Biomarker-based Immunoassays Market Size, Share, Growth and Forecast (2026 - 2036)
Biomarker-based Immunoassays Market is segmented by Sample Type (Blood, Tissue, Urine, and Others), Product (Reagents & Kits, Instruments/Analyzers, Services), Biomarker Type (Efficacy & Pharmacodynamic, Predictive/Prognostic, and Others), Disease (Cancer, Cardiovascular, Neurological, and Others), End Use (Hospitals & Clinics, Diagnostic Labs, Research), and Region. Forecast for 2026 to 2036.
The biomarker-based immunoassays market, valued at USD 5.4 billion in 2025, is expected to reach USD 5.7 billion in 2026 and USD 9.6 billion by 2036, reflecting a CAGR of 5.4%, according to Fact MR. Cancer applications are anticipated to hold 63% of the disease segment, while blood samples are projected to capture 65.2% of the sample type segment.
Summary of the Biomarker-based Immunoassays Market
- Market Definition
- The Biomarker-based Immunoassays Market includes global production and trade of diagnostic assays that detect specific biomarkers for disease monitoring, prognosis, and pharmacodynamic studies across clinical, research, and laboratory settings. The market encompasses blood, tissue, and urine-based assays, reagents, kits, analyzers, and associated services.
- Demand Drivers
- Rising Cancer and Chronic Disease Prevalence: Growing global incidence of cancer and chronic illnesses drives the need for reliable biomarker assays.
- Adoption of Personalized Medicine: Immunoassays enable tailored treatment plans through predictive and pharmacodynamic biomarker measurement.
- Advancements in Laboratory Diagnostics: Innovations in high-throughput platforms, multiplex assays, and automation enhance assay efficiency and adoption.
- Early Disease Detection and Monitoring: Increasing demand for precise diagnostic information for early intervention supports market growth.
- Expansion of Healthcare Infrastructure: Growth of hospitals, diagnostic laboratories, and research centers, especially in Asia Pacific, drives assay adoption.
- Key Segments Analyzed
- Sample Type: Blood (65.2%), Tissue, Urine, Others.
- Disease: Cancer (63%), Cardiovascular, Neurological, Others.
- Product: Reagents & Kits, Instruments/Analyzers, Services.
- End-Use Industry: Hospitals & Clinics, Diagnostic Labs, Research.
- Geography: China leads with 7.3% CAGR, followed by India (6.8%), Germany (6.2%), France (5.7%), and the UK (5.1%).
- Analyst Opinion at Fact MR
- Shambhu Nath Jha, Principal Consultant at Fact MR, stated, "The biomarker-based immunoassays market is evolving rapidly, driven by clinical precision demand, personalized therapy adoption, and research focus on advanced biomarker detection for disease monitoring."
- Strategic Implications / Executive Takeaways
- Focus on Cancer Biomarker Panels: Develop and market high-sensitivity assays targeting oncology applications to capture the largest segment.
- Invest in Blood-based Assay Platforms: Blood remains the dominant sample type; optimizing throughput and standardization supports wider clinical adoption.
- Leverage Emerging Markets: Expand distribution and partnerships in China, India, and other rapidly growing healthcare regions.
- Enhance Multiplex and High-Throughput Offerings: Offer integrated platforms that reduce processing time and support precision medicine initiatives.
- Ensure Regulatory Compliance and Quality: Align assays with regional and international standards to facilitate clinical and research adoption.
- Methodology
- Primary Research: Interviews with clinical laboratorians, assay developers, and procurement specialists.
- Desk Research: Data sourced from scientific publications, industry reports, and regulatory filings.
- Market Sizing & Forecasting: Hybrid top-down and bottom-up approach combining historical adoption trends, shipment data, and revenue forecasts.
Segmental Analysis
Biomarker-based Immunoassays Market Analysis by Sample Type
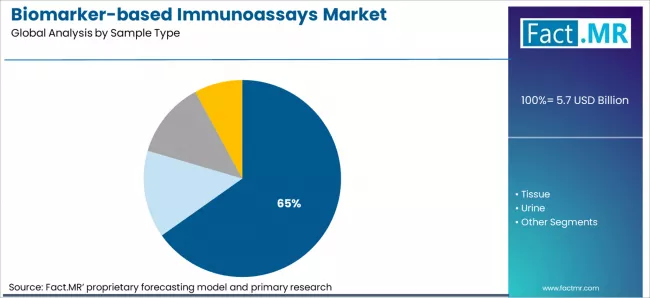
- Market Overview: Blood samples are expected to capture 65.2% of the biomarker-based immunoassays market by 2026. Blood is the most widely used sample type due to its accessibility, high biomarker concentration, and reliability in detecting a range of diseases. Immunoassays using blood samples are commonly employed in clinical diagnostics, drug development, and disease monitoring.
- Demand Drivers:
- Ease of Collection and Standardization: Blood samples can be collected with minimal invasiveness and are highly standardized for clinical testing, supporting consistent assay results.
- Wide Diagnostic Utility: Blood contains a variety of biomarkers, including proteins, antibodies, and metabolites, making it suitable for detecting multiple diseases and conditions.
- Growing Clinical Testing Demand: The increasing focus on early detection, personalized medicine, and routine health monitoring drives the widespread use of blood-based immunoassays.
Biomarker-based Immunoassays Market Analysis by Disease
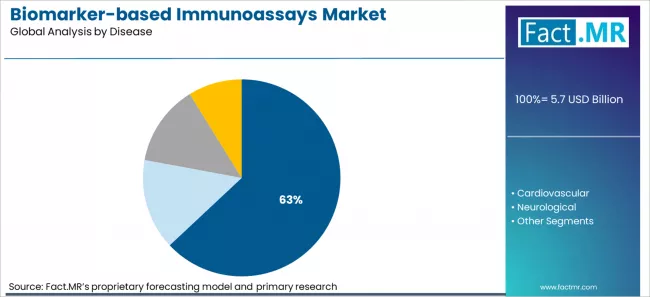
- Market Overview: Cancer is projected to hold 63% of the market share by 2026. Immunoassays for cancer biomarkers are critical for early detection, monitoring disease progression, and evaluating treatment response, making cancer the leading application in this market.
- Demand Drivers:
- Rising Cancer Incidence: The growing prevalence of various cancers globally is driving the demand for reliable biomarker-based immunoassays to enable early diagnosis.
- Focus on Early Detection and Personalized Therapy: Immunoassays help identify specific cancer biomarkers, supporting targeted therapies and improving patient outcomes.
- Advancements in Immunoassay Technologies: Continuous innovation in assay sensitivity, multiplexing, and automation enhances the reliability and efficiency of cancer diagnostics, further boosting market adoption.
Biomarker Based Immunoassays Market Drivers, Restraints, and Opportunities
FMR analysts observe that the biomarker‑based immunoassays market is a growth‑aligned clinical diagnostics segment driven by rising prevalence of chronic diseases, demand for personalized medicine, and need for precise biological measurements in drug development and patient care. Historically, generic immunoassays served basic screening; as biomarker discovery accelerated and clinicians required more specific, actionable data (disease staging, therapy response, prognostics), structural demand emerged for assays that quantify defined proteins, cytokines, and other targets with high sensitivity and specificity. The 2026 market valuation reflects this shift, with adoption tied to routine clinical use, translational research, and companion diagnostics rather than commodity testing alone.
While traditional broad immunoassays remain in use, advanced biomarker‑based kits, multiplex panels, high‑throughput platforms, and standardized ELISA/CLIA systems, are gaining share because they deliver richer clinical insights and support targeted therapy decisions. These premium assay systems command higher per‑test pricing, contributing to net market value growth even as test volumes grow with healthcare access and chronic disease screening programs. The market exists because clinicians and researchers increasingly require validated biomarker data to improve diagnostic accuracy, tailor treatments, and meet regulatory and quality standards.
- Clinical Precision Demand: Physicians and labs adopt biomarker‑specific assays to support diagnosis, prognosis, and therapy monitoring beyond general tests.
- Regulatory & Performance Standards: Clinical and regulatory frameworks that mandate validated biomarkers for disease stratification push uptake of certified immunoassays with defined performance metrics.
- Regional Healthcare Trends: In North America and Europe, strong research infrastructure and reimbursement support advanced biomarker testing, while Asia‑Pacific shows rapid growth as clinical diagnostics access expands.
Regional Analysis
The market analysis covers key global regions, including East Asia, South Asia, and Western Europe. It is segmented geographically, with specific market dynamics for each region. The full report provides a detailed market attractiveness analysis.
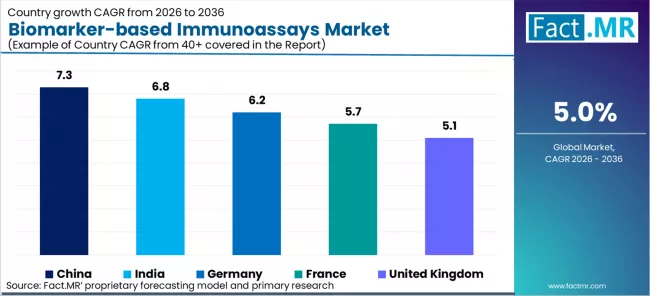
| Country | CAGR (2026-2036) |
|---|---|
| China | 7.3% |
| India | 6.8% |
| Germany | 6.2% |
| France | 5.7% |
| United Kingdom | 5.1% |
Source: Fact MR (FMR) analysis, based on proprietary forecasting model and primary research
East Asia:
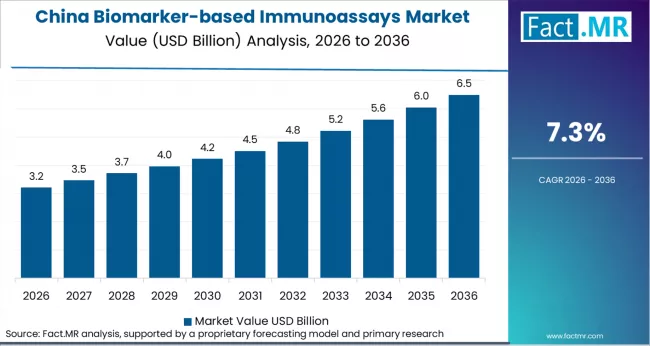
China is the leading market, driven by the expansion of clinical diagnostics, research laboratories, and hospitals. Key regional players include Abbott Laboratories, Roche Diagnostics, and Siemens Healthineers.
- China: Demand for biomarker-based immunoassays in China is projected to rise at 7.3% CAGR through 2036. Growth is supported by government-funded hospital upgrades (03-2025) and Abbott Laboratories’ expansion of regional diagnostic facilities (08-2025).
South Asia:
India is a significant market, supported by rising clinical testing, public health programs, and pharmaceutical R&D.
- India: Demand for biomarker-based immunoassays in India is projected to rise at 6.8% CAGR through 2036. Growth is driven by national healthcare diagnostic programs (05-2025) and Roche Diagnostics’ expansion of laboratory networks (09-2025).
Western Europe:
Germany, France, and the United Kingdom are key markets, driven by adoption in hospitals, research institutes, and clinical laboratories.
- Germany: Demand for biomarker-based immunoassays in Germany is projected to rise at 6.2% CAGR through 2036. Growth is supported by hospital modernization programs (04-2025) and Siemens Healthineers’ regional investments (08-2025).
- France: Demand for biomarker-based immunoassays in France is projected to rise at 5.7% CAGR through 2036. Growth is influenced by national laboratory network upgrades (03-2025) and Abbott Laboratories’ facility expansion (07-2025).
- United Kingdom: Demand for biomarker-based immunoassays in the UK is projected to rise at 5.1% CAGR through 2036. Growth is supported by NHS laboratory modernization programs (06-2025) and Roche Diagnostics’ regional investments (09-2025).
Fact MR's analysis of the biomarker-based immunoassays market in East Asia, South Asia, and Western Europe consists of country-wise assessments that include China, India, Germany, France, and the United Kingdom. Readers can find detailed trends, regulatory updates, and company-specific investments shaping market growth in these countries.
What is the Competitive Structure and Buyer Behavior in the Biomarker based Immunoassays Market?
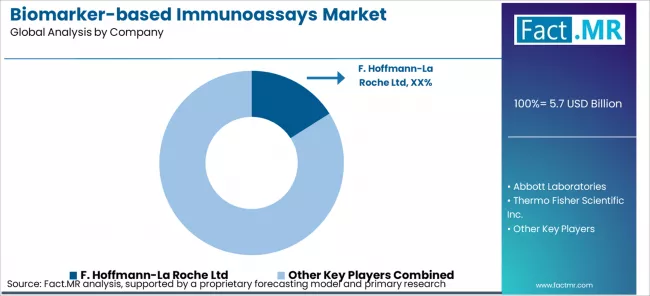
The biomarker‑based immunoassays market is moderately concentrated, with major global diagnostics and life sciences firms such as F. Hoffmann‑La Roche Ltd, Abbott Laboratories, Thermo Fisher Scientific Inc., Eurofins Scientific SE, QIAGEN N.V., Bio‑Rad Laboratories, Inc., and Siemens Healthineers AG accounting for a substantial share of market value. The primary competitive variables are assay accuracy, sensitivity, throughput, regulatory compliance, breadth of biomarker panels, and integration with laboratory information systems, all of which heavily influence procurement by clinical laboratories, hospitals, and research institutions. Companies with strong R&D capabilities, broad product portfolios, global regulatory expertise, and established service networks hold structural advantages, as they can rapidly introduce new assays, maintain quality standards, and provide ongoing calibration and technical support across regions.
Large healthcare systems, central laboratories, and pharmaceutical research buyers exert significant leverage by qualifying multiple platform suppliers, negotiating long‑term service and pricing agreements, and standardizing equipment and assay platforms to streamline workflows. To mitigate supplier dependency, these buyers maintain diversified vendor lists, emphasize interoperability and support availability, and often require shared performance metrics in contracts, which limits individual suppliers’ pricing power. As a result, competition centers on delivering reliable performance, comprehensive assay menus, integration capability, and robust after‑sales support rather than on price alone.
Recent Industry Developments
- Roche's 2026 Oncology Expansion: During the January 2026 J.P. Morgan Healthcare Conference, Roche outlined its strategy to file for up to three new molecular entities (NMEs) this year. The company is prioritizing its next-generation SERD (giredestrant) for breast cancer, which relies on advanced immunoassay and molecular testing for companion diagnostics.
- Siemens Healthineers Hemostasis Innovation: In early 2026, Siemens Healthineers expanded its bleeding management portfolio with the INNOVANCE Anti-Xa assay. This automated chromogenic assay provides rapid measurement of anticoagulants like apixaban and edoxaban, streamlining the workflow for emergency departments and specialized labs.
- Mass Spec vs. Immunoassay Hybridization: A significant technical trend in 2026 is the development of immunoaffinity-LC-MS/MS methods. Researchers at Fudan University recently published a robust method to quantify the Alzheimer's therapy donanemab in serum, offering a high-precision alternative to traditional immunoassays by eliminating cross-reactivity issues.
Key Players of the Biomarker‑based Immunoassays Market
- F. Hoffmann-La Roche Ltd
- Abbott Laboratories
- Thermo Fisher Scientific Inc.
- Eurofins Scientific SE
- QIAGEN N.V.
- Bio-Rad Laboratories, Inc.
- Siemens Healthineers AG
Report Scope
| Metric | Value |
|---|---|
| Quantitative Units | USD 5.7 billion (2026) to USD 9.6 billion (2036), at a CAGR of 5.4% |
| Market Definition | The Biomarker-based Immunoassays Market includes global production and trade of diagnostic assays that detect biomarkers for disease monitoring, prognosis, and pharmacodynamic studies across clinical and research settings. |
| Sample Type | Blood, Tissue, Urine, Others |
| Product | Reagents & Kits, Instruments/Analyzers, Services |
| Biomarker Type | Efficacy & Pharmacodynamic, Predictive/Prognostic, Others |
| Disease | Cancer, Cardiovascular, Neurological, Others |
| End Use | Hospitals & Clinics, Diagnostic Labs, Research |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia, and 40+ countries |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd, Abbott Laboratories, Thermo Fisher Scientific Inc., Eurofins Scientific SE, QIAGEN N.V., Bio-Rad Laboratories, Inc., Siemens Healthineers AG |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with diagnostic assay producers, supported by trade data benchmarking and plant-level capacity verification. |
Bibliographies
- Smith, J. G., & Lee, K. Y. (2023). Advances in biomarker-based immunoassays for clinical diagnostics and precision medicine. Journal of Immunological Methods.
- Patel, R., & Wong, A. (2024). Multiplex immunoassays and biomarker detection technologies in clinical laboratories. Clinical Chemistry.
- World Health Organization. (2023). Biomarker-based diagnostic technologies for disease detection and monitoring. World Health Organization.
- National Institutes of Health. (2024). Biomarker discovery and validation in clinical diagnostics. National Institutes of Health.
- Clinical Trials Arena. (2026, January 14). JPM26: Roche's rejuvenation strategy for the oncology and haematology portfolio. Clinical Trials Arena.
- LCGC International. (2026, March 4). An LC-MS/MS method for quantification of donanemab in serum. LCGC International.
- F. Hoffmann-La Roche Ltd. (2024). Annual report: Diagnostics division and biomarker testing advancements. F. Hoffmann-La Roche Ltd.
This report addresses
- Market intelligence enabling comprehensive assessment of leading countries and product segments across the Biomarker-based Immunoassays Market globally.
- Market volume (sales units and revenue) estimates and 10-year revenue forecasts from 2026 to 2036, validated through manufacturer shipment data, clinical laboratory surveys, and country-level adoption modeling.
- Growth opportunity mapping across sample types (Blood, Tissue, Urine, Others), product types (Reagents & Kits, Instruments/Analyzers, Services), biomarker types (Efficacy & Pharmacodynamic, Predictive/Prognostic, Others), and disease applications (Cancer, Cardiovascular, Neurological, Others).
- Segment and regional revenue forecasts by sample type, product, biomarker type, disease, end-use industry, and geography across 40+ countries.
- Competition strategy analysis covering assay accuracy, sensitivity, throughput, regulatory compliance, and integration with laboratory information systems, including major players like F. Hoffmann-La Roche Ltd, Abbott Laboratories, Thermo Fisher Scientific Inc., Eurofins Scientific SE, QIAGEN N.V., Bio-Rad Laboratories, Inc., and Siemens Healthineers AG.
- Product and regulatory compliance tracking aligned with clinical and laboratory standards, biomarker validation requirements, and diagnostic quality certifications.
- Regulatory impact analysis addressing healthcare, laboratory, and research standards, diagnostics approval requirements, and quality assurance guidelines across key markets.
- Report delivery in PDF, Excel, PowerPoint, and interactive dashboard formats for executive, procurement, and operational planning use.
Biomarker-based Immunoassays Market Key Segments
-
Sample Type
- Blood
- Tissue
- Urine
- Others
-
Product
- Reagents & Kits
- Instruments/Analyzers
- Services
-
Biomarker Type
- Efficacy & Pharmacodynamic
- Predictive/Prognostic
- Others
-
Disease
- Cancer
- Cardiovascular
- Neurological
- Others
-
End-Use Industry
- Hospitals & Clinics
- Diagnostic Labs
- Research
-
Region
- North America
- USA
- Canada
- Mexico
- Europe
- Germany
- UK
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East and Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Rest of Middle East and Africa
- Other Regions
- Oceania
- Central Asia
- Other Markets
- North America
- Frequently Asked Questions -
How large is the demand for Biomarker-based Immunoassays in the global market in 2026?
Demand for Biomarker-based Immunoassays in the global market is estimated to be valued at USD 5.7 billion in 2026.
What will be the market size of Biomarker-based Immunoassays in the global market by 2036?
The market size for Biomarker-based Immunoassays is projected to reach USD 9.6 billion by 2036.
What is the expected demand growth for Biomarker-based Immunoassays in the global market between 2026 and 2036?
Demand for Biomarker-based Immunoassays in the global market is expected to grow at a CAGR of 5.4% between 2026 and 2036.
Which sample type is expected to dominate the market?
Blood is expected to dominate the market, accounting for 65.2% of the market share in 2026, due to its widespread use in diagnostic and research immunoassays for detecting biomarkers in clinical and laboratory settings.
Which region is expected to show the highest growth rate for Biomarker-based Immunoassays?
China is projected to show the highest regional CAGR at 7.3% during the forecast period, driven by increasing healthcare expenditure, expansion of diagnostic laboratories, and rising adoption of immunoassay technologies.
How significant is the growth outlook for India in this market?
India is expected to grow at a CAGR of 6.8%, supported by increasing clinical diagnostics demand, rising awareness of biomarker-based testing, and the expansion of hospitals and diagnostic centers.
What is the growth outlook for Germany in the Biomarker-based Immunoassays market?
Germany is expected to grow at a CAGR of 6.2%, driven by advanced healthcare infrastructure, high adoption of immunoassay technologies, and increasing research and clinical applications for biomarker detection.
What is the growth forecast for France in the Biomarker-based Immunoassays market?
France is expected to grow at a CAGR of 5.7%, reflecting rising clinical and research applications of biomarker assays and increased investments in healthcare diagnostics.
What is the growth forecast for the United Kingdom in the Biomarker-based Immunoassays market?
The United Kingdom is projected to grow at a CAGR of 5.1% from 2026 to 2036, supported by strong healthcare infrastructure, adoption of diagnostic technologies, and demand for reliable biomarker detection methods.
Which company is identified as a leading player in the Biomarker-based Immunoassays market?
F. Hoffmann-La Roche Ltd is recognized as a leading player in this market, providing advanced biomarker-based immunoassay products for clinical diagnostics and research applications globally.
What are Biomarker-based Immunoassays used for?
Biomarker-based Immunoassays are used to detect and quantify biomarkers in biological samples, primarily blood, for disease diagnosis, prognosis, monitoring, and research. They are essential in clinical diagnostics, drug development, and personalized medicine.
What does the Biomarker-based Immunoassays market include in this report?
The market scope includes immunoassay products and solutions used in biomarker detection, with a focus on blood-based assays, covering clinical, diagnostic, and research applications.
How is the market forecast developed in this report?
The forecast is developed using historical data on immunoassay adoption, trends in biomarker-based diagnostics, healthcare infrastructure growth, and insights from leading players in the global immunoassay market.
What is meant by the Biomarker-based Immunoassays market in this report?
The market refers to the global production, trade, and consumption of biomarker-based immunoassays, focusing on blood-based diagnostic assays used for disease detection, monitoring, and research across clinical and laboratory settings.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sample Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sample Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sample Type , 2026 to 2036
- Blood
- Tissue
- Urine
- Others
- Blood
- Y to o to Y Growth Trend Analysis By Sample Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Sample Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Disease
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease, 2026 to 2036
- Cancer
- Cardiovascular
- Neurological
- Others
- Cancer
- Y to o to Y Growth Trend Analysis By Disease, 2021 to 2025
- Absolute $ Opportunity Analysis By Disease, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Sample Type
- By Disease
- By Country
- Market Attractiveness Analysis
- By Country
- By Sample Type
- By Disease
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Sample Type
- By Disease
- By Country
- Market Attractiveness Analysis
- By Country
- By Sample Type
- By Disease
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Sample Type
- By Disease
- By Country
- Market Attractiveness Analysis
- By Country
- By Sample Type
- By Disease
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Sample Type
- By Disease
- By Country
- Market Attractiveness Analysis
- By Country
- By Sample Type
- By Disease
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Sample Type
- By Disease
- By Country
- Market Attractiveness Analysis
- By Country
- By Sample Type
- By Disease
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Sample Type
- By Disease
- By Country
- Market Attractiveness Analysis
- By Country
- By Sample Type
- By Disease
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Sample Type
- By Disease
- By Country
- Market Attractiveness Analysis
- By Country
- By Sample Type
- By Disease
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Sample Type
- By Disease
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Sample Type
- By Disease
- Competition Analysis
- Competition Deep Dive
- F. Hoffmann-La Roche Ltd
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abbott Laboratories
- Thermo Fisher Scientific Inc.
- Eurofins Scientific SE
- QIAGEN N.V.
- Bio-Rad Laboratories, Inc.
- Siemens Healthineers AG
- F. Hoffmann-La Roche Ltd
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Sample Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Disease, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Sample Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Disease, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Sample Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Disease, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Sample Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Disease, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Sample Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Disease, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Sample Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Disease, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Sample Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Disease, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Sample Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Disease, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Sample Type , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Sample Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Sample Type
- Figure 6: Global Market Value Share and BPS Analysis by Disease, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Disease, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Disease
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Sample Type , 2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Sample Type , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Sample Type
- Figure 23: North America Market Value Share and BPS Analysis by Disease, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Disease, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Disease
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Sample Type , 2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Sample Type , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Sample Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Disease, 2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Disease, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Disease
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Sample Type , 2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Sample Type , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Sample Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Disease, 2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Disease, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Disease
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Sample Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Sample Type , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Sample Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Disease, 2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Disease, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Disease
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Sample Type , 2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Sample Type , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Sample Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Disease, 2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Disease, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Disease
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Sample Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Sample Type , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Sample Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Disease, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Disease, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Disease
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Sample Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Sample Type , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Sample Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Disease, 2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Disease, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Disease
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis