Mosquito-Borne Infections Testing Market Size, Share, Growth and Forecast (2026 - 2036)
Mosquito-Borne Infections Testing Market is segmented by Test Type (Molecular Diagnostics including PCR, RT-PCR, Isothermal Amplification, and Next-Generation Sequencing; Immunoassay Systems including Chemiluminescence and Fluorescence Immunoassay; ELISA; Rapid Tests including Rapid Diagnostic Tests and Lateral Flow Assays; Blot Systems and Evaluation Software; and Microspot Array Diagnostics), Indication (Malaria including Plasmodium falciparum, Plasmodium vivax, and Mixed/Other Species; Zika Virus; Chikungunya; Dengue including Dengue NS1 Antigen and Dengue IgM/IgG Antibodies; and Others), End User (Hospitals and Clinics, Private Commercial Laboratories, Academic and Research Centers, and Others), and Region. Forecast for 2026 to 2036.
Segmental Analysis
Mosquito Borne Infections Testing Market Analysis by Test Type
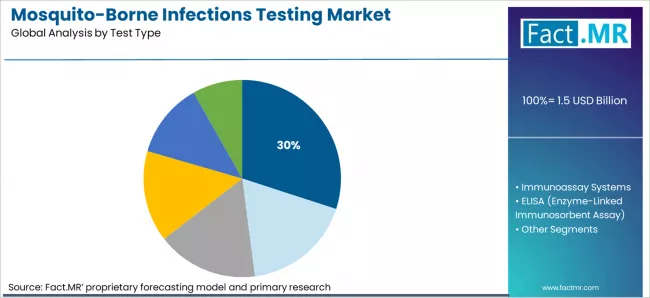
Molecular diagnostics hold about 30.0% value share in 2025. They command premium pricing. RT PCR is required for dengue serotype surveillance and malaria resistance monitoring. Multiplex platforms detect dengue, chikungunya, and Zika from one sample. This supports differential diagnosis in endemic hospitals.
- Roche Elecsys Dengue Panel Automated Platform Launch: Roche received CE mark in October 2025 for Elecsys Dengue Ag on cobas platforms. [1] The test shows 94.90% sensitivity and 99.96% specificity. The panel will expand to include IgM and IgG assays. It offers a complete automated dengue workflow in Europe.
- Abbott Bioline Malaria RDT Sensitivity Investigation: A December 2024 Malaria Journal study found 24% sensitivity for Abbott Bioline P. vivax RDTs in Myanmar. [4] WHO circulated internal guidance. The incident highlights RDT reliability risks and strengthens demand for molecular confirmation.
- QIAGEN QIAstat Dx Syndromic Panel India Launch: QIAGEN launched QIAstat Dx febrile panels in India in January 2024. [5] The system enables simultaneous detection of multiple pathogens. It supports differential diagnosis in high burden hospitals.
Mosquito Borne Infections Testing Market Analysis by Indication
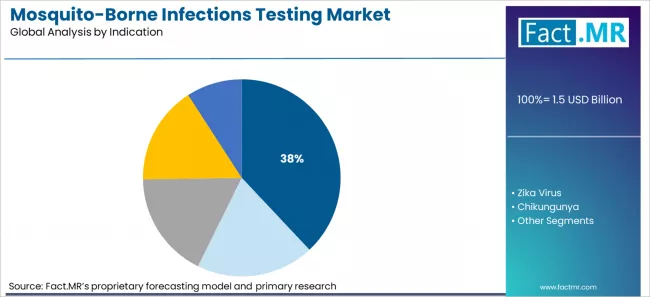
Malaria testing holds about 38.0% value share in 2025. It benefits from structured procurement through Global Fund and donor programmes. Laboratory confirmation is institutionalized in endemic regions. Demand is stable and less seasonal than dengue.
- WHO World Malaria Report 2025 and Diagnostics Investment: WHO reported 282 million malaria cases and 610,000 deaths in 2024. [6] Drug resistance in eight African countries requires PCR surveillance. This expands molecular testing beyond RDT use.
- Brazil Dengue Epidemic and NS1 Procurement Surge: Brazil’s 2024 epidemic exceeded 10 million cases. [3] Emergency NS1 and PCR procurement followed. Updated protocols require lab confirmation for hospitalised patients.
- Dengue Geographic Expansion in Europe and Travel Medicine: Germany reported local dengue transmission in 2024. [7] Testing demand rose in hospitals and travel clinics. High income settings favor automated platforms such as Roche Elecsys.
Drivers, Restraints, and Opportunities
Fact.MR estimates the mosquito borne infections testing market reached USD 1.4 billion in 2025. Growth reflects record disease burden and expanding lab infrastructure in endemic regions. Malaria RDT procurement once dominated revenues. Now dengue testing has become an equal driver. WHO reported 282 million malaria cases and 610,000 deaths in 2024.
The key tension lies in geography and pricing. Disease burden is highest in low and middle income countries. These markets sustain low pricing. Malaria RDTs cost below USD 1.00 in Global Fund procurement. In contrast, RT PCR dengue panels cost USD 15 to 50 per test. This creates two market tiers. One is high volume, low ASP public RDT demand.
- WHO Global Strategic Plan for Arboviral Diseases: WHO launched a global plan in October 2024 requiring USD 55 million for dengue and other arboviral diseases. [2] The plan aligns with the Global Vector Control Response. It creates structured procurement commitments for diagnostics. This reduces supplier risk and supports product registration in endemic markets.
- Roche Elecsys Dengue Ag CE Mark and Laboratory Automation Shift: Roche received CE mark in October 2025 for Elecsys Dengue Ag. [1] The test delivers 18 minute results with 94.90% sensitivity and high throughput. It sets a new benchmark in Europe and IVD regulated markets. Automated immunoassays are expected to replace manual ELISA and RDT protocols in hospitals.
- Brazil's Record Dengue Burden and Diagnostic Infrastructure Investment: Brazil reported over 10 million dengue cases and 6,321 deaths in 2024. [3] The Ministry of Health expanded lab confirmation and NS1 procurement. Brazil also launched Wolbito do Brasil in July 2024 for mosquito control. Diagnostics remain central to outbreak monitoring.
Regional Analysis
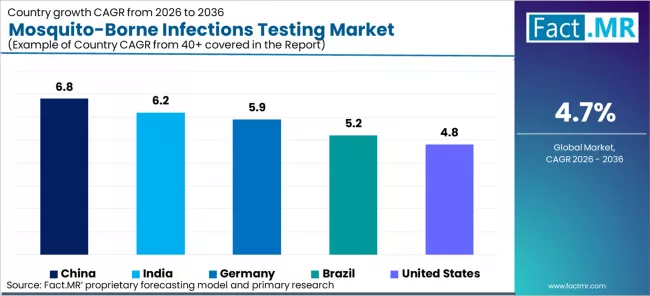
The mosquito-borne infections testing market is analysed across North America, Europe, Asia Pacific, Latin America, and Middle East and Africa, with each region assessed by disease burden intensity, national diagnostic programme procurement structure, laboratory infrastructure maturity, and private sector testing market development. Country-level coverage includes China, India, Germany, Brazil, and the United States. The full report provides market attractiveness analysis benchmarked against WHO prequalification requirements, national insurance reimbursement coverage for arboviral diagnostics, and outbreak response procurement trigger mechanisms.
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 6.8% |
| India | 6.2% |
| Germany | 5.9% |
| Brazil | 5.2% |
| United States | 4.8% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
Asia Pacific Mosquito-Borne Infections Testing Market Analysis
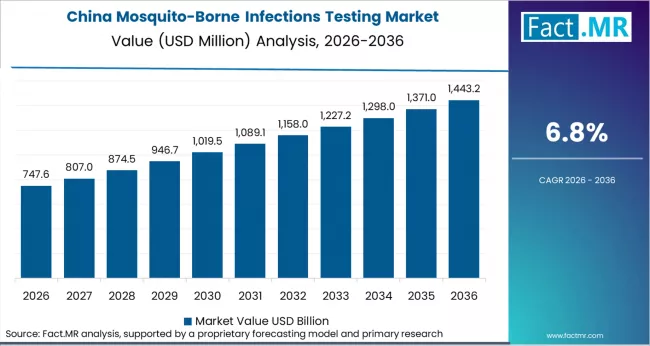
Asia Pacific carries the highest disease burden globally. Malaria remains endemic across much of the region. Dengue incidence is rising across Southeast Asia. Private hospital laboratory networks are expanding in India and China. Thermo Fisher, Roche, Abbott, and QIAGEN lead the premium tier. Domestic manufacturers supply high-volume public procurement.
- China: Demand for mosquito-borne infections testing in China is projected to rise at 6.8% CAGR through 2036. Growth is driven by dengue resurgence in Guangdong, Yunnan, and Fujian. CDC-China mandates laboratory confirmation under national reporting rules. China remains malaria free but maintains PCR surveillance for imported cases. The 2024 National Health Commission update expanded RT-PCR capacity at provincial CDCs. Domestic firms such as BGI Genomics and Bioperfectus expanded multiplex PCR registrations in 2024 and 2025.
- India: Demand in India is projected to rise at 6.2% CAGR through 2036. Procurement is supported by the National Vector Borne Disease Control Programme. Government centres distribute malaria RDTs and dengue NS1 kits. ICMR guidelines define approved platforms. Private labs such as Metropolis, SRL, Thyrocare, and Dr. Lal PathLabs expanded testing after record 2024 outbreaks.
FACT.MR covers China, India, Japan, South Korea, Australia and New Zealand, ASEAN, and Rest of Asia Pacific. The report includes procurement data, regulatory timelines, and private lab expansion trends.
Europe Mosquito-Borne Infections Testing Market Analysis
Europe is the regulatory benchmark under EU IVD Regulation. It is also experiencing local dengue transmission. Roche, bioMerieux, and Abbott lead the laboratory market. Roche’s October 2025 Elecsys Dengue Ag CE mark defines the premium automated tier.
- Germany: Demand in Germany is projected to rise at 5.9% CAGR through 2036. Autochthonous dengue cases were confirmed in 2024. Testing now includes patients without travel history. Roche’s CE mark in October 2025 allows easy upgrades on cobas platforms. Updated 2024 clinical guidelines widened NS1 testing indications.
FACT.MR covers Germany, France, United Kingdom, Italy, Spain, Netherlands, Nordics, and Rest of Europe. The report includes EU IVD timelines and surveillance data.
North America Mosquito-Borne Infections Testing Market Analysis
North America is driven by surveillance and travel medicine demand. CDC infrastructure and FDA cleared platforms shape the market. Abbott, Thermo Fisher, Hologic, and QuidelOrtho are key suppliers.
- United States: Demand is projected to rise at 4.8% CAGR through 2036. Puerto Rico extended its dengue emergency through 2025. CDC issued updated testing guidance in March 2025 under HAN-00523. Climate modelling suggests mosquito range expansion. West Nile virus remains the leading domestic infection with 1,791 cases in 2024 and over 2,000 in 2025.
FACT.MR covers the United States, Canada, and Mexico. The report includes CDC guidance and FDA approval timelines.
Latin America Mosquito-Borne Infections Testing Market Analysis
Latin America carries the highest dengue burden globally. Brazil accounts for most 2024 cases. Malaria remains endemic in Amazon regions. Roche, Abbott, and bioMerieux serve the institutional tier. Domestic firms supply RDT volumes.
- Brazil: Demand is projected to rise at 5.2% CAGR through 2036. Brazil recorded over 10 million dengue cases and 6,321 deaths in 2024. ANVISA expanded diagnostic confirmation requirements. Public procurement of NS1 and PCR increased across SUS laboratories. Wolbito do Brasil opened in July 2024 to support vector control. WHO reported Brazil drove most regional dengue cases in 2024.
FACT.MR covers Brazil, Mexico, Colombia, Argentina, Chile, and Rest of Latin America. The report includes SUS pricing benchmarks, ANVISA approvals, and epidemic cycle data.
Competitive Aligners for Market Players
The global mosquito-borne infections testing market is moderately concentrated in the premium platform segment. It is highly fragmented in rapid diagnostic tests. Abbott, Roche, bioMerieux, Thermo Fisher, QIAGEN, and Siemens dominate molecular and automated immunoassay revenues. These segments command the highest average selling prices. In contrast, more than 20 manufacturers compete in the WHO-prequalified RDT space. Many are based in India, South Korea, and China. In the premium tier, platform integration is the key advantage. Suppliers offering dengue, malaria, and febrile panels on installed automation systems gain stronger customer loyalty.
Vertically integrated platform providers hold structural advantages. Roche benefits from its October 2025 Elecsys Dengue Ag CE mark on the widely installed cobas platform. This enables hospitals to adopt new assays without buying new instruments. Abbott operates across both RDT and automated tiers through SD BIOLINE and ARCHITECT platforms. bioMerieux strengthened its rapid test reach through the GenBody acquisition, expanding beyond its VIDAS base. Buyer behavior is split. Public health agencies use multi-year tenders with limited pricing power. Private hospitals choose platforms that lock in reagent supply for several years.
Recent Developments
- In 2025, Roche received CE mark approval for its fully automated Elecsys Dengue Ag test, a high-throughput immunoassay that detects dengue NS1 antigen with high sensitivity and specificity on cobas e platforms. This launch targets laboratory workflows in Europe and other CE-recognition markets, supporting faster acute dengue diagnosis.
- In June 2025, bioMérieux agreed to acquire Day Zero Diagnostics’ sequencing assets for under USD 25 million, strengthening its rapid infectious disease testing pipeline and advancing next-generation diagnostics for antibiotic-resistant infections.
Key Players
- Abbott Laboratories
- F. Hoffmann-La Roche Ltd.
- Bio-Rad Laboratories, Inc.
- Siemens Healthineers AG
- Qiagen N.V.
- Thermo Fisher Scientific Inc.
- Cepheid (Danaher Corporation)
- DiaSorin S.p.A.
- Hologic, Inc.
- Meridian Bioscience, Inc.
- bioMerieux SA
- QuidelOrtho Corporation
- BD (Becton, Dickinson and Company)
Bibliography
- [1] F. Hoffmann-La Roche Ltd. (2025, October). Roche Receives CE Mark for Novel Automated High-Throughput Elecsys Dengue Ag Test to Diagnose Dengue.
- [2] World Health Organization. (2024, October). WHO Launches Global Strategic Plan to Fight Rising Dengue and Other Aedes-Borne Arboviral Diseases.
- [3] World Health Organization. (2025, December). Dengue: Global Situation, Surveillance and Progress - 2024
- [4] Shoklo Malaria Research Unit / Malaria Journal. (2024, December). The Underperforming Abbott-Bioline Malaria Ag P.f/P.v Rapid Diagnostic Test: A Whiter Shade of Pale.
- [5] QIAGEN N.V. (2024, January). QIAGEN Launches Two Syndromic Testing Panels for QIAstat-Dx Instruments in India Including Gastrointestinal Panel 2 and Meningitis/Encephalitis Panel.
- [6] World Health Organization. (2025, January). World Malaria Report 2025. WHO Global Malaria Programme.
- [7] Robert Koch Institut. (2024). SurvStat - Dengue Virus Infections Including Autochthonous Cases in Germany.
This Report Addresses
- Market intelligence for strategic planning across mosquito-borne infections diagnostic test type economics, multiplex versus singleplex platform adoption, WHO prequalification procurement dynamics, and competitive response to the 2024 record dengue epidemic and climate-driven geographic arboviral expansion reshaping endemic and non-endemic country testing demand.
- Market size and forecast: mosquito-borne infections testing market valued at USD 1.5 billion in 2026, projected to reach USD 2.3 billion by 2036 at a CAGR of 4.7%, with segmentation by test type, indication, end user, and region.
- Growth opportunity mapping across multiplex RT-PCR arboviral panel adoption, automated immunoassay platform expansion for dengue in European and high-income Asia Pacific markets, WHO prequalification pipeline tracking, and private diagnostic laboratory network growth in India, China, and Brazil.
- Segment and regional forecasts: country-level CAGR analysis for China (6.8%), India (6.2%), Germany (5.9%), Brazil (5.2%), and United States (4.8%), with test type, indication, and end user splits by regional healthcare system structure.
- Competition strategy assessment: positioning of Roche, Abbott, bioMerieux, Thermo Fisher, QIAGEN, and Siemens Healthineers covering platform integration strategy, WHO prequalification investment, Roche Elecsys Dengue Ag CE mark competitive implications, and Abbott Bioline RDT quality management response.
- Product and regulatory tracking: EU IVD Regulation CE mark requirements, FDA EUA and 510(k) clearance status for arboviral platforms, WHO prequalification list updates, dengue NS1 inclusion on WHO Essential Diagnostics List, and ICMR and CDC guideline updates for mosquito-borne infection diagnostic protocols.
- Regulatory and policy impact analysis: WHO October 2024 global strategic plan for arboviral diseases USD 55 million investment framework, Brazil Ministry of Health dengue epidemic response procurement requirements, CDC HAN-00523 updated testing recommendations, and India NVBDCP procurement programme evolution.
- Report delivery in Excel data tables, PowerPoint executive summary, and detailed PDF with verified WHO, CDC, ECDC, and company press release sources for executive strategy, investor relations, and product planning teams.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD 1.5 billion (2026) to USD 2.3 billion (2036), at a CAGR of 4.7% |
| Market Definition | Mosquito-borne infections testing encompasses in-vitro diagnostic assays, rapid tests, molecular platforms, and immunoassay systems used to detect and differentiate Plasmodium malaria parasites, dengue, Zika, chikungunya, West Nile, and other mosquito-transmitted pathogens in clinical, public health, blood safety, and travel medicine settings globally. |
| Test Type Segmentation | Molecular Diagnostics (PCR, RT-PCR, Isothermal Amplification, NGS), Immunoassay Systems (Chemiluminescence, Fluorescence), ELISA, Rapid Tests (RDTs, Lateral Flow Assays), Blot Systems and Evaluation Software, Microspot Array Diagnostics |
| Indication Segmentation | Malaria (P. falciparum, P. vivax, Mixed Species), Zika Virus, Chikungunya, Dengue (NS1 Antigen, IgM/IgG Antibodies), Others (West Nile, Yellow Fever, Japanese Encephalitis) |
| End User Segmentation | Hospitals and Clinics, Private Commercial Laboratories, Academic and Research Centers, Others (Blood Banks, Public Health Laboratories, Travel Medicine Centers) |
| Application Coverage | Acute febrile illness diagnosis, outbreak surveillance and response, blood safety screening, travel medicine return testing, malaria elimination programme confirmatory testing, and arboviral co-infection differential diagnosis |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East and Africa |
| Countries Covered | China, India, Japan, South Korea, Australia and New Zealand, ASEAN, Rest of Asia Pacific, Germany, France, United Kingdom, Italy, Spain, Netherlands, Nordics, Rest of Europe, United States, Canada, Mexico, Brazil, Colombia, Argentina, Chile, Rest of Latin America, Kingdom of Saudi Arabia, United Arab Emirates, South Africa, Nigeria, Ethiopia, Rest of Middle East and Africa |
| Key Companies Profiled | Abbott Laboratories, F. Hoffmann-La Roche Ltd., Bio-Rad Laboratories Inc., Siemens Healthineers AG, Qiagen N.V., Thermo Fisher Scientific Inc., Cepheid (Danaher), DiaSorin S.p.A., Hologic Inc., Meridian Bioscience Inc., bioMerieux SA, QuidelOrtho Corporation, BD (Becton Dickinson) |
| Forecast Period | 2026 to 2036 |
| Approach | A hybrid top-down and bottom-up modelling approach was used, incorporating WHO global disease burden reports, national diagnostic programme budget allocations, WHO prequalification procurement volume estimates, and per-test ASP benchmarks by assay type and geography. Estimates were validated through primary interviews with clinical laboratory directors, public health procurement officers, and company press release cross-referencing against WHO, CDC, and ECDC surveillance data. |
Mosquito-Borne Infections Testing Market by Segment
-
By Test Type :
- Molecular Diagnostics
- PCR (Polymerase Chain Reaction)
- RT-PCR (Real-Time PCR)
- Isothermal Amplification
- Next-Generation Sequencing
- Immunoassay Systems
- Chemiluminescence Immunoassay
- Fluorescence Immunoassay
- ELISA (Enzyme-Linked Immunosorbent Assay)
- Rapid Tests
- Rapid Diagnostic Tests (RDTs)
- Lateral Flow Assays
- Blot Systems and Evaluation Software
- Microspot Array Diagnostics
- Molecular Diagnostics
-
By Indication :
- Malaria
- Plasmodium falciparum
- Plasmodium vivax
- Mixed/Other Species
- Zika Virus
- Chikungunya
- Dengue
- Dengue NS1 Antigen
- Dengue IgM/IgG Antibodies
- Others (West Nile, Yellow Fever, Japanese Encephalitis)
- Malaria
-
By End User :
- Hospitals and Clinics
- Infectious Disease Departments
- Emergency Departments
- Outpatient Clinics
- Private Commercial Laboratories
- Reference Laboratories
- Regional Diagnostic Centers
- Academic and Research Centers
- Universities
- Research Institutes
- Others
- Blood Banks
- Public Health Laboratories
- Travel Medicine Centers
- Hospitals and Clinics
-
By Region :
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Netherlands
- Nordics
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- South Korea
- Australia and New Zealand
- ASEAN
- Rest of Asia Pacific
- Latin America
- Brazil
- Mexico
- Colombia
- Argentina
- Chile
- Rest of Latin America
- Middle East and Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Nigeria
- Ethiopia
- Rest of Middle East and Africa
- North America
- Frequently Asked Questions -
How large is the mosquito-borne infections testing market in 2025?
The global mosquito-borne infections testing market was valued at USD 1.4 billion in 2025.
What will the market size be in 2026?
The market is estimated to grow to USD 1.5 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 2.3 billion by 2036, representing an absolute gain of USD 0.8 billion over the forecast period.
What is the expected CAGR for the forecast period 2026 to 2036?
Fact.MR projects a CAGR of 4.7% from 2026 to 2036 for the global mosquito-borne infections testing market.
Which test type segment is poised to lead the mosquito-borne infections testing market?
Molecular Diagnostics are forecast to lead with an estimated 30.0% value share.
Which indication segment holds the largest market share?
Malaria holds the largest indication share at approximately 38.0% share in market.
Which country shows the fastest growth rate in the market?
China projects the fastest country-level CAGR at 6.8% through 2036.
Why is Brazil projected to grow at 5.2%?
Record dengue outbreaks and expanded public procurement of NS1 and PCR tests through the national health system are sustaining demand.
What is driving 4.8% growth in the United States?
Updated CDC testing guidelines, Puerto Rico’s dengue emergency, and steady West Nile surveillance are supporting consistent diagnostic demand.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Type , 2026 to 2036
- Molecular Diagnostics
- Immunoassay Systems
- ELISA (Enzyme-Linked Immunosorbent Assay)
- Rapid Tests
- Blot Systems and Evaluation Software
- Microspot Array Diagnostics
- Molecular Diagnostics
- Y to o to Y Growth Trend Analysis By Test Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Test Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Malaria
- Zika Virus
- Chikungunya
- Dengue
- Others (West Nile, Yellow Fever, Japanese Encephalitis)
- Malaria
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Indication
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Indication
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Indication
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Indication
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Indication
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Indication
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By Indication
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Type
- By Indication
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Type
- By Indication
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- F. Hoffmann-La Roche Ltd.
- Bio-Rad Laboratories, Inc.
- Siemens Healthineers AG
- Qiagen N.V.
- Thermo Fisher Scientific Inc.
- Cepheid (Danaher Corporation)
- DiaSorin S.p.A.
- Hologic, Inc.
- Meridian Bioscience, Inc.
- bioMerieux SA
- QuidelOrtho Corporation
- BD (Becton, Dickinson and Company)
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Test Type, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Test Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Test Type
- Figure 6: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Test Type, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Test Type
- Figure 23: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Indication
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Test Type, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Test Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Indication
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Test Type, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Test Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Indication
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Test Type, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Test Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Test Type, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Test Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Indication
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Test Type, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Test Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Test Type, 2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Test Type, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Test Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis