Self-Administered Biologics Market Size, Share, Growth and Forecast (2026 - 2036)
Self-administered Biologics Market is segmented by Product Type (Prefilled Syringes, Auto injectors, Pen Injectors, Wearable Injectors, and Others), by Therapeutic Area (Autoimmune & Chronic Diseases, Oncology Support Therapy, Infectious Diseases, Endocrinology, and Others), by Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, and Specialty Clinics) and Region. Forecast for 2026 to 2036.
Fact.MR opines the self-administered biologics market stands at a Device-Driven Differentiation Inflection, where the clinical and commercial value of a biologic is increasingly inseparable from its delivery platform, compelling manufacturers to compete not just on molecule efficacy but on injection experience, adherence technology, and patient-centric device design that payers are beginning to reward through formulary access and reimbursement differentiation.
Segmental Analysis
Self-administered Biologics Market Analysis by Product Type
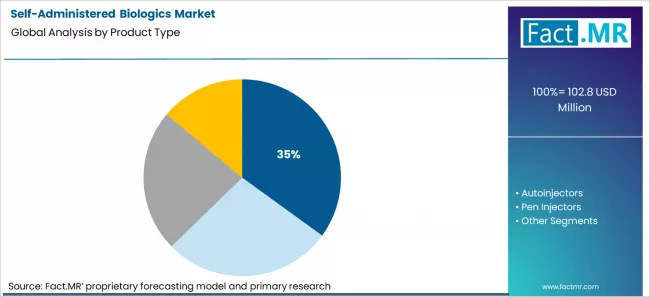
Prefilled syringes are projected to hold 35% share in 2026. They remain dominant because they are simple, widely compatible with existing manufacturing lines, and easier to launch globally without proprietary device complexity. This makes them the preferred format for biosimilars and rapid new product entry.
- Becton Dickinson announced a USD 1.2 billion investment in 2023 to expand prefilled syringe capacity and develop polymer based platforms suited for high concentration biologics [5].
- Auto injector innovation is advancing as well. Antares Pharma, now part of Halozyme Therapeutics, expanded commercial use of its VIBEX QuickShot platform to improve injection comfort and patient adherence [6].
Self-administered Biologics Market Analysis by Therapeutic Area
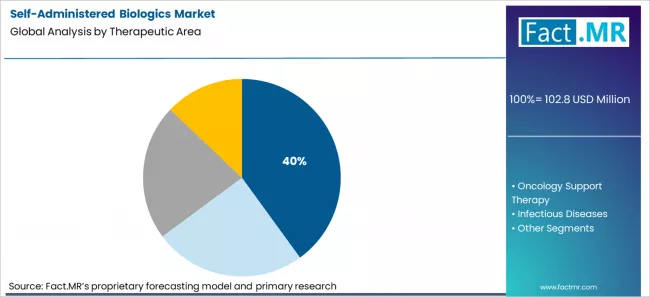
Autoimmune and chronic diseases are expected to hold 40% market share in 2026. Long treatment durations and high adherence needs make device experience critical in this segment. The large installed base of patients using self-injection continues to drive demand.
- In oncology, Roche and Genentech completed EU rollout of subcutaneous trastuzumab and pertuzumab, with rapid patient transition from IV infusion to faster injection formats [7].
- In endocrinology, Eli Lilly launched the connected Tempo Pen system in 2023, integrating insulin dosing with digital monitoring platforms and reinforcing the shift toward data enabled self-administration [8].
Drivers, Restraints, and Opportunities
FACT.MR analysts note that the self-administered biologics market has grown over two decades on the back of major autoimmune and endocrine blockbusters such as adalimumab, etanercept, and insulin glargine. The sector is now entering a second transition phase, where biosimilar competition and next generation biologics in oncology and rare diseases are reshaping revenue mix and growth patterns.
The central dynamic is clear. Biosimilar are lowering prices in mature autoimmune categories, reducing per unit revenue. At the same time, lower prices are expanding patient access and increasing total treatment volumes. Originator companies are shifting strategy toward device innovation, high concentration formulations, and subcutaneous versions of IV drugs. This product and device integration allows manufacturers to offset drug price pressure with device services, adherence tools, and patient support programs that generate recurring value.
- Biosimilar reimbursement mandates in Canada have accelerated switching from originators to lower cost alternatives, directly increasing autoinjector volumes across provinces [2].
- China’s National Healthcare Security Administration expanded its reimbursement list in 2023 to include more subcutaneous biologics, significantly enlarging the eligible patient pool for rheumatoid arthritis and psoriasis treatments [3].
- Wearable injectors are also gaining traction. High volume biologics now require delivery systems beyond traditional syringes. BD Medical’s Libertas and Enable Injections’ enJogo platforms entered commercial agreements in 2023 and 2024, marking the shift from pilot programs to scaled adoption [4].
Regional Analysis
The self-administered biologics market is analyzed across North America, Europe, Asia-Pacific, Latin America, and Middle East and Africa. Regional variation is driven by national reimbursement policy, biosimilar adoption rates, chronic disease prevalence, and specialty pharmacy infrastructure maturity. The full report offers detailed market attractiveness analysis comparing payer dynamics, device platform preferences, and competitive intensity by region.
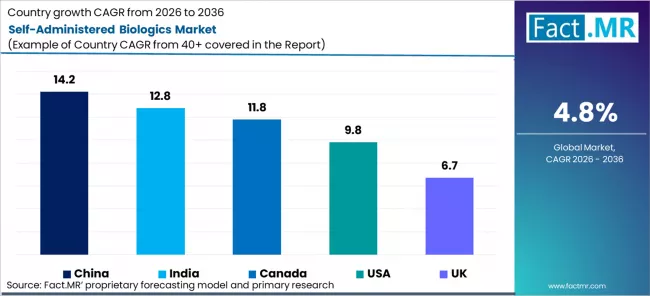
| Country | CAGR% |
|---|---|
| China | 14.2% |
| India | 12.8% |
| Canada | 11.9% |
| United States | 9.8% |
| United Kingdom | 6.7% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
North America Self-administered Biologics Market Analysis
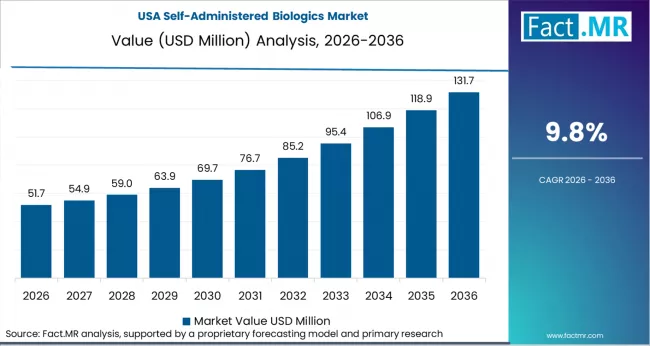
North America remains the largest revenue contributor to the self-administered biologics market. High list prices, strong specialty pharmacy networks, and complex payer systems protect originator revenues even as biosimilar gain share. Leading companies such as Amgen, AbbVie, and Eli Lilly and Company maintain dominance through payer contracts and proprietary delivery devices.
- United States: The U.S. market is projected to grow at 9.8% CAGR through 2036. Around five million patients use self-injectable autoimmune biologics supported by specialty pharmacies and pharmacy benefit managers. The Inflation Reduction Act will introduce Medicare drug price negotiations from 2026, affecting select biologics including insulin. Manufacturers are responding with device innovation and expanded domestic production to defend formulary position.
Europe Self-administered Biologics Market Analysis
Europe acts as a global testing ground for biosimilar policy. Reference pricing and national substitution programs have accelerated the shift from originators to biosimilars. Companies such as Sandoz and Samsung Bioepis compete strongly in tender driven systems where device compatibility influences procurement outcomes.
- United Kingdom: The UK market is expected to grow at 6.7% CAGR through 2036. NHS England’s biosimilar programme has rapidly shifted patients to lower cost adalimumab and etanercept autoinjectors. By 2024, biosimilars captured most rheumatology share. Updated NHS frameworks and NICE guidance support high concentration subcutaneous formulations, reinforcing device upgrades and standardizing biosimilar self-injection across secondary care.
Asia Pacific Self-administered Biologics Market Analysis
Asia Pacific offers the strongest volume growth potential. Rising chronic disease prevalence and expanding reimbursement are creating new patient populations. Domestic companies such as Biocon Biologics and 3SBio compete alongside multinational firms, with pricing and local manufacturing scale playing a central role.
- China: China is projected to grow at 14.2% CAGR through 2036. Expanded reimbursement under the National Healthcare Security Administration has widened access to subcutaneous biologics for rheumatology patients. Faster regulatory reviews for drug device combinations are reducing approval timelines. Domestic manufacturers are scaling autoinjector production, increasing competition and raising quality standards across the biologics sector.
- India: India is forecast to grow at 12.8% CAGR through 2036. Biocon Biologics expanded biosimilar insulin and trastuzumab pen injector distribution through government tenders in 2023. Updated guidance from the Central Drugs Standard Control Organisation clarified approval pathways for combination devices. National non communicable disease programmes are also expanding insulin access at primary care level, strengthening public demand.
- Canada: Canada is expected to grow at 11.9% CAGR through 2036. Provincial biosimilar transition policies have moved large patient populations onto lower cost autoinjectors. Health Canada updated combination product regulations in 2023, aligning with global standards. The Canadian Agency for Drugs and Technologies in Health now evaluates device features in reimbursement reviews, making delivery technology more central to market access decisions.
Competitive Aligners for Market Players
The self-administered biologics market is moderately concentrated among leading originator companies. Major manufacturers such as AbbVie, Amgen, Johnson & Johnson, Eli Lilly and Company, Pfizer, Roche, Sanofi, Bristol Myers Squibb, Merck & Co., and Novartis together generate roughly two thirds of global revenue. High development costs, long clinical timelines, and complex regulatory pathways limit entry to well capitalized firms.
The biosimilar segment is more fragmented. Companies such as Samsung Bioepis, Sandoz, Celltrion, Biocon Biologics, and Pfizer Biosimilars compete across selected molecules.
Competitive advantage increasingly depends on delivery devices, patient support programs, and payer contracts rather than molecule patents alone. AbbVie’s Humira lifecycle strategy showed how device upgrades can extend market leadership. Similarly, connected insulin pens and new subcutaneous formats help companies maintain reimbursement strength even after exclusivity ends.
Large pharmacy benefit managers shape access through formulary control and substitution mandates. As a result, manufacturers must prove that device features improve outcomes or reduce total care costs to secure preferred positioning at scale.
Key Players
- Amgen Inc.
- AbbVie Inc.
- Genentech, Inc.
- Johnson & Johnson
- Pfizer Inc.
- Sanofi S.A.
- Bristol Myers Squibb Company
- Merck & Co., Inc.
- GSK plc
- Regeneron Pharmaceuticals, Inc.
- Novartis AG
- Eli Lilly and Company
Report Address
- Market intelligence for strategic planning with analysis of self-injection platform migration from prefilled syringes toward autoinjectors, pen devices, and wearable injectors across originator and biosimilar segments.
- Market size and forecast showing valuation of USD 102.8 billion in 2026 and projected USD 164.2 billion by 2036 at 4.8% CAGR.
- Growth opportunity mapping across biosimilar autoinjector platforms, connected device programmes, high-concentration subcutaneous formulations, and online pharmacy distribution channel expansion.
- Segment and regional forecasts covering all major countries with product type, therapeutic area, and distribution channel breakdowns.
- Competition strategy assessment of originator and biosimilar manufacturers including payer contracting models, device platform investment strategies, and specialty pharmacy partnership structures.
- Product and compliance tracking of FDA drug-device combination approval pathways, EMA biosimilar guidance updates, and national health technology assessment reimbursement policy changes affecting self-injection biologic access.
- Biosimilar market penetration analysis comparing transition rate timelines and device substitution dynamics across North America, Europe, and Asia-Pacific.
- Report delivery in Excel, PowerPoint, and PDF formats with fully validated data, primary research evidence, and source documentation.
Bibliography
- [1] Stevanato Group. (2026, January 16). Datwyler, LTS Device Technologies, and Stevanato Group collaborate to expand self-delivery options for large-volume drugs
- [2] British Columbia Ministry of Health, Biosimilar Drugs Policy Update, May 2019 (updated through 2023).
- [3] National Healthcare Security Administration, People's Republic of China, 2023 National Reimbursement Drug List Announcement, December 2023.
- [4] Enable Injections, Commercial Partnership Announcements 2024, March 2024.
- [5] Becton Dickinson and Company, Fiscal Year 2023 Annual Report and Investor Day Presentation, November 2023.
- [6] Halozyme Therapeutics, VIBEX QuickShot Partnership Programme Update, Annual Report 2023, February 2024.
- [7] Enable Injections, enJogo Wearable Injector Commercial Agreement Announcement, January 2024.
- [8] AbbVie Inc., Annual Report 2023 - Immunology Pipeline Section, February 2024.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD 102.8 billion (2026) to USD 164.2 billion (2036), at a CAGR of 4.8% |
| Market Definition | Self-administered biologics are injectable biological medicines, including monoclonal antibodies, insulin analogues, and cytokine inhibitors, packaged in patient-operable autoinjectors, prefilled syringes, pen devices, or wearable pumps for long-term home management of chronic autoimmune, endocrine, oncology, and infectious disease indications. |
| Product Type | Prefilled Syringes, Autoinjectors, Pen Injectors, Wearable, Injectors |
| Therapeutic Area | Autoimmune & Chronic Diseases, Oncology Support Therapy, Infectious Diseases, Endocrinology |
| Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Clinics |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, Australia & New Zealand, India, ASEAN, Rest of Asia Pacific, Norway, Germany, United Kingdom, France, Spain, Netherlands, Nordics, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Ecuador, Rest of Latin America, Kingdom of Saudi Arabia, United Arab Emirates, South Africa, Turkey, Rest of Middle East & Africa |
| Key Companies Profiled | Amgen Inc., AbbVie Inc., Genentech, Inc., Johnson & Johnson, Pfizer Inc., Sanofi S.A., Bristol Myers Squibb Company, Merck & Co., Inc., GSK plc, Regeneron Pharmaceuticals, Inc., Novartis AG, and Eli Lilly and Company. |
| Forecast Period | 2026 to 2036 |
| Approach | Top down and bottom up market modeling validated through primary interviews with biologic manufacturers, biosimilar developers, specialty pharmacy executives, hospital procurement heads, and regulatory consultants, supported by national reimbursement data, health technology assessment reports, biologic sales disclosures, clinical trial databases, and company reported revenue and manufacturing investment figures. |
Self-administered Biologics Market by Segment
-
By Product Type :
- Prefilled Syringes
- Autoinjectors
- Pen Injectors
- Wearable Injectors
-
By Therapeutic Area :
- Autoimmune & Chronic Diseases
- Oncology Support Therapy
- Infectious Diseases
- Endocrinology
-
By Distribution Channel :
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Specialty Clinics
-
Region :
- Asia Pacific
- China
- Japan
- South Korea
- Australia & New Zealand
- India
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Spain
- Italy
- Nordics
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- United Arab Emirates
- Kingdom of Saudi Arabia
- South Africa
- Rest of Middle East & Africa
- Asia Pacific
- Frequently Asked Questions -
How large is the self-administered biologics market in 2025?
The self-administered biologics market was valued at USD 98.3 billion in 2025.
What will the market size be in 2026?
The market is estimated to grow to USD 102.8 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 164.2 billion by 2036.
What is the expected CAGR for the forecast period 2026 to 2036?
FACT.MR projects a CAGR of 4.8% from 2026 to 2036.
Which Product Type segment holds the largest market share?
Prefilled Syringes lead the Product Type segment with 35% share in 2026.
Which Therapeutic Area segment dominates in 2026?
Autoimmune & Chronic Diseases holds 40% therapeutic area share.
Which Distribution Channel holds the largest share?
Hospital Pharmacies account for 40% of distribution channel share in 2026.
Which country shows the fastest CAGR?
China projects the fastest country-level CAGR at 14.2% through 2036.
What is the absolute dollar growth from 2026 to 2036?
The absolute dollar growth from 2026 to 2036 represents an incremental gain of USD 61.4 billion.
How significant is the United States self-administered biologics market?
The United States records 9.8% CAGR through 2036.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Prefilled Syringes
- Autoinjectors
- Pen Injectors
- Wearable Injectors
- Prefilled Syringes
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapeutic Area
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapeutic Area, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapeutic Area, 2026 to 2036
- Autoimmune & Chronic Diseases
- Oncology Support Therapy
- Infectious Diseases
- Endocrinology
- Autoimmune & Chronic Diseases
- Y to o to Y Growth Trend Analysis By Therapeutic Area, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapeutic Area, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Therapeutic Area
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Therapeutic Area
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Therapeutic Area
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Therapeutic Area
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Therapeutic Area
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Therapeutic Area
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Therapeutic Area
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Therapeutic Area
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Therapeutic Area
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Therapeutic Area
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Therapeutic Area
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Therapeutic Area
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Therapeutic Area
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Therapeutic Area
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Therapeutic Area
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Therapeutic Area
- Competition Analysis
- Competition Deep Dive
- Amgen Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- AbbVie Inc.
- Genentech, Inc.
- Johnson & Johnson
- Pfizer Inc.
- Sanofi S.A.
- Bristol Myers Squibb Company
- Merck & Co., Inc.
- GSK plc
- Amgen Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Therapeutic Area, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Therapeutic Area
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Therapeutic Area
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 31: Latin America Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Therapeutic Area
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 38: Western Europe Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Therapeutic Area
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 45: Eastern Europe Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Therapeutic Area
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 52: East Asia Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Therapeutic Area
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Therapeutic Area
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y to o to Y Growth Comparison by Product Type , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Therapeutic Area, 2026 and 2036
- Figure 66: Middle East & Africa Market Y to o to Y Growth Comparison by Therapeutic Area, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Therapeutic Area
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis