Pyrogen Testing Market Size, Share, Growth and Forecast (2026 - 2036)
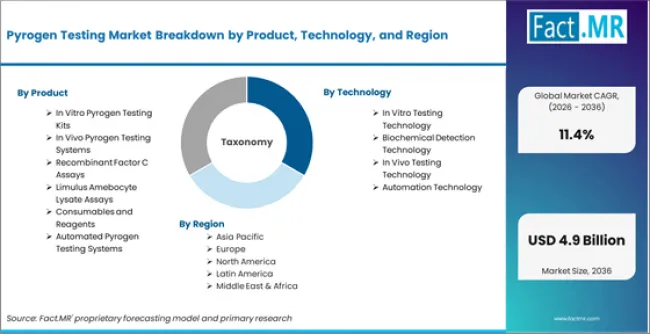
Pyrogen Testing Market is segmented by Product (In Vitro Pyrogen Testing Kits, In Vivo Pyrogen Testing Systems, Recombinant Factor C Assays, Limulus Amebocyte Lysate Assays, Consumables and Reagents, Automated Pyrogen Testing Systems), Technology (In Vitro Testing Technology, Biochemical Detection Technology, In Vivo Testing Technology, Automation Technology), Method (LAL Test Method, MAT Method, Rabbit Test Method, Recombinant Protein Method), and Region, with forecasts covering the period from 2026 to 2036.
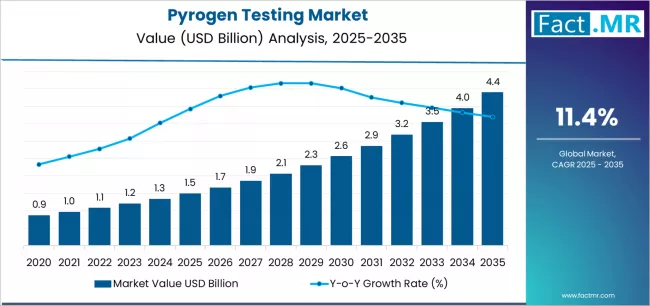
According to Fact.MR estimates, the global pyrogen testing Market was valued at USD 1.5 billion in 2025. The market is projected to reach USD 1.7 billion in 2026 and is expected to grow to USD 4.9 billion by 2036, expanding at a CAGR of 11.4%. In Vitro Pyrogen Testing Kits is anticipated to account for 38.6% of the product segment in 2026, while In Vitro Testing Technology is expected to remain the leading application with around 41.3% share.
Pyrogen Testing Market Analysis and Forecast by Fact.MR
- The global pyrogen testing market is estimated at US$ 1.5 billion in 2025 and has been forecast to expand at a CAGR of 11.4% to reach US$ 4.9 billion by 2036.
- Pyrogen testing validates that pharmaceutical products, medical devices, and biologics are free from fever-inducing contaminants before patient administration.
- In vitro pyrogen testing kits, including monocyte activation tests (MAT) and endotoxin detection kits, account for the largest product share. LAL (Limulus Amebocyte Lysate) test methods remain the most widely used, while recombinant Factor C (rFC) assays are growing as an animal-free alternative. Pharmaceutical manufacturing is the dominant application.
- Pharmaceutical companies and biotechnology firms form the primary buyer base.
Summary of Pyrogen Testing Market
- Market Snapshot
- In 2025, the global Pyrogen Testing Market was valued at approximately USD 1.5 billion.
- The market is estimated to reach USD 1.7 billion in 2026 and is projected to attain USD 4.9 billion by 2036.
- The pyrogen testing market is likely to expand at a CAGR of 11.4% during the forecast period.
- The market is anticipated to create an absolute dollar opportunity of USD 3.2 billion between 2026 and 2036.
- In Vitro Pyrogen Testing Kits accounts for 38.6% of product share in 2026.
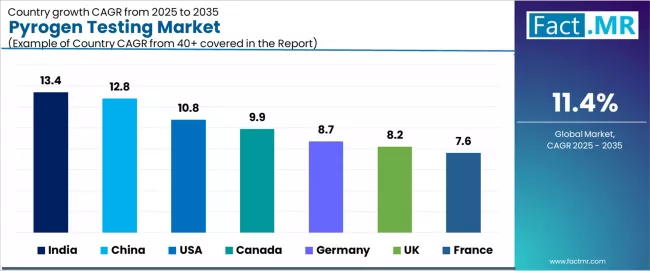
- India (13.4%) and China (12.8%) are the key growth markets during the forecast period.
- Demand and Growth Drivers
- Biopharmaceutical production volume growth, including biologics, cell therapies, and gene therapies, is generating proportionally higher pyrogen testing demand at multiple manufacturing stages.
- Regulatory mandate for endotoxin and pyrogen testing by FDA, EMA, and pharmacopoeial standards creates non-discretionary demand for validated testing products across all parenteral drug manufacturing.
- Recombinant Factor C (rFC) assay adoption as a synthetic, animal-free alternative to LAL testing is addressing horseshoe crab conservation concerns and supply chain vulnerability.
- Monocyte Activation Test (MAT) adoption for non-endotoxin pyrogen detection is expanding the testing scope beyond traditional LAL methods that only detect gram-negative bacterial endotoxin.
- Medical device pyrogen testing requirements, with growing implantable and parenteral device production, are expanding the addressable market beyond pharmaceutical manufacturing.
- Product and Segment View
- In Vitro Pyrogen Testing Kits accounts for 38.6% of the product segment in 2026, supported by strong installed base and continued procurement across primary end use channels.
- In Vitro Testing Technology accounts for 41.3% of the technology segment in 2026, reflecting concentrated demand across established application areas.
- LAL Test Method accounts for 35.9% of the method segment in 2026, with adoption rising across both developed and emerging markets.
- Key segmentation includes:
- Product: In Vitro Pyrogen Testing Kits, In Vivo Pyrogen Testing Systems, Recombinant Factor C Assays
- Technology: In Vitro Testing Technology, Biochemical Detection Technology, In Vivo Testing Technology
- Method: LAL Test Method, MAT Method, Rabbit Test Method
- Application: Pharmaceutical Manufacturing, Medical Device Testing, Biotechnology Research
- These systems enable:
- Targeted procurement and specification alignment across buyer segments
- Performance benchmarking across product and application categories
- Regional demand mapping across 30 plus countries
- Geography and Competitive Outlook
- North America accounts for a large share of global demand, supported by established procurement channels and a mature buyer base.
- Europe shows stable demand, with Germany (8.7%) contributing through regulatory compliance and institutional procurement for pyrogen testing products.
- Asia Pacific demand is led by India (13.4%) and China (12.8%).
- India (13.4%) and China (12.8%) are the fastest growing country markets during the forecast period.
- Leading companies include Charles River Laboratories, Novo Nordisk, Merz KGaA, GenScript, bioMérieux SA.
- Analyst Opinion
- Shambhu Nath Jha, Principal Consultant at Fact.MR, says 'The pyrogen testing market is structurally linked to parenteral pharmaceutical manufacturing volume and regulatory enforcement. Charles River Laboratories leads through its Endosafe LAL and rFC platform and broadest pharmaceutical quality control testing portfolio. The LAL-to-rFC transition is the defining competitive dynamic, and companies that establish rFC as the validated standard across their customer base will capture recurring revenue as manufacturing volumes grow. The monocyte activation test represents a separate growth vector addressing non-endotoxin pyrogen detection that LAL and rFC cannot identify.'
Key Growth Drivers, Constraints, and Opportunities
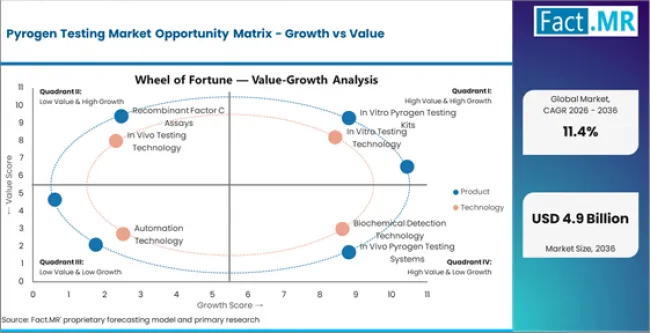
Key Factors Driving Growth
- Biopharmaceutical production volume growth, including biologics, cell therapies, and gene therapies, is generating proportionally higher pyrogen testing demand at multiple manufacturing stages.
- Regulatory mandate for endotoxin and pyrogen testing by FDA, EMA, and pharmacopoeial standards creates non-discretionary demand for validated testing products across all parenteral drug manufacturing.
- Recombinant Factor C (rFC) assay adoption as a synthetic, animal-free alternative to LAL testing is addressing horseshoe crab conservation concerns and supply chain vulnerability.
Key Market Constraints
- LAL reagent supply dependency on horseshoe crab blood harvesting creates sustainability concerns and potential supply limitations that motivate alternative test development.
- Regulatory transition period from LAL to rFC methods, where some agencies still require LAL as the compendial reference, creates parallel testing obligations that increase cost.
- Small batch manufacturing in cell and gene therapy creates testing cost per batch that is proportionally higher than large-scale pharmaceutical production.
Key Opportunity Areas
- Complete regulatory acceptance of rFC as a compendial method replacement for LAL would accelerate market transition toward sustainable, synthetic pyrogen testing.
- Automated pyrogen testing platforms integrated with manufacturing quality control systems are enabling real-time batch release testing that reduces manufacturing cycle time.
- Emerging market pharmaceutical manufacturing capacity expansion is creating first-time procurement demand for validated pyrogen testing systems and reagents.
Segment-wise Analysis of the Pyrogen Testing Market
- In Vitro Pyrogen Testing Kits holds 38.6% of the product segment in 2026, supported by established demand channels and continued procurement across primary end use applications.
- In Vitro Testing Technology holds 41.3% of the technology segment in 2026, supported by established demand channels and continued procurement across primary end use applications.
- LAL Test Method holds 35.9% of the method segment in 2026, supported by established demand channels and continued procurement across primary end use applications.
The pyrogen testing market is segmented by product, technology, method, application, and region.
By product, the market covers In Vitro Pyrogen Testing Kits, In Vivo Pyrogen Testing Systems, Recombinant Factor C Assays.
By technology, coverage includes In Vitro Testing Technology, Biochemical Detection Technology, In Vivo Testing Technology.
Which Product Segment Dominates the Pyrogen Testing Market?
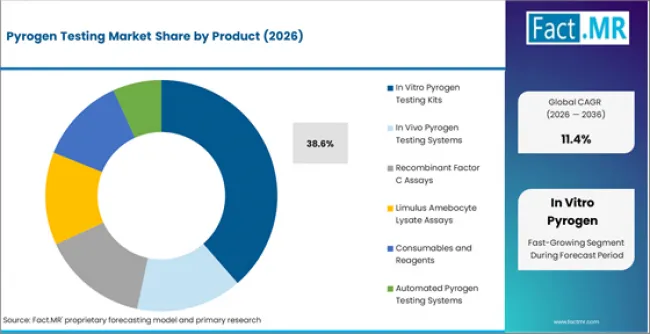
In Vitro Pyrogen Testing Kits is expected to account for 38.6% of the product segment in 2026. Within this category, Monocyte Activation Test Kits, Endotoxin Detection Kits, Biological Assay Based Kits represent the primary sub-segments generating procurement activity. The depth of the in vitro pyrogen testing kits installed base across product applications sustains replacement demand and limits share erosion from newer alternatives in the pyrogen testing market.
In Vivo Pyrogen Testing Systems is the second largest category in the product segment, with demand concentrated in Rabbit Pyrogen Test Systems and Laboratory Animal Test Systems. Growth in in vivo pyrogen testing systems reflects shifting buyer requirements within the pyrogen testing market toward this category.
Which Technology Segment Dominates the Pyrogen Testing Market?
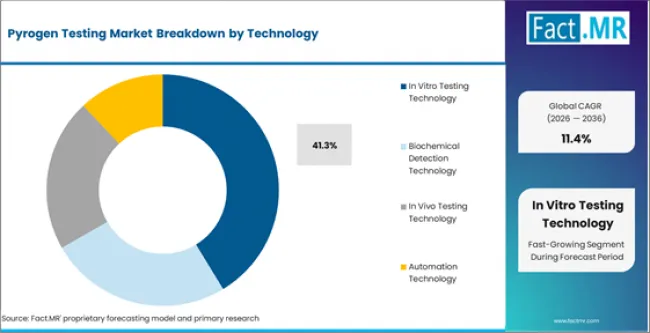
In Vitro Testing Technology is expected to account for 41.3% of the technology segment in 2026. Within this category, Cell Based Assay Technology, Monocyte Activation Technology represent the primary sub-segments generating procurement activity. The depth of the in vitro testing technology installed base across technology applications sustains replacement demand and limits share erosion from newer alternatives in the pyrogen testing market.
Biochemical Detection Technology is the second largest category in the technology segment, with demand concentrated in Limulus Amebocyte Lysate Technology and Recombinant Factor C Technology. Growth in biochemical detection technology reflects shifting buyer requirements within the pyrogen testing market toward this category.
Which Method Segment Dominates the Pyrogen Testing Market?
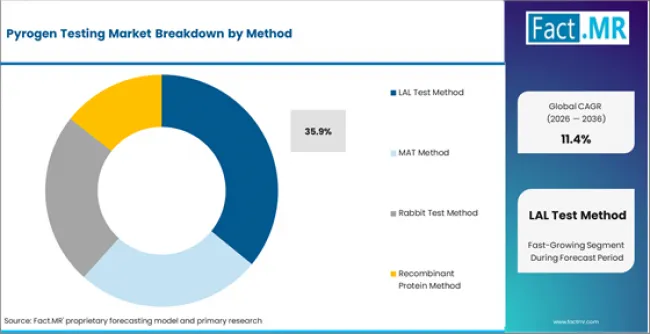
LAL Test Method is expected to account for 35.9% of the method segment in 2026. Within this category, Gel Clot Method, Turbidimetric Method, Chromogenic Method represent the primary sub-segments generating procurement activity. The depth of the lal test method installed base across method applications sustains replacement demand and limits share erosion from newer alternatives in the pyrogen testing market.
MAT Method is the second largest category in the method segment, with demand concentrated in Human Blood Based Monocyte Activation Test and Cell Mediated Immune Response Testing. Growth in mat method reflects shifting buyer requirements within the pyrogen testing market toward this category.
Which Product Trend is Shaping the Next Phase of Growth in the Pyrogen Testing Market?
Recombinant Factor C (rFC) assays represent the most important technology transition within the pyrogen testing market. rFC is a synthetic alternative to LAL that detects endotoxin using a recombinant version of the horseshoe crab clotting enzyme, eliminating dependence on wild-caught horseshoe crab blood. This addresses both the supply sustainability concern and the ethical considerations around horseshoe crab harvesting that have pressured the LAL industry.
Charles River Laboratories, Lonza (through its Endosafe platform), and bioMerieux are leading rFC adoption. European Pharmacopoeia has accepted rFC as a compendial method, and FDA acceptance is progressing through individual product validations. The market transition from LAL to rFC is gradual because pharmaceutical companies must validate the new method against their existing products, but the trajectory is clear: rFC is becoming the default choice for new product development while LAL maintains its installed base in established manufacturing processes.
Regional Outlook Across Key Markets
.webp)
- India is growing at 13.4%, supported by pharmaceutical export quality compliance, biologic production expansion, and domestic reagent development.
- China is growing at 12.8%, supported by biopharmaceutical manufacturing growth, CDE regulatory standards, and pharmacopoeial updates.
- USA is growing at 10.8%, supported by FDA enforcement standards, cell therapy manufacturing growth, and domestic reagent production.
- Canada is growing at 9.9%, supported by Health Canada GMP requirements, pharmaceutical export alignment, and U.S. supply chain proximity.
CAGR Table
| Country | CAGR (%) |
|---|---|
| India | 13.4% |
| China | 12.8% |
| USA | 10.8% |
| Canada | 9.9% |
| Germany | 8.7% |
| UK | 8.2% |
| France | 7.6% |
Source: Fact MR (FMR) analysis, based on proprietary forecasting model and primary research.
Market Outlook for Pyrogen Testing in India
The India Pyrogen Testing market is projected to grow at a CAGR of 13.4% through 2036. India is the fastest-growing pyrogen testing market, driven by pharmaceutical manufacturing expansion and export quality compliance requirements.
- Indian pharmaceutical manufacturers require pyrogen testing to meet FDA, EMA, and WHO prequalification standards for export products.
- Growing biologic and biosimilar production by Biocon, Dr. Reddys, and others generates incremental pyrogen testing demand.
- Domestic testing reagent companies are developing cost-competitive LAL and rFC products for the Indian pharmaceutical market.
Pyrogen Testing Demand Trends in China
The China Pyrogen Testing market is projected to grow at a CAGR of 12.8% through 2036. Biopharmaceutical manufacturing growth and CDE regulatory standards support pyrogen testing demand in China.
- Domestic biopharmaceutical production expansion by BeiGene, Hengrui, and WuXi Biologics generates growing pyrogen testing volumes.
- CDE regulatory standards aligning with ICH guidelines mandate validated pyrogen testing for pharmaceutical product release.
- Chinese pharmacopoeial updates are incorporating rFC and MAT methods alongside traditional LAL standards.
USA Pyrogen Testing Market Growth Outlook
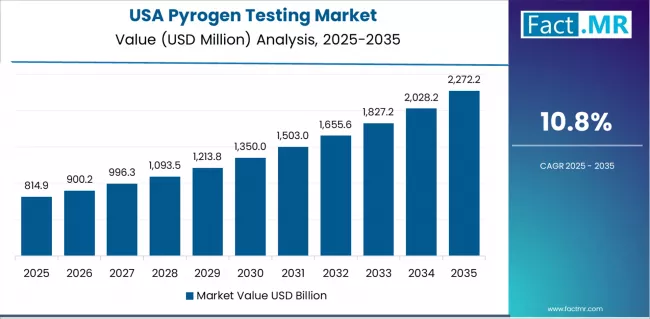
The USA Pyrogen Testing market is projected to grow at a CAGR of 10.8% through 2036. The U.S. is the largest pyrogen testing market, driven by pharmaceutical manufacturing concentration and FDA enforcement standards.
- FDA Bacterial Endotoxins Test requirements (USP <85>) mandate validated pyrogen testing for all parenteral pharmaceutical products.
- Cell and gene therapy manufacturing expansion generates high per-batch testing demand at FDA-registered manufacturing facilities.
- Charles River Laboratories and Lonza maintain major U.S. testing reagent production and distribution operations.
Canada Pyrogen Testing Market Demand Analysis
The Canada Pyrogen Testing market is projected to grow at a CAGR of 9.9% through 2036. Pharmaceutical manufacturing quality standards and Health Canada requirements support pyrogen testing in Canada.
- Health Canada GMP requirements mandate pyrogen testing aligned with USP and EP pharmacopoeial standards.
- Canadian pharmaceutical manufacturing operations require validated testing for both domestic and export markets.
- Proximity to U.S. pharmaceutical supply chain supports aligned testing practices and reagent procurement.
Pyrogen Testing Market Growth Assessment in Germany
The Germany Pyrogen Testing market is projected to grow at a CAGR of 8.7% through 2036. Pharmaceutical manufacturing and PEI regulatory oversight sustain pyrogen testing demand in Germany.
- Paul Ehrlich Institut regulatory oversight of biologic products requires validated pyrogen testing for German-manufactured biologics.
- European Pharmacopoeia rFC method acceptance (EP 2.6.32) provides compendial support for rFC adoption in German manufacturing.
- Merck KGaA domestic operations provide pyrogen testing reagents through established European pharmaceutical supply channels.
Pyrogen Testing Market Opportunity Outlook in UK
The UK Pyrogen Testing market is projected to grow at a CAGR of 8.2% through 2036. MHRA standards and pharmaceutical manufacturing support pyrogen testing demand in the United Kingdom.
- MHRA GMP inspection requirements mandate validated pyrogen testing for UK-manufactured parenteral pharmaceutical products.
- Post-Brexit MHRA regulatory independence maintains alignment with EP pyrogen testing standards.
- Cell and gene therapy manufacturing hubs in Stevenage and other locations generate specialized pyrogen testing demand.
Demand Landscape for Pyrogen Testing in France
The France Pyrogen Testing market is projected to grow at a CAGR of 7.6% through 2036. Pharmaceutical manufacturing and ANSM regulatory standards support pyrogen testing in France.
- Sanofi, Servier, and other French pharmaceutical manufacturers require pyrogen testing across parenteral production lines.
- ANSM regulatory oversight mandates compliance with European Pharmacopoeia pyrogen testing standards.
- Vaccine production by Sanofi Pasteur in Lyon generates substantial endotoxin and pyrogen testing volumes.
Competitive Benchmarking and Company Positioning
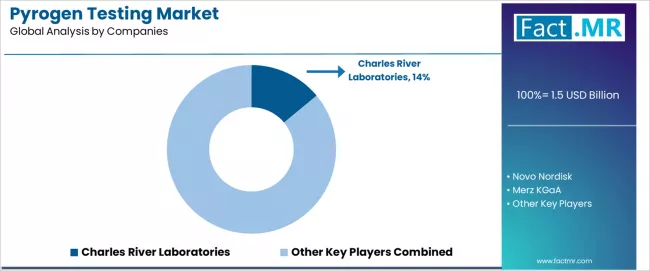
Pyrogen Testing Market Analysis By Company
- Charles River Laboratories, Novo Nordisk, Merz KGaA, GenScript hold established positions in the global pyrogen testing market through broad product portfolios, geographic reach, and growing capabilities in higher specification categories.
- bioMérieux SA, Lonza Group, Thermo Fisher Scientific, Inc., Seikagaku Biobusiness Corporation participate across specific price tiers, end use segments, and regional channels, with competition shaped by product quality, service support, and distribution access.
- Pyrogen testing platform breadth spanning LAL, rFC, and MAT methods, regulatory validation support capability, and pharmaceutical manufacturing customer relationship depth are the primary competitive differentiators.
Charles River Laboratories leads the pyrogen testing market through its Endosafe LAL and rFC platform and broad pharmaceutical quality testing portfolio. Lonza competes through its PyroGene rFC assay. Merck KGaA serves through its Millipore pyrogen detection product line. GenScript provides recombinant endotoxin detection reagents. bioMerieux SA participates through its microbial quality control testing platform.
The market is concentrated among a small number of pharmaceutical quality testing companies with deep regulatory validation expertise. Competition is shaped by the LAL-to-rFC transition, where companies with both legacy LAL and next-generation rFC products can manage customer migration. Pure rFC suppliers compete on sustainability messaging and supply reliability. MAT testing introduces a separate competitive dimension where human blood-based testing offers broader pyrogen detection than endotoxin-specific LAL or rFC methods.
Key Companies in the Pyrogen Testing Market
Charles River Laboratories, Novo Nordisk, Merz KGaA, GenScript are among the leading players in the pyrogen testing market globally, supported by strong brand visibility and broad product portfolios.
bioMérieux SA, Lonza Group, Thermo Fisher Scientific, Inc., Seikagaku Biobusiness Corporation hold established positions across specific categories, supported by durable products and regional distribution.
Recent Industry Developments
- Charles River Laboratories - rFC Compendial Validation (January 2026)
- Charles River Laboratories completed compendial validation of its recombinant Factor C endotoxin detection assay across 50 pharmaceutical product types, supporting regulatory acceptance as a LAL replacement method.
- Novo Nordisk - Automated MAT Platform (2025)
- Novo Nordisk launched an automated monocyte activation test platform that standardizes non-endotoxin pyrogen detection across pharmaceutical quality control laboratories.
- Merz KGaA - Rapid Endotoxin Device (2025)
- Merz KGaA introduced a portable rapid endotoxin detection device delivering quantitative results in under 15 minutes, targeting in-process manufacturing quality checks.
- GenScript - Sustainable Testing Initiative (February 2026)
- GenScript announced a sustainability program transitioning its entire endotoxin testing portfolio from horseshoe crab-derived LAL to synthetic recombinant Factor C reagents by 2028.
Leading Companies Shaping the Pyrogen Testing Market
- Charles River Laboratories
- Novo Nordisk
- Merz KGaA
- GenScript
- bioMérieux SA
- Lonza Group
- Thermo Fisher Scientific, Inc.
- Seikagaku Biobusiness Corporation
- FUJIFILM Wako Chemicals USA Corporation (Pyrostar)
- MiCAN Technologies Inc.
Sources and Research References
- Charles River Laboratories. Product portfolio and corporate announcements.
- Novo Nordisk. Product portfolio and corporate announcements.
- Merz KGaA. Product portfolio and corporate announcements.
- Industry association statistics, regulatory publications, trade databases, and company product catalogs.
- Primary interviews with manufacturers, distributors, buyers, and institutional users.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Key Questions This Report Addresses
- What is the current and future size of the Pyrogen Testing Market?
- How fast is the Pyrogen Testing Market expected to grow between 2026 and 2036?
- Which product is likely to lead the market by 2026?
- Which technology segment is expected to account for the highest demand by 2026?
- What factors are driving demand in this market globally?
- How is technology adoption influencing the market structure?
- Which countries are projected to show the fastest growth through 2036?
- What is driving market expansion in India and China?
- Who are the key companies active in the Pyrogen Testing Market?
- How does Fact.MR estimate and validate the market forecast?
Pyrogen Testing Market Definition
The pyrogen testing market covers products, technologies, and services associated with product, technology, method categories. The scope includes global and regional market size and forecasts for 2026 to 2036 across product, technology, method, application, and region.
Pyrogen Testing Market Inclusions
- The scope covers global and regional market size and forecasts for 2026 to 2036 across product, technology, method, application, and region.
- It includes regional demand analysis across major geographies, based on factors such as technology adoption, regulatory requirements, institutional spending, and commercial demand patterns.
- The report includes pricing and cost analysis across entry-level, mid-range, and premium categories, including hardware, software, and service components where applicable.
- It covers technology trends, market drivers and constraints, and the competitive landscape, including product portfolios, pricing strategy, geographic reach, and distribution expansion of leading players.
Pyrogen Testing Market Exclusions
- The scope excludes adjacent categories, general-purpose alternatives, and products not specifically classified under the pyrogen testing market taxonomy.
- It excludes custom-built in-house solutions, informal unbranded products with low market visibility, and rental or leasing services without direct product sales.
- The report excludes military or defense-grade variants, industrial systems outside the defined scope, and non-commercial applications without market transaction data.
- It also excludes software-only platforms sold independently from hardware, unless they are part of the defined product or service taxonomy.
Pyrogen Testing Market Research Methodology
- The methodology combines secondary research, primary interviews, and forecast modelling.
- It draws on 120 plus secondary sources and benchmarks 65 plus company product portfolios.
- Market sizing covers 30 plus countries through a demand-side model, supported by top-down validation.
- Key inputs include end use adoption rates, technology penetration, pricing benchmarks, and regional procurement patterns.
- The model cross-checks demand with shipments, pricing trends, channel mix, and product launches.
- Primary validation includes 25 plus interviews across manufacturers, distributors, buyers, and institutional users.
- Final estimates go through trade-flow checks, sales-pattern review, and sensitivity testing.
Scope of Analysis
| Parameter | Details |
|---|---|
| Quantitative Units | USD 1.7 billion to USD 4.9 billion, at a CAGR of 11.4% |
| Market Definition | The Pyrogen Testing Market covers products, technologies, and services across product, technology, method categories for commercial, institutional, and consumer applications. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, Canada, UK, Germany, China, India, 30 plus countries |
| Key Companies | Charles River Laboratories, Novo Nordisk, Merz KGaA, GenScript, bioMérieux SA, Lonza Group, Thermo Fisher Scientific, Inc., Seikagaku Biobusiness Corporation, FUJIFILM Wako Chemicals USA Corporation (Pyrostar), MiCAN Technologies Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid demand-side and top-down methodology built on country-level application demand, product benchmarking, pricing analysis, shipment validation, and primary interviews across manufacturers, distributors, and end users |
Pyrogen Testing Market Segmentation
-
Pyrogen Testing Market Segmented by Product:
- In Vitro Pyrogen Testing Kits
- Monocyte Activation Test Kits
- Endotoxin Detection Kits
- Biological Assay Based Kits
- In Vivo Pyrogen Testing Systems
- Rabbit Pyrogen Test Systems
- Laboratory Animal Test Systems
- Recombinant Factor C Assays
- Chromogenic rFC Assays
- Fluorometric rFC Assays
- Limulus Amebocyte Lysate Assays
- Gel Clot LAL Assays
- Turbidimetric LAL Assays
- Chromogenic LAL Assays
- Consumables and Reagents
- Endotoxin Standards
- Cell Culture Reagents
- Test Controls and Calibrators
- Automated Pyrogen Testing Systems
- High Throughput Screening Systems
- Integrated Laboratory Platforms
- In Vitro Pyrogen Testing Kits
-
Pyrogen Testing Market Segmented by Technology:
- In Vitro Testing Technology
- Cell Based Assay Technology
- Monocyte Activation Technology
- Biochemical Detection Technology
- Limulus Amebocyte Lysate Technology
- Recombinant Factor C Technology
- In Vivo Testing Technology
- Animal Based Pyrogen Testing
- Rabbit Test Methodology
- Automation Technology
- Robotic Sample Handling Systems
- Automated Detection Platforms
- In Vitro Testing Technology
-
Pyrogen Testing Market Segmented by Method:
- LAL Test Method
- Gel Clot Method
- Turbidimetric Method
- Chromogenic Method
- MAT Method
- Human Blood Based Monocyte Activation Test
- Cell Mediated Immune Response Testing
- Rabbit Test Method
- Temperature Rise Based Detection
- In Vivo Biological Response Testing
- Recombinant Protein Method
- rFC Based Endotoxin Detection
- Fluorescence Based Detection
- LAL Test Method
-
Pyrogen Testing Market Segmented by Application:
- Pharmaceutical Manufacturing
- Vaccine Production Testing
- Injectable Drug Testing
- Biologics Quality Control
- Medical Device Testing
- Implantable Device Testing
- Parenteral Device Validation
- Biotechnology Research
- Cell Therapy Development
- Gene Therapy Research
- Food and Beverage Testing
- Quality Assurance Testing
- Novel Ingredient Safety Testing
- Pharmaceutical Manufacturing
-
Pyrogen Testing Market Segmented by End Use:
- Pharmaceutical Companies
- Multinational Pharma Manufacturers
- Generic Drug Manufacturers
- Biotechnology Companies
- Contract Biotech Developers
- Research Based Biotech Firms
- Contract Research Organizations
- Preclinical Testing Providers
- Regulatory Compliance Labs
- Medical Device Manufacturers
- Diagnostic Laboratories
- Pharmaceutical Companies
-
Pyrogen Testing Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
How big is the pyrogen testing market in 2026?
The global pyrogen testing market is estimated to be valued at USD 1.7 billion in 2026.
What will be the size of pyrogen testing market in 2036?
The market size for the pyrogen testing market is projected to reach USD 4.9 billion by 2036.
How much will be the pyrogen testing market growth between 2026 and 2036?
The pyrogen testing market is expected to grow at a 11.4% CAGR between 2026 and 2036.
What are the key product types in the pyrogen testing market?
The key product types in pyrogen testing market are in vitro pyrogen testing kits, in vivo pyrogen testing systems, recombinant factor c assays, limulus amebocyte lysate assays, consumables and reagents and automated pyrogen testing systems.
Which technology segment is expected to contribute significant share in the pyrogen testing market in 2026?
In terms of technology, in vitro testing technology segment is expected to command 41.3% share in the pyrogen testing market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- In Vitro Pyrogen Testing Kits
- Monocyte Activation Test Kits
- Endotoxin Detection Kits
- Biological Assay Based Kits
- In Vivo Pyrogen Testing Systems
- Rabbit Pyrogen Test Systems
- Laboratory Animal Test Systems
- Recombinant Factor C Assays
- Chromogenic rFC Assays
- Fluorometric rFC Assays
- Limulus Amebocyte Lysate Assays
- Gel Clot LAL Assays
- Turbidimetric LAL Assays
- Chromogenic LAL Assays
- Consumables and Reagents
- Endotoxin Standards
- Cell Culture Reagents
- Test Controls and Calibrators
- Automated Pyrogen Testing Systems
- High Throughput Screening Systems
- Integrated Laboratory Platforms
- In Vitro Pyrogen Testing Kits
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- In Vitro Testing Technology
- Cell Based Assay Technology
- Monocyte Activation Technology
- Biochemical Detection Technology
- Limulus Amebocyte Lysate Technology
- Recombinant Factor C Technology
- In Vivo Testing Technology
- Animal Based Pyrogen Testing
- Rabbit Test Methodology
- Automation Technology
- Robotic Sample Handling Systems
- Automated Detection Platforms
- In Vitro Testing Technology
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Method
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Method, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Method, 2026 to 2036
- LAL Test Method
- Gel Clot Method
- Turbidimetric Method
- Chromogenic Method
- MAT Method
- Human Blood Based Monocyte Activation Test
- Cell Mediated Immune Response Testing
- Rabbit Test Method
- Temperature Rise Based Detection
- In Vivo Biological Response Testing
- Recombinant Protein Method
- rFC Based Endotoxin Detection
- Fluorescence Based Detection
- LAL Test Method
- Y to o to Y Growth Trend Analysis By Method, 2021 to 2025
- Absolute $ Opportunity Analysis By Method, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Pharmaceutical Manufacturing
- Vaccine Production Testing
- Injectable Drug Testing
- Biologics Quality Control
- Medical Device Testing
- Implantable Device Testing
- Parenteral Device Validation
- Biotechnology Research
- Cell Therapy Development
- Gene Therapy Research
- Food and Beverage Testing
- Quality Assurance Testing
- Novel Ingredient Safety Testing
- Pharmaceutical Manufacturing
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical Companies
- Multinational Pharma Manufacturers
- Generic Drug Manufacturers
- Biotechnology Companies
- Contract Biotech Developers
- Research Based Biotech Firms
- Contract Research Organizations
- Preclinical Testing Providers
- Regulatory Compliance Labs
- Medical Device Manufacturers
- Diagnostic Laboratories
- Pharmaceutical Companies
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Technology
- By Method
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Technology
- By Method
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Technology
- By Method
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Technology
- By Method
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Technology
- By Method
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Technology
- By Method
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Technology
- By Method
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Technology
- By Method
- By Application
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Technology
- By Method
- By Application
- By End Use
- Competition Analysis
- Competition Deep Dive
- Charles River Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Novo Nordisk
- Merz KGaA
- GenScript
- bioMérieux SA
- Lonza Group
- Thermo Fisher Scientific, Inc.
- Seikagaku Biobusiness Corporation
- FUJIFILM Wako Chemicals USA Corporation (Pyrostar)
- MiCAN Technologies Inc.
- Charles River Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Method, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Method, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Method, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Method, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Method, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Method, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Method, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Method, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Technology
- Figure 9: Global Market Value Share and BPS Analysis by Method, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Method, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Method
- Figure 12: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Application
- Figure 15: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by End Use
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Product
- Figure 32: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Technology
- Figure 35: North America Market Value Share and BPS Analysis by Method, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Method, 2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Method
- Figure 38: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by Application
- Figure 41: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by End Use
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Product
- Figure 48: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by Technology
- Figure 51: Latin America Market Value Share and BPS Analysis by Method, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Method, 2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Method
- Figure 54: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Application
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product
- Figure 64: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 66: Western Europe Market Attractiveness Analysis by Technology
- Figure 67: Western Europe Market Value Share and BPS Analysis by Method, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Method, 2026 to 2036
- Figure 69: Western Europe Market Attractiveness Analysis by Method
- Figure 70: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Application
- Figure 73: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by End Use
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Method, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Method, 2026 to 2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Method
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Application
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 95: East Asia Market Attractiveness Analysis by Product
- Figure 96: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 98: East Asia Market Attractiveness Analysis by Technology
- Figure 99: East Asia Market Value Share and BPS Analysis by Method, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Method, 2026 to 2036
- Figure 101: East Asia Market Attractiveness Analysis by Method
- Figure 102: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 104: East Asia Market Attractiveness Analysis by Application
- Figure 105: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 107: East Asia Market Attractiveness Analysis by End Use
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Method, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Method, 2026 to 2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Method
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026 to 2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Method, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Method, 2026 to 2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Method
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis