Cancer Biomarkers Market Size, Share, Growth and Forecast (2026 - 2036)
Cancer Biomarkers Market is segmented by Test (PSA Tests, CTC Tests, AFP Tests, CA Tests, HER2 Tests, BRCA Tests, ALK Tests, CEA Tests, EGFR Mutation Tests, KRAS Mutation Tests, Others), Disease Indication (Breast Cancer, Lung Cancer, Colorectal Cancer, Melanoma, Blood Cancer, Prostate Cancer, Ovarian Cancer, Stomach Cancer, Liver Cancer, Others), Application (Diagnostics, Prognostics, Predictive Testing, Drug Discovery and Development), and Region, with forecasts covering the period from 2026 to 2036.
According to Fact.MR estimates, the global cancer biomarkers market market was valued at USD 14.2 billion in 2025. The market is projected to reach USD 16.0 billion in 2026 and is expected to grow to USD 54.5 billion by 2036, expanding at a CAGR of 13.0%. PSA Tests is anticipated to account for 35.4% of the product segment in 2026, while Breast Cancer is expected to remain the leading application with around 24.9% share.
Cancer Biomarkers Market Analysis and Forecast by Fact.MR
The cancer biomarkers market, valued at USD 14.2 billion in 2025, is expanding at a 13.0% CAGR to reach USD 54.469 billion by 2036. Cancer biomarkers are measurable biological indicators, including proteins, gene mutations, and circulating tumor cells, used in cancer diagnosis, prognosis, treatment selection, and therapy monitoring. Growth reflects accelerating clinical adoption of precision oncology protocols, expansion of liquid biopsy platforms, and integration of multi-omics data into treatment decision frameworks. The USD 38.423 billion incremental opportunity between 2026 and 2036 is concentrated in diagnostic applications across North America, East Asia, and Europe.
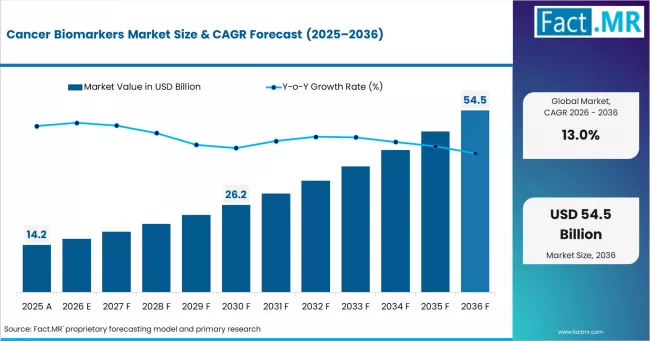
Summary of Cancer Biomarkers Market
- Market Snapshot
- Market value in 2025: USD 14.2 billion
- Projected value in 2026: USD 16.046 billion; forecast to reach USD 54.469 billion by 2036
- CAGR from 2026 to 2036: 13.0%
- Incremental opportunity (2026 to 2036): USD 38.423 billion
- PSA tests lead the test-type segment with 35.4% share in 2025
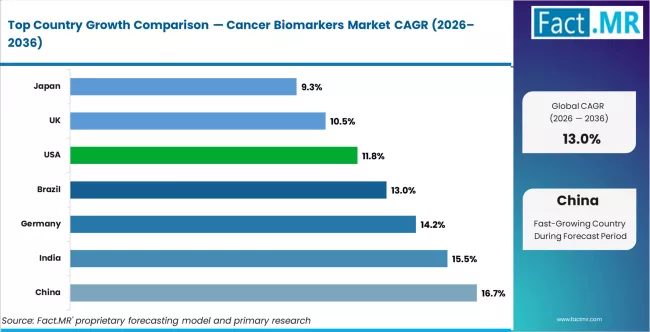
- China and India lead country-level growth, followed by Germany and Brazil
- Demand and Growth Drivers
- Precision oncology protocols are replacing empirical treatment selection, creating structured demand for biomarker-based companion diagnostics that match patients to targeted therapies.
- Liquid biopsy platforms are enabling non-invasive cancer detection and monitoring, reducing dependence on tissue biopsies and expanding the addressable patient population for biomarker testing.
- National cancer screening programs in the USA, China, and several European countries are mandating or incentivizing biomarker-based early detection, generating recurring test volumes at scale.
- Pharmaceutical companies developing targeted therapies require validated companion diagnostic biomarkers for regulatory approval, creating a parallel demand stream from drug development pipelines.
- Product and Segment View
- PSA tests account for 35.4% of test-type demand, driven by established prostate cancer screening protocols and reimbursement coverage in North America and Europe.
- Breast cancer holds 24.9% of disease indication share, reflecting the maturity of HER2, BRCA, and hormone receptor biomarker testing in treatment selection.
- Diagnostics lead application demand at 31.5%, anchored by routine screening, early detection panels, and companion diagnostic requirements.
- Key segmentation includes:
- Test: PSA Tests, CTC Tests, AFP Tests, CA Tests, HER2 Tests, BRCA Tests, ALK Tests, CEA Tests, EGFR Mutation Tests, KRAS Mutation Tests, Others
- Disease Indication: Breast Cancer, Lung Cancer, Colorectal Cancer, Melanoma, Blood Cancer, Prostate Cancer, Ovarian Cancer, Stomach Cancer, Liver Cancer, Others
- Application: Diagnostics, Prognostics, Predictive Testing, Drug Discovery and Development
- End Use: Hospitals, Diagnostic Laboratories, Pharmaceutical and Biotechnology Companies, Research Institutes
- Geography and Competitive Outlook
- North America is the largest revenue region, with the USA contributing USD 6.035 billion in 2025 through established reimbursement pathways and precision oncology infrastructure.
- East Asia is the fastest-growing region, with China recording the highest country CAGR, supported by national cancer screening expansion and genomics infrastructure investment.
- Europe is shaped by regulatory harmonization under IVDR, with Germany leading regional growth through strong molecular diagnostics adoption in university hospitals.
- F. Hoffmann-La Roche Ltd. leads the competitive field with a 21.0% market share, followed by Thermo Fisher Scientific, Illumina, QIAGEN, and Abbott Laboratories.
- Analyst Opinion
- The cancer biomarkers market is transitioning from single-analyte testing toward multi-omics panels that combine genomic, proteomic, and metabolomic data for comprehensive tumor profiling. This shift favors platform companies with integrated sequencing, assay development, and data analytics capabilities. The commercialization of multi-cancer early detection tests is expected to create a new demand category, distinct from established companion diagnostics. Fact.MR anticipates that regulatory approval of blood-based MCED panels in the USA and EU will be a major inflection point, expanding the addressable testing population from symptomatic patients to asymptomatic screening cohorts.
Key Growth Drivers, Constraints, and Opportunities
Key Factors Driving Growth
- Precision medicine mandates: Oncology treatment guidelines from NCCN, ESMO, and NICE increasingly require biomarker testing before prescribing targeted therapies, converting clinical recommendations into structured test demand.
- Liquid biopsy commercialization: FDA clearance and payer coverage expansion for blood-based cancer detection tests are reducing the invasiveness barrier to biomarker testing, broadening the eligible patient population beyond those undergoing surgical biopsy.
- Pharmaceutical companion diagnostic requirements: Regulatory agencies now mandate validated companion diagnostics for most targeted oncology drug approvals, creating a parallel biomarker test demand stream tied to drug development and launch timelines.
Key Market Constraints
- Reimbursement uncertainty: Payer coverage for newer biomarker tests, particularly multi-gene panels and MCED assays, remains inconsistent across markets, limiting adoption in price-sensitive healthcare systems.
- Standardization gaps: Variability in biomarker assay performance, reference ranges, and laboratory protocols across testing facilities creates reproducibility challenges that slow clinical adoption of newer test modalities.
- Data integration complexity: Translating multi-omics biomarker data into actionable treatment decisions requires computational infrastructure and clinical informatics expertise that many hospitals and laboratories lack.
Key Opportunity Areas
- Multi-cancer early detection (MCED) panels: Blood-based tests capable of detecting multiple cancer types from a single draw are creating a new screening category with the potential to expand the testing population from millions to hundreds of millions of asymptomatic adults.
- AI-assisted biomarker discovery: Machine learning applied to genomic and proteomic datasets is accelerating the identification of novel biomarker signatures, shortening the path from discovery to clinical validation.
- Emerging market screening infrastructure: National cancer screening programs in China, India, and Brazil are expanding biomarker test access beyond tertiary hospitals to secondary-care facilities and community health centers.
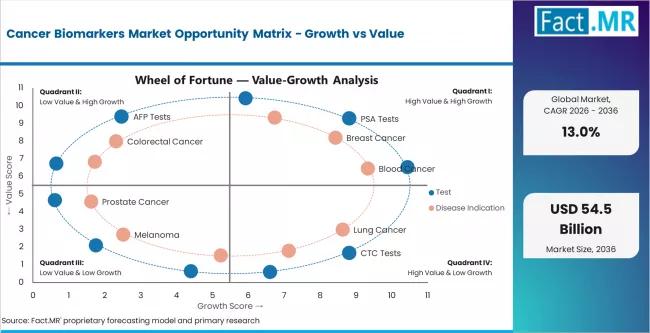
Segment-wise Analysis of the Cancer Biomarkers Market
- PSA tests lead the test-type segment at 35.4% share, driven by established prostate cancer screening guidelines and broad reimbursement coverage.
- Breast cancer holds 24.9% of disease indication demand, reflecting the maturity of HER2, BRCA, and hormone receptor testing protocols.
- Diagnostics account for 31.5% of application share, anchored by routine screening and companion diagnostic requirements tied to targeted therapy prescribing.
Which test type dominates the Cancer Biomarkers Market?
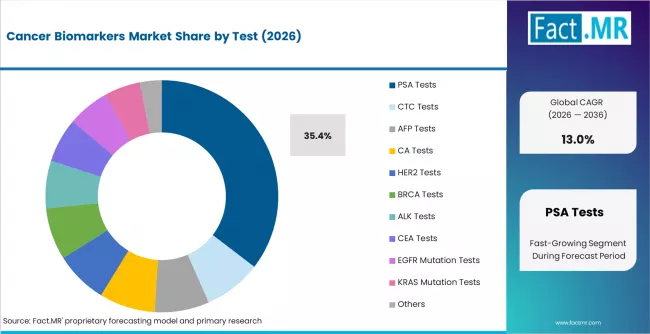
PSA tests hold a 35.4% share of test-type demand in 2025, making prostate-specific antigen testing the largest single biomarker test category. The dominance reflects decades of clinical integration into prostate cancer screening protocols, broad physician familiarity, established reimbursement coverage, and high-volume test ordering patterns in urology and primary care settings across North America, Europe, and Japan.
HER2, BRCA, and EGFR mutation tests are growing faster than PSA, driven by expanding companion diagnostic requirements for targeted therapy prescribing in breast cancer, lung cancer, and colorectal cancer. CA tests hold approximately 11% share, used primarily in ovarian and gastrointestinal cancer monitoring. The test-type mix is shifting toward multi-gene panel tests that consolidate several biomarker assays into a single workflow, favoring platform-based sequencing approaches over standalone immunoassay formats.
Which disease indication dominates the Cancer Biomarkers Market?
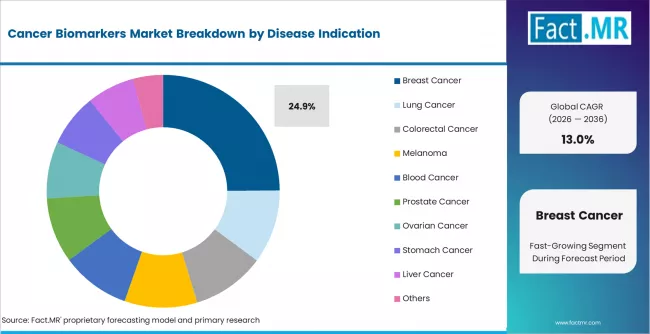
Breast cancer accounts for 24.9% of disease indication demand in 2025, the largest single-indication category. The share reflects the clinical maturity of HER2-targeted therapy, BRCA mutation testing for risk stratification, and hormone receptor profiling for endocrine therapy selection. These biomarker tests are embedded in standard-of-care treatment algorithms across developed markets, generating high and recurring test volumes.
Lung cancer is the fastest-growing disease indication, driven by the expansion of EGFR, ALK, and PD-L1 biomarker testing tied to immunotherapy and targeted kinase inhibitor prescribing. Colorectal cancer biomarker testing is expanding through KRAS and microsatellite instability assays. Prostate cancer biomarker demand is sustained by PSA screening volumes, while liver cancer and ovarian cancer indications are growing from smaller bases as new biomarker tests enter clinical use.
Which application dominates the Cancer Biomarkers Market?
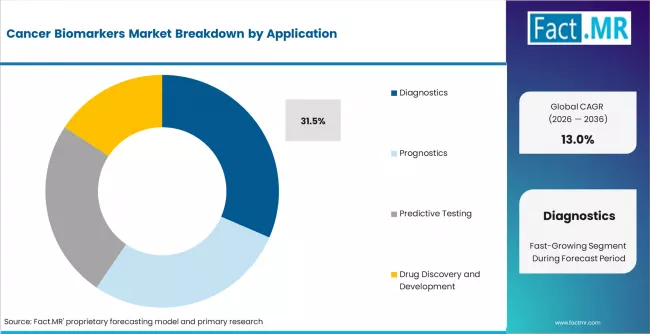
Diagnostics lead at 31.5% of application share in 2025, reflecting the primary use of biomarker tests in cancer detection, confirmation, and subtyping at the point of initial clinical workup. Diagnostic applications include both tissue-based and liquid biopsy platforms used to identify tumor presence, classify tumor type, and determine molecular subtype for treatment planning.
Predictive testing is growing rapidly as companion diagnostic requirements expand the scope of biomarker testing beyond diagnosis into therapy selection. Prognostic applications use biomarkers to estimate disease trajectory and recurrence risk. Drug discovery and development is the smallest but fastest-growing application, as pharmaceutical companies increase investment in biomarker-driven clinical trial design and patient stratification.
Which Product Trend is Shaping the Next Phase of Growth in the Cancer Biomarkers Market?
Multi-cancer early detection (MCED) testing is the product trend shaping the next growth phase. MCED panels use blood-based assays to detect methylation patterns, cell-free DNA fragments, or protein signatures associated with multiple cancer types from a single blood draw. This approach moves biomarker testing from a diagnostic confirmation role to a proactive screening function, expanding the addressable population from patients with suspected cancer to asymptomatic individuals in routine health checks. Companies including GRAIL (Illumina subsidiary), Exact Sciences, and Guardant Health are advancing MCED platforms through large-scale clinical validation studies.
The commercial impact of MCED adoption will be substantial if payer coverage aligns with clinical evidence timelines. Current MCED tests target 50+ cancer types and are designed for annual screening in adults over 50, creating a potential test volume base measured in hundreds of millions of annual tests globally. Regulatory pathways in the USA and EU are under active development, with FDA premarket approval submissions expected to set the precedent for coverage decisions. Fact.MR projects that MCED commercialization will become the single largest growth accelerator in the cancer biomarkers market from 2028 onward, provided analytical sensitivity improvements and reimbursement frameworks keep pace with clinical adoption demand.
Regional Outlook Across Key Markets
.webp)
- North America is the largest revenue region, with the USA contributing USD 6.035 billion in 2025, supported by established reimbursement infrastructure and precision oncology leadership.
- East Asia is the fastest-growing region, led by China, where national cancer screening expansion and genomics investment are accelerating biomarker test adoption.
- Europe is shaped by IVDR regulatory harmonization and strong molecular diagnostics infrastructure in Germany, France, and the UK.
- South Asia and Pacific is an emerging growth corridor, with India recording strong CAGR driven by cancer incidence growth and expanding diagnostic laboratory networks.
CAGR Table
| Country | CAGR (%) |
|---|---|
| China | 16.7% |
| India | 15.5% |
| Germany | 14.2% |
| Brazil | 13.0% |
| USA | 11.8% |
| UK | 10.5% |
| Japan | 9.3% |
Source: Fact.MR analysis, based on proprietary forecasting model and primary research.
China: National screening programs and genomics investment
China records the highest country-level CAGR in the cancer biomarkers market, driven by government-funded cancer screening programs targeting lung, colorectal, and liver cancers in high-prevalence regions. The National Health Commission has expanded biomarker-based screening protocols from pilot cities to provincial-level implementation, creating structured test volumes through public health infrastructure. Domestic genomics companies including BGI and Berry Oncology are scaling locally manufactured NGS platforms that reduce per-test costs below import-equivalent pricing, accelerating adoption in secondary-care hospitals.
- Growth anchor: Government-funded screening expansion and domestic genomics manufacturing are creating volume-driven demand at price points below Western-equivalent testing.
India: Diagnostic laboratory expansion and oncology burden growth
India records the second-highest country CAGR, reflecting the intersection of rising cancer incidence, expanding private diagnostic laboratory networks, and growing physician awareness of biomarker-guided treatment. National Cancer Registry data indicates sustained increases in breast, cervical, and oral cancer diagnoses, each of which is driving demand for specific biomarker test panels. Private laboratory chains including Metropolis, SRL, and Thyrocare are adding oncology biomarker test menus as competitive differentiators, expanding access beyond tertiary oncology centers.
- Growth anchor: Private diagnostic laboratory investment in oncology test menus is bringing biomarker testing to tier-2 and tier-3 city populations.
Germany: Molecular diagnostics leadership in Europe
Germany leads European biomarker market growth, supported by a dense network of university hospitals with established molecular pathology departments, favorable reimbursement for NGS-based oncology testing under statutory health insurance, and strong clinical trial infrastructure that generates early demand for novel biomarker assays. The German Molecular Pathology Network coordinates multi-center biomarker testing programs, ensuring standardized quality across testing sites and supporting rapid adoption of newly approved companion diagnostics.
- Growth anchor: Statutory health insurance coverage of NGS-based oncology panels removes the payer barrier that slows adoption in other European markets.
USA: Precision oncology infrastructure and MCED pipeline
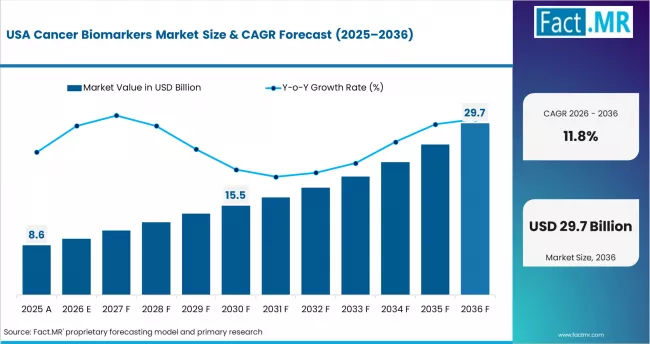
The USA contributes USD 6.035 billion to the global cancer biomarkers market in 2025, the largest single-country revenue pool. Growth is transitioning from established companion diagnostic volume toward next-generation applications including MCED screening, minimal residual disease monitoring, and therapy response assessment using liquid biopsy. Medicare coverage decisions on MCED tests and multi-gene panel reimbursement expansion are the primary near-term catalysts. The concentration of oncology drug development activity in the USA creates a persistent companion diagnostic demand stream tied to FDA drug-device co-approval requirements.
- Growth anchor: FDA regulatory decisions on MCED coverage and expanded companion diagnostic co-approvals are the primary near-term demand catalysts.
Japan: Aging demographics and precision oncology adoption
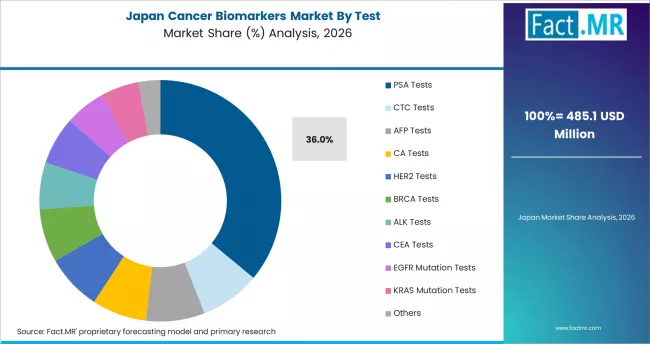
Japan records the lowest CAGR among tracked markets but remains a significant revenue contributor, reflecting its aging population with high cancer incidence rates and well-established diagnostic infrastructure. The Japanese government's cancer genomic medicine program, operated through designated core hospitals, provides structured biomarker testing for patients with advanced cancers, generating sustained NGS panel test volumes. Growth is moderated by the slower pace of reimbursement expansion for newer assay types compared to the USA and Germany.
- Growth anchor: The national cancer genomic medicine program provides structured demand through designated testing centers in core hospitals.
Competitive Benchmarking and Company Positioning
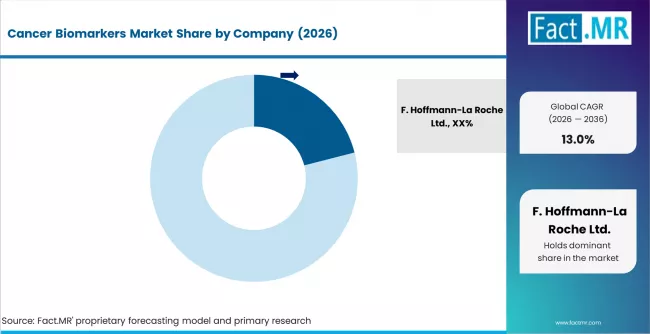
Cancer Biomarkers Market Analysis By Company
- F. Hoffmann-La Roche Ltd. leads with 21.0% market share, built on its integrated diagnostics-pharma model that links companion diagnostic development directly to its oncology drug pipeline.
- Thermo Fisher Scientific and Illumina compete in the platform tier, supplying NGS instruments, reagents, and assay kits to hospitals and reference laboratories operating high-throughput biomarker testing programs.
- QIAGEN and Abbott Laboratories maintain strong positions in molecular diagnostics consumables and immunoassay-based biomarker testing respectively, serving high-volume clinical laboratory workflows.
The competitive field is concentrated at the top, with the five largest players accounting for a combined share exceeding 50%. Roche's leadership position reflects its unique diagnostics-pharmaceutical integration, where companion diagnostic development is aligned with its oncology drug pipeline, creating co-approval advantages that reinforce both the therapeutics and diagnostics businesses. Thermo Fisher and Illumina compete on sequencing platform installed base, where laboratory switching costs create durable revenue streams from reagent and consumable sales.
Competition is intensifying in the liquid biopsy and MCED segments, where specialized companies including Guardant Health, Exact Sciences, and Natera are challenging established diagnostics players. These companies have invested heavily in clinical validation studies and are pursuing direct-to-payer contracting strategies that bypass traditional laboratory distribution channels.
In emerging markets, domestic companies in China (BGI, Berry Oncology) and India (Strand Life Sciences) are gaining share by offering locally manufactured platforms at price points below multinational competitors, targeting government-funded screening programs as anchor customers.
Key Companies in the Cancer Biomarkers Market
F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Illumina Inc., QIAGEN, Abbott Laboratories, Merck and Co. Inc., bioMerieux SA, Agilent Technologies Inc., Hologic Inc., Novartis AG, and Genomic Health Inc. are the principal companies operating in the global cancer biomarkers market. These players compete across diagnostic platform breadth, companion diagnostic co-approval portfolios, sequencing throughput, and clinical evidence generation.
Recent Industry Developments
- Illumina: MCED clinical validation milestone (2025)
- Illumina advanced its multi-cancer early detection program through completion of a large-scale prospective clinical validation study in 2025, generating data intended to support FDA premarket approval submission for a blood-based screening test targeting 50+ cancer types.
- Roche Diagnostics: companion diagnostic expansion for immunotherapy (2025)
- Roche Diagnostics received regulatory approvals in 2025 for expanded companion diagnostic indications linked to PD-L1 and tumor mutational burden biomarkers, supporting prescribing decisions for immunotherapy combinations in lung and bladder cancer.
- Guardant Health: liquid biopsy Medicare coverage expansion (2025)
- Guardant Health secured expanded Medicare coverage for its blood-based liquid biopsy platform in 2025, broadening reimbursement eligibility from advanced-stage to earlier-stage colorectal cancer screening applications.
- Thermo Fisher Scientific: NGS oncology panel launch (2026)
- Thermo Fisher Scientific launched an expanded next-generation sequencing oncology panel in early 2026, consolidating multiple biomarker assays into a single workflow designed for community hospital laboratories with lower throughput requirements.
Leading Companies Shaping the Cancer Biomarkers Market
-
Global Players
- F. Hoffmann-La Roche Ltd.
- Thermo Fisher Scientific Inc.
- Illumina, Inc.
- QIAGEN
- Abbott Laboratories
- Merck and Co., Inc.
-
Regional Players
- bioMerieux SA
- Agilent Technologies, Inc.
- Hologic, Inc.
- Genomic Health, Inc.
-
Emerging / Start-up Players
- Guardant Health
- Exact Sciences
- Natera, Inc.
Sources and Research References
- F. Hoffmann-La Roche Ltd. Annual Report 2024 and Diagnostics Division Investor Presentations
- Thermo Fisher Scientific Inc. Annual Report 2024, SEC 10-K Filing
- Illumina Inc. Investor Presentations and FDA Submission Press Releases
- WHO GLOBOCAN Cancer Incidence and Mortality Database (2024 update)
- FDA Premarket Approval (PMA) Database for In Vitro Diagnostics
- EMA European Public Assessment Reports for Companion Diagnostics
- National Cancer Institute (NCI) Cancer Biomarker Research Program Publications
All numerical data in this report is derived from Fact.MR proprietary research. Source references are provided for contextual and directional validation only.
Key Questions This Report Addresses
- What is the projected size of the cancer biomarkers market by 2036?
- Which test type holds the largest share in the cancer biomarkers market?
- What CAGR is the cancer biomarkers market expected to register from 2026 to 2036?
- Which country is the fastest-growing market for cancer biomarkers?
- Who is the leading company in the global cancer biomarkers market?
- What is the incremental opportunity in the cancer biomarkers market between 2026 and 2036?
- Which disease indication dominates cancer biomarker testing demand?
- How is multi-cancer early detection testing expected to affect market growth?
- What role does companion diagnostic co-approval play in biomarker test demand?
- What is the market value of cancer biomarkers in the USA in 2025?
Cancer Biomarkers Market Definition
The cancer biomarkers market covers molecular, protein, genetic, and cellular indicators used in cancer diagnosis, prognosis, predictive testing, and drug discovery. These biomarkers are detected through laboratory tests including immunoassays, PCR, next-generation sequencing, and liquid biopsy platforms, and are used by hospitals, diagnostic laboratories, pharmaceutical companies, and research institutes to guide clinical decisions in oncology.
Cancer Biomarkers Market Inclusions
- Diagnostic, prognostic, and predictive biomarker tests for cancer detection and treatment selection
- Companion diagnostics linked to targeted therapy and immunotherapy drug approvals
- Liquid biopsy platforms detecting circulating tumor DNA, circulating tumor cells, and exosomes
- Research-use-only biomarker assays and panels used in drug discovery and clinical trial stratification
Cancer Biomarkers Market Exclusions
- General clinical chemistry tests not specific to oncology applications
- Imaging-based diagnostic modalities including PET, CT, and MRI scans
- Therapeutic drugs, biologics, and treatment devices used in cancer care
- Population health screening tools not based on molecular or protein biomarker detection
Cancer Biomarkers Market Research Methodology
- Bottom-up revenue modeling using test volume, average reimbursement rates, and installed base data across 20+ countries
- Primary interviews with laboratory directors, oncology department heads, and diagnostic procurement managers at hospitals and reference laboratories
- Cross-referencing regulatory approval databases (FDA, EMA, NMPA) for companion diagnostic and biomarker test clearances
- Validation of segment shares through published clinical guidelines, payer coverage policies, and laboratory test menu disclosures
- Forecasting calibrated against cancer incidence projections (WHO GLOBOCAN), precision medicine adoption rates, and pharmaceutical pipeline analysis
- Company-level revenue triangulation using annual reports, SEC filings, and investor presentations from listed diagnostics and life sciences companies
Scope of Analysis

| Parameter | Details |
|---|---|
| Market size (2025) | USD 14.2 billion |
| Forecast period | 2026 to 2036 |
| CAGR | 13.0% |
| Base year | 2025 |
| Units | USD Billion |
| Segments covered | Test (PSA Tests, CTC Tests, AFP Tests, CA Tests, HER2 Tests, BRCA Tests, ALK Tests, CEA Tests, EGFR Mutation Tests, KRAS Mutation Tests, Others); Disease Indication (Breast Cancer, Lung Cancer, Colorectal Cancer, Melanoma, Blood Cancer, Prostate Cancer, Ovarian Cancer, Stomach Cancer, Liver Cancer, Others); Application (Diagnostics, Prognostics, Predictive Testing, Drug Discovery and Development); End Use (Hospitals, Diagnostic Laboratories, Pharmaceutical and Biotechnology Companies, Research Institutes) |
| Regions covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries profiled | USA, China, India, Germany, UK, Brazil, Japan |
| Companies profiled | F. Hoffmann-La Roche Ltd., Thermo Fisher Scientific Inc., Illumina Inc., QIAGEN, Abbott Laboratories, Merck and Co. Inc. |
| Forecasting approach | Bottom-up revenue modeling validated through primary research, regulatory database cross-referencing, and company-level financial triangulation |
Market Segmentation Analysis
-
Cancer Biomarkers Market Market Segmented by Test:
- PSA Tests
- Total PSA Tests
- Free PSA Tests
- CTC Tests
- CellSearch Based Tests
- Microfluidics Based Tests
- AFP Tests
- Total AFP Tests
- AFP L3 Tests
- CA Tests
- CA 125 Tests
- CA 19 9 Tests
- CA 15 3 Tests
- HER2 Tests
- IHC Testing
- FISH Testing
- BRCA Tests
- BRCA1 Testing
- BRCA2 Testing
- ALK Tests
- FISH Based ALK Testing
- NGS Based ALK Testing
- CEA Tests
- Serum CEA Tests
- Plasma CEA Tests
- EGFR Mutation Tests
- PCR Based EGFR Testing
- NGS Based EGFR Testing
- KRAS Mutation Tests
- PCR Based KRAS Testing
- NGS Based KRAS Testing
- Others
- Biomarker Panel Tests
- Multi Gene Assays
- PSA Tests
-
Cancer Biomarkers Market Market Segmented by Disease Indication:
- Breast Cancer
- Hormone Receptor Positive
- HER2 Positive
- Triple Negative
- Lung Cancer
- Non Small Cell Lung Cancer
- Small Cell Lung Cancer
- Colorectal Cancer
- Early Stage Detection
- Metastatic Cancer
- Melanoma
- Cutaneous Melanoma
- Metastatic Melanoma
- Blood Cancer
- Leukemia
- Lymphoma
- Myeloma
- Prostate Cancer
- Localized Prostate Cancer
- Advanced Prostate Cancer
- Ovarian Cancer
- Epithelial Ovarian Cancer
- Germ Cell Tumors
- Stomach Cancer
- Gastric Adenocarcinoma
- Gastrointestinal Stromal Tumors
- Liver Cancer
- Hepatocellular Carcinoma
- Cholangiocarcinoma
- Others
- Pancreatic Cancer
- Cervical Cancer
- Breast Cancer
-
Cancer Biomarkers Market Market Segmented by Application:
- Diagnostics
- Early Detection
- Screening Programs
- Risk Assessment
- Prognostics
- Disease Progression
- Survival Analysis
- Recurrence Monitoring
- Predictive Testing
- Therapy Selection
- Targeted Therapy Selection
- Immunotherapy Response
- Drug Discovery and Development
- Clinical Trials
- Biomarker Validation
- Companion Diagnostics
- Diagnostics
-
Cancer Biomarkers Market Market Segmented by End Use:
- Hospitals
- Oncology Departments
- Clinical Diagnostics
- Treatment Monitoring
- Diagnostic Laboratories
- Independent Labs
- Centralized Labs
- Specialty Labs
- Pharmaceutical and Biotechnology Companies
- Drug Development
- Clinical Trials
- Companion Diagnostics
- Research Institutes
- Academic Research
- Cancer Research Centers
- Translational Research
- Hospitals
-
Cancer Biomarkers Market Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
What is the current size of the cancer biomarkers market?
The global cancer biomarkers market is valued at USD 14.2 billion in 2025.
What growth rate is expected for the cancer biomarkers market?
The market is projected to grow at a CAGR of 13.0% from 2026 to 2036.
What will the cancer biomarkers market be worth by 2036?
The market is forecast to reach USD 54.469 billion by 2036.
Which test type leads the cancer biomarkers market?
PSA tests lead with a 35.4% share of test-type demand in 2025.
Which disease indication has the largest biomarker testing demand?
Breast cancer holds 24.9% of disease indication share in 2025.
Who is the market leader in cancer biomarkers?
F. Hoffmann-La Roche Ltd. leads with a 21.0% market share in 2025.
Which country is growing fastest in the cancer biomarkers market?
China records the highest country-level CAGR among tracked markets.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- CTC Tests
- CellSearch Based Tests
- Microfluidics Based Tests
- AFP Tests
- Total AFP Tests
- AFP L3 Tests
- CA Tests
- CA 125 Tests
- CA 19 9 Tests
- CA 15 3 Tests
- HER2 Tests
- IHC Testing
- FISH Testing
- BRCA Tests
- BRCA1 Testing
- BRCA2 Testing
- ALK Tests
- FISH Based ALK Testing
- NGS Based ALK Testing
- CEA Tests
- Serum CEA Tests
- Plasma CEA Tests
- EGFR Mutation Tests
- PCR Based EGFR Testing
- NGS Based EGFR Testing
- KRAS Mutation Tests
- PCR Based KRAS Testing
- NGS Based KRAS Testing
- Others
- Biomarker Panel Tests
- Multi Gene Assays
- Y to o to Y Growth Trend Analysis By Test , 2021 to 2025
- Absolute $ Opportunity Analysis By Test , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Disease Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease Indication, 2026 to 2036
- Breast Cancer
- Hormone Receptor Positive
- HER2 Positive
- Triple Negative
- Lung Cancer
- Non Small Cell Lung Cancer
- Small Cell Lung Cancer
- Colorectal Cancer
- Early Stage Detection
- Metastatic Cancer
- Melanoma
- Cutaneous Melanoma
- Metastatic Melanoma
- Blood Cancer
- Leukemia
- Lymphoma
- Myeloma
- Prostate Cancer
- Localized Prostate Cancer
- Advanced Prostate Cancer
- Ovarian Cancer
- Epithelial Ovarian Cancer
- Germ Cell Tumors
- Stomach Cancer
- Gastric Adenocarcinoma
- Gastrointestinal Stromal Tumors
- Liver Cancer
- Hepatocellular Carcinoma
- Cholangiocarcinoma
- Others
- Pancreatic Cancer
- Cervical Cancer
- Breast Cancer
- Y to o to Y Growth Trend Analysis By Disease Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Disease Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Diagnostics
- Early Detection
- Screening Programs
- Risk Assessment
- Prognostics
- Disease Progression
- Survival Analysis
- Recurrence Monitoring
- Predictive Testing
- Therapy Selection
- Targeted Therapy Selection
- Immunotherapy Response
- Drug Discovery and Development
- Clinical Trials
- Biomarker Validation
- Companion Diagnostics
- Diagnostics
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Oncology Departments
- Clinical Diagnostics
- Treatment Monitoring
- Diagnostic Laboratories
- Independent Labs
- Centralized Labs
- Specialty Labs
- Pharmaceutical and Biotechnology Companies
- Drug Development
- Clinical Trials
- Companion Diagnostics
- Research Institutes
- Academic Research
- Cancer Research Centers
- Translational Research
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test
- By Disease Indication
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Disease Indication
- By Application
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test
- By Disease Indication
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Disease Indication
- By Application
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test
- By Disease Indication
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Disease Indication
- By Application
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test
- By Disease Indication
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Disease Indication
- By Application
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test
- By Disease Indication
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Disease Indication
- By Application
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test
- By Disease Indication
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Disease Indication
- By Application
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test
- By Disease Indication
- By Application
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Disease Indication
- By Application
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Disease Indication
- By Application
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test
- By Disease Indication
- By Application
- By End Use
- Competition Analysis
- Competition Deep Dive
- F. Hoffmann-La Roche Ltd.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Thermo Fisher Scientific Inc.
- Illumina, Inc.
- QIAGEN
- Abbott Laboratories
- Other Players (combined)
- F. Hoffmann-La Roche Ltd.
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Test , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Test , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Test , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Test , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Test , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Test , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Test , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Disease Indication, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Test,2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Test
- Figure 6: Global Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Disease Indication
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region,2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 19: Latin America Market Incremental Dollar Opportunity,2026 to 2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity,2026 to 2036
- Figure 22: East Asia Market Incremental Dollar Opportunity,2026 to 2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity,2026 to 2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity,2026 to 2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Test,2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Test
- Figure 29: North America Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Disease Indication
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by End Use
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Test,2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by Test
- Figure 42: Latin America Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 44: Latin America Market Attractiveness Analysis by Disease Indication
- Figure 45: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 47: Latin America Market Attractiveness Analysis by Application
- Figure 48: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 50: Latin America Market Attractiveness Analysis by End Use
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Test,2026 to 2036
- Figure 54: Western Europe Market Attractiveness Analysis by Test
- Figure 55: Western Europe Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 57: Western Europe Market Attractiveness Analysis by Disease Indication
- Figure 58: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 60: Western Europe Market Attractiveness Analysis by Application
- Figure 61: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 63: Western Europe Market Attractiveness Analysis by End Use
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Test,2026 to 2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Test
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Disease Indication
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Application
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Test,2026 to 2036
- Figure 80: East Asia Market Attractiveness Analysis by Test
- Figure 81: East Asia Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 83: East Asia Market Attractiveness Analysis by Disease Indication
- Figure 84: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 86: East Asia Market Attractiveness Analysis by Application
- Figure 87: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 89: East Asia Market Attractiveness Analysis by End Use
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Test,2026 to 2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Test
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Disease Indication
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Test,2026 to 2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Test
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Disease Indication, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Disease Indication,2026 to 2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Disease Indication
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Application,2026 to 2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by End Use,2026 to 2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis