Kidney Cancer Diagnostics Market Size, Share, Growth and Forecast (2026 - 2036)
The Kidney Cancer Diagnostics Market is segmented by Modality (Imaging Diagnostics, Molecular/Biomarker Testing, Biopsy/Histopathology, Nuclear Medicine Imaging), Cancer Type (Renal Cell Carcinoma Diagnostics, Transitional Cell Carcinoma Diagnostics, Wilms Tumor Diagnostics, Other Kidney Cancer Types), and Region. Forecast for 2026 to 2036.
FACT.MR opines the Kidney Cancer Diagnostics market was valued at USD 1,096.22 million in 2026 and is projected to reach USD 2,156.42 million by 2036, expanding at a 7.0% CAGR. Imaging Diagnostics leads by Modality with 54.0% share in 2026.
Kidney Cancer Diagnostics Market Size, Market Forecast and Outlook By FACT.MR
The Kidney Cancer Diagnostics market was valued at USD 1,096.22 million in 2026 and is forecast to reach USD 2,156.42 million by 2036, advancing at a 7.0% CAGR. As per FACT.MR's analysis, expanding clinical adoption, regulatory modernisation, and technology integration across global healthcare systems are accelerating market growth through the forecast decade.
The absolute value expansion of USD 1060.20 million over the 2026 to 2036 period reflects structural investment in diagnostic and therapeutic infrastructure across established and emerging markets. Supply chain localisation initiatives, increased healthcare capital budgets in Asia Pacific, and regulatory compliance investment cycles in North America and Europe are collectively sustaining market momentum across the forecast window.
Country-level CAGR projections span 6.8% in Japan to 7.4% in Germany. Germany advances at 7.4% driven by EU IVDR Regulation 2017/746 compliance driving renal oncology diagnostic system upgrades and DKG-certified cancer centre investment. USA advances at 7.1% driven by CMS reimbursement expansion for ctDNA-based kidney cancer monitoring and FDA companion diagnostic approvals for targeted therapies. Japan advances at 6.8% driven by National Cancer Centre Japan precision oncology programme broadening molecular diagnostic access for renal cell carcinoma staging.
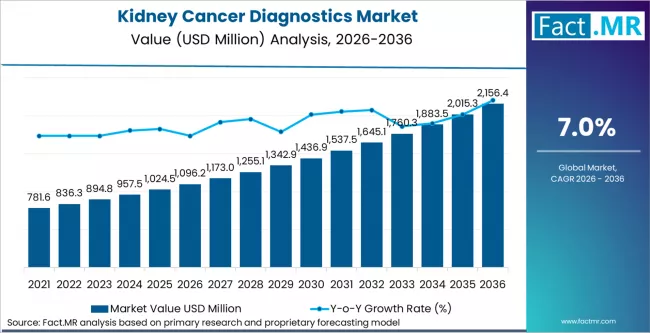
Kidney Cancer Diagnostics Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1,096.22 Million |
| Industry Value (2036) | USD 2,156.42 Million |
| CAGR (2026-2036) | 7.0% |
Kidney Cancer Diagnostics Market Definition
The Kidney Cancer Diagnostics encompasses all products, platforms, and services deployed for renal cell carcinoma diagnostics and related imaging diagnostics applications within clinical, research, and public health settings. Market scope covers Imaging Diagnostics, Molecular/Biomarker Testing, Biopsy/Histopathology, and associated consumables and software systems compliant with applicable regulatory frameworks.
Kidney Cancer Diagnostics Market Inclusions
The report covers Imaging Diagnostics, Molecular/Biomarker Testing, Biopsy/Histopathology, Nuclear Medicine Imaging across all commercial formulations. Segment breakdowns by Cancer Type and region, pricing trend analysis, competitive landscape assessment, and regulatory compliance tracking are fully incorporated across 40-plus countries over the 2026 to 2036 forecast period.
Kidney Cancer Diagnostics Market Exclusions
Combination products incorporating kidney cancer diagnostics as a minor sub-component within a broader platform system are excluded. Experimental or pre-clinical stage technologies without regulatory clearance or established commercial distribution, and niche applications representing less than 1% of total market revenue, fall outside the defined scope.
Kidney Cancer Diagnostics Market Research Methodology
- Primary Research: FACT.MR analysts conducted structured interviews with clinical specialists, procurement directors, and regulatory affairs professionals across 40 countries to validate adoption timelines and pricing dynamics.
- Desk Research: Data aggregation covered regulatory body approvals, institutional procurement records, company annual reports, investor presentations, and official clinical guideline publications.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up methodology applies installed base metrics and regional adoption velocity curves to project market value trajectories through 2036.
- Data Validation and Update Cycle: Segment forecasts are cross-validated against quarterly revenue disclosures from publicly listed market participants and reconciled with procurement data from institutional sourcing bodies.
Summary of Kidney Cancer Diagnostics Market
- Kidney Cancer Diagnostics Market Definition
- The Kidney Cancer Diagnostics covers imaging diagnostics, molecular/biomarker testing, and related platforms used for renal cell carcinoma diagnostics across clinical and research settings globally.
- Demand Drivers in the Market
- Regulatory compliance cycles in major markets drive procurement upgrades and technology refresh investment across imaging diagnostics categories.
- Rising chronic disease burden and ageing population demographics in high-income markets expand the volume of renal cell carcinoma diagnostics procedures requiring advanced kidney cancer diagnostics solutions.
- Greenfield healthcare infrastructure investment in Asia Pacific and Africa creates first-time adoption opportunities for imaging diagnostics and biopsy/histopathology platforms.
- Key Segments Analyzed in the FACT.MR Report
- Imaging Diagnostics holds 54.0% of Modality segment revenue in 2026, anchored by its established clinical utility and broad reimbursement coverage across major markets.
- Renal Cell Carcinoma Diagnostics commands the highest Cancer Type revenue share in 2026, reflecting institutional procurement concentration in high-volume healthcare settings.
- Germany leads regional growth at 7.4% CAGR through 2036, driven by EU IVDR Regulation 2017/746 compliance driving renal oncology diagnostic system upgrades and DKG-certified cancer centre investment.
- Analyst Opinion at FACT.MR
- FACT.MR analysts observe that the Kidney Cancer Diagnostics market is entering a technology consolidation phase where integrated platforms with regulatory-validated performance data command systematic procurement preference over standalone legacy devices. Capital project directors at leading healthcare systems face binary decisions between extending ageing equipment service contracts and committing to next-generation platform upgrades that satisfy both clinical performance and compliance requirements simultaneously.
- Strategic Implications / Executive Takeaways
- Manufacturers must secure regulatory clearance and reimbursement code establishment in the USA, Germany, and Japan simultaneously to qualify for multi-market tender cycles that now specify cross-jurisdictional compliance as a minimum shortlisting criterion.
- Distribution partners and group purchasing organisations that consolidate kidney cancer diagnostics procurement across hospital networks gain disproportionate negotiating leverage, requiring suppliers to invest in GPO contract management capabilities.
- Emerging market entry strategies must account for local manufacturing content requirements and halal or country-specific certification obligations that add 12 to 18 months to standard market entry timelines.
What are the Drivers of Kidney Cancer Diagnostics Market?
The rising global incidences of kidney cancer drive the growth of the kidney cancer diagnostics market. According to the World Health Organization (WHO), more than 431,000 new cases of kidney cancer were reported globally in 2022.
Factors such as an aging population, increased rates of smoking, obesity, hypertension, and environmental exposures are contributing to a steady rise in renal cancer cases worldwide. This increasing burden is translating into a greater demand for early and accurate diagnostic tools to detect the disease in its initial stages, when treatment is most effective. Moreover, heightened awareness of kidney health and the inclusion of kidney screenings in routine checkups are pushing more patients to seek early diagnosis.
Technological advancements in diagnostic imaging and molecular testing are propelling the growth of the kidney cancer diagnostics market. Innovations in CT, MRI, PET scans, and contrast-enhanced ultrasound (CEUS) have vastly improved the ability to detect and differentiate kidney tumors.
The rise of molecular diagnostics and biomarker-based tools is transforming traditional approaches to diagnosis. Liquid biopsies, genomic profiling, and urine-based diagnostic kits are offering non-invasive, accurate, and personalized insights into tumor behavior, enabling clinicians to tailor treatments more effectively. These technologies are being increasingly adopted due to their precision and patient-friendly nature.
The integration of artificial intelligence (AI) and machine learning (ML) into diagnostic workflows is another key factor driving market growth. AI tools are now capable of analyzing imaging scans and histopathological data with impressive speed and accuracy, facilitating early detection and reducing diagnostic errors. This technology also supports predictive analytics and risk stratification, enabling clinicians to make more informed decisions.
The growth of healthcare infrastructure in emerging markets, combined with rising investments by both public and private entities, is expanding diagnostic access. Countries across the Asia-Pacific, Latin America, and the Middle East are enhancing their diagnostic capabilities through hospital expansions, increased insurance coverage, and public health initiatives focused on cancer awareness. Moreover, strategic collaborations among healthcare providers, diagnostic firms, and tech companies are fostering innovation and market expansion.
What are the Regional Trends of Kidney Cancer Diagnostics Market?
North America dominates the global kidney cancer diagnostics market, primarily due to its well-established healthcare infrastructure, high adoption of advanced diagnostic technologies, and increased public awareness regarding early cancer detection. The USA contributes the largest share, driven by the presence of major diagnostic companies, a high prevalence of kidney cancer, and extensive research and development activities.
Factors such as rising healthcare expenditures, government initiatives for early diagnosis, and robust public health infrastructure are driving market growth in Europe. The European Union’s emphasis on personalized medicine and support for oncology research is encouraging the adoption of molecular diagnostic tools. Increasing geriatric populations and growing cancer awareness programs are also contributing to greater diagnostic uptake across the region.
The market growth in Asia Pacific region is driven by improving healthcare infrastructure, growing incidence of kidney-related disorders, and rising patient awareness in countries like China, India, Japan, and South Korea. Moreover, government initiatives to boost cancer screening programs, increasing disposable incomes, and the entry of international diagnostic companies are accelerating access to advanced diagnostics in the region. The demand for cost-effective, portable, and non-invasive testing solutions is also shaping innovation in local markets.
In the Middle East and Southeast Asia, the kidney cancer diagnostics market is still in its early stages of development. Nevertheless, it shows promise due to increasing healthcare investments, a growing middle-class population, and rising awareness of cancer symptoms. However, challenges such as limited access to advanced diagnostics, insufficiently trained professionals, and infrastructure gaps may restrict rapid adoption in the short term.
Kidney Cancer Diagnostics Market Drivers, Restraints, and Opportunities
FACT.MR analysis of the Kidney Cancer Diagnostics market identifies regulatory-driven procurement cycles, technology consolidation among major OEMs, and greenfield healthcare infrastructure expansion in Asia Pacific as the three primary structural forces shaping market velocity through 2036. Organisations that align product development roadmaps with confirmed reimbursement pathways and clinical guideline integration timelines will capture disproportionate share in the early forecast years.
Restraining forces include capital expenditure cycles that extend replacement timelines in budget-constrained health systems, and technical integration complexity that slows the adoption of next-generation platforms in facilities with legacy IT infrastructure. Opportunities are concentrated in point-of-care and decentralised settings where lower-cost platforms with simplified operation interfaces are expanding the addressable market beyond traditional hospital procurement channels.
- Regulatory Compliance Investment: EU MDR/IVDR and US FDA 510(k) modernisation requirements force systematic equipment upgrades across European and North American hospital networks, generating replacement demand independent of clinical performance lifecycle considerations.
- AI Integration and Workflow Automation: Leading OEMs are embedding AI-assisted decision support and automated measurement tools directly into imaging diagnostics platforms, expanding their clinical value proposition and justifying premium pricing in hospital capital procurement budgets.
- Emerging Market Healthcare Access: WHO Universal Health Coverage targets and national health insurance expansions in India, Indonesia, and sub-Saharan Africa are directly funding first-time kidney cancer diagnostics procurement in district and primary care facilities that previously had no access to these diagnostic tools.
Regional Analysis
The Kidney Cancer Diagnostics Market is analysed across North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East & Africa covering 40-plus countries. The full FACT.MR report provides market attractiveness analysis and country-level CAGR projections for each region.
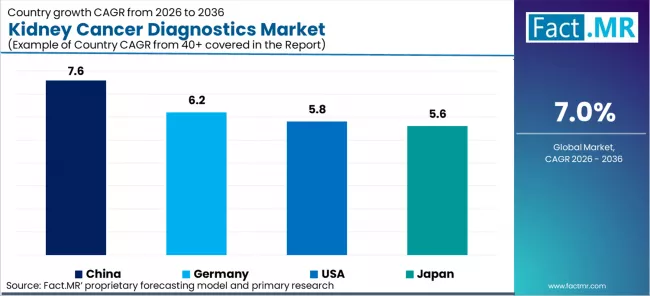
| Country | CAGR (2026 to 2036) |
|---|---|
| Germany | 7.4% |
| USA | 7.1% |
| Japan | 6.8% |
Source: FACT.MR analysis, based on proprietary forecasting model and primary research
Germany: Kidney Cancer Diagnostics Market Analysis
Germany: Demand for kidney cancer diagnostics in Germany is projected to grow at a 7.4% CAGR through 2036. EU IVDR Regulation 2017/746 compliance driving renal oncology diagnostic system upgrades and DKG-certified cancer centre investment. Healthcare procurement agencies in Germany are prioritising imaging diagnostics and molecular/biomarker testing platforms that meet local regulatory standards while delivering measurable performance improvements over legacy systems. Government-funded health programmes and private sector hospital expansion projects jointly support technology adoption across both primary and tertiary care settings in Germany. As per FACT.MR's regional assessment, Germany represents a priority market for kidney cancer diagnostics suppliers seeking exposure to above-average growth trajectories during the 2026 to 2036 forecast period.
USA: Kidney Cancer Diagnostics Market Analysis
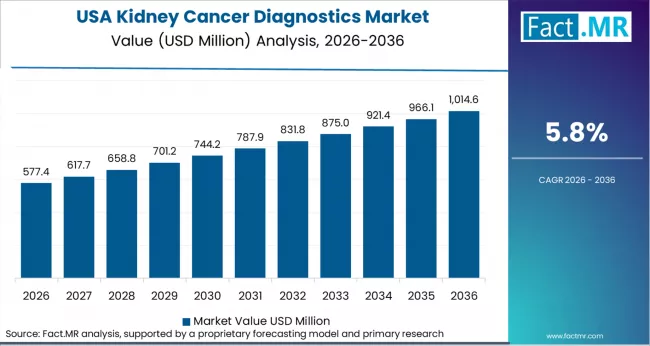
USA: Demand for kidney cancer diagnostics in USA is projected to grow at a 7.1% CAGR through 2036. CMS reimbursement expansion for ctDNA-based kidney cancer monitoring and FDA companion diagnostic approvals for targeted therapies. Healthcare procurement agencies in USA are prioritising imaging diagnostics and molecular/biomarker testing platforms that meet local regulatory standards while delivering measurable performance improvements over legacy systems. Government-funded health programmes and private sector hospital expansion projects jointly support technology adoption across both primary and tertiary care settings in USA. As per FACT.MR's regional assessment, USA represents a priority market for kidney cancer diagnostics suppliers seeking exposure to above-average growth trajectories during the 2026 to 2036 forecast period.
Japan: Kidney Cancer Diagnostics Market Analysis
Japan: Demand for kidney cancer diagnostics in Japan is projected to grow at a 6.8% CAGR through 2036. National Cancer Centre Japan precision oncology programme broadening molecular diagnostic access for renal cell carcinoma staging. Healthcare procurement agencies in Japan are prioritising imaging diagnostics and molecular/biomarker testing platforms that meet local regulatory standards while delivering measurable performance improvements over legacy systems. Government-funded health programmes and private sector hospital expansion projects jointly support technology adoption across both primary and tertiary care settings in Japan. As per FACT.MR's regional assessment, Japan represents a priority market for kidney cancer diagnostics suppliers seeking exposure to above-average growth trajectories during the 2026 to 2036 forecast period.
Category-wise Analysis
Kidney Cancer Diagnostics Market Analysis by Modality
Based on FACT.MR analysis, Imaging Diagnostics hold 54.0% of kidney cancer diagnostics market revenue in 2026. Cross-sectional imaging with CT and MRI forms the clinical cornerstone of renal mass detection, characterisation, and staging, with contrast-enhanced CT recommended as the primary modality in NCCN and EAU kidney cancer guidelines. The transition from analogue to digital pathology and AI-assisted image analysis is expanding the diagnostic throughput of hospital radiology departments without proportional staffing increases.
- AI-Assisted Radiology Adoption: GE HealthCare and Siemens Healthineers both disclosed AI-enhanced renal mass characterisation software clearances from the FDA in 2024, reducing radiologist reporting time for incidentally detected kidney lesions and improving inter-reader consistency in T-staging classifications.
- ctDNA Monitoring Approval: The FDA granted breakthrough device designation to ctDNA-based kidney cancer recurrence monitoring assays in 2024, enabling non-invasive post-nephrectomy surveillance that generates repeat testing episodes throughout the post-treatment follow-up period.
- EU IVDR Compliance Impact: EU In Vitro Diagnostic Regulation 2017/746 mandatory compliance timelines required kidney cancer biomarker test manufacturers to complete performance evaluation recertification, replacing legacy self-certification and raising the evidential bar for new renal oncology assay market entry.
Kidney Cancer Diagnostics Market Analysis by Cancer Type
Renal Cell Carcinoma diagnostics account for 70.0% of kidney cancer diagnostics market revenue in 2026. RCC represents approximately 90% of all kidney cancers in adults, generating the highest testing volume across imaging, biopsy, and molecular modalities. The availability of multiple targeted therapy and immunotherapy approvals for metastatic RCC has created a companion diagnostic testing ecosystem requiring PD-L1, VHL mutation, and mTOR pathway assessment before treatment selection.
- Targeted Therapy Companion Testing: Bristol Myers Squibb's nivolumab plus ipilimumab approval for first-line metastatic RCC established PD-L1 testing as a routine diagnostic step, with Roche's VENTANA PD-L1 assay approved as the companion diagnostic, directly linking RCC diagnosis with immunohistochemistry testing volume.
- VHL Gene Mutation Testing: The FDA approval of Merck's belzutifan for VHL disease-associated RCC in 2021 created a mandatory genetic testing requirement, establishing VHL mutation analysis as a new diagnostic indication distinct from standard RCC staging workup.
- Liquid Biopsy Pipeline: Guardant Health's 2024 investor presentations disclosed clinical validation data for cfDNA panels covering clear cell RCC recurrence biomarkers, positioning ctDNA as a complement to radiological surveillance for post-surgical monitoring in high-risk patients.
Competitive Aligners for Kidney Cancer Diagnostics Market Players
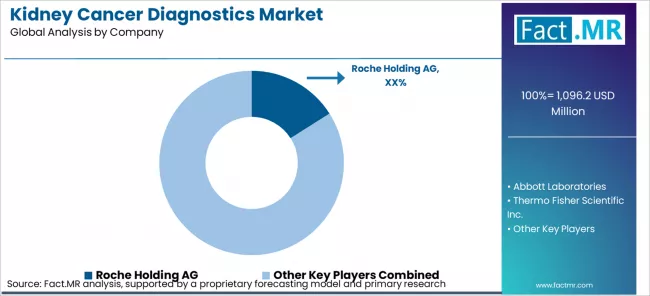
The Kidney Cancer Diagnostics Market is moderately concentrated, with the top five companies collectively holding approximately 55 to 65% of global revenue. Competitive differentiation is primarily driven by regulatory clearance breadth, clinical evidence portfolios, and the depth of integration with hospital information systems and electronic health record platforms. First-mover advantage in securing companion diagnostic designations and reimbursement code establishment creates durable competitive moats in oncology and specialty diagnostic sub-segments.
Vertically integrated manufacturers that control both the instrument platform and consumable supply chain maintain superior gross margins and switching cost advantages compared to device-only players dependent on third-party reagent or consumable supply agreements. Long-term service contracts and application specialist support programmes are becoming the primary competitive battleground as hardware performance gaps narrow between tier-one and tier-two suppliers in established markets.
FMI analysts note that buyer leverage is increasing as hospital group purchasing organisations consolidate procurement across larger facility networks, forcing suppliers to offer volume-tiered pricing and bundled service agreements that compress unit margins. This trend is accelerating platform standardisation within health systems, reducing the frequency of competitive re-evaluation but increasing the stakes of initial contract awards. Suppliers investing in digital health integration and outcome data analytics are best positioned to sustain premium pricing through contract renewal cycles.
Key Players in the Market
- F. Hoffmann-La Roche AG
- Siemens Healthineers AG
- GE HealthCare Technologies Inc.
- Philips Healthcare
- Thermo Fisher Scientific Inc.
- QIAGEN N.V.
- Guardant Health Inc.
- Becton Dickinson & Company
- Bio-Rad Laboratories Inc.
- Illumina Inc.
Bibliography
- European Parliament and Council. (2017). Regulation (EU) 2017/746 on in vitro diagnostic medical devices. Official Journal of the European Union.
- World Health Organization. (2024). Global cancer observatory: Kidney cancer incidence and mortality statistics 2024. IARC.
- National Comprehensive Cancer Network. (2024). NCCN clinical practice guidelines in oncology: Kidney cancer (Version 2.2024). NCCN.
- European Association of Urology. (2024). EAU guidelines on renal cell carcinoma. EAU Annual Congress.
- National Human Genome Research Institute. (2024). DNA sequencing costs and renal oncology genomic panel data. NIH NHGRI.
- NHS England. (2024). Genomic Medicine Service renal cancer pathway update. NHS England.
This Report Addresses
- Market sizing and quantitative forecast metrics detailing kidney cancer diagnostics revenue projections through 2036 across all key segments and regions.
- Segmentation analysis mapping adoption velocity of specific imaging diagnostics and molecular/biomarker testing technologies and evaluating the technical factors driving procurement decisions.
- Regional deployment intelligence comparing growth dynamics in Asia Pacific greenfield markets against brownfield upgrade cycles in European and North American healthcare networks.
- Regulatory compliance assessment analysing how MDR, IVDR, and FDA modernisation requirements are forcing equipment refresh cycles and creating structured replacement demand.
- Competitive landscape evaluation tracking supplier positioning, companion diagnostic designations, reimbursement code coverage, and key account contract award patterns.
- Capital project strategic guidance defining procurement specifications, integration requirements, and total cost of ownership parameters for next-generation platform selection.
- Supply chain vulnerability analysis identifying logistics bottlenecks, component sourcing concentration risks, and lead time pressures affecting market supply.
- Custom data delivery formats including interactive dashboards, raw Excel datasets, and comprehensive PDF narrative reports with regional sub-market detail.
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 1,096.22 million to USD 2,156.42 million, at a CAGR of 7.0% |
| Market Definition | Kidney Cancer Diagnostics encompasses imaging diagnostics, molecular/biomarker testing, and related systems for renal cell carcinoma diagnostics applications across clinical and research settings. |
| Modality Segmentation | Imaging Diagnostics, Molecular/Biomarker Testing, Biopsy/Histopathology, Nuclear Medicine Imaging |
| Cancer Type Segmentation | Renal Cell Carcinoma Diagnostics, Transitional Cell Carcinoma Diagnostics, Wilms Tumor Diagnostics, Other Kidney Cancer Types |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | Germany, USA, Japan, and 40 plus countries |
| Key Companies Profiled | F. Hoffmann-La Roche AG, Siemens Healthineers AG, GE HealthCare Technologies Inc., Philips Healthcare, Thermo Fisher Scientific Inc., QIAGEN N.V., Guardant Health Inc., Becton Dickinson & Company, Bio-Rad Laboratories Inc., Illumina Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up methodology starting with installed base metrics and projecting adoption velocity through 2036 |
Kidney Cancer Diagnostics Market Analysis by Segments
-
Modality:
- Imaging Diagnostics
- Molecular/Biomarker Testing
- Biopsy/Histopathology
- Nuclear Medicine Imaging
-
Cancer Type:
- Renal Cell Carcinoma Diagnostics
- Transitional Cell Carcinoma Diagnostics
- Wilms Tumor Diagnostics
- Other Kidney Cancer Types
-
Region:
- North America (USA, Canada, Mexico)
- Latin America (Brazil, Argentina, Rest of Latin America)
- Europe (Germany, UK, France, Italy, Spain, Nordics, Rest of Europe)
- East Asia (China, Japan, South Korea)
- South Asia & Pacific (India, ASEAN, Australia & New Zealand)
- Middle East & Africa (Saudi Arabia, UAE, South Africa, Rest of MEA)
- Frequently Asked Questions -
How large is the Kidney Cancer Diagnostics Market in 2026?
The Kidney Cancer Diagnostics market is valued at USD 1,096.22 million in 2026.
What will the Kidney Cancer Diagnostics Market reach by 2036?
The market is projected to reach USD 2,156.42 million by 2036.
What is the expected CAGR for the Kidney Cancer Diagnostics Market between 2026 and 2036?
The market is expected to grow at a CAGR of 7.0% between 2026 and 2036.
Which Modality leads the Kidney Cancer Diagnostics Market in 2026?
Imaging Diagnostics leads the Modality segment with 54.0% share in 2026, driven by established clinical utility and broad reimbursement coverage.
What is the fastest-growing country in the Kidney Cancer Diagnostics Market?
Germany leads regional growth at a 7.4% CAGR through 2036, driven by EU IVDR Regulation 2017/746 compliance driving renal oncology diagnostic system upgrades and DKG-certified cancer centre investment.
What does the FACT.MR Kidney Cancer Diagnostics Market report cover?
The report covers market sizing, segmentation analysis, regional intelligence, regulatory compliance assessment, competitive landscape evaluation, and strategic guidance for the 2026 to 2036 forecast period.
What is excluded from the Kidney Cancer Diagnostics Market scope?
Combination products where kidney cancer diagnostics is a minor sub-component, pre-clinical stage technologies, and niche applications below 1% revenue threshold are excluded.
How does FACT.MR forecast the Kidney Cancer Diagnostics Market?
FACT.MR applies a hybrid top-down and bottom-up methodology starting with installed base metrics and cross-validates projections against quarterly revenue disclosures from listed market participants.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test, 2026 to 2036
- Immunohistochemistry
- Complete Blood Count
- Urine Test
- Blood Chemistry
- Immunohistochemistry
- Y to o to Y Growth Trend Analysis By Test, 2021 to 2025
- Absolute $ Opportunity Analysis By Test, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Wilms Tumor
- Transitional Cell Carcinoma
- Kidney Sarcoma
- Renal Cell Carcinoma
- Kidney Lymphoma
- Wilms Tumor
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-User, 2026 to 2036
- Hospital-associated Labs
- Independent Diagnostic Laboratories
- Cancer Research Institutes
- Others
- Hospital-associated Labs
- Y to o to Y Growth Trend Analysis By End-User, 2021 to 2025
- Absolute $ Opportunity Analysis By End-User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test
- By Indication
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Indication
- By End-User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test
- By Indication
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Indication
- By End-User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test
- By Indication
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Indication
- By End-User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test
- By Indication
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Indication
- By End-User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test
- By Indication
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Indication
- By End-User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test
- By Indication
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Indication
- By End-User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test
- By Indication
- By End-User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test
- By Indication
- By End-User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test
- By Indication
- By End-User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test
- By Indication
- By End-User
- Competition Analysis
- Competition Deep Dive
- Roche Holding AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abbott Laboratories
- Thermo Fisher Scientific Inc.
- Illumina Inc.
- Quest Diagnostics Incorporated
- NeoGenomics Laboratories
- Myriad Genetics Inc.
- Sysmex Corporation
- NanoString Technologies Inc.
- Glenmark Pharmaceuticals
- Rosetta Genomics
- Cobolong
- Roche Holding AG
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End-User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Test, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End-User, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 4: Global Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Test
- Figure 6: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 7: Global Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 10: Global Market Y to o to Y Growth Comparison by End-User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End-User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 24: North America Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Test
- Figure 26: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 27: North America Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Indication
- Figure 29: North America Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 30: North America Market Y to o to Y Growth Comparison by End-User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End-User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Test
- Figure 36: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 37: Latin America Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Indication
- Figure 39: Latin America Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 40: Latin America Market Y to o to Y Growth Comparison by End-User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End-User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Test
- Figure 46: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Indication
- Figure 49: Western Europe Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by End-User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End-User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Test
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by End-User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End-User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Test
- Figure 66: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 67: East Asia Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Indication
- Figure 69: East Asia Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 70: East Asia Market Y to o to Y Growth Comparison by End-User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End-User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Test
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by End-User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End-User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Test, 2026 and 2036
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Test, 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Test
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by Indication, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End-User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by End-User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End-User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis