific provides specialty peptide chemistry and complex peptide manufacturing. WuXi AppTec has emerged as a significant peptide player through aggressive capacity expansion serving global demand. Piramal Pharma Solutions and Auro Peptides are scaling rapidly in India, capturing commercial supply demand that cannot be served by constrained Western CDMOs.
The competitive dynamics favor firms with three attributes: commercial-scale SPPS capacity, complex peptide chemistry capability, and integrated sterile fill-finish services. Bachem and Lonza lead on scale and integrated services. WuXi AppTec and Piramal lead on cost-competitive expansion. CordenPharma and CPC Scientific lead on specialty chemistry. Commercial winners over the next decade will combine scale, complex chemistry, and integrated services while managing capacity expansion investment risk carefully as multiple large programs compete for limited infrastructure.
Competitive Analysis
Strategic positioning is shaped by the choice between scale and specialty differentiation. Leading CDMOs with commercial-scale SPPS capacity and long-term sponsor relationships capture the largest GLP-1 and metabolic disease peptide volumes. Specialty CDMOs with complex chemistry expertise capture higher per-kilogram economics for differentiated peptide classes. Integrated CDMOs offering end-to-end API through fill-finish services capture additional value and customer stickiness. Capacity expansion investment decisions are the dominant strategic question, because building new SPPS infrastructure is capital-intensive and multi-year, and CDMOs must balance commercial demand visibility against the risk of capacity overhang if GLP-1 or other peptide programs miss expectations.
Benchmarking Table
| Company | SPPS Capacity | Chemistry Complexity | Integrated Services | Geographic Reach |
|---|---|---|---|---|
| Bachem | Very High | Very High | High | Global |
| Lonza Group | High | High | Very High | Global |
| Polypeptide Group | High | High | Medium-High | Global |
| WuXi AppTec | High (Expanding) | High | High | Global |
| Piramal Pharma Solutions | Medium-High | Medium-High | Medium-High | Global |
Recent Developments
- Bachem announced multi-hundred-million-dollar capacity expansion at its Bubendorf and Sisseln facilities in 2025, adding substantial SPPS capacity and sterile fill-finish infrastructure to serve GLP-1 and peptide therapeutic demand.
- Lonza expanded peptide manufacturing capacity at its Visp, Switzerland site in 2025, adding commercial-scale SPPS infrastructure and integrated services to serve long-term peptide supply contracts.
- Polypeptide Group announced capacity expansion across multiple global sites in 2025 and 2026, responding to sustained commercial peptide demand and locking in multi-year sponsor supply agreements.
Leading Companies Shaping Peptide Therapeutics CDMO Market
-
Major Players
- Bachem
- Lonza Group
- Polypeptide Group
- AmbioPharm
- CordenPharma
- WuXi AppTec
- Piramal Pharma Solutions
-
Emerging Players
- CPC Scientific
- Fresenius Kabi
- Almac
- Wacker Chemie
- PolyPeptide Laboratories
- Auro Peptides
- Samsung Biologics
- Catalent
- Thermo Fisher Scientific
- Siegfried
Sources and Research References
- World Health Organization (WHO) essential medicines and peptide therapeutic publications
- U.S. Food and Drug Administration (FDA) Center for Drug Evaluation and Research peptide therapeutic guidance and approval data
- European Medicines Agency (EMA) guidelines on peptide active substances and peptide drug products
- Bachem, Lonza Group, Polypeptide Group, WuXi AppTec, AmbioPharm, CordenPharma, and Piramal Pharma Solutions corporate filings and investor presentations
- FDA Orange Book and approved peptide therapeutic pipeline data
- Peer-reviewed peptide manufacturing and therapeutic journals including Journal of Peptide Science, Molecular Pharmaceutics, and Organic Process Research and Development
- Trade publications including Pharmaceutical Technology, Contract Pharma, and BioPharma Reporter
- Primary interviews with CDMO executives, biopharma manufacturing and supply chain leaders, specialty amino acid suppliers, and regulatory affairs professionals
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Key Questions This Report Addresses
- What is the size of the peptide therapeutics CDMO market in 2026 and 2036?
- What CAGR is the market expected to record between 2026 and 2036?
- Which service and technology segments are projected to lead the market in 2026?
- Which therapeutic peptide class accounts for the largest share of CDMO demand?
- How are GLP-1 peptides reshaping global SPPS capacity and CDMO economics?
- Which countries are the fastest-growing markets through 2036?
- Who are the leading companies in peptide CDMO manufacturing, and how are they competitively differentiated?
- How does Fact.MR estimate and validate the market forecast?
Peptide Therapeutics CDMO Market Definition
The peptide therapeutics CDMO market covers contract development and manufacturing services for peptide active pharmaceutical ingredients, peptide drug products, and related analytical and release activities delivered by specialized contract organizations to pharmaceutical and biotechnology sponsors. The scope includes peptide API manufacturing via solid-phase peptide synthesis, liquid-phase peptide synthesis, and hybrid approaches, along with peptide drug product formulation, sterile fill-finish services, lyophilization, analytical characterization, stability testing, and cGMP release testing.
Peptide Therapeutics CDMO Market Inclusions
The market includes peptide API manufacturing services across chain lengths and complexity levels, including short peptides, medium and long peptides, lipidated peptides, PEGylated peptides, cyclic peptides, and peptide conjugates. It covers process development and scale-up services, clinical-scale and commercial-scale cGMP manufacturing, sterile fill-finish services for peptide drug products, analytical characterization, stability testing, and peptide-specific QC and release services. Work done for GLP-1 agonists, metabolic disease peptides, oncology peptides, antimicrobial peptides, and peptide vaccines is in scope.
Peptide Therapeutics CDMO Market Exclusions
The market excludes internal peptide manufacturing done by biopharma sponsors in their own facilities, merchant peptide reagent supply outside of contract manufacturing engagements, and small-scale research peptide synthesis not tied to therapeutic program development. Generic sterile fill-finish services not associated with peptide drug products, therapeutic revenue for the sponsor brand, and unrelated small-molecule API CDMO work are also excluded. Academic research peptide services and bench-scale peptide synthesis without cGMP deliverables fall outside the scope.
Peptide Therapeutics CDMO Market Research Methodology
- Primary Research: Interviews with CDMO business development and operations leads at leading peptide manufacturers, biopharma manufacturing and supply chain executives running active peptide programs, specialty amino acid and resin suppliers, and regulatory affairs professionals specializing in peptide therapeutics.
- Desk Research: Peptide therapeutic pipeline databases, FDA and EMA approval records for peptide products, CDMO capacity expansion announcements, annual reports of Bachem, Lonza, Polypeptide Group, WuXi AppTec, and Piramal Pharma Solutions, and peer-reviewed peptide manufacturing literature.
- Market Sizing: Demand-side model built on active clinical and commercial peptide programs, estimated per-kilogram SPPS manufacturing costs and contract values, CDMO capacity utilization across peptide-specific infrastructure, and commercial supply contract pricing benchmarks for GLP-1 and adjacent peptide classes.
- Data Validation: Triangulated across CDMO reported peptide segment revenues, sponsor pipeline disclosures, capital expenditure announcements tied to peptide capacity, and primary interviews. Sensitivity testing on capacity utilization, pricing, and commercial-to-clinical program mix.
Scope of Analysis
| Parameter | Details |
|---|---|
| Quantitative Units | USD 3.2132 billion in 2026 to USD 8.961 billion by 2036, at a CAGR of 10.8% |
| Market Definition | Contract development and manufacturing services for peptide active pharmaceutical ingredients and peptide drug products, including solid-phase peptide synthesis, liquid-phase synthesis, hybrid synthesis, process development, sterile fill-finish, analytical characterization, and cGMP release services delivered by specialized contract organizations. |
| Regions | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries | India, China, South Korea, Japan, Germany, Switzerland, United States, and 25+ additional countries |
| Companies | Bachem, Lonza Group, Polypeptide Group, AmbioPharm, CordenPharma, CPC Scientific, WuXi AppTec, Fresenius Kabi, Piramal Pharma Solutions, Almac, Wacker Chemie, Auro Peptides |
| Forecast Period | 2026 to 2036 |
| Approach | Demand-side and top-down methodology built on active peptide pipeline program counts, per-kilogram SPPS manufacturing contract values, CDMO capacity utilization, commercial supply contract pricing benchmarks, and primary interviews with CDMO executives, pharma sponsors, and regulators. |
Analysis by Service Type, Peptide Class, End Use, Technology, Distribution Channel, and Region
-
Peptide Therapeutics CDMO Market Market Segmented by Product:
- Peptide API Manufacturing Services
- Solid-Phase Peptide Synthesis (SPPS)
- Long-Chain Peptide Synthesis
- High-Purity Peptide APIs
- Solid-Phase Peptide Synthesis (SPPS)
- Peptide Formulation Services
- Injectable Peptide Formulations
- Oral & Nasal Peptide Delivery Systems
- Injectable Peptide Formulations
- Process Development Services
- Scale-Up & Optimization Services
- Analytical Method Development
- Scale-Up & Optimization Services
- GMP Manufacturing Services
- Clinical & Commercial Production
- Sterile Fill-Finish Services
- Clinical & Commercial Production
- Peptide API Manufacturing Services
-
Peptide Therapeutics CDMO Market Market Segmented by Application:
- Oncology
- Targeted Cancer Therapies
- Peptide-Based Immunotherapies
- Targeted Cancer Therapies
- Metabolic Disorders
- Diabetes Management
- Obesity & GLP-1 Analog Development
- Diabetes Management
- Cardiovascular Diseases
- Peptide Hormone Therapies
- Hypertension & Lipid Control
- Peptide Hormone Therapies
- Infectious Diseases
- Antimicrobial Peptides
- Vaccine Adjuvant Applications
- Antimicrobial Peptides
- Oncology
-
Peptide Therapeutics CDMO Market Market Segmented by End Use:
- Pharmaceutical & Biotechnology Companies
- Large Pharma Enterprises
- Mid-Sized Biotech Firms
- Large Pharma Enterprises
- Contract Development & Manufacturing Organizations (CDMOs)
- Peptide Manufacturing Specialists
- Integrated Biologics CDMOs
- Peptide Manufacturing Specialists
- Academic & Research Institutes
- Drug Discovery Labs
- Translational Research Centers
- Drug Discovery Labs
- Contract Research Organizations (CROs)
- Clinical Development Support
- Preclinical Testing Providers
- Clinical Development Support
- Pharmaceutical & Biotechnology Companies
-
Peptide Therapeutics CDMO Market Market Segmented by Technology:
- Solid-Phase Peptide Synthesis (SPPS)
- Automated Peptide Synthesizers
- High-Throughput Synthesis Platforms
- Automated Peptide Synthesizers
- Liquid-Phase Peptide Synthesis
- Hybrid Synthesis Systems
- Purification Chromatography Tech
- Hybrid Synthesis Systems
- Recombinant Peptide Production
- Biotech Expression Systems
- Microbial & Cell-Based Production
- Biotech Expression Systems
- Advanced Purification Technologies
- HPLC & UHPLC Systems
- Mass Spectrometry Quality Control
- HPLC & UHPLC Systems
- Solid-Phase Peptide Synthesis (SPPS)
-
Peptide Therapeutics CDMO Market Market Segmented by Distribution Channel:
- Direct CDMO Contracts
- Long-Term Pharma Partnerships
- Integrated Manufacturing Agreements
- Long-Term Pharma Partnerships
- Licensing & Strategic Collaborations
- R&D Co-Development Deals
- Technology Transfer Agreements
- R&D Co-Development Deals
- Outsourced Manufacturing Networks
- Global CDMO Partnerships
- Specialized Peptide Vendors
- Global CDMO Partnerships
- Direct CDMO Contracts
-
Peptide Therapeutics CDMO Market Market Segmented by Packaging:
- Sterile Bulk Packaging
- API Powder Containers
- GMP-Compliant Sealed Vials
- API Powder Containers
- Injectable Drug Packaging
- Prefilled Syringes
- Single-Dose Vials
- Prefilled Syringes
- Cold Chain Packaging
- Temperature-Controlled Transport
- Cryogenic Storage Systems
- Temperature-Controlled Transport
- Lyophilized Product Packaging
- Freeze-Dried Peptide Kits
- Reconstitution Packaging Systems
- Freeze-Dried Peptide Kits
- Sterile Bulk Packaging
-
Peptide Therapeutics CDMO Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
How big is the peptide therapeutics CDMO market in 2026?
The global peptide therapeutics CDMO market is valued at USD 3.2132 billion in 2026.
What will be the size of the peptide therapeutics CDMO market in 2036?
The market is projected to reach USD 8.961 billion by 2036.
How fast is the peptide therapeutics CDMO market expected to grow between 2026 and 2036?
The market is expected to expand at a CAGR of 10.8% between 2026 and 2036.
Which service segment leads the peptide therapeutics CDMO market in 2026?
Peptide API Manufacturing Services lead the service segment with 42% share in 2026.
Which country is the fastest-growing market for peptide CDMOs?
India is the fastest-growing major market with a 15.8% CAGR through 2036.
Which is the second-fastest-growing market?
China is the second-fastest-growing market, expanding at a 14.2% CAGR through 2036.
What is driving demand in the peptide therapeutics CDMO market?
Demand is driven primarily by the unprecedented commercial success of GLP-1 peptide therapeutics including semaglutide, tirzepatide, and the next wave of candidates, combined with sponsor preference for outsourced peptide manufacturing and rising chemistry complexity across therapeutic peptide classes.
How does Fact.MR estimate and validate the market forecast?
The forecast is built on a demand-side and top-down methodology using active peptide pipeline program counts, per-kilogram SPPS manufacturing contract values, CDMO capacity utilization, commercial supply contract pricing benchmarks, and primary interviews with CDMO executives, pharma sponsors, and regulators.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Peptide API Manufacturing Services
- Solid-Phase Peptide Synthesis (SPPS)
- _Long-Chain Peptide Synthesis
- _High-Purity Peptide APIs
- Peptide Formulation Services
- Injectable Peptide Formulations
- _Oral & Nasal Peptide Delivery Systems
- Process Development Services
- Scale-Up & Optimization Services
- _Analytical Method Development
- GMP Manufacturing Services
- Clinical & Commercial Production
- _Sterile Fill-Finish Services
- Peptide API Manufacturing Services
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Oncology
- Targeted Cancer Therapies
- _Peptide-Based Immunotherapies
- Metabolic Disorders
- Diabetes Management
- _Obesity & GLP-1 Analog Development
- Cardiovascular Diseases
- Peptide Hormone Therapies
- _Hypertension & Lipid Control
- Infectious Diseases
- Antimicrobial Peptides
- _Vaccine Adjuvant Applications
- Oncology
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical & Biotechnology Companies
- Large Pharma Enterprises
- _Mid-Sized Biotech Firms
- Contract Development & Manufacturing Organizations (CDMOs)
- Peptide Manufacturing Specialists
- _Integrated Biologics CDMOs
- Academic & Research Institutes
- Drug Discovery Labs
- _Translational Research Centers
- Contract Research Organizations (CROs)
- Clinical Development Support
- _Preclinical Testing Providers
- Pharmaceutical & Biotechnology Companies
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Solid-Phase Peptide Synthesis (SPPS)
- Automated Peptide Synthesizers
- _High-Throughput Synthesis Platforms
- Liquid-Phase Peptide Synthesis
- Hybrid Synthesis Systems
- _Purification Chromatography Tech
- Recombinant Peptide Production
- Biotech Expression Systems
- _Microbial & Cell-Based Production
- Advanced Purification Technologies
- HPLC & UHPLC Systems
- _Mass Spectrometry Quality Control
- Solid-Phase Peptide Synthesis (SPPS)
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct CDMO Contracts
- Long-Term Pharma Partnerships
- _Integrated Manufacturing Agreements
- Licensing & Strategic Collaborations
- R&D Co-Development Deals
- _Technology Transfer Agreements
- Outsourced Manufacturing Networks
- Global CDMO Partnerships
- _Specialized Peptide Vendors
- Direct CDMO Contracts
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging, 2026 to 2036
- Sterile Bulk Packaging
- API Powder Containers
- _GMP-Compliant Sealed Vials
- Injectable Drug Packaging
- Prefilled Syringes
- _Single-Dose Vials
- Cold Chain Packaging
- Temperature-Controlled Transport
- _Cryogenic Storage Systems
- Lyophilized Product Packaging
- Freeze-Dried Peptide Kits
- _Reconstitution Packaging Systems
- Sterile Bulk Packaging
- Y to o to Y Growth Trend Analysis By Packaging, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Competition Analysis
- Competition Deep Dive
- Bachem AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- PolyPeptide Group
- CPC Scientific Inc.
- Lonza Group Ltd.
- CordenPharma International
- GenScript Biotech Corporation
- Almac Group
- Thermo Fisher Scientific Inc.
- AmbioPharm Inc.
- Peptides International Inc.
- Bachem AG
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Packaging, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Distribution Channel
- Figure 18: Global Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Packaging
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 28: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 46: North America Market Attractiveness Analysis by Distribution Channel
- Figure 47: North America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 49: North America Market Attractiveness Analysis by Packaging
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Application
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 62: Latin America Market Attractiveness Analysis by Technology
- Figure 63: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 65: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 66: Latin America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 68: Latin America Market Attractiveness Analysis by Packaging
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Application
- Figure 76: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 78: Western Europe Market Attractiveness Analysis by End Use
- Figure 79: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 81: Western Europe Market Attractiveness Analysis by Technology
- Figure 82: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 84: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 85: Western Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 87: Western Europe Market Attractiveness Analysis by Packaging
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Application
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Packaging
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 113: East Asia Market Attractiveness Analysis by Application
- Figure 114: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 116: East Asia Market Attractiveness Analysis by End Use
- Figure 117: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 119: East Asia Market Attractiveness Analysis by Technology
- Figure 120: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 122: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 123: East Asia Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 125: East Asia Market Attractiveness Analysis by Packaging
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Packaging
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Packaging
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis