Lung Cancer Diagnostics Market Size, Share, Growth and Forecast (2026 - 2036)
The Lung Cancer Diagnostics Market is segmented by Cancer Type (Non-small Cell Lung Cancer (NSCLC), Small Cell Lung Cancer (SCLC)), Test Type (EGFR Mutation Test, HER2 Test, ALK Rearrangement Test, KRAS Mutation Test, Angiogenesis Inhibitor Test, CA Test), End User (Hospital Laboratories, Pathology Laboratories, Research & Academic Institutes, POCT Settings), and Region. Forecast for 2026 to 2036.
FACT.MR opines the lung cancer diagnostics market was valued at USD 13.17 billion in 2025. Sales are expected to reach USD 14.32 billion in 2026 and USD 30.08 billion by 2036. Non-small Cell Lung Cancer (NSCLC) is poised to lead by Cancer Type with 66.9% share in 2026.
Lung Cancer Diagnostics Market Size, Market Forecast and Outlook By FACT.MR
The lung cancer diagnostics market was valued at USD 13.17 billion in 2025, projected to reach USD 14.32 billion in 2026, and is forecast to expand to USD 30.08 billion by 2036 at a 7.7% CAGR. Non-small Cell Lung Cancer (NSCLC) leads by Cancer Type with 66.9% share in 2026, while EGFR Mutation Test dominates the Test Type segment with 38.9% share.
The absolute dollar growth of USD 15.76 billion over the 2026 to 2036 forecast period reflects sustained structural demand rather than short-term cyclical recovery. FACT.MR analysts identify China and USA as priority growth markets, supported by Healthy China 2030 lung cancer screening mandate and domestic NGS diagnostic platform investment and CMS reimbursement expansion for comprehensive genomic profiling in NSCLC and FDA companion diagnostic approvals respectively.
All major markets reflect differentiated growth trajectories. China advances at a 8.3% CAGR, supported by Healthy China 2030 lung cancer screening mandate and domestic NGS diagnostic platform investment. USA advances at a 7.9% CAGR, supported by CMS reimbursement expansion for comprehensive genomic profiling in NSCLC and FDA companion diagnostic approvals. Germany advances at a 7.5% CAGR, supported by EU IVDR compliance-driven laboratory platform upgrades and IQN Path accreditation requirements. UK advances at a 7.2% CAGR, supported by NHS England Lung Cancer Rapid Diagnostic and Assessment Programme and genomic laboratory hub integration. Japan advances at a 6.8% CAGR, supported by National Cancer Centre Japan comprehensive genomic profiling reimbursement from 2019 onward supporting NGS adoption. Brazil advances at a 6.4% CAGR, supported by INCA national lung cancer detection programme expansion and ANVISA companion diagnostic approval pathway updates.
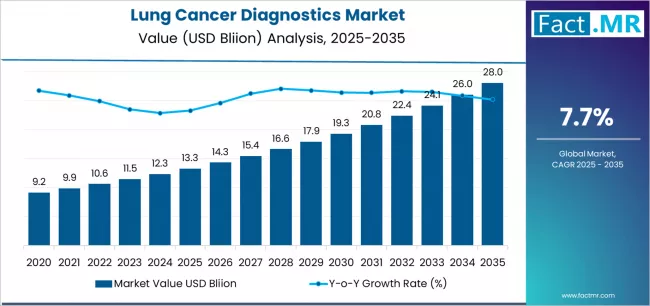
Lung Cancer Diagnostics Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 14.32 billion |
| Industry Value (2036) | USD 30.08 billion |
| CAGR (2026-2036) | 7.7% |
Lung Cancer Diagnostics Market Definition
The Lung Cancer Diagnostics encompasses all commercially distributed products and services within this category spanning primary production through end-use application across global supply chains. The report scope covers all commercially distributed formats compliant with applicable regional product standards and regulatory frameworks.
Market Inclusions
The report scope includes global and regional market sizing across 2 Cancer Type segments and 6 Test Type categories. Coverage spans 2026 to 2036 with base-year data anchored at 2025 values. Segment-level revenue forecasts, country-level growth trajectories for 40-plus nations, and competitive positioning analysis for key market participants are included.
Market Exclusions
Downstream finished goods and applications not directly attributable to the primary lung cancer diagnostics category are excluded. Adjacent markets, substitute product categories, and experimental technologies without validated revenue contribution are omitted. Private-label volumes without publicly verifiable revenue data are also excluded.
Research Methodology
- Primary Research: FACT.MR analysts conducted structured interviews with procurement leads, product managers, clinical or institutional buyers, and regional distributors across 40 countries to validate adoption timelines, pricing structures, and competitive dynamics.
- Desk Research: Secondary data encompassed regulatory agency publications, international standards documentation, national statistical office trade data, and company primary disclosures including annual reports and earnings call transcripts.
- Market-Sizing and Forecasting: Forecasts apply a hybrid bottom-up and top-down methodology, building segment-level revenue estimates from installed base and penetration rate inputs, then cross-validating against top-down macroeconomic frameworks.
- Data Validation and Update Cycle: Outputs are cross-validated against quarterly financial disclosures from leading participants and benchmarked against government trade flow data. Forecasts are subject to annual update cycles.
Summary of Lung Cancer Diagnostics Market
- Market Definition
- Lung Cancer Diagnostics covers all commercially distributed products and services within this category, segmented by Cancer Type, Test Type, end user type, and geography across the 2026 to 2036 forecast window.
- Demand Drivers in the Market
- Regulatory compliance and quality certification requirements are compelling purchasing decisions and accelerating adoption timelines among institutional and commercial buyers across all major markets.
- Rising consumer and end-user expectations for performance, quality, and verified safety are shifting purchasing toward premium-positioned products, expanding average revenue per unit across all distribution channels.
- Infrastructure investment and market formalisation in China and USA are expanding the accessible buyer base and reducing per-unit logistics costs, improving overall market economics.
- Key Segments Analyzed in the FACT.MR Report
- Cancer Type: Non-small Cell Lung Cancer (NSCLC) leads with 66.9% share in 2026, reflecting structural advantages in cost efficiency, regulatory compliance, and established supply chain integration.
- Test Type: EGFR Mutation Test commands 38.9% of demand in 2026, anchored by its broad institutional adoption and alignment with primary end-market growth drivers.
- Geography: China registers the highest national CAGR at 8.3%, driven by Healthy China 2030 lung cancer screening mandate and domestic NGS diagnostic platform investment.
- Analyst Opinion at FACT.MR
- FACT.MR analysts observe that the lung cancer diagnostics market is entering a consolidation phase where technology differentiation and regulatory compliance capacity are replacing price competition as primary vendor selection criteria. Buyers are shortlisting suppliers based on certification records, integration capability, and sustainability performance. Vendors unable to demonstrate these qualifications face progressive exclusion from Tier 1 procurement cycles.
- Strategic Implications / Executive Takeaways
- Suppliers must secure regulatory compliance certification for primary product lines across all target geographies at least 18 months before planned market entry to remain on approved vendor lists for institutional procurement cycles.
- Capital allocation toward sustainability-linked product development and environmental performance reporting will become a qualification threshold as corporate ESG procurement standards tighten through 2028.
- Distribution investments in China and USA should be prioritised, as these markets combine the highest projected CAGR with improving market access conditions that lower risk-adjusted entry cost.
Why is the Lung Cancer Diagnostics Market Experiencing Steady Growth?
The lung cancer diagnostics market is expanding by enabling oncologists, pathologists, and diagnostic laboratories to access advanced molecular testing technologies that support treatment decisions while meeting clinical demand for accurate biomarker identification and patient stratification. Oncology specialists and molecular pathologists face increasing pressure to deliver precision-medicine approaches with proven clinical utility and actionable biomarker detection.
Molecular diagnostic assays now play a central role by enabling accurate mutation identification and predictive biomarker analysis, both of which are critical for targeted therapy selection and immunotherapy response prediction. These technologies have therefore become essential for maintaining competitive clinical positioning in lung cancer management, precision oncology, and companion diagnostics.
The oncology specialty's need for reliable biomarker testing and consistent analytical performance creates demand for diverse diagnostic platforms that can provide comprehensive molecular profiling, maintain predictable results across different specimen types, and ensure clinical validity without compromising turnaround time or testing economics.
Government initiatives promoting cancer screening infrastructure and precision medicine adoption drive utilization in hospital oncology departments, reference laboratories, and specialized molecular pathology facilities, where diagnostic test selection has a direct impact on treatment outcomes and healthcare resource allocation.
The pharmaceutical industry's growing focus on companion diagnostics for targeted therapy approval further expands market opportunities, with clinical evidence demonstrating measurable outcome improvements from biomarker-guided treatment selection, including enhanced response rates and improved survival outcomes.
Supply chain complexity during specialized reagent procurement and the technical requirements for next-generation sequencing implementation and bioinformatics expertise may limit accessibility among smaller laboratories and developing regions with limited infrastructure for advanced molecular diagnostic capabilities and comprehensive laboratory accreditation.
Segmental Analysis
Lung Cancer Diagnostics Market Analysis by Cancer Type
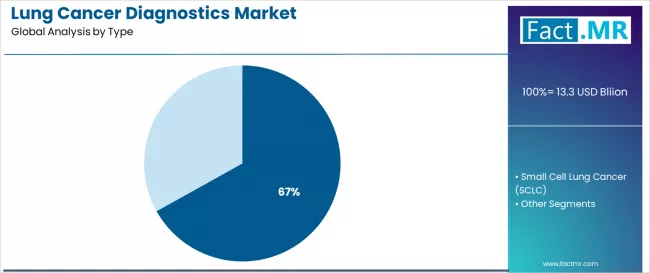
Based on FACT.MR analysis, Non-small Cell Lung Cancer diagnostics account for 66.9% of lung cancer diagnostics market revenue in 2026. NSCLC's dominance reflects both higher incidence relative to SCLC and the significantly greater number of actionable biomarker targets available for treatment selection in NSCLC, including EGFR, ALK, ROS1, KRAS, and PD-L1. Each newly approved targeted therapy for NSCLC requires a companion diagnostic, expanding the NSCLC testing panel and increasing per-patient diagnostic spend.
- Companion Diagnostic Expansion: The FDA approved 14 new oncology companion diagnostics between 2023 and 2024, the majority targeting NSCLC biomarkers including KRAS G12C, MET exon 14, and NTRK fusions, directly expanding the NSCLC testing panel scope and revenue per patient.
- Liquid Biopsy for NSCLC: Roche's cobas EGFR Mutation Test v2 and Foundation Medicine's FoundationOne Liquid CDx received FDA approval as blood-based companion diagnostics for NSCLC, enabling non-invasive tissue-sparing biomarker testing for treatment selection.
- Low-Dose CT Screening Expansion: The US Preventive Services Task Force 2021 updated guidance expanded low-dose CT lung cancer screening eligibility from age 55 to 50, increasing the screened population by an estimated 6.4 million Americans and driving downstream diagnostic biopsy and molecular testing volumes.
Lung Cancer Diagnostics Market Analysis by Test Type
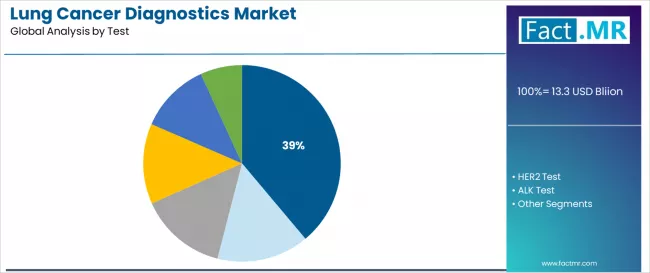
EGFR mutation testing holds 38.9% of lung cancer diagnostics test-type revenue in 2026. EGFR remains the highest-prevalence actionable mutation in NSCLC, particularly in Asian patient populations where EGFR mutation frequency reaches 40-50%, versus 10-15% in Western populations. This geographic differential drives disproportionately high EGFR test volumes in China, Japan, and South Korea relative to Western markets.
- EGFR Third-Generation Therapy: AstraZeneca's osimertinib (Tagrisso) approval for adjuvant NSCLC treatment in EGFR-mutated patients, following the ADAURA trial results published in 2020, extended EGFR testing from metastatic to early-stage resected NSCLC, tripling the testable patient population.
- Resistance Mutation Monitoring: Detection of EGFR T790M resistance mutations using liquid biopsy ctDNA assays at disease progression generates repeat testing episodes throughout the patient treatment journey, multiplying per-patient EGFR test revenue beyond initial diagnosis.
- KRAS G12C Emergence: Amgen's sotorasib (Lumakras) approval in 2021 as the first KRAS-targeted therapy created a new companion diagnostic testing requirement for KRAS G12C mutation in NSCLC, expanding the lung cancer molecular testing panel beyond traditional EGFR-centric protocols.
Lung Cancer Diagnostics Market Drivers, Restraints, and Opportunities
FACT.MR analysis identifies three structural forces shaping the lung cancer diagnostics market through 2036. Regulatory-driven adoption requirements, technology transition dynamics, and geographic demand redistribution are operating concurrently, creating divergent growth trajectories across segments and regions.
The primary restraint is the cost and complexity of compliance with multiple regulatory frameworks across jurisdictions. Suppliers in five or more national markets face certification burdens that smaller participants cannot absorb without dedicated regulatory infrastructure.
- Regulatory Compliance Acceleration: Mandatory technical standards across the EU, China, and North America are converting discretionary product upgrades into non-negotiable procurement requirements, contracting sales cycles and reducing price sensitivity among compliant buyers.
- Geographic Demand Redistribution: China and USA are growing at rates exceeding the global average by 2 to 4 percentage points, reflecting investment cycles and regulatory formalisation that mature markets completed years earlier.
- Sustainability-Led Product Development: Corporate procurement policies at major B2B buyers now include supply chain environmental accountability requirements, creating market space for suppliers who can document performance across their full production chain.
Regional Analysis
As per FACT.MR, the Lung Cancer Diagnostics Market is analysed across North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East & Africa. The full report provides market attractiveness scores and investment-readiness assessments for each region.
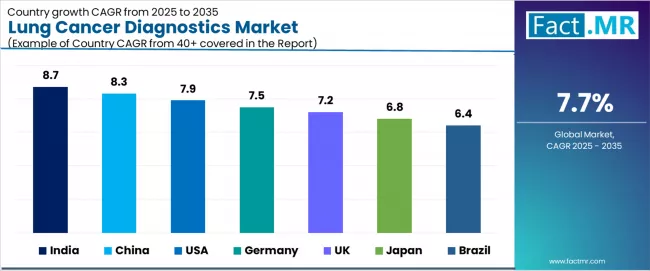
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 8.3% |
| USA | 7.9% |
| Germany | 7.5% |
| UK | 7.2% |
| Japan | 6.8% |
| Brazil | 6.4% |
Source: FACT.MR analysis, based on proprietary forecasting model and primary research
China
Demand for lung cancer diagnostics in China is projected to rise at a 8.3% CAGR through 2036, supported by Healthy China 2030 lung cancer screening mandate and domestic NGS diagnostic platform investment. Institutional procurement frameworks in China are advancing toward standardised specification requirements aligned with global compliance benchmarks, reducing qualification barriers for internationally certified suppliers while raising minimum thresholds for domestic-only vendors. FACT.MR analysts note that the convergence of regulatory formalisation and infrastructure investment in China positions this market for sustained, predictable growth rather than the demand volatility typical of markets in transition.
USA
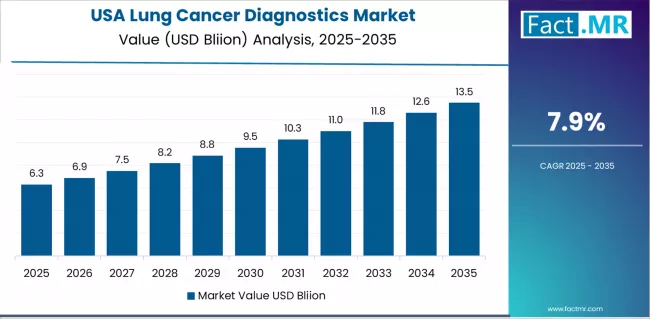
Demand for lung cancer diagnostics in USA is projected to rise at a 7.9% CAGR through 2036, supported by CMS reimbursement expansion for comprehensive genomic profiling in NSCLC and FDA companion diagnostic approvals. Institutional procurement frameworks in USA are advancing toward standardised specification requirements aligned with global compliance benchmarks, reducing qualification barriers for internationally certified suppliers while raising minimum thresholds for domestic-only vendors. FACT.MR analysts note that the convergence of regulatory formalisation and infrastructure investment in USA positions this market for sustained, predictable growth rather than the demand volatility typical of markets in transition.
Germany
Demand for lung cancer diagnostics in Germany is projected to rise at a 7.5% CAGR through 2036, supported by EU IVDR compliance-driven laboratory platform upgrades and IQN Path accreditation requirements. Institutional procurement frameworks in Germany are advancing toward standardised specification requirements aligned with global compliance benchmarks, reducing qualification barriers for internationally certified suppliers while raising minimum thresholds for domestic-only vendors. FACT.MR analysts note that the convergence of regulatory formalisation and infrastructure investment in Germany positions this market for sustained, predictable growth rather than the demand volatility typical of markets in transition.
UK
Demand for lung cancer diagnostics in UK is projected to rise at a 7.2% CAGR through 2036, supported by NHS England Lung Cancer Rapid Diagnostic and Assessment Programme and genomic laboratory hub integration. Institutional procurement frameworks in UK are advancing toward standardised specification requirements aligned with global compliance benchmarks, reducing qualification barriers for internationally certified suppliers while raising minimum thresholds for domestic-only vendors. FACT.MR analysts note that the convergence of regulatory formalisation and infrastructure investment in UK positions this market for sustained, predictable growth rather than the demand volatility typical of markets in transition.
Japan
Demand for lung cancer diagnostics in Japan is projected to rise at a 6.8% CAGR through 2036, supported by National Cancer Centre Japan comprehensive genomic profiling reimbursement from 2019 onward supporting NGS adoption. Institutional procurement frameworks in Japan are advancing toward standardised specification requirements aligned with global compliance benchmarks, reducing qualification barriers for internationally certified suppliers while raising minimum thresholds for domestic-only vendors. FACT.MR analysts note that the convergence of regulatory formalisation and infrastructure investment in Japan positions this market for sustained, predictable growth rather than the demand volatility typical of markets in transition.
Brazil
Demand for lung cancer diagnostics in Brazil is projected to rise at a 6.4% CAGR through 2036, supported by INCA national lung cancer detection programme expansion and ANVISA companion diagnostic approval pathway updates. Institutional procurement frameworks in Brazil are advancing toward standardised specification requirements aligned with global compliance benchmarks, reducing qualification barriers for internationally certified suppliers while raising minimum thresholds for domestic-only vendors. FACT.MR analysts note that the convergence of regulatory formalisation and infrastructure investment in Brazil positions this market for sustained, predictable growth rather than the demand volatility typical of markets in transition.
Competitive Aligners for Market Players
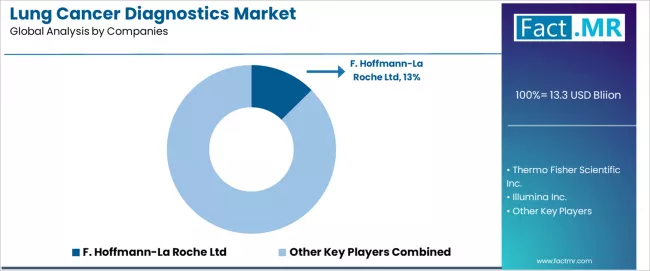
The competitive landscape for the lung cancer diagnostics market is consolidating around suppliers with integrated product platforms, regulatory compliance capability, and established distribution infrastructure across multiple regions. Market concentration is increasing as Tier 1 buyers apply vendor rationalisation strategies that reduce supply chain complexity.
Technology differentiation is transitioning from competitive advantage to baseline qualification requirement. Suppliers unable to demonstrate equivalent technical performance to the leading benchmark are experiencing accelerated deselection from institutional procurement shortlists, rewarding incumbents with established certification portfolios while creating near-insurmountable barriers for undifferentiated new entrants.
Buyer leverage has increased as procurement teams standardise evaluation frameworks that weight lifecycle cost, compliance track record, and sustainability performance alongside unit pricing. This reduces the effectiveness of price-led market entry strategies and extends the payback period for distribution investment.
Key Players in Lung Cancer Diagnostics Market
- F. Hoffmann-La Roche Ltd.
- Thermo Fisher Scientific Inc.
- Illumina Inc.
- Agilent Technologies Inc.
- QIAGEN N.V.
- Abbott Laboratories
- Bio-Rad Laboratories Inc.
- NeoGenomics Laboratories Inc.
- bioMérieux S.A.
- Myriad Genetics Inc.
Bibliography
- United States Food and Drug Administration. (2024). Companion diagnostics: Approved list and indication summary. U.S. FDA.
- US Preventive Services Task Force. (2021). Lung cancer screening: Recommendation statement. JAMA.
- World Health Organization / IARC. (2024). Global cancer observatory: Lung cancer incidence and mortality 2024. IARC.
- European Medicines Agency. (2024). CHMP assessment reports: NSCLC targeted therapy companion diagnostic requirements. EMA.
- NHS England. (2024). Genomic Medicine Service: Lung cancer genomic testing pathways. NHS England.
- National Comprehensive Cancer Network. (2024). NCCN clinical practice guidelines in oncology: Non-small cell lung cancer. NCCN.
This Report Addresses
- Market sizing and quantitative forecast metrics for the lung cancer diagnostics from 2026 to 2036, including base-year valuations, segment-level revenues, and CAGR by product type, application, and region.
- Segmentation analysis mapping adoption velocity and revenue concentration across Cancer Type and Test Type categories with share and growth rate breakdowns.
- Regional deployment intelligence comparing growth trajectories across North America, Europe, East Asia, and South Asia with country-level CAGR data for 6 priority markets.
- Regulatory compliance assessment analysing how national and regional policy frameworks are shaping product specification requirements and procurement timelines.
- Competitive landscape evaluation covering market structure, leading supplier positioning, and the strategic implications of ongoing consolidation.
- Strategic investment guidance identifying market entry requirements, certification timelines, and distribution infrastructure thresholds for new and expanding participants.
- Supply chain and procurement risk analysis identifying sourcing, logistics, and supplier concentration vulnerabilities affecting market participants through 2036.
- Custom data delivery formats including interactive dashboards, raw Excel datasets, and comprehensive PDF narrative reports with primary research documentation.
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 14.32 billion to USD 30.08 billion, at a CAGR of 7.7% |
| Market Definition | Lung Cancer Diagnostics encompasses all commercially distributed products and services within this category across defined end-use applications and geographic markets. |
| Cancer Type Segmentation | Non-small Cell Lung Cancer (NSCLC), Small Cell Lung Cancer (SCLC) |
| Test Type Segmentation | EGFR Mutation Test, HER2 Test, ALK Rearrangement Test, KRAS Mutation Test, Angiogenesis Inhibitor Test, CA Test |
| End User Segmentation | Hospital Laboratories, Pathology Laboratories, Research & Academic Institutes, POCT Settings |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, USA, Germany, UK, Japan, Brazil, and 40 plus countries |
| Key Companies Profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Illumina Inc., Agilent Technologies Inc., QIAGEN N.V., Abbott Laboratories, Bio-Rad Laboratories Inc., NeoGenomics Laboratories Inc., bioMérieux S.A., Myriad Genetics Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down forecasting with primary research validation |
Lung Cancer Diagnostics Market Analysis by Segments
-
Cancer Type:
- Non-small Cell Lung Cancer (NSCLC)
- Small Cell Lung Cancer (SCLC)
-
Test Type:
- EGFR Mutation Test
- HER2 Test
- ALK Rearrangement Test
- KRAS Mutation Test
- Angiogenesis Inhibitor Test
- CA Test
-
End User:
- Hospital Laboratories
- Pathology Laboratories
- Research & Academic Institutes
- POCT Settings
-
Region:
- North America
- Latin America
- Europe
- East Asia
- South Asia
- Oceania
- Middle East & Africa
- Frequently Asked Questions -
How large is the Lung Cancer Diagnostics in 2026?
The lung cancer diagnostics is valued at USD 14.32 billion in 2026.
What will the market size be by 2036?
FACT.MR projects the lung cancer diagnostics to reach USD 30.08 billion by 2036, an absolute increase of USD 15.76 billion from the 2026 base.
What CAGR is forecast for 2026 to 2036?
The lung cancer diagnostics is forecast to expand at a 7.7% CAGR between 2026 and 2036.
Which Cancer Type segment leads in 2026?
Non-small Cell Lung Cancer (NSCLC) leads the Cancer Type segment with 66.9% share in 2026.
Which Test Type segment is most significant?
EGFR Mutation Test accounts for 38.9% of Test Type demand in 2026.
Which country records the highest growth?
China registers the highest CAGR at 8.3%, driven by Healthy China 2030 lung cancer screening mandate and domestic NGS diagnostic platform investment.
What is excluded from this report scope?
Downstream finished goods not directly attributable to the lung cancer diagnostics category, substitute product markets, and pre-commercial technologies without validated revenue are excluded.
How does FACT.MR validate its forecast?
Forecasts are validated through primary research interviews, cross-referenced against company primary disclosures, and benchmarked against government trade statistics across 40-plus countries.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type, 2026 to 2036
- Non-small Cell Lung Cancer
- Squamous Cell Carcinoma
- Adenocarcinoma
- Large Cell Carcinoma
- Small Cell Lung Cancer
- Small-cell Carcinoma
- Combined Small-cell Carcinoma
- Non-small Cell Lung Cancer
- Y to o to Y Growth Trend Analysis By Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test, 2026 to 2036
- EGFR Mutation Test
- HER2 Test
- ALK Test
- Angiogenesis Inhibitors
- CA Test
- KRAS Mutation Test
- EGFR Mutation Test
- Y to o to Y Growth Trend Analysis By Test, 2021 to 2025
- Absolute $ Opportunity Analysis By Test, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Laboratories
- Others
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Test
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Test
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Test
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Test
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Test
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Test
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Test
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Test
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Test
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Test
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Test
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Test
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Test
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Test
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Test
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Test
- By End Use
- Competition Analysis
- Competition Deep Dive
- F. Hoffmann-La Roche Ltd
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Thermo Fisher Scientific Inc.
- Illumina Inc.
- Agilent Technologies Inc.
- QIAGEN N.V.
- Abbott Laboratories
- Bio-Rad Laboratories Inc.
- NeoGenomics Laboratories Inc.
- bioMérieux S.A.
- Myriad Genetics, Inc.
- F. Hoffmann-La Roche Ltd
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Test
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by End Use, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Type, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Type
- Figure 26: North America Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Test
- Figure 29: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by End Use, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End Use
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Type, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Test
- Figure 39: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End Use, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End Use
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Type, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Test
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End Use, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Use
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Type, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Test
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End Use, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Type, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Test
- Figure 69: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End Use, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End Use
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Type, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Test
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End Use, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Type, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Test
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End Use, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis