Allergy Clinical Trial Services Market Size, Share, Growth and Forecast (2026 - 2036)
Allergy Clinical Trial Services Market Size and Share Forecast Outlook 2026 to 2036
Allergy clinical trial services are witnessing accelerated market expansion across forecast horizons, with valuation anticipated to climb from USD 1.4 billion in 2026 to USD 2.9 billion by 2036, exhibiting sustained CAGR of 7.4%. By service line, Clinical operations and monitoring account for 24.0% share. By trial focus indication, Allergic rhinitis captures 24.0% of market share.
Allergy Clinical Trial Services Market
| Metric | Value |
|---|---|
| Estimated Value in (2026E) | USD 1.4 billion |
| Forecast Value in (2036F) | USD 2.9 billion |
| Forecast CAGR (2026 to 2036) | 7.4 % |
Category
| Category | Segments |
|---|---|
| Service Line | Study design & protocol development, Site management & patient recruitment, Clinical operations & monitoring, Bioanalytical & immunoassay services, Data management & biostatistics, Regulatory & medical writing |
| Trial Focus / Indication | Allergic rhinitis, Asthma (allergic), Atopic dermatitis, Food allergy, Urticaria / anaphylaxis |
| End User | Pharma sponsors, Biotech sponsors, Academic medical centers, Device / diagnostic companies, Government / NGO sponsors |
| Region | North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
Segmental Analysis
Why do clinical operations and monitoring services command lead position in allergy trial service lines?
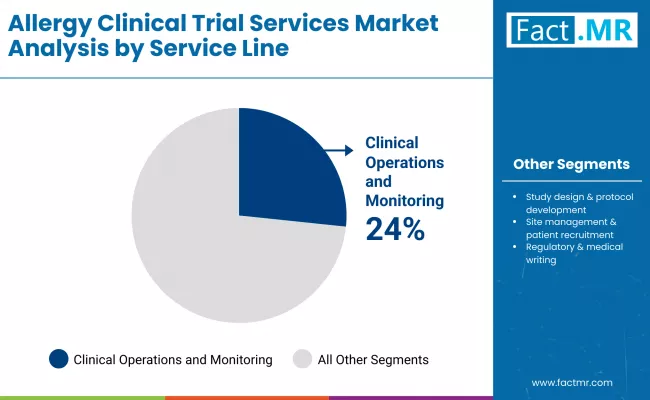
Clinical operations and monitoring account for 24.0% share because allergic reactions require intensive real-time oversight that exceeds standard trial monitoring protocols. Hypersensitivity manifestations can escalate rapidly from mild symptoms to anaphylaxis, necessitating on-site medical personnel during allergen exposure challenges and continuous safety surveillance throughout treatment phases. Food allergy trials conducting oral immunotherapy require medically supervised dose escalations where participants consume increasing allergen quantities under emergency-equipped conditions, creating labor-intensive monitoring requirements. Corporate investment reflects strategic positioning: CROs deploy specialized allergy monitoring platforms combining electronic adverse event capture with immediate clinician notification systems, establishing premium pricing through safety infrastructure that prevents trial interruptions while maintaining participant protection standards exceeding general therapeutic area requirements.
How does allergic rhinitis establish dominance within allergy trial focus indications?
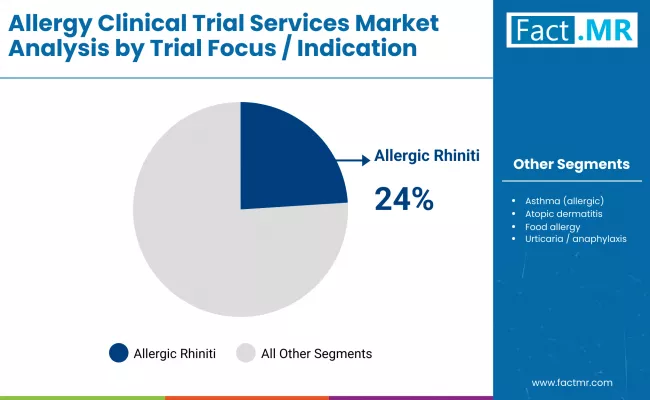
Allergic rhinitis captures 24.0% of trial focus because high disease prevalence creates commercially viable patient populations supporting large-scale efficacy trials across multiple geographies. Unlike food allergies requiring carefully controlled exposure protocols, seasonal rhinitis enables natural allergen challenges during pollen seasons, reducing protocol complexity while maintaining clinical relevance. Market research demonstrates that sublingual immunotherapy tablets targeting grass, ragweed, and dust mite allergens generate sustained pharmaceutical investment due to favorable regulatory pathways and chronic treatment models supporting recurring revenue. Trial availability spans early-phase dose-finding through post-marketing surveillance, creating multiple engagement points for CRO services matching sponsor development stage and regulatory requirements across regional health authorities.
Market Dynamics
- Drivers: Rising pediatric food allergy prevalence has intensified beyond traditional allergen avoidance strategies toward active desensitization protocols. Educational campaigns from allergy advocacy organizations and ingredient-focused parental communities have elevated understanding of multi-allergen sensitization patterns, oral immunotherapy mechanisms, and sustained unresponsiveness endpoints. This knowledge expansion creates demand for transformative interventions rather than symptom management approaches. Simultaneously, regulatory innovation in needle-free delivery and home-use epinephrine systems are democratizing access to emergency treatments previously confined to injectable formats, expanding addressable trial populations beyond procedurally comfortable participants to anxiety-driven avoiders seeking alternative administration routes. FDA approval of neffy nasal spray has accelerated investment in human factors trials and comparative bioavailability studies, providing regulatory pathways that encourage innovation in patient-centered delivery technologies.
- Restraints: Patient recruitment challenges create trial timeline extensions and enrollment failures across specialized allergic populations. Geographic variation in allergen exposure patterns limits site viability for seasonal rhinitis trials, while competition for pediatric food allergy participants intensifies across simultaneous studies targeting identical sensitization profiles. This recruitment bottleneck drives premature study termination before achieving statistical power, exemplified by multiple grass pollen immunotherapy trials abandoned due to insufficient enrollment. Additionally, biomarker validation complexity continues expanding with mechanistic endpoints including allergen-specific IgG4 ratios and regulatory T cell populations requiring extensive preliminary evidence demonstrating clinical relevance before regulatory acceptance, limiting innovation adoption beyond traditional symptom scores that lack differentiation capacity for disease-modifying therapies.
- Trend 1: Component-Resolved Diagnostic Integration Trial design is shifting from crude allergen extract testing toward molecular allergen component identification that addresses specific protein epitopes driving patient reactions. This precision diagnostics evolution enables targeted immunotherapy selection matching individual sensitization patterns rather than population-level allergen exposure assumptions. CROs incorporating component-resolved testing platforms differentiate through mechanistic patient stratification supporting personalized protocol adaptations, positioning early adopters for premium trial segments where sponsors invest in biomarker-guided development over empirical dose escalation approaches.
- Trend 2: Artificial Intelligence Patient Recruitment Machine learning algorithms analyzing electronic health records are accelerating patient identification for complex allergic phenotype criteria. Natural language processing extracts allergy-relevant information from clinical documentation including specific food reactions and anaphylaxis histories, enabling targeted recruitment outreach that reduces screening failures. CROs investing in AI-powered recruitment platforms demonstrate enrollment timeline reductions approaching 30% compared to traditional site-based identification methods, creating competitive advantages in sponsor selection decisions where trial startup speed influences development program success.
Allergy Clinical Trial Services Market Analysis by Key Country
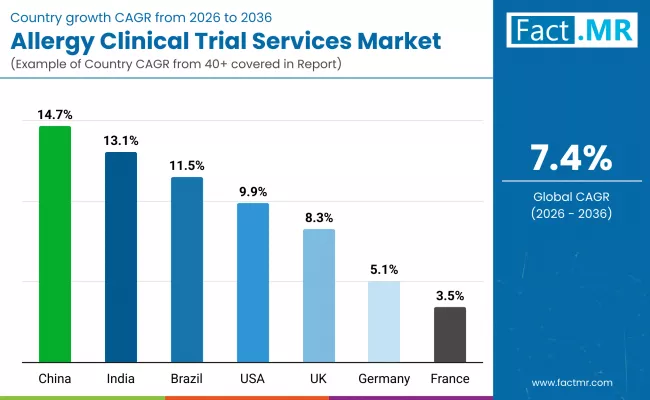
| Country | CAGR (2026-2036) |
| China | 14.7% |
| India | 13.1% |
| Brazil | 11.5% |
| USA | 9.9% |
| UK | 8.3% |
| Germany | 5.1% |
| France | 3.5% |
What factors drive China to lead regional expansion in allergy clinical trial services?
China leads regional growth at 14.7 percent CAGR following NMPA implementation of the 30-day IND pathway for innovative drugs in 2025, reducing clinical startup timelines by 50 percent compared to previous 60-day standards. This regulatory acceleration dramatically shortened the interval from protocol submission to first patient enrollment, making China increasingly attractive for first-in-human trials and early-phase allergy therapeutic development. The pathway applies to Category I innovative drugs including biologics, small molecules, and traditional Chinese medicines addressing allergic conditions, with eligibility extending to nationally prioritized research programs and globally synchronized clinical trials. China maintains approximately one-third of the global new drug pipeline and initiated 39 percent of worldwide oncology trials in 2024, demonstrating infrastructure and expertise transferable to allergy research and creating substantial opportunity for service providers with regional regulatory knowledge.
How do regulatory frameworks and clinical infrastructure position India for growth?
India achieves significant expansion at 13.1 percent CAGR through implementation of New Drugs and Clinical Trials Rules 2019 and subsequent 2025 amendments strengthening ethical oversight and data integrity standards. These regulatory enhancements increased global biopharma confidence in Indian trial quality, prompting companies including Novartis and Bristol Myers Squibb to double clinical site footprints within the country by 2026. The massive patient population provides access to treatment-naive subjects and diverse genetic backgrounds enabling pharmacogenomics analyses, while cost advantages relative to Western markets reduce per-patient enrollment expenses without compromising quality.
How is Brazil emerging within allergy clinical trial services?
Brazil exhibits robust market expansion at 11.5 percent CAGR, driven by ANVISA regulatory reforms streamlining clinical trial approval processes. These reforms increased registration of Phase III trials for respiratory allergies and infectious disease-linked allergic conditions, positioning Brazil for greater participation in multinational development programs. Rapid urbanization and industrial growth have elevated asthma and allergic rhinitis rates, creating large patient populations for recruitment while environmental pollutant exposure patterns enable studies examining pollution-allergy interactions. The combination of regulatory agility, growing disease burden, and cost-competitive trial execution relative to North American and European markets positions Brazil for sustained growth in Phase IV post-marketing surveillance and observational studies.
What accounts for the United States achieving the highest regional growth rate in allergy clinical trial services?
The United States demonstrates exceptional expansion at 9.9 percent CAGR, propelled by FDA regulatory focus on non-injectable emergency treatments and precision medicine initiatives. FDA guidance emphasizing patient-centered endpoints and real-world evidence has created demand for novel trial designs incorporating telemedicine-enabled monitoring and patient-reported outcome measures. Rising pediatric food allergy prevalence, particularly multi-allergen sensitivities, drives investment in oral immunotherapy protocols requiring extensive safety monitoring across multiple treatment phases, substantially expanding the scope of clinical services required per study.
How does United Kingdom regulatory innovation support in allergy trial services?
United Kingdom achieves notable expansion at 8.3 percent CAGR through MHRA introduction of 14-day Phase I assessment procedures in early 2026, creating fast-track routes for early-phase research and development. This regulatory acceleration increased healthy volunteer trials by 16 percent, positioning the UK for first-choice consideration in dose-finding studies and initial safety evaluations for novel allergy therapeutics. Imperial College National Heart and Lung Institute has advanced pioneering research including virus-like particle technology for peanut allergy vaccines, demonstrating UK academic leadership in innovative immunotherapy approaches.
How do Germany-specific regulatory requirements influence its growth trajectory?
Germany exhibits steady expansion at 5.1 percent CAGR, anchored by Therapy Allergen Ordinance programs requiring rigorous registration data for allergy immunotherapies. The Paul-Ehrlich-Institute oversees comprehensive review processes demanding pivotal Phase III evidence for sublingual and subcutaneous immunotherapy products, creating sustained demand for large-scale efficacy trials. Companies including Allergy Therapeutics conduct multi-year studies evaluating grass pollen allergen extracts and other seasonal allergen formulations to satisfy German regulatory standards, which influence broader European approval strategies. The established nature of Germany allergy immunotherapy market creates competitive pressure for incremental improvements rather than breakthrough innovations, moderating growth rates while maintaining stable trial volumes for companies seeking market access in Europe largest economy.
What factors contribute to France exhibiting the most modest regional growth?
France demonstrates the slowest regional expansion at 3.5 percent CAGR, reflecting mature pharmaceutical infrastructure with limited recent regulatory innovation specific to allergy trials. The country maintains established clinical research capabilities through academic medical centers and contract research organizations, but has not implemented accelerated pathways comparable to UK MHRA reforms or emerging market regulatory streamlining. Competition from other European Union member states offering faster approval timelines or cost advantages has moderated France share of multinational trial activity, while domestic pharmaceutical focus on oncology and rare diseases limits local sponsor investment in allergy therapeutic development. Despite slower growth, France retains importance for European registration trials requiring multi-country participation and contributes specialized expertise in pediatric allergology through centers of excellence in Paris and Lyon.
Competitive Landscape
IQVIA maintains market leadership through comprehensive service portfolios spanning bioanalytical capabilities, extensive global site networks, and specialized regulatory expertise in immunology and allergic disease trials. Company leverages integrated technology platforms connecting electronic data capture systems, patient recruitment databases, and real-time monitoring dashboards providing sponsors with visibility across multi-regional allergy studies. IQVIA bioanalytical laboratories offer specialized immunoassay capabilities including allergen-specific IgE testing, component-resolved diagnostics, and cytokine profiling required for mechanistic allergy research. Organization maintains therapeutic area centers of excellence staffed by allergists and immunologists advising on protocol design, endpoint selection, and regulatory strategy, differentiating IQVIA from generalist CROs lacking allergy-specific expertise. Strategic acquisitions expanded capabilities in real-world evidence generation and patient registry development, enabling post-approval studies examining long-term outcomes of allergy immunotherapies in routine clinical practice settings.
ICON competes through specialized pediatric research capabilities particularly relevant to food allergy and atopic dermatitis trials predominantly affecting children. Company operates dedicated pediatric units within clinical research sites, employing staff trained in child-friendly recruitment techniques and age-appropriate consent procedures required for studies involving minor subjects. ICON developed expertise managing complex food challenge protocols exposing allergic children to incremental allergen doses under medical supervision, requiring detailed safety monitoring and immediate access to emergency medications.
Recent Developments
- In February 2025, researchers at Imperial College National Heart and Lung Institute published Phase 1 first-in-human trial results for a novel peanut allergy vaccine in the Journal of Allergy and Clinical Immunology.
- In September 2024, DBV Technologies announced completion of patient screening for the Phase 3 VITESSE trial evaluating Viaskin Peanut Immunotherapy. This immunotherapy approach delivers allergen through a skin patch, representing an alternative to oral immunotherapy that avoids gastrointestinal side effects.
Key Players in in Allergy Clinical Trial Services Market
- IQVIA
- ICON
- Parexel
- Syneos Health
- Labcorp Drug Development
Bibliography
- U.S. Food and Drug Administration. 2024. Guidance for Industry: Allergic Rhinitis Clinical Development Programs for Drug Products. FDA Center for Drug Evaluation and Research.
- National Medical Products Administration (NMPA). 2025. Technical guidelines for clinical trial approval: 30-day IND pathway for innovative drugs in allergic disease treatment.
- European Medicines Agency. 2023. Guideline on clinical development of products for specific immunotherapy for treatment of allergic diseases. EMA Committee for Medicinal Products for Human Use.
- Journal of Allergy and Clinical Immunology. 2025. Phase 1 first-in-human trial results for peanut allergy vaccine employing virus-like particle technology. Imperial College National Heart and Lung Institute.
- Centers for Disease Control and Prevention. 2024. Food Allergy Among U.S. Children: Trends in Prevalence and Hospitalizations. National Center for Health Statistics.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 1.4 Billion |
| Service Line | Study design & protocol development, Site management & patient recruitment, Clinical operations & monitoring, Bioanalytical & immunoassay services, Data management & biostatistics, Regulatory & medical writing |
| Trial Focus / Indication | Allergic rhinitis, Asthma (allergic), Atopic dermatitis, Food allergy, Urticaria / anaphylaxis |
| End User | Pharma sponsors, Biotech sponsors, Academic medical centers, Device / diagnostic companies, Government / NGO sponsors |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | U.S., Canada, Germany, Italy, UK, France, Japan, Australia, and other countries |
| Key Companies Profiled | IQVIA, ICON, Parexel, Syneos Health, Labcorp Drug Development |
| Additional Attributes | Dollar revenue in the allergy clinical trial services market is segmented by trial phase, allergy type, study design, service type, end user, price tier, and region. Regional demand patterns are driven by rising prevalence of allergic and immunologic disorders, increasing development of biologics and immunotherapies, stricter regulatory requirements for safety and efficacy data, and growing outsourcing of complex clinical trials to specialized CROs with expertise in allergy focused study design and patient recruitment. |
Allergy Clinical Trial Services Market by Segments
-
Service Line :
- Study design & protocol development
- Site management & patient recruitment
- Clinical operations & monitoring
- Bioanalytical & immunoassay services
- Data management & biostatistics
- Regulatory & medical writing
-
Trial Focus / Indication :
- Allergic rhinitis
- Asthma (allergic)
- Atopic dermatitis
- Food allergy
- Urticaria / anaphylaxis
-
End User :
- Pharma sponsors
- Biotech sponsors
- Academic medical centers
- Device / diagnostic companies
- Government / NGO sponsors
-
Region :
- Asia Pacific
- Japan
- South Korea
- China
- India
- Australia
- New Zealand
- Rest of Asia Pacific
- North America
- United States
- Canada
- Mexico
- Europe
- France
- Italy
- Spain
- Germany
- UK
- Rest of Europe
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Asia Pacific
- Frequently Asked Questions -
How big is the global allergy clinical trial services market in 2026?
The market is valued at approximately USD 1.4 billion in 2026.
What will be the size of the allergy clinical trial services market in 2036?
The market is projected to reach about USD 2.9 billion by 2036.
How fast will the allergy clinical trial services market grow between 2026 and 2036?
The market is expected to grow at a CAGR of 7.4% during the forecast period.
Which service line dominates the allergy clinical trial services market?
Clinical operations and monitoring services lead with a 24.0% share in 2026.
Which trial focus represents the largest indication segment?
Allergic rhinitis dominates, accounting for 24.0% of trial focus in clinical development programs.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual Reports and Clinical Pipelines
- Peer-reviewed Journals and Academic Literature
- Regulatory Agency Guidelines and Filings
- Clinical Trial Registries
- Investor Presentations and Earnings Briefs
- Industry White Papers and Technical Notes
- Trade Journals and Analyst Reports
- Conference Proceedings and Scientific Congresses
- Government Health Statistics
- Press Releases and Media Coverage
- FMI Internal Knowledge Base
- Paid Databases and Subscription Sources
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Expert Interviews
- Quantitative Surveys
- Hybrid Research Approach
- Why Primary Evidence is Used
- Field Techniques
- In-depth Interviews
- Structured Surveys
- Focus Groups
- Observational Research
- Stakeholder Universe Engaged
- CRO Executives
- Pharmaceutical Sponsors
- Biotechnology Firms
- Clinical Investigators
- Allergists and Immunologists
- Regulatory Experts
- Governance, Ethics, and Data Stewardship
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Validation
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Scenario Forecast
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Value Chain Analysis
- PESTLE and Porter’s Five Forces Analysis
- Regulatory Landscape
- Market Dynamics
- Global Allergy Clinical Trial Services Market Analysis 2021-2025 and Forecast 2026-2036
- Historical Market Size (USD Billion), 2021-2025
- Market Forecast (USD Billion), 2026-2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021-2036
- Global Market Analysis by Service Line
- Study Design & Protocol Development
- Site Management & Patient Recruitment
- Clinical Operations & Monitoring
- Bioanalytical & Immunoassay Services
- Data Management & Biostatistics
- Regulatory & Medical Writing
- Global Market Analysis by Trial Focus / Indication
- Allergic Rhinitis
- Allergic Asthma
- Atopic Dermatitis
- Food Allergy
- Urticaria / Anaphylaxis
- Global Market Analysis by End User
- Pharma Sponsors
- Biotech Sponsors
- Academic Medical Centers
- Device / Diagnostic Companies
- Government & NGO Sponsors
- Global Market Analysis by Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
- Key Country Market Analysis
- United States
- China
- India
- Brazil
- United Kingdom
- Germany
- France
- Market Structure Analysis
- Competition Dashboard
- Market Share Analysis
- Benchmarking of Leading CROs
- Competitive Landscape
- IQVIA - Company Profile
- ICON - Company Profile
- Parexel - Company Profile
- Syneos Health - Company Profile
- Labcorp Drug Development - Company Profile
- Assumptions & Acronyms Used
List Of Table
- Allergy Clinical Trial Services Market Key Takeaways
- Global Allergy Clinical Trial Services Market Size (USD Billion), 2021 to 2036
- Global Allergy Clinical Trial Services Market Y-o-Y Growth (%), 2021 to 2036
- Global Allergy Clinical Trial Services Market Absolute $ Opportunity Analysis (USD Billion), 2026 to 2036
- Global Allergy Clinical Trial Services Market Pricing Analysis (USD/Study), 2021 to 2036
- Global Allergy Clinical Trial Services Market Size (USD Billion) By Service Line, 2021 to 2036
- Global Allergy Clinical Trial Services Market Size (USD Billion) By Trial Focus / Indication, 2021 to 2036
- Global Allergy Clinical Trial Services Market Size (USD Billion) By End User, 2021 to 2036
- Global Allergy Clinical Trial Services Market Size (USD Billion) By Region, 2021 to 2036
- North America Allergy Clinical Trial Services Market Size (USD Billion) By Country, 2021 to 2036
- North America Allergy Clinical Trial Services Market Size (USD Billion) By Service Line, 2021 to 2036
- North America Allergy Clinical Trial Services Market Size (USD Billion) By Trial Focus / Indication, 2021 to 2036
- North America Allergy Clinical Trial Services Market Size (USD Billion) By End User, 2021 to 2036
- North America Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- North America Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- North America Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- North America Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Latin America Allergy Clinical Trial Services Market Size (USD Billion) By Country, 2021 to 2036
- Latin America Allergy Clinical Trial Services Market Size (USD Billion) By Service Line, 2021 to 2036
- Latin America Allergy Clinical Trial Services Market Size (USD Billion) By Trial Focus / Indication, 2021 to 2036
- Latin America Allergy Clinical Trial Services Market Size (USD Billion) By End User, 2021 to 2036
- Latin America Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- Latin America Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- Latin America Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- Latin America Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Western Europe Allergy Clinical Trial Services Market Size (USD Billion) By Country, 2021 to 2036
- Western Europe Allergy Clinical Trial Services Market Size (USD Billion) By Service Line, 2021 to 2036
- Western Europe Allergy Clinical Trial Services Market Size (USD Billion) By Trial Focus / Indication, 2021 to 2036
- Western Europe Allergy Clinical Trial Services Market Size (USD Billion) By End User, 2021 to 2036
- Western Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- Western Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- Western Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- Western Europe Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Eastern Europe Allergy Clinical Trial Services Market Size (USD Billion) By Country, 2021 to 2036
- Eastern Europe Allergy Clinical Trial Services Market Size (USD Billion) By Service Line, 2021 to 2036
- Eastern Europe Allergy Clinical Trial Services Market Size (USD Billion) By Trial Focus / Indication, 2021 to 2036
- Eastern Europe Allergy Clinical Trial Services Market Size (USD Billion) By End User, 2021 to 2036
- Eastern Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- Eastern Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- Eastern Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- Eastern Europe Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- East Asia Allergy Clinical Trial Services Market Size (USD Billion) By Country, 2021 to 2036
- East Asia Allergy Clinical Trial Services Market Size (USD Billion) By Service Line, 2021 to 2036
- East Asia Allergy Clinical Trial Services Market Size (USD Billion) By Trial Focus / Indication, 2021 to 2036
- East Asia Allergy Clinical Trial Services Market Size (USD Billion) By End User, 2021 to 2036
- East Asia Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- East Asia Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- East Asia Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- East Asia Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- South Asia and Pacific Allergy Clinical Trial Services Market Size (USD Billion) By Country, 2021 to 2036
- South Asia and Pacific Allergy Clinical Trial Services Market Size (USD Billion) By Service Line, 2021 to 2036
- South Asia and Pacific Allergy Clinical Trial Services Market Size (USD Billion) By Trial Focus / Indication, 2021 to 2036
- South Asia and Pacific Allergy Clinical Trial Services Market Size (USD Billion) By End User, 2021 to 2036
- South Asia and Pacific Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- South Asia and Pacific Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- South Asia and Pacific Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- South Asia and Pacific Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Middle East & Africa Allergy Clinical Trial Services Market Size (USD Billion) By Country, 2021 to 2036
- Middle East & Africa Allergy Clinical Trial Services Market Size (USD Billion) By Service Line, 2021 to 2036
- Middle East & Africa Allergy Clinical Trial Services Market Size (USD Billion) By Trial Focus / Indication, 2021 to 2036
- Middle East & Africa Allergy Clinical Trial Services Market Size (USD Billion) By End User, 2021 to 2036
- Middle East & Africa Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- Middle East & Africa Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- Middle East & Africa Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- Middle East & Africa Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Allergy Clinical Trial Services Market Competition Dashboard
- Allergy Clinical Trial Services Market Competition Benchmarking Analysis
- Allergy Clinical Trial Services Market Share Analysis of Top Players By Regional
- Allergy Clinical Trial Services Market Share Analysis of Top Players By Service Line
- Allergy Clinical Trial Services Market Share Analysis of Top Players By Trial Focus / Indication
- Allergy Clinical Trial Services Market Share Analysis of Top Players By End User
- IQVIA - Company Profile
- ICON - Company Profile
- Parexel - Company Profile
- Syneos Health - Company Profile
- Labcorp Drug Development - Company Profile
- Assumptions & Acronyms Used in Allergy Clinical Trial Services Market Report
List Of Figures
- Allergy Clinical Trial Services Market Size (USD Billion), 2021 to 2036
- Allergy Clinical Trial Services Market Y-o-Y Growth (%), 2021 to 2036
- Allergy Clinical Trial Services Market Absolute $ Opportunity Analysis (USD Billion), 2026 to 2036
- Allergy Clinical Trial Services Market Value Chain Analysis
- Allergy Clinical Trial Services Market Supply Chain Analysis
- Allergy Clinical Trial Services Market Investment Feasibility Matrix
- Allergy Clinical Trial Services Market PESTLE Analysis
- Allergy Clinical Trial Services Market Porter’s Five Forces Analysis
- Allergy Clinical Trial Services Market Product Life Cycle Analysis
- Allergy Clinical Trial Services Market Opportunity Map Analysis
- Allergy Clinical Trial Services Market Scenario Forecast Analysis
- Allergy Clinical Trial Services Market Production and Consumption Statistics
- Allergy Clinical Trial Services Market Import and Export Statistics
- Global Allergy Clinical Trial Services Market Share (%) By Service Line, 2026
- Global Allergy Clinical Trial Services Market Share (%) By Service Line, 2036
- Global Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2026
- Global Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2036
- Global Allergy Clinical Trial Services Market Share (%) By End User, 2026
- Global Allergy Clinical Trial Services Market Share (%) By End User, 2036
- Global Allergy Clinical Trial Services Market Share (%) By Region, 2026
- Global Allergy Clinical Trial Services Market Share (%) By Region, 2036
- Global Allergy Clinical Trial Services Market Attractiveness Analysis By Region
- North America Allergy Clinical Trial Services Market Size (USD Billion), 2021 to 2036
- North America Allergy Clinical Trial Services Market Share (%) By Country, 2026
- North America Allergy Clinical Trial Services Market Share (%) By Country, 2036
- North America Allergy Clinical Trial Services Market Share (%) By Service Line, 2026
- North America Allergy Clinical Trial Services Market Share (%) By Service Line, 2036
- North America Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2026
- North America Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2036
- North America Allergy Clinical Trial Services Market Share (%) By End User, 2026
- North America Allergy Clinical Trial Services Market Share (%) By End User, 2036
- North America Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- North America Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- North America Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- North America Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Latin America Allergy Clinical Trial Services Market Size (USD Billion), 2021 to 2036
- Latin America Allergy Clinical Trial Services Market Share (%) By Country, 2026
- Latin America Allergy Clinical Trial Services Market Share (%) By Country, 2036
- Latin America Allergy Clinical Trial Services Market Share (%) By Service Line, 2026
- Latin America Allergy Clinical Trial Services Market Share (%) By Service Line, 2036
- Latin America Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2026
- Latin America Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2036
- Latin America Allergy Clinical Trial Services Market Share (%) By End User, 2026
- Latin America Allergy Clinical Trial Services Market Share (%) By End User, 2036
- Latin America Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- Latin America Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- Latin America Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- Latin America Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Western Europe Allergy Clinical Trial Services Market Size (USD Billion), 2021 to 2036
- Western Europe Allergy Clinical Trial Services Market Share (%) By Country, 2026
- Western Europe Allergy Clinical Trial Services Market Share (%) By Country, 2036
- Western Europe Allergy Clinical Trial Services Market Share (%) By Service Line, 2026
- Western Europe Allergy Clinical Trial Services Market Share (%) By Service Line, 2036
- Western Europe Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2026
- Western Europe Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2036
- Western Europe Allergy Clinical Trial Services Market Share (%) By End User, 2026
- Western Europe Allergy Clinical Trial Services Market Share (%) By End User, 2036
- Western Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- Western Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- Western Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- Western Europe Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Eastern Europe Allergy Clinical Trial Services Market Size (USD Billion), 2021 to 2036
- Eastern Europe Allergy Clinical Trial Services Market Share (%) By Country, 2026
- Eastern Europe Allergy Clinical Trial Services Market Share (%) By Country, 2036
- Eastern Europe Allergy Clinical Trial Services Market Share (%) By Service Line, 2026
- Eastern Europe Allergy Clinical Trial Services Market Share (%) By Service Line, 2036
- Eastern Europe Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2026
- Eastern Europe Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2036
- Eastern Europe Allergy Clinical Trial Services Market Share (%) By End User, 2026
- Eastern Europe Allergy Clinical Trial Services Market Share (%) By End User, 2036
- Eastern Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- Eastern Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- Eastern Europe Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- Eastern Europe Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- East Asia Allergy Clinical Trial Services Market Size (USD Billion), 2021 to 2036
- East Asia Allergy Clinical Trial Services Market Share (%) By Country, 2026
- East Asia Allergy Clinical Trial Services Market Share (%) By Country, 2036
- East Asia Allergy Clinical Trial Services Market Share (%) By Service Line, 2026
- East Asia Allergy Clinical Trial Services Market Share (%) By Service Line, 2036
- East Asia Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2026
- East Asia Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2036
- East Asia Allergy Clinical Trial Services Market Share (%) By End User, 2026
- East Asia Allergy Clinical Trial Services Market Share (%) By End User, 2036
- East Asia Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- East Asia Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- East Asia Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- East Asia Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- South Asia and Pacific Allergy Clinical Trial Services Market Size (USD Billion), 2021 to 2036
- South Asia and Pacific Allergy Clinical Trial Services Market Share (%) By Country, 2026
- South Asia and Pacific Allergy Clinical Trial Services Market Share (%) By Country, 2036
- South Asia and Pacific Allergy Clinical Trial Services Market Share (%) By Service Line, 2026
- South Asia and Pacific Allergy Clinical Trial Services Market Share (%) By Service Line, 2036
- South Asia and Pacific Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2026
- South Asia and Pacific Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2036
- South Asia and Pacific Allergy Clinical Trial Services Market Share (%) By End User, 2026
- South Asia and Pacific Allergy Clinical Trial Services Market Share (%) By End User, 2036
- South Asia and Pacific Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- South Asia and Pacific Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- South Asia and Pacific Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- South Asia and Pacific Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Middle East & Africa Allergy Clinical Trial Services Market Size (USD Billion), 2021 to 2036
- Middle East & Africa Allergy Clinical Trial Services Market Share (%) By Country, 2026
- Middle East & Africa Allergy Clinical Trial Services Market Share (%) By Country, 2036
- Middle East & Africa Allergy Clinical Trial Services Market Share (%) By Service Line, 2026
- Middle East & Africa Allergy Clinical Trial Services Market Share (%) By Service Line, 2036
- Middle East & Africa Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2026
- Middle East & Africa Allergy Clinical Trial Services Market Share (%) By Trial Focus / Indication, 2036
- Middle East & Africa Allergy Clinical Trial Services Market Share (%) By End User, 2026
- Middle East & Africa Allergy Clinical Trial Services Market Share (%) By End User, 2036
- Middle East & Africa Allergy Clinical Trial Services Market Attractiveness Analysis By Country
- Middle East & Africa Allergy Clinical Trial Services Market Attractiveness Analysis By Service Line
- Middle East & Africa Allergy Clinical Trial Services Market Attractiveness Analysis By Trial Focus / Indication
- Middle East & Africa Allergy Clinical Trial Services Market Attractiveness Analysis By End User
- Allergy Clinical Trial Services Market Competitive Landscape
- Allergy Clinical Trial Services Market Structure Analysis
- Allergy Clinical Trial Services Market Competition Dashboard
- Allergy Clinical Trial Services Market Competition Benchmarking Analysis
- Allergy Clinical Trial Services Market Share Analysis of Top Players By Regional
- Allergy Clinical Trial Services Market Share Analysis of Top Players By Service Line
- Allergy Clinical Trial Services Market Share Analysis of Top Players By Trial Focus / Indication
- Allergy Clinical Trial Services Market Share Analysis of Top Players By End User
- IQVIA - Revenue Analysis (USD Billion), 2019-2025
- ICON - Revenue Analysis (USD Billion), 2019-2025
- Parexel - Revenue Analysis (USD Billion), 2019-2025
- Syneos Health - Revenue Analysis (USD Billion), 2019-2025
- Labcorp Drug Development - Revenue Analysis (USD Billion), 2019-2025
- IQVIA - Market Position Analysis
- ICON - Market Position Analysis
- Parexel - Market Position Analysis
- Syneos Health - Market Position Analysis
- Labcorp Drug Development - Market Position Analysis