Neurological Rare Disease Biologics Market Size, Share, Growth and Forecast (2026 - 2036)
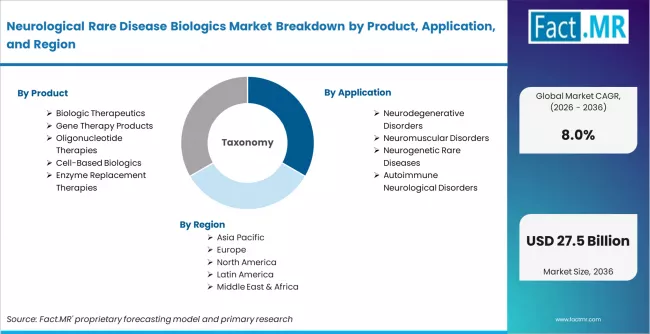
Neurological Rare Disease Biologics Market is segmented by Product (Biologic Therapeutics, Gene Therapy Products, Oligonucleotide Therapies, Cell-Based Biologics, Enzyme Replacement Therapies), Application (Neurodegenerative Disorders, Neuromuscular Disorders, Neurogenetic Rare Diseases, Autoimmune Neurological Disorders), End Use (Pharmaceutical & Biotechnology Companies, Specialty Clinics & Hospitals, Academic & Research Institutes, Contract Research Organizations (CROs)), and Region, with forecasts covering the period from 2026 to 2036.
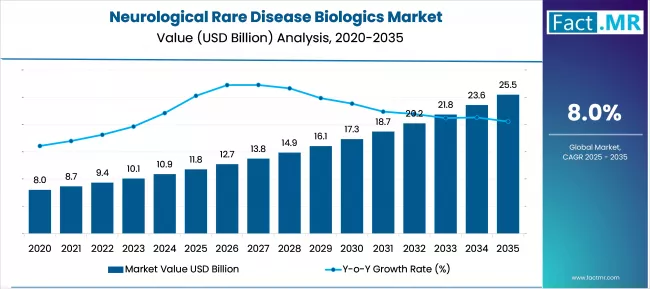
According to Fact.MR estimates, the global neurological rare disease biologics market market was valued at USD 11.8 billion in 2025. The market is projected to reach USD 12.7 billion in 2026 and is expected to grow to USD 27.5 billion by 2036, expanding at a CAGR of 8.0%. Biologic Therapeutics is anticipated to account for 44.0% of the product segment in 2026, while Neurodegenerative Disorders is expected to remain the leading application with around 41.0% share.
Neurological Rare Disease Biologics Market Analysis and Forecast by Fact.MR
According to Fact.MR estimates, the global neurological rare disease biologics market market was valued at USD 11.8 billion in 2025. The market is projected to reach USD 12.7 billion in 2026 and is expected to grow to USD 27.5 billion by 2036, expanding at a CAGR of 8.0%. Biologic Therapeutics is anticipated to account for 44.0% of the product segment in 2026, while Neurodegenerative Disorders is expected to remain the leading application with around 41.0% share.
Summary of Neurological Rare Disease Biologics Market
- Market Snapshot
- The Neurological Rare Disease Biologics market was valued at approximately USD 11.8 billion in 2025.
- By 2036, the Neurological Rare Disease Biologics market is expected to be worth USD 27.51 billion.
- From 2026 to 2036, the market is projected to expand at a CAGR of 8.0%.
- The market is projected to create an incremental opportunity of USD 14.77 billion between 2026 and 2036.
- In 2026, Biologic Therapeutics are expected to account for 44% of the product segment, driven by expanding commercial adoption of monoclonal antibodies, recombinant proteins, and fusion proteins targeting rare neurological indications.
- China (12.4%) and India (11.8%) are two of the fastest growing markets in the world.
- Demand and Growth Drivers
- Demand is shaped by expanding commercial launches of disease-modifying biologics for rare neurological indications including spinal muscular atrophy (SMA), Duchenne muscular dystrophy (DMD), Huntington's disease, and amyotrophic lateral sclerosis (ALS), where premium pricing and orphan drug exclusivity underpin commercial returns.
- Growth reflects regulatory support under the U.S. Orphan Drug Act, EU Orphan Regulation, Japan's Sakigake designation, and China's Priority Review pathways, which shorten approval timelines and extend market exclusivity for rare disease biologics.
- Adoption is increasing due to advances in genetic diagnostic capability, newborn screening expansion, and patient advocacy infrastructure that accelerates identification of eligible patients and builds early commercial momentum for newly approved therapies.
- Product and Segment View
- Biologic Therapeutics lead the product segment at 44% share in 2026, anchored by monoclonal antibodies, recombinant proteins, and fusion proteins targeting rare neurological indications.
- Oligonucleotide therapies including antisense oligonucleotides (ASOs) and siRNA-based biologics represent the fastest-growing product pool, supported by commercial success of Spinraza, Evrysdi-adjacent platforms, and next-generation ASO programs.
- Gene therapy products including AAV-based therapies and CRISPR-enabled therapeutics anchor the premium price tier, with Zolgensma-style one-time treatments reshaping commercial and payer dynamics.
- Geography and Competitive Outlook
- Asia Pacific leads growth, with China at a 12.4% CAGR driven by rare disease policy reform, National Reimbursement Drug List expansion, and accelerated regulatory approval pathways.
- India (11.8%) and South Korea (10.6%) follow, supported by rare disease policy expansion, government financial assistance schemes, and growing biotech clinical infrastructure.
- Germany (8.9%) leads European growth through strong reimbursement for orphan drugs, while the United States (7.4%) remains the largest absolute market. Leading players include Biogen, Novartis, Roche, Pfizer, Sanofi, Eli Lilly, AstraZeneca, Ionis Pharmaceuticals, Alnylam, BioMarin, Vertex, and PTC Therapeutics.
- Analyst Opinion
- Neurological rare disease biologics is one of the most commercially attractive segments in pharmaceutical therapeutics, anchored by premium pricing, orphan drug exclusivity, and structurally underserved patient populations. The segment has been reshaped over the past decade by commercial breakthroughs including Biogen's Spinraza and Roche's Evrysdi in SMA, and by gene therapy entrants including Novartis Zolgensma. Ionis Pharmaceuticals anchors the antisense oligonucleotide platform, while Alnylam leads in siRNA. The most significant commercial shift over the forecast period will come from three sources: first, the maturation of AAV gene therapy platforms that enable one-time dosing economics; second, the commercial expansion of oligonucleotide platforms into Huntington's disease, ALS, and prion diseases; and third, the gradual commercial viability of neurological indications in emerging markets where patient populations are large but historical reimbursement has been limited. The competitive risks are payer pushback against premium pricing, durability uncertainty for gene therapies, and the growing commercial importance of diagnostic infrastructure that determines addressable patient populations.
Why is the Neurological Rare Disease Biologics Market Growing?
- Orphan drug regulatory pathways combined with premium pricing create strong commercial returns that sustain biotech and specialty pharma investment.
- Genetic diagnostic expansion and newborn screening programs accelerate patient identification and build early commercial momentum for approved therapies.
- Advancing oligonucleotide and gene therapy platforms extend therapeutic reach into previously untreatable neurological indications.
The structural growth driver is the commercial attractiveness of orphan drug regulatory pathways, which combine shortened development timelines, market exclusivity, and premium pricing to generate strong returns on investment in rare disease biologics. The U.S. Orphan Drug Act provides seven years of market exclusivity and tax credits for qualified clinical testing, while the EU Orphan Regulation provides ten years of market exclusivity. Japan's Sakigake designation and China's Priority Review pathways have emerged as important additional regulatory accelerators. These frameworks support biotech and specialty pharma investment despite the small patient populations, because per-patient pricing of USD 300,000 to USD 3 million annually supports commercial viability.
The second driver is the expansion of genetic diagnostic capability and newborn screening programs, which accelerate patient identification and build early commercial momentum for newly approved therapies. Universal SMA newborn screening implementation across the United States, most European countries, and multiple Asian markets has dramatically changed the commercial trajectory for SMA biologics, enabling pre-symptomatic treatment initiation. Similar diagnostic expansion is emerging for Duchenne muscular dystrophy, lysosomal storage disorders, and rare epilepsies, creating structural demand growth as identification rates improve.
The third driver is the maturation of oligonucleotide and gene therapy platforms that extend therapeutic reach into previously untreatable neurological indications. Antisense oligonucleotide platforms developed by Ionis Pharmaceuticals, Biogen, and others are extending into Huntington's disease, ALS, and prion diseases. AAV gene therapy platforms are advancing in Duchenne muscular dystrophy (Sarepta Elevidys), Batten disease, and other pediatric neurological conditions. CRISPR-enabled therapeutics are entering clinical development for rare neurogenetic conditions. Each platform expansion opens new addressable patient populations and supports long-term market growth.
Segment-wise Analysis of Neurological Rare Disease Biologics Market
- Biologic Therapeutics lead the product segment at 44% share in 2026, anchored by monoclonal antibodies, recombinant proteins, and fusion proteins.
- Oligonucleotide therapies represent the fastest-growing segment, with antisense oligonucleotides and siRNA-based biologics expanding commercial reach.
- Gene therapy products anchor the premium price tier with Zolgensma and Elevidys-style one-time treatments reshaping commercial and payer dynamics.
The market segments across product type, therapeutic indication, patient population, technology platform, distribution channel, and region. Product coverage spans biologic therapeutics (monoclonal antibodies, recombinant proteins, fusion proteins), gene therapy products (AAV-based gene therapies, CRISPR-enabled therapeutics), oligonucleotide therapies (antisense oligonucleotides, siRNA-based biologics), cell-based biologics (stem cell-derived therapies, neural cell replacement therapies), and enzyme replacement therapies (recombinant enzymes, neuroprotective enzyme therapies).
Biologic Therapeutics Lead the Product Segment
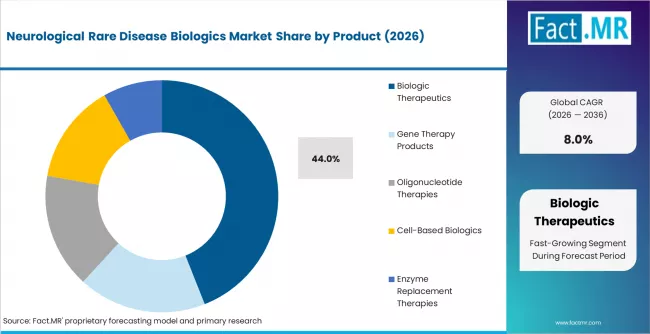
Biologic therapeutics account for 44% of product revenue in 2026. The subsegment is anchored by monoclonal antibodies, recombinant proteins used in enzyme replacement therapy for lysosomal storage disorders with neurological manifestations, and fusion proteins in late-stage development. Premium pricing ranging from USD 300,000 to USD 700,000 annually per patient supports strong revenue per unit despite small patient populations.
Commercial leaders in this subsegment include Roche, Biogen with multiple programs, Sanofi in lysosomal storage disorders, Eli Lilly with recently acquired assets, and emerging biotechs with novel monoclonal antibody and fusion protein approaches. The commercial dynamics favor established biopharma companies with global commercial infrastructure for rare disease products, which is a significant barrier to entry for emerging biotech competitors.
Oligonucleotide Therapies Are the Fastest-Growing Segment
Oligonucleotide therapies represent approximately 27% of 2026 revenue but are the fastest-growing segment by CAGR. Antisense oligonucleotides (ASOs) are anchored by Biogen's Spinraza in SMA and a growing pipeline across Huntington's disease, ALS, and rare epilepsies. siRNA-based biologics are led by Alnylam's platform extending into rare neurological indications. The commercial attraction is the platform generalizability, which allows a single technology platform to serve multiple rare indications, improving capital efficiency for developers.
Gene therapy products account for approximately 15% of 2026 revenue, anchored by Novartis Zolgensma in SMA and Sarepta Elevidys in Duchenne muscular dystrophy. The premium pricing of USD 2 to 3 million per one-time treatment reshapes commercial and payer dynamics, with outcome-based contracting and annuity payment structures becoming more common. Cell-based biologics and enzyme replacement therapies round out the segmentation with specialized applications in specific rare neurological indications.
Key Growth Drivers, Constraints, and Market Scope
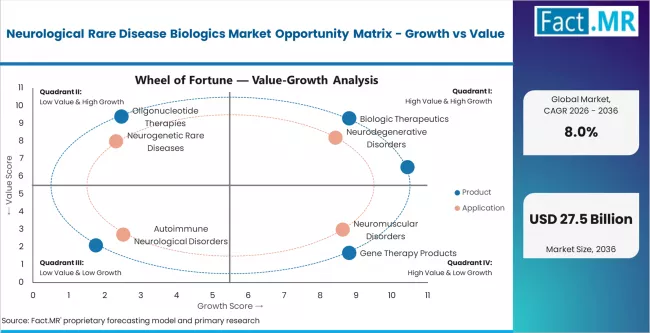
- Orphan drug regulatory incentives, genetic diagnostic expansion, and platform technology maturation drive sustained commercial growth through 2036.
- Payer pushback on premium pricing, small patient populations, and durability uncertainty for gene therapies constrain commercial upside.
- Emerging market expansion, newborn screening broadening, and next-generation platform technologies create meaningful upside opportunities.
The neurological rare disease biologics market has matured from a niche therapeutic category into a major commercial opportunity anchored by premium pricing and orphan drug exclusivity. The key variables over the forecast period are payer willingness to sustain current pricing levels, diagnostic infrastructure development pace in emerging markets, and the clinical and commercial success rate of next-generation gene therapy and oligonucleotide platforms.
Growth Drivers
Three primary forces drive growth. First, orphan drug regulatory pathways combined with premium pricing create strong commercial returns that sustain biotech and specialty pharma investment. Second, genetic diagnostic capability expansion and newborn screening program growth accelerate patient identification and build early commercial momentum for approved therapies. Third, maturation of oligonucleotide platforms, AAV gene therapy, and emerging CRISPR-enabled therapeutics extends therapeutic reach into previously untreatable neurological indications, creating structural market expansion.
Constraints
Payer pushback on premium pricing is the most significant commercial constraint. European reimbursement authorities and the U.S. Inflation Reduction Act price negotiation provisions create downward pricing pressure, and some rare disease products face coverage restrictions or step-therapy requirements that slow commercial uptake. Small patient populations create absolute market size ceilings that limit commercial scale for individual products. Durability uncertainty for gene therapies creates payer concerns about long-term value, particularly when annual therapies are available as alternatives. Manufacturing complexity for gene and cell therapies creates supply constraints and cost pressure that affect commercial economics.
Opportunities
The largest near-term opportunity is in emerging market expansion, where rare disease policy frameworks in China, India, Brazil, and other large markets are maturing and creating commercial viability for premium-priced rare disease biologics. A second opportunity is in newborn screening program expansion, which structurally grows addressable patient populations. A third opportunity is in next-generation platform technologies including base editing, prime editing, and improved AAV serotypes that enable new indications and improved commercial outcomes. Combination therapy approaches including biologic-gene therapy combinations represent a fourth opportunity area for differentiated clinical and commercial positioning.
Regional Outlook Across Key Markets
.webp)
- Asia Pacific leads growth, with China at a 12.4% CAGR driven by rare disease policy reform and NRDL expansion.
- India (11.8%) and South Korea (10.6%) follow, anchored by rare disease policy expansion and growing biotech infrastructure.
- Japan (9.7%) and Germany (8.9%) support strong mid-tier growth through established reimbursement frameworks.
- Brazil (8.2%) anchors Latin America, while the United States (7.4%) remains the largest absolute market.
Country CAGR Table
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 12.4% |
| India | 11.8% |
| South Korea | 10.6% |
| Japan | 9.7% |
| Germany | 8.9% |
| Brazil | 8.2% |
| United States | 7.4% |
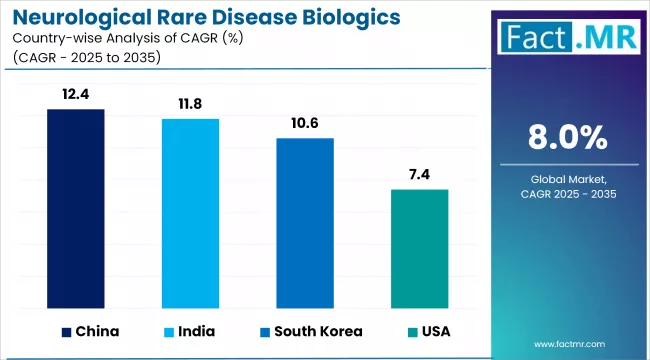
Regional performance reflects the interplay between rare disease policy frameworks, reimbursement coverage, diagnostic infrastructure, and healthcare system maturity. Asia Pacific leads growth because it combines recent policy reform with large untreated patient populations. Developed markets in North America and Europe grow more slowly but represent larger absolute revenue bases, driven by established reimbursement frameworks and commercial infrastructure.
China
China is the fastest-growing major market, with a 12.4% CAGR through 2036. The country's rare disease policy reform including the First List of Rare Diseases (2018) and Second List (2023), combined with National Reimbursement Drug List (NRDL) expansion for orphan drugs, has dramatically changed commercial viability. Multiple rare disease biologics are now NRDL-listed, with substantial commercial uptake following coverage.
- Rare Disease Catalog expansion and NRDL orphan drug coverage drive structural commercial uptake.
- Priority Review pathways and conditional approval under the National Medical Products Administration accelerate market entry.
- Domestic biotech growth combined with multinational commercial partnerships supports rapid commercial scaling.
India
India posts an 11.8% CAGR through 2036. The country's National Policy for Rare Diseases (NPRD 2021) and Umbrella Scheme for rare disease treatment funding have created commercial viability for premium-priced rare disease biologics. Patient advocacy groups including the Organization for Rare Diseases India (ORDI) anchor patient identification and access initiatives.
- National Policy for Rare Diseases 2021 and government funding schemes support commercial viability.
- Genetic diagnostic infrastructure expansion through public programs and private laboratories accelerates patient identification.
- Patient advocacy and managed access programs anchor commercial uptake in the absence of comprehensive insurance coverage.
South Korea
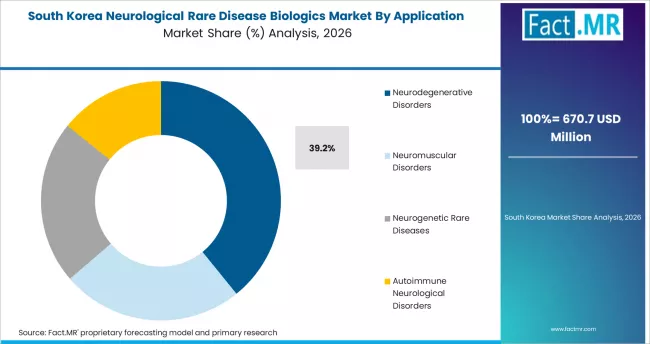
South Korea records a 10.6% CAGR through 2036. The country's Rare and Intractable Disease Support Program combined with national health insurance coverage for designated rare diseases creates strong commercial infrastructure. Samsung Medical Center, Seoul National University Hospital, and Asan Medical Center anchor specialty care delivery.
- National health insurance coverage for designated rare diseases supports commercial uptake.
- Samsung Medical Center, Asan Medical Center, and SNU Hospital anchor specialty care delivery infrastructure.
- K-biotech policy and clinical trial infrastructure support rapid adoption of newly approved biologics.
Japan
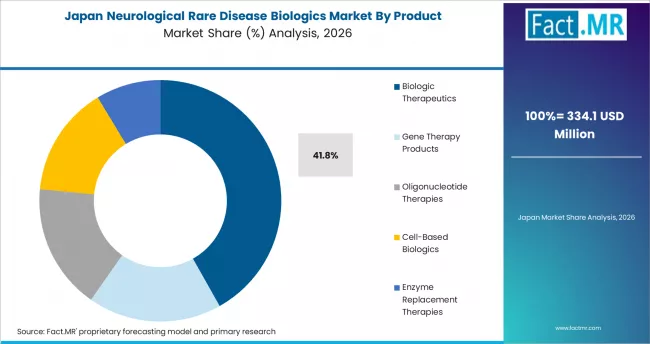
Japan posts a 9.7% CAGR through 2036. The country's Sakigake designation, Orphan Drug Program, and comprehensive national health insurance coverage create strong commercial infrastructure for rare disease biologics. Multiple global biologics have launched in Japan with PMDA approval and reimbursement coverage.
- Sakigake designation and Orphan Drug Program accelerate regulatory approval and market entry.
- Comprehensive national health insurance coverage supports premium-priced biologics commercial viability.
- Takeda, Astellas, and Daiichi Sankyo anchor domestic rare disease biologics commercial infrastructure.
Germany
Germany records an 8.9% CAGR through 2036. The country anchors European commercial leadership through strong G-BA reimbursement assessment framework, comprehensive statutory health insurance coverage, and established rare disease centers of excellence. Multiple rare disease biologics launch in Germany as first European market following EMA approval.
- G-BA reimbursement assessment and statutory health insurance coverage support premium biologics pricing.
- Rare disease centers of excellence including Charité, Heidelberg, and Munich anchor specialty care delivery.
- Domestic commercial infrastructure and orphan drug policy framework support first-to-launch dynamics.
Brazil
Brazil posts an 8.2% CAGR through 2036. The country's National Policy for the Comprehensive Attention to People with Rare Diseases (Portaria 199/2014) and SUS rare disease centers create public sector commercial access. High-cost biologics are covered through specialty pharmaceutical programs at the federal level.
- Portaria 199/2014 rare disease policy and SUS specialty pharmaceutical programs support public sector access.
- Federal high-cost drug programs anchor commercial viability for premium-priced rare disease biologics.
- Patient advocacy coordination and genetic diagnostic expansion support growing patient identification.
United States
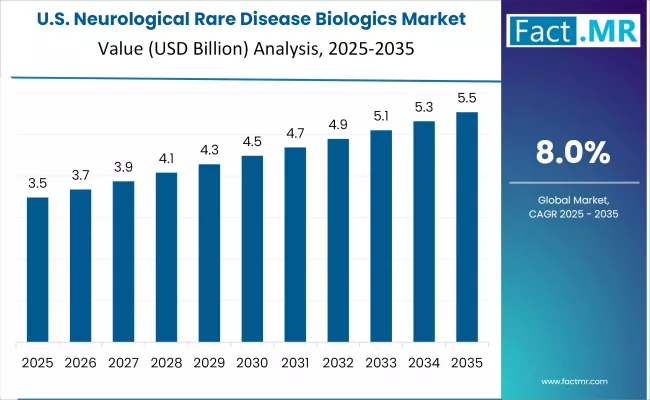
The United States is the largest absolute market, with 2026 revenue estimated at approximately USD 4.1 billion and a 7.4% CAGR through 2036. The country anchors global commercial infrastructure for rare disease biologics through FDA orphan drug designation, premium commercial pricing, private insurance and Medicare coverage, and established specialty pharmacy distribution.
- FDA orphan drug designation, Breakthrough Therapy, and Accelerated Approval pathways anchor global regulatory leadership.
- Private insurance and Medicare coverage combined with premium commercial pricing support strong per-patient revenue.
- Biogen, Ionis, Alnylam, BioMarin, Vertex, and PTC Therapeutics anchor commercial platform leadership.
Competitive Benchmarking and Company Positioning
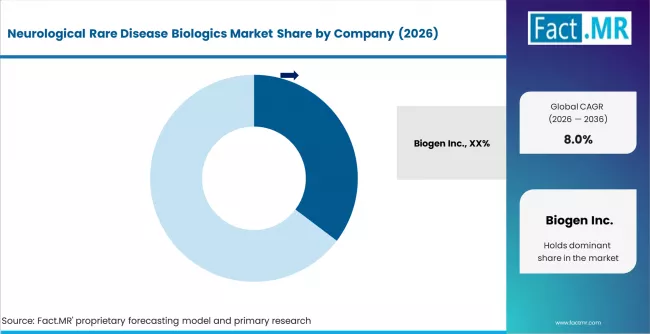
- Biogen, Roche, Novartis, and Sanofi anchor global commercial leadership in neurological rare disease biologics through broad product portfolios and established specialty sales infrastructure.
- Ionis Pharmaceuticals, Alnylam Pharmaceuticals, and BioMarin anchor platform-technology leadership in oligonucleotides, siRNA, and enzyme replacement therapies.
- Vertex, PTC Therapeutics, and specialty biotechs compete in specific indications with differentiated clinical and commercial positioning.
The neurological rare disease biologics market is moderately concentrated, with the top ten companies collectively controlling approximately 65 to 70% of global revenue. Biogen holds the largest commercial position, driven by Spinraza in SMA and a broad rare neurological pipeline. Roche has emerged as a major player through Evrysdi and PTC Therapeutics partnership. Novartis anchors gene therapy leadership through Zolgensma, and Sanofi leads in lysosomal storage disorders with neurological manifestations.
Ionis Pharmaceuticals holds a differentiated position as the leading antisense oligonucleotide platform developer, with Spinraza (partnered with Biogen), Qalsody for SOD1-ALS, and a broad pipeline across Huntington's disease, ALS, and rare epilepsies. Alnylam Pharmaceuticals leads in siRNA platforms, with programs extending into rare neurological indications alongside established TTR amyloidosis franchise. BioMarin anchors enzyme replacement therapy leadership for lysosomal storage disorders.
Vertex Pharmaceuticals has expanded into rare neurological indications through recent pipeline development and acquisitions. PTC Therapeutics anchors specific rare indication leadership through approved products and advancing pipeline. Eli Lilly, Pfizer, and AstraZeneca compete in selected rare neurological indications through internal development and partnerships. Emerging biotechs including Sarepta Therapeutics in Duchenne muscular dystrophy, BioCryst Pharmaceuticals, and others compete in specific indications.
The competitive dynamics favor companies with three attributes: established specialty sales and managed access infrastructure for rare disease products, platform technology that supports multiple rare indications efficiently, and strong regulatory and reimbursement capability for orphan drug commercial launches. Biogen leads on specialty sales infrastructure. Ionis and Alnylam lead on platform technology. Novartis leads on gene therapy commercial experience. The commercial winners over the next decade will combine these attributes with strong clinical development execution and payer engagement capability.
Competitive Analysis
Strategic positioning is shaped by the choice between platform-technology leadership and indication-specific commercial excellence. Platform companies including Ionis, Alnylam, and gene therapy specialists pursue capital-efficient development across multiple rare indications using shared technology foundations. Indication-specific specialists including Sarepta in Duchenne muscular dystrophy and PTC Therapeutics in specific rare conditions pursue deep commercial engagement with concentrated patient populations. Large biopharma companies including Biogen, Roche, Novartis, and Sanofi pursue both approaches in parallel, with broad portfolios and platform technology investments. The commercial winners over the next decade will combine platform efficiency with indication-specific commercial depth, while maintaining payer and advocacy engagement capability that supports premium commercial positioning.
Benchmarking Table
| Company | Portfolio Breadth | Platform Leadership | Specialty Sales | Payer Engagement |
|---|---|---|---|---|
| Biogen | High | High (ASO) | Very High | Very High |
| Roche | Very High | High | Very High | Very High |
| Novartis | Very High | Very High (Gene Tx) | Very High | Very High |
| Ionis | Medium-High | Very High (ASO) | Medium | High |
| Alnylam | Medium | Very High (siRNA) | High | High |
Recent Developments
- Biogen and Ionis advanced multiple antisense oligonucleotide programs in 2025 and 2026 across Huntington's disease, ALS, and rare epilepsies, building on Spinraza commercial platform leadership.
- Novartis expanded Zolgensma commercial access globally in 2025 and 2026, with expanded pediatric coverage and outcome-based contracting arrangements in additional markets.
- Roche and PTC Therapeutics advanced Evrysdi commercial expansion and next-generation SMA programs in 2025 and 2026, reinforcing their position across the commercial franchise.
Leading Companies Shaping Neurological Rare Disease Biologics Market
-
Major Players
- Biogen Inc.
- F. Hoffmann-La Roche AG
- Novartis AG
- Pfizer Inc.
- Sanofi S.A.
- Eli Lilly and Company
- AstraZeneca PLC
- Ionis Pharmaceuticals Inc.
-
Emerging Players
- Alnylam Pharmaceuticals Inc.
- BioMarin Pharmaceutical Inc.
- Vertex Pharmaceuticals Inc.
- PTC Therapeutics Inc.
- Sarepta Therapeutics Inc.
- BioCryst Pharmaceuticals Inc.
- Arcturus Therapeutics Holdings Inc.
- Stoke Therapeutics Inc.
- Wave Life Sciences Ltd.
- Ultragenyx Pharmaceutical Inc.
Sources and Research References
- U.S. Food and Drug Administration (FDA) orphan drug designation database and rare disease approval tracking
- European Medicines Agency (EMA) orphan drug database and COMP Committee reports
- World Health Organization (WHO) rare disease classification and prevalence estimates
- National Organization for Rare Disorders (NORD), EURORDIS, and Organization for Rare Diseases India (ORDI) publications
- Biogen, Roche, Novartis, Ionis, and Alnylam corporate filings, clinical trial reports, and investor presentations
- ClinicalTrials.gov clinical trial registry and rare disease patient registry publications
- Primary interviews with neurologists, rare disease advocacy group leaders, payer medical directors, and biotech commercial executives
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Key Questions This Report Addresses
- What is the size of the neurological rare disease biologics market in 2026 and 2036?
- What CAGR is the market expected to record between 2026 and 2036?
- Which product segments lead the market in 2026?
- How are orphan drug regulatory pathways shaping commercial economics?
- Which countries are the fastest-growing markets through 2036?
- How are newborn screening programs affecting commercial uptake?
- Who are the leading companies, and how are they competitively differentiated?
- How does Fact.MR estimate and validate the market forecast?
Neurological Rare Disease Biologics Market Definition
The Neurological Rare Disease Biologics market covers biologic therapeutics approved or in late-stage development for rare neurological indications with prevalence below regulatory thresholds for orphan drug designation. The scope spans biologic therapeutics (monoclonal antibodies, recombinant proteins, fusion proteins), gene therapy products (AAV-based and CRISPR-enabled), oligonucleotide therapies (antisense oligonucleotides and siRNA-based biologics), cell-based biologics (stem cell-derived and neural cell replacement therapies), and enzyme replacement therapies for lysosomal storage disorders with neurological manifestations.
Neurological Rare Disease Biologics Market Inclusions
The market includes biologic therapeutics commercially available or in Phase 2/3 clinical development for rare neurological indications, covering SMA, Duchenne muscular dystrophy, Huntington's disease, ALS, rare epilepsies, lysosomal storage disorders with neurological manifestations, rare leukodystrophies, and ultra-rare neurogenetic conditions. Revenue streams include drug product sales, managed access programs, expanded access revenue, and contracted patient support services. Orphan drug designated products with neurological indications are included regardless of whether the indication is the lead or follow-on approval.
Neurological Rare Disease Biologics Market Exclusions
The market excludes biologics for common neurological conditions such as multiple sclerosis, Alzheimer's disease, Parkinson's disease, and common epilepsies where patient populations exceed orphan drug prevalence thresholds. Small molecule therapies for rare neurological indications are outside the scope, as are supportive care products, diagnostic imaging agents, and medical devices. Early-stage clinical programs before Phase 2 proof-of-concept are excluded from near-term commercial forecasts, though they may be tracked for pipeline visibility.
Neurological Rare Disease Biologics Market Research Methodology
- Primary Research: Interviews with neurologists and neuromuscular specialists, rare disease advocacy group leaders, hospital pharmacy directors, payer medical directors, and commercial heads at biotech and specialty pharma companies.
- Desk Research: FDA, EMA, PMDA, NMPA, and KFDA approval databases, orphan drug designation lists, ClinicalTrials.gov data, company annual reports and investor presentations, and rare disease patient registry publications.
- Market Sizing: Forward-looking model built on epidemiology-based patient population estimates, diagnosis and treatment rate assumptions, pricing data, reimbursement coverage analysis, and product-level revenue projections.
- Data Validation: Cross-checked against company financial disclosures, rare disease registry data, payer formulary coverage reports, and primary interview triangulation. Sensitivity testing on diagnosis rates, treatment uptake, and pricing assumptions.
Scope of Analysis
| Parameter | Details |
|---|---|
| Quantitative Units | USD 12.74 billion in 2026 to USD 27.51 billion by 2036, at a CAGR of 8.0% |
| Market Definition | Biologic therapeutics approved or in late-stage development for rare neurological indications with prevalence below regulatory thresholds for orphan drug designation, covering biologic therapeutics, gene therapy, oligonucleotide therapies, cell-based biologics, and enzyme replacement therapies. |
| Regions | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries | China, India, South Korea, Japan, Germany, Brazil, United States, and 25+ additional countries |
| Companies | Biogen, Roche, Novartis, Pfizer, Sanofi, Eli Lilly, AstraZeneca, Ionis Pharmaceuticals, Alnylam, BioMarin, Vertex, PTC Therapeutics |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid demand-side and top-down methodology built on epidemiology-based patient population estimates, treatment rate assumptions, pricing and reimbursement data, and primary interviews with neurologists, payer medical directors, and biotech commercial leads. |
Analysis by Product, Indication, Patient Population, Platform, Distribution Channel, and Region
-
Neurological Rare Disease Biologics Market Market Segmented by Product:
- Biologic Therapeutics
- Monoclonal Antibodies
- Recombinant Proteins
- Fusion Proteins
- Monoclonal Antibodies
- Gene Therapy Products
- AAV-Based Gene Therapies
- CRISPR-Enabled Therapeutics
- AAV-Based Gene Therapies
- Oligonucleotide Therapies
- Antisense Oligonucleotides (ASOs)
- siRNA-Based Biologics
- Antisense Oligonucleotides (ASOs)
- Cell-Based Biologics
- Stem Cell-Derived Therapies
- Neural Cell Replacement Therapies
- Stem Cell-Derived Therapies
- Enzyme Replacement Therapies
- Recombinant Enzymes
- Neuroprotective Enzyme Therapies
- Recombinant Enzymes
- Biologic Therapeutics
-
Neurological Rare Disease Biologics Market Market Segmented by Application:
- Neurodegenerative Disorders
- Alzheimer’s Disease
- Parkinson’s Disease
- Huntington’s Disease
- Alzheimer’s Disease
- Neuromuscular Disorders
- Spinal Muscular Atrophy (SMA)
- Amyotrophic Lateral Sclerosis (ALS)
- Spinal Muscular Atrophy (SMA)
- Neurogenetic Rare Diseases
- Lysosomal Storage Disorders
- CNS Developmental Disorders
- Lysosomal Storage Disorders
- Autoimmune Neurological Disorders
- Multiple Sclerosis (Rare Subtypes)
- Neuroinflammatory Conditions
- Multiple Sclerosis (Rare Subtypes)
- Neurodegenerative Disorders
-
Neurological Rare Disease Biologics Market Market Segmented by End Use:
- Pharmaceutical & Biotechnology Companies
- Rare Disease Drug Developers
- Neurobiology-Focused Biotech Firms
- Rare Disease Drug Developers
- Specialty Clinics & Hospitals
- Neurology Specialty Centers
- Rare Disease Treatment Centers
- Neurology Specialty Centers
- Academic & Research Institutes
- Neuroscience Research Labs
- Translational Medicine Centers
- Neuroscience Research Labs
- Contract Research Organizations (CROs)
- Clinical Trial Service Providers
- Biologics Development CROs
- Clinical Trial Service Providers
- Pharmaceutical & Biotechnology Companies
-
Neurological Rare Disease Biologics Market Market Segmented by Technology:
- Gene Therapy Platforms
- AAV Vector Technology
- Lentiviral Systems
- AAV Vector Technology
- RNA-Based Therapeutics
- ASO & siRNA Platforms
- mRNA Therapeutic Systems
- ASO & siRNA Platforms
- Monoclonal Antibody Engineering
- Humanized Antibody Platforms
- Bispecific Antibodies
- Humanized Antibody Platforms
- Cell & Regenerative Therapy Tech
- Neural Stem Cell Engineering
- Induced Pluripotent Stem Cells (iPSC)
- Neural Stem Cell Engineering
- Precision Medicine & Biomarker Tech
- Genomic Profiling Tools
- AI-Driven Target Discovery
- Genomic Profiling Tools
- Gene Therapy Platforms
-
Neurological Rare Disease Biologics Market Market Segmented by Distribution Channel:
- Direct Pharma Partnerships
- Biotech Licensing Agreements
- Orphan Drug Collaborations
- Biotech Licensing Agreements
- Specialty Pharmacies
- Rare Disease Drug Distributors
- Hospital Specialty Pharmacies
- Rare Disease Drug Distributors
- Hospital Procurement
- Institutional Supply Contracts
- Neurology Department Networks
- Institutional Supply Contracts
- Research Collaborations
- Academic-Industry Partnerships
- Government Funding Programs
- Academic-Industry Partnerships
- Direct Pharma Partnerships
-
Neurological Rare Disease Biologics Market Market Segmented by Packaging:
- Sterile Injectable Packaging
- Single-Dose Vials
- Prefilled Syringes
- Single-Dose Vials
- Cold Chain Packaging
- Ultra-Low Temperature Storage
- Cryogenic Transport Systems
- Ultra-Low Temperature Storage
- Lyophilized Drug Packaging
- Freeze-Dried Biologic Vials
- Reconstitution Kits
- Freeze-Dried Biologic Vials
- Gene Therapy Delivery Packaging
- Viral Vector Storage Containers
- Secure Biologic Transport Kits
- Viral Vector Storage Containers
- Sterile Injectable Packaging
-
Neurological Rare Disease Biologics Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
How big is the neurological rare disease biologics market in 2026?
The global neurological rare disease biologics market is valued at USD 12.74 billion in 2026.
What will be the size of the market in 2036?
The market is projected to reach USD 27.51 billion by 2036.
How fast is the market expected to grow between 2026 and 2036?
The market is expected to expand at a CAGR of 8.0% between 2026 and 2036.
Which product segment leads the market in 2026?
Biologic Therapeutics including monoclonal antibodies, recombinant proteins, and fusion proteins lead the product segment with 44% share in 2026.
Which country is the fastest-growing market?
China is the fastest-growing major market with a 12.4% CAGR through 2036.
Which is the second-fastest-growing market?
India is the second-fastest-growing market, expanding at an 11.8% CAGR through 2036.
What does the neurological rare disease biologics market include?
The market covers biologic therapeutics for rare neurological indications including SMA, Duchenne muscular dystrophy, Huntington's disease, ALS, rare epilepsies, and lysosomal storage disorders with neurological manifestations, spanning monoclonal antibodies, gene therapy, oligonucleotide therapies, cell-based biologics, and enzyme replacement therapies.
How does Fact.MR estimate and validate the market forecast?
The forecast is built on a hybrid demand-side and top-down methodology using epidemiology-based patient population estimates, treatment rate assumptions, pricing and reimbursement data, and primary interviews with neurologists, payer medical directors, and biotech commercial leads.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Biologic Therapeutics
- Monoclonal Antibodies
- _Recombinant Proteins
- _Fusion Proteins
- Gene Therapy Products
- AAV-Based Gene Therapies
- _CRISPR-Enabled Therapeutics
- Oligonucleotide Therapies
- Antisense Oligonucleotides (ASOs)
- _siRNA-Based Biologics
- Cell-Based Biologics
- Stem Cell-Derived Therapies
- _Neural Cell Replacement Therapies
- Enzyme Replacement Therapies
- Recombinant Enzymes
- _Neuroprotective Enzyme Therapies
- Biologic Therapeutics
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Neurodegenerative Disorders
- Alzheimer’s Disease
- _Parkinson’s Disease
- _Huntington’s Disease
- Neuromuscular Disorders
- Spinal Muscular Atrophy (SMA)
- _Amyotrophic Lateral Sclerosis (ALS)
- Neurogenetic Rare Diseases
- Lysosomal Storage Disorders
- _CNS Developmental Disorders
- Autoimmune Neurological Disorders
- Multiple Sclerosis (Rare Subtypes)
- _Neuroinflammatory Conditions
- Neurodegenerative Disorders
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical & Biotechnology Companies
- Rare Disease Drug Developers
- _Neurobiology-Focused Biotech Firms
- Specialty Clinics & Hospitals
- Neurology Specialty Centers
- _Rare Disease Treatment Centers
- Academic & Research Institutes
- Neuroscience Research Labs
- _Translational Medicine Centers
- Contract Research Organizations (CROs)
- Clinical Trial Service Providers
- _Biologics Development CROs
- Pharmaceutical & Biotechnology Companies
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Gene Therapy Platforms
- AAV Vector Technology
- _Lentiviral Systems
- RNA-Based Therapeutics
- ASO & siRNA Platforms
- _mRNA Therapeutic Systems
- Monoclonal Antibody Engineering
- Humanized Antibody Platforms
- _Bispecific Antibodies
- Cell & Regenerative Therapy Tech
- Neural Stem Cell Engineering
- _Induced Pluripotent Stem Cells (iPSC)
- Precision Medicine & Biomarker Tech
- Genomic Profiling Tools
- _AI-Driven Target Discovery
- Gene Therapy Platforms
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct Pharma Partnerships
- Biotech Licensing Agreements
- _Orphan Drug Collaborations
- Specialty Pharmacies
- Rare Disease Drug Distributors
- _Hospital Specialty Pharmacies
- Hospital Procurement
- Institutional Supply Contracts
- _Neurology Department Networks
- Research Collaborations
- Academic-Industry Partnerships
- _Government Funding Programs
- Direct Pharma Partnerships
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging, 2026 to 2036
- Sterile Injectable Packaging
- Single-Dose Vials
- _Prefilled Syringes
- Cold Chain Packaging
- Ultra-Low Temperature Storage
- _Cryogenic Transport Systems
- Lyophilized Drug Packaging
- Freeze-Dried Biologic Vials
- _Reconstitution Kits
- Gene Therapy Delivery Packaging
- Viral Vector Storage Containers
- _Secure Biologic Transport Kits
- Sterile Injectable Packaging
- Y to o to Y Growth Trend Analysis By Packaging, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- By End Use
- By Technology
- By Distribution Channel
- By Packaging
- Competition Analysis
- Competition Deep Dive
- Biogen Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Novartis AG
- Pfizer Inc.
- Merck & Co. Inc.
- Johnson & Johnson
- Bayer AG
- Genzyme Corporation (Sanofi)
- Alexion Pharmaceuticals Inc.
- BioMarin Pharmaceutical Inc.
- Roche Holding AG
- Biogen Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Packaging, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Packaging, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Distribution Channel
- Figure 18: Global Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by Packaging
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 28: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 46: North America Market Attractiveness Analysis by Distribution Channel
- Figure 47: North America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 49: North America Market Attractiveness Analysis by Packaging
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 56: Latin America Market Attractiveness Analysis by Application
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 62: Latin America Market Attractiveness Analysis by Technology
- Figure 63: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 65: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 66: Latin America Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 68: Latin America Market Attractiveness Analysis by Packaging
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 75: Western Europe Market Attractiveness Analysis by Application
- Figure 76: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 78: Western Europe Market Attractiveness Analysis by End Use
- Figure 79: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 81: Western Europe Market Attractiveness Analysis by Technology
- Figure 82: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 84: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 85: Western Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 87: Western Europe Market Attractiveness Analysis by Packaging
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Application
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Packaging
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 113: East Asia Market Attractiveness Analysis by Application
- Figure 114: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 116: East Asia Market Attractiveness Analysis by End Use
- Figure 117: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 119: East Asia Market Attractiveness Analysis by Technology
- Figure 120: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 122: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 123: East Asia Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 125: East Asia Market Attractiveness Analysis by Packaging
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Packaging
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product,2026 to 2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Technology,2026 to 2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel,2026 to 2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Packaging, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging,2026 to 2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Packaging
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis