CTC Diagnostics Market Size, Share, Growth and Forecast (2025 - 2035)
CTC Diagnostics Market Size and Share Forecast Outlook 2025 to 2035
The Global CTC Diagnostics Market Is Slated To Reach USD 13.5 Million In 2025, Expected To Further Rise To USD 41.4 Million, Expanding At An 11.8% CAGR. By Product, CTC Diagnostics Kits & Reagents Demand Is Rising Whereas Cancer Stem Cell Research Comprises Bulk Of All Applications.
CTC Diagnostics Market Size and Share Forecast Outlook 2025 to 2035
The global CTC diagnostics market is forecast to reach USD 41.4 million by 2035, up from USD 13.5 million in 2025. During the forecast period, the industry is projected to register at a CAGR of 11.8%.
Growing cancer prevalence drives demand for early detection, technological advances in liquid biopsy improve accuracy and efficiency, regulatory approvals enable clinical adoption; and personalized medicine encourages CTC analysis in treatment monitoring.
Quick Stats of CTC Diagnostics Market
- CTC Diagnostics Market Size (2025): USD 13.5 million
- Projected CTC Diagnostics Market Size (2035): USD 41.4 million
- Forecast CAGR of CTC Diagnostics Market (2025 to 2035): 11.8%
- Leading Product Segment of CTC Diagnostics Market: Kits & Reagents
- Leading Application Segment of CTC Diagnostics Market: Cancer Stem Cell Research
- Key Growth Regions of CTC Diagnostics Market: United Kingdom, China, Japan
- Prominent Players in the CTC Diagnostics Market: Qiagen, Bio-Techne Corporation, Janssen Diagnostics LLC, Advanced Cell Diagnostics Inc., ApoCell Inc., Others
2025-to-2035.webp)
| Metric | Value |
|---|---|
| Industry Size (2025E) | USD 13.5 million |
| Industry Size (2035F) | USD 41.4 million |
| CAGR (2025-2035) | 11.8% |
The CTC diagnostics market is projected to expand from USD 13.5 million in 2025 to USD 41.4 million by 2035, advancing at a CAGR of 11.8%. This trajectory highlights rapid adoption of circulating tumor cell (CTC)-based diagnostic tools in oncology. By 2030, the market is expected to cross USD 23.5 million, with growth intensifying toward 2035 as clinical validation, regulatory approvals, and reimbursement frameworks improve.
The market is gaining importance as cancer detection increasingly shifts toward minimally invasive and liquid biopsy techniques. Between 2025 and 2029, growth will be driven by rising cancer prevalence, greater awareness of personalized medicine, and the demand for non-invasive alternatives to traditional biopsies.
As oncologists emphasize early detection and treatment monitoring, CTC-based platforms will play a critical role in bridging diagnostic precision with patient comfort. This early phase will also see heightened collaboration between academic research institutes and diagnostic companies, which will accelerate the translation of laboratory findings into clinical applications.
From 2030 to 2035, the industry is expected to move into a stronger commercialization phase, with larger adoption in hospitals and diagnostic centers. Technological innovations in microfluidics, next-generation sequencing, and AI-based data analytics will further enhance detection sensitivity and clinical utility. The projected rise to USD 41.4 million underscores the potential of CTC diagnostics to become a core component of oncology practice.
However, the market’s long-term trajectory will depend on addressing cost-effectiveness, ensuring scalability, and securing widespread regulatory endorsements. By 2035, CTC diagnostics are expected to be integrated into precision oncology workflows globally, supporting both therapeutic decisions and patient monitoring.
Key Dynamics of CTC Diagnostics Market Analyzed
The CTC diagnostics market is gaining traction as liquid biopsy technologies emerge as vital tools in oncology. Growth is being fueled by the shift toward non-invasive cancer detection, personalized medicine adoption, and technological progress in cell detection and analysis. At the same time, cost barriers, regulatory challenges, and technical limitations are shaping the pace of adoption across regions.
Rising Cancer Incidence and Focus on Early Detection
The growing prevalence of cancer worldwide is a primary driver of the CTC diagnostics market. Traditional biopsy methods are invasive, often painful, and limited in repeatability. CTC-based diagnostics offer a minimally invasive alternative, enabling real-time monitoring of tumor progression and treatment response through liquid biopsies. As cancer survival rates improve with early diagnosis, demand for advanced diagnostic tools is expected to expand significantly. Governments and health organizations are increasingly promoting early screening, further encouraging adoption.
Clinical demand for non-invasive approaches is particularly high in cases of metastatic cancers, where repeated biopsies are often required. CTC diagnostics provide dynamic insights without the risks associated with invasive tissue extraction. This ability to offer longitudinal monitoring makes CTC platforms highly attractive for oncologists seeking precision-guided treatment strategies, reinforcing the market’s long-term growth outlook.
Integration with Personalized Medicine and Targeted Therapies
The rise of personalized medicine has significantly boosted the relevance of CTC diagnostics. Precision oncology relies heavily on understanding tumor heterogeneity, treatment resistance, and genetic mutations, all of which can be tracked through circulating tumor cells. By enabling personalized therapy adjustments, CTC diagnostics enhance patient outcomes and reduce unnecessary exposure to ineffective drugs. Pharmaceutical companies are increasingly incorporating CTC-based monitoring into clinical trials, strengthening adoption in both research and clinical practice.
As targeted therapies and immunotherapies continue to grow, real-time assessment tools such as CTC platforms are becoming indispensable. They allow clinicians to evaluate treatment efficacy, modify drug regimens promptly, and identify emerging resistance patterns. This integration with next-generation cancer therapies positions CTC diagnostics as a crucial enabler of precision healthcare, driving both clinical adoption and industry investment.
Technological Advancements in Detection and Analysis
Continuous improvements in detection platforms, including microfluidics, immunoassays, and AI-driven analytics, are fueling growth. These innovations are making CTC diagnostics more accurate, sensitive, and scalable, reducing false negatives and increasing clinical confidence. Advances in automation are also lowering turnaround times, making the technology more suitable for routine hospital use.
Beyond detection, improvements in downstream analysis such as genomic sequencing and proteomic profiling are unlocking new applications. These technologies enable deeper insights into tumor biology, offering opportunities for predictive diagnostics and drug development. As cost barriers gradually decline with scaling, CTC diagnostics are expected to transition from niche adoption to mainstream oncology practices across global healthcare systems.
High Cost of Implementation and Limited Accessibility
One of the most pressing challenges is the high cost associated with CTC diagnostic platforms. Advanced equipment, specialized reagents, and skilled personnel make the tests significantly more expensive compared to traditional diagnostic options. This creates accessibility gaps, particularly in low- and middle-income countries where healthcare budgets are constrained. Limited reimbursement policies in several regions further slow adoption, as patients and hospitals are often unwilling to absorb the high upfront costs.
The disparity between advanced economies and developing regions continues to limit global penetration. While the technology shows promise, achieving cost-effectiveness at scale remains a hurdle. Without widespread reimbursement coverage and pricing strategies, the adoption of CTC diagnostics will remain concentrated in well-funded hospitals and research institutions.
Regulatory Hurdles and Clinical Validation Gaps
Regulatory approvals remain a major barrier for CTC diagnostics. Unlike conventional diagnostic methods, liquid biopsy technologies face stricter scrutiny for demonstrating sensitivity, specificity, and reproducibility. Lengthy approval timelines slow down market entry, discouraging smaller innovators from scaling solutions. These challenges make the pathway from research validation to clinical adoption longer and more resource-intensive.
In addition, many CTC-based technologies are still in the translational stage, with limited clinical validation for large-scale deployment. While early studies show promise, more evidence is required to standardize protocols and achieve broad physician confidence. Until consistent validation across diverse patient populations is achieved, market growth may be constrained by skepticism and regulatory bottlenecks.
Technical Limitations and Reliability Concerns
Despite progress, CTC diagnostics still face technical barriers related to sensitivity and reliability. Circulating tumor cells are rare in the bloodstream, and capturing them consistently poses scientific challenges. False negatives and variability in results reduce physician confidence, especially when treatment decisions depend on diagnostic accuracy.
Moreover, integrating CTC results with existing diagnostic workflows such as imaging and pathology remains complex. Without seamless integration, adoption rates in routine oncology practice may remain modest. Overcoming these limitations through technological refinement and standardized practices is essential for ensuring CTC diagnostics become a trusted component of precision medicine.
Regional Trends of CTC Diagnostics Market
North America dominates the CTC diagnostics market, with the United States contributing significantly due to high cancer incidence, strong R&D activity, and frequent product approvals. Leading players are reinforcing their presence through strategic partnerships and clinical trials, positioning the region as the hub for early adoption of liquid biopsy technologies.
Europe is witnessing steady growth, driven by wider use of CTC technologies in cancer prognosis, early detection, and therapy monitoring. Strong regulatory frameworks, coupled with funding for cancer research and innovation, are accelerating clinical uptake. Research institutions across Germany, France, and the United Kingdom are playing an active role in expanding applications of CTC diagnostics.
Asia Pacific is emerging as the fastest-growing region, supported by rising cancer burden, government initiatives, and expanding healthcare infrastructure. China and Japan are investing heavily in liquid biopsy solutions, creating fertile ground for adoption.
The Middle East and Africa are showing moderate growth, with improved healthcare investments, partnerships with global biotech firms, and growing awareness of personalized medicine fostering adoption.
Country-Wise Outlook
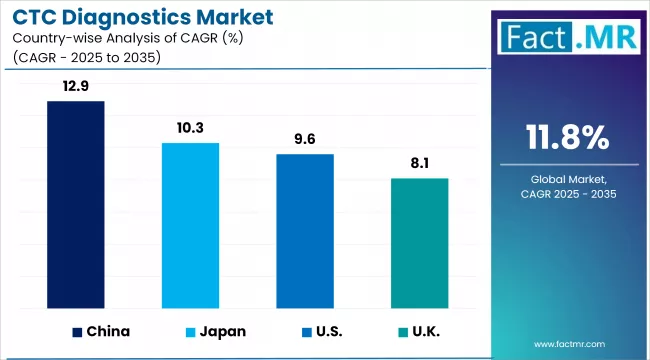
| Countries | CAGR (2025-2035) |
|---|---|
| United Kingdom | 8.1% |
| China | 12.9% |
| Japan | 10.3% |
United Kingdom CTC Diagnostics Market sees Growth Driven by Strong Healthcare Infrastructure
The United Kingdom is at the forefront of integrating Circulating Tumor Cell (CTC) diagnostics into clinical oncology, due to a strong healthcare infrastructure and a commitment to personalized medicine.
The National Health Service (NHS) has launched programs that use liquid biopsy technology to detect cancer-related genetic mutations, including the ESR1 mutation in breast cancer patients. This method allows for earlier intervention and access to targeted therapies, minimizing the need for invasive procedures.
The UK government's emphasis on precision medicine and early cancer detection has resulted in increased funding and support for research into CTC diagnostics. This environment promotes innovation and the creation of novel diagnostic tools.
China’s CTC Diagnostics Market Growth Driven by Rising Cancer Incidence and Healthcare Investments
China's circulating tumor cell (CTC) diagnostics market is expanding rapidly, thanks to a number of key factors. The country's rising cancer incidence is a major driver, necessitating advanced diagnostic techniques. China's strong healthcare infrastructure and major investments in medical research and development drive the market's growth.
Technological advancements play a key role in improving the sensitivity and accuracy of CTC detection techniques. Microfluidics, immunoaffinity-based enrichment, and next-generation sequencing enhance CTC diagnostic efficiency. This progress allows for earlier cancer detection and better monitoring of treatment responses.
China's regulations are changing to support the growth of the CTC diagnostics market. The National Medical Products Administration (NMPA) is responsible for the approval and regulation of medical devices and in-vitro diagnostic reagents. They ensure new diagnostic tools are safe and effective.
The market presents many opportunities, especially in developing personalized medicine. CTC diagnostics enable clinicians to customize treatments based on individual tumor profiles, which leads to better therapeutic outcomes. Additionally, integrating artificial intelligence and machine learning into diagnostic platforms could further improve the accuracy and efficiency of CTC analysis.
Japan’s CTC Diagnostics Market Expands Due to Early Cancer Detection Demand and Innovative Technologies
The Japanese circulating tumor cell (CTC) diagnostics market is growing rapidly because of developments in molecular diagnostics and a strong focus on detecting cancer early.
Several key factors are driving this growth. Japan has rising cancer rates and needs non-invasive diagnostic options. CTC diagnostics provide a promising method to detect and monitor various types of cancer, including breast, prostate, and lung cancer. New technologies, such as microfluidic devices and immunoaffinity-based enrichment techniques, improve the sensitivity and specificity of CTC detection.
Japan's regulatory frameworks are changing to support the use of CTC diagnostics in clinical practice. The Pharmaceuticals and Medical Devices Agency (PMDA) oversees the approval and regulation of medical devices, including in vitro diagnostics. The PMDA's guidelines ensure that new diagnostic tools meet safety and effectiveness standards, making it easier to use them in healthcare settings.
The Japanese market presents many opportunities, especially for developing companion diagnostics. CTC analysis can help identify patients who will gain the most from specific therapies, enabling more personalized treatment plans. Furthermore, integrating artificial intelligence and machine learning into diagnostic platforms could enhance the accuracy and efficiency of CTC analysis.
Category-wise Analysis
Kits & Reagents are Driving Market Growth through Essential CTC Detection Tools
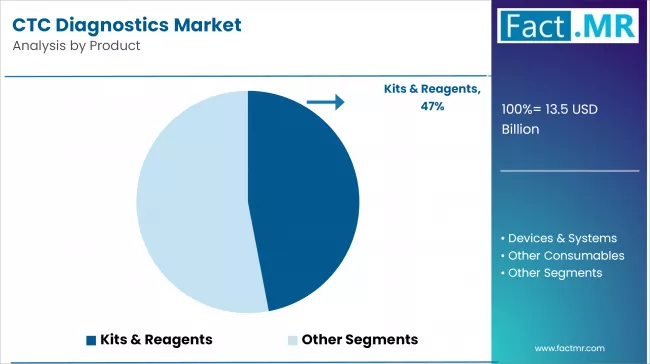
The kits & reagents segment holds the largest market share because they are essential for detecting and enriching CTCs in blood samples. These products are vital for clinical and research applications, allowing laboratories and healthcare providers to perform tests with high accuracy and reliability.
The growing use of liquid biopsy techniques in oncology is driving up the demand for these products. They allow for non-invasive cancer detection and treatment monitoring. The kits and reagents segment is also seeing benefits from ongoing technological improvements that improve test sensitivity and usability.
Demand for CTC Diagnostics Devices and Systems is Rising Simultaneously
The devices and systems segment is expanding rapidly because of the increasing adoption of automated and efficient systems for isolating, detecting, and analyzing circulating tumor cells (CTCs). Systems like microfluidic devices and immunomagnetic separation tools enhance the speed and accuracy of CTC diagnostics.
As healthcare providers seek more efficient and automated solutions for cancer diagnosis and monitoring, the demand for better devices and systems is expected to rise significantly. This growth is also fueled by the increasing trend toward personalized and targeted cancer therapies, which rely on improved CTC technologies to support clinical decision-making.
CTC Enrichment Methods Remains the Preferred Technology
CTC enrichment methods hold the largest market share because they are essential for isolating and purifying CTCs from blood samples for future analysis. Immunomagnetic separation, microfluidics, and density gradient centrifugation are widely used technologies for isolating viable CTCs due to their efficiency and sensitivity. This segment is growing because of the rising demand for early cancer detection and non-invasive diagnostics, along with ongoing improvements in isolation techniques.
The CTC analysis segment is also growing rapidly, largely due to the use of new technologies like artificial intelligence and machine learning in data analysis. These technologies are changing how CTCs are analyzed. They allow for more precise molecular profiling and a deeper understanding of tumor dynamics.
With the increasing focus on personalized medicine, being able to analyze CTCs and identify genetic mutations or resistance markers is becoming crucial for developing tailored treatment strategies. This trend is expected to drive growth in the CTC analysis segment, especially as AI and data-driven insights become more prevalent in clinical oncology.
Cancer Stem Cell Research Leads by Application in the Market
Cancer stem cell research plays an important role in the CTC diagnostics market. It aims to find and isolate circulating tumor cells that behave like stem cells. These cells are believed to fuel tumor growth, spread, and recurrence. Understanding these cells helps in creating targeted therapies and tracking how cancer patients respond to treatment.
EMT biomarker development is another important area. This involves studying epithelial-mesenchymal transition (EMT) markers on circulating tumor cells. This research helps us understand tumor growth and its potential to spread. Analyzing EMT biomarkers allows for the early detection of aggressive cancers and supports doctors in creating personalized treatment plans.
Blood is the Leading Sample Choice for Non-Invasive CTC Detection
Blood samples hold the largest market share for circulating tumor cells because it is the most commonly used sample for CTC detection. Blood-based CTC testing is well-known for being non-invasive, making it the preferred choice for early cancer detection, monitoring, and evaluating treatment.
The high volume of blood samples collected for diagnosis and the established methods for blood-based CTC analysis significantly contribute to this segment’s dominance. Continuous improvements in liquid biopsy technologies focusing on blood samples have further strengthened the market position of the blood specimen segment.
Bone Marrow Specimen Samples are Equally Preferred
Bone marrow specimen samples are equally demanded. This is due to the increasing demand for specific and localized tumor analysis. Bone marrow is a primary site for blood cancers like leukemia and lymphoma, providing important information about CTCs in these conditions.
As biopsy techniques improve, isolating CTCs from bone marrow is becoming more common in research and clinical settings. The rising focus on precision oncology and the need for detailed cancer profiling are also expected to boost the growth of the bone marrow segment, especially concerning targeted therapies and monitoring treatment responses.
Competitive Analysis
The CTC diagnostics market features a competitive landscape with several major players. The regulatory environment is evolving to meet the growing need for CTC-based diagnostic solutions.
Regulatory bodies are working on guidelines and standards to maintain the safety and effectiveness of these diagnostic tools. However, the speed of regulatory approval can differ, which may impact how quickly new technologies become available.
The increasing demand for non-invasive cancer diagnostics, particularly liquid biopsies that use blood samples to detect and monitor cancer, is a key factor driving this growth. These methods provide benefits like reduced discomfort for patients and a better chance of detecting cancer early. However, there are still challenges, including technical issues and inconsistent standards among various platforms and methods.
New technology is crucial for the growth of the CTC diagnostics market. Better microfluidics, immunoaffinity-based enrichment techniques, and next-generation sequencing are increasing the sensitivity and accuracy of CTC detection. These improvements help create more precise and trustworthy diagnostic tools, broadening the clinical applications of CTC diagnostics.
Key Players in the Market
- Qiagen
- Bio-Techne Corporation
- Janssen Diagnostics LLC
- Advanced Cell Diagnostics Inc.
- ApoCell Inc.
- Celltraffix Inc.
- Aviva Biosciences
- Biocept Inc.
- Clearbridge Biomedics
- Fluxion Biosciences
- IVDiagnostics
- Stemcell Technologies Inc.
- Sysmex Corporation
Key Developments
- In March 2024, Bio-Rad Laboratories, Inc. introduced validated antibodies for rare cell and circulating tumor cell (CTC) enumeration, expanding the tools available for CTC research and diagnostics.
- In August 2023, Cell Microsystems, a biotechnology company based in the United States, paid an undisclosed sum to acquire Fluxion Biosciences. This acquisition aims to improve Cell Microsystems' cell analysis capabilities while also providing advanced solutions for research and clinical applications. Fluxion Biosciences is a company based in the United States that develops technologies for isolating and analyzing circulating tumor cells.
Segmentation of CTC Diagnostics Market
-
By Product :
- Devices & Systems
- Kits & Reagents
- Other Consumables
-
By Technology :
- CTC Enrichment Methods
- CTC Detection Methods
- CTC Analysis
-
By Application :
- Cancer Stem Cell Research
- EMT Biomarkers Development
- Tumorigenesis Research
- Others
-
By Sample Type :
- Blood
- Bone Marrow
- Others
-
By Region :
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
- Frequently Asked Questions -
What is the Global CTC Diagnostics Market size in 2025?
The CTC diagnostics market is valued at USD 13.5 million in 2025.
Who are the Major Players Operating in the CTC Diagnostics Market?
Prominent players in the market include ApoCell In, Celltraffix Inc., Aviva Biosciences, Biocept Inc, Clearbridge Biomedics, Fluxion Biosciences, and IVDiagnostics.
What is the Estimated Valuation of the CTC Diagnostics Market by 2035?
The market is expected to reach a valuation of USD 41.4 million by 2035.
At what CAGR is the CTC Diagnostics Market slated to grow during the study period?
The growth rate of the CTC Diagnostics market is 11.8% from 2025-2035.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Supply Side Participants and their Roles
- Producers
- Mid-Level Participants (Traders/ Agents/ Brokers)
- Wholesalers and Distributors
- Value Added and Value Created at Node in the Supply Chain
- List of Components Suppliers
- List of Existing and Potential Buyers
- Supply Side Participants and their Roles
- Investment Feasibility Matrix
- Value Chain Analysis
- Profit Margin Analysis
- Wholesalers and Distributors
- Retailers
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- By Key Regions
- By Key Countries
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis, 2020-2024
- Current and Future Market Size Value (USD Mn) & Volume (Units) Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020-2024 and Forecast 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis By Product, 2020-2024
- Current and Future Market Size Value (USD Mn) & Volume (Units) Analysis and Forecast By Product, 2025-2035
- Devices & Systems
- Kits & Reagents
- Other Consumables
- Y-o-Y Growth Trend Analysis By Product, 2020-2024
- Absolute $ Opportunity Analysis By Product, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis By Technology, 2020-2024
- Current and Future Market Size Value (USD Mn) & Volume (Units) Analysis and Forecast By Technology, 2025-2035
- CTC Enrichment Methods
- CTC Detection Methods
- CTC Analysis
- Y-o-Y Growth Trend Analysis By Technology, 2020-2024
- Absolute $ Opportunity Analysis By Technology, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis By Application, 2020-2024
- Current and Future Market Size Value (USD Mn) & Volume (Units) Analysis and Forecast By Application, 2025-2035
- Cancer Stem Cell Research
- EMT Biomarkers Development
- Tumorigenesis Research
- Others
- Y-o-Y Growth Trend Analysis By Application, 2020-2024
- Absolute $ Opportunity Analysis By Application, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Sample Type
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis By Sample Type, 2020-2024
- Current and Future Market Size Value (USD Mn) & Volume (Units) Analysis and Forecast By Sample Type, 2025-2035
- Blood
- Bone Marrow
- Others
- Y-o-Y Growth Trend Analysis By Sample Type, 2020-2024
- Absolute $ Opportunity Analysis By Sample Type, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Mn) & Volume (Units) Analysis By Region, 2020-2024
- Current Market Size Value (USD Mn) & Volume (Units) Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- East Asia
- South Asia Pacific
- Eastern Europe
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- U.S.
- Canada
- Mexico
- By Product
- By Technology
- By Application
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Application
- By Sample Type
- Key Takeaways
- Latin America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Technology
- By Application
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Application
- By Sample Type
- Key Takeaways
- Western Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- Italy
- France
- U.K.
- Spain
- Russia
- BENELUX
- Rest of Europe
- By Product
- By Technology
- By Application
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Application
- By Sample Type
- Key Takeaways
- East Asia Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Product
- By Technology
- By Application
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Application
- By Sample Type
- Key Takeaways
- South Asia Pacific Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia Pacific
- By Product
- By Technology
- By Application
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Application
- By Sample Type
- Key Takeaways
- Eastern Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltics
- Rest of Eastern Europe
- By Product
- By Technology
- By Application
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Application
- By Sample Type
- Key Takeaways
- Middle East & Africa Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) & Volume (Units) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) & Volume (Units) Forecast By Market Taxonomy, 2025-2035
- By Country
- KSA
- Other GCC Countries
- Turkiye
- South Africa
- Rest of MEA
- By Product
- By Technology
- By Application
- By Sample Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Technology
- By Application
- By Sample Type
- Key Takeaways
- Key Countries Market Analysis
- Value (USD Mn) & Volume (Units)ed States
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Technology
- By Application
- By Sample Type
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Technology
- By Application
- By Sample Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Technology
- By Application
- By Sample Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Technology
- By Application
- By Sample Type
- Value (USD Mn) & Volume (Units)ed States
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Technology
- By Application
- By Sample Type
- Competition Analysis
- Competition Deep Dive
- Qiagen
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Bio-Techne Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Janssen Diagnostics LLC
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Advanced Cell Diagnostics Inc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- ApoCell Inc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Celltraffix Inc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Aviva Biosciences
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Biocept Inc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Clearbridge Biomedics
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Fluxion Biosciences
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- IVDiagnostics
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Stemcell Technologies Inc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Sysmex Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Qiagen
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List Of Table
- Table 1: Global Market Value (USD Mn) Forecast by Region, 2020 to 2035
- Table 2: Global Market Volume (Units) Forecast by Region, 2020 to 2035
- Table 3: Global Market Value (USD Mn) Forecast by Product, 2020 to 2035
- Table 4: Global Market Volume (Units) Forecast by Product, 2020 to 2035
- Table 5: Global Market Value (USD Mn) Forecast by Technology, 2020 to 2035
- Table 6: Global Market Volume (Units) Forecast by Technology, 2020 to 2035
- Table 7: Global Market Value (USD Mn) Forecast by Application, 2020 to 2035
- Table 8: Global Market Volume (Units) Forecast by Application, 2020 to 2035
- Table 9: Global Market Value (USD Mn) Forecast by Sample Type, 2020 to 2035
- Table 10: Global Market Volume (Units) Forecast by Sample Type, 2020 to 2035
- Table 11: North America Market Value (USD Mn) Forecast by Country, 2020 to 2035
- Table 12: North America Market Volume (Units) Forecast by Country, 2020 to 2035
- Table 13: North America Market Value (USD Mn) Forecast by Product, 2020 to 2035
- Table 14: North America Market Volume (Units) Forecast by Product, 2020 to 2035
- Table 15: North America Market Value (USD Mn) Forecast by Technology, 2020 to 2035
- Table 16: North America Market Volume (Units) Forecast by Technology, 2020 to 2035
- Table 17: North America Market Value (USD Mn) Forecast by Application, 2020 to 2035
- Table 18: North America Market Volume (Units) Forecast by Application, 2020 to 2035
- Table 19: North America Market Value (USD Mn) Forecast by Sample Type, 2020 to 2035
- Table 20: North America Market Volume (Units) Forecast by Sample Type, 2020 to 2035
- Table 21: Latin America Market Value (USD Mn) Forecast by Country, 2020 to 2035
- Table 22: Latin America Market Volume (Units) Forecast by Country, 2020 to 2035
- Table 23: Latin America Market Value (USD Mn) Forecast by Product, 2020 to 2035
- Table 24: Latin America Market Volume (Units) Forecast by Product, 2020 to 2035
- Table 25: Latin America Market Value (USD Mn) Forecast by Technology, 2020 to 2035
- Table 26: Latin America Market Volume (Units) Forecast by Technology, 2020 to 2035
- Table 27: Latin America Market Value (USD Mn) Forecast by Application, 2020 to 2035
- Table 28: Latin America Market Volume (Units) Forecast by Application, 2020 to 2035
- Table 29: Latin America Market Value (USD Mn) Forecast by Sample Type, 2020 to 2035
- Table 30: Latin America Market Volume (Units) Forecast by Sample Type, 2020 to 2035
- Table 31: Western Europe Market Value (USD Mn) Forecast by Country, 2020 to 2035
- Table 32: Western Europe Market Volume (Units) Forecast by Country, 2020 to 2035
- Table 33: Western Europe Market Value (USD Mn) Forecast by Product, 2020 to 2035
- Table 34: Western Europe Market Volume (Units) Forecast by Product, 2020 to 2035
- Table 35: Western Europe Market Value (USD Mn) Forecast by Technology, 2020 to 2035
- Table 36: Western Europe Market Volume (Units) Forecast by Technology, 2020 to 2035
- Table 37: Western Europe Market Value (USD Mn) Forecast by Application, 2020 to 2035
- Table 38: Western Europe Market Volume (Units) Forecast by Application, 2020 to 2035
- Table 39: Western Europe Market Value (USD Mn) Forecast by Sample Type, 2020 to 2035
- Table 40: Western Europe Market Volume (Units) Forecast by Sample Type, 2020 to 2035
- Table 41: East Asia Market Value (USD Mn) Forecast by Country, 2020 to 2035
- Table 42: East Asia Market Volume (Units) Forecast by Country, 2020 to 2035
- Table 43: East Asia Market Value (USD Mn) Forecast by Product, 2020 to 2035
- Table 44: East Asia Market Volume (Units) Forecast by Product, 2020 to 2035
- Table 45: East Asia Market Value (USD Mn) Forecast by Technology, 2020 to 2035
- Table 46: East Asia Market Volume (Units) Forecast by Technology, 2020 to 2035
- Table 47: East Asia Market Value (USD Mn) Forecast by Application, 2020 to 2035
- Table 48: East Asia Market Volume (Units) Forecast by Application, 2020 to 2035
- Table 49: East Asia Market Value (USD Mn) Forecast by Sample Type, 2020 to 2035
- Table 50: East Asia Market Volume (Units) Forecast by Sample Type, 2020 to 2035
- Table 51: South Asia Pacific Market Value (USD Mn) Forecast by Country, 2020 to 2035
- Table 52: South Asia Pacific Market Volume (Units) Forecast by Country, 2020 to 2035
- Table 53: South Asia Pacific Market Value (USD Mn) Forecast by Product, 2020 to 2035
- Table 54: South Asia Pacific Market Volume (Units) Forecast by Product, 2020 to 2035
- Table 55: South Asia Pacific Market Value (USD Mn) Forecast by Technology, 2020 to 2035
- Table 56: South Asia Pacific Market Volume (Units) Forecast by Technology, 2020 to 2035
- Table 57: South Asia Pacific Market Value (USD Mn) Forecast by Application, 2020 to 2035
- Table 58: South Asia Pacific Market Volume (Units) Forecast by Application, 2020 to 2035
- Table 59: South Asia Pacific Market Value (USD Mn) Forecast by Sample Type, 2020 to 2035
- Table 60: South Asia Pacific Market Volume (Units) Forecast by Sample Type, 2020 to 2035
- Table 61: Eastern Europe Market Value (USD Mn) Forecast by Country, 2020 to 2035
- Table 62: Eastern Europe Market Volume (Units) Forecast by Country, 2020 to 2035
- Table 63: Eastern Europe Market Value (USD Mn) Forecast by Product, 2020 to 2035
- Table 64: Eastern Europe Market Volume (Units) Forecast by Product, 2020 to 2035
- Table 65: Eastern Europe Market Value (USD Mn) Forecast by Technology, 2020 to 2035
- Table 66: Eastern Europe Market Volume (Units) Forecast by Technology, 2020 to 2035
- Table 67: Eastern Europe Market Value (USD Mn) Forecast by Application, 2020 to 2035
- Table 68: Eastern Europe Market Volume (Units) Forecast by Application, 2020 to 2035
- Table 69: Eastern Europe Market Value (USD Mn) Forecast by Sample Type, 2020 to 2035
- Table 70: Eastern Europe Market Volume (Units) Forecast by Sample Type, 2020 to 2035
- Table 71: Middle East & Africa Market Value (USD Mn) Forecast by Country, 2020 to 2035
- Table 72: Middle East & Africa Market Volume (Units) Forecast by Country, 2020 to 2035
- Table 73: Middle East & Africa Market Value (USD Mn) Forecast by Product, 2020 to 2035
- Table 74: Middle East & Africa Market Volume (Units) Forecast by Product, 2020 to 2035
- Table 75: Middle East & Africa Market Value (USD Mn) Forecast by Technology, 2020 to 2035
- Table 76: Middle East & Africa Market Volume (Units) Forecast by Technology, 2020 to 2035
- Table 77: Middle East & Africa Market Value (USD Mn) Forecast by Application, 2020 to 2035
- Table 78: Middle East & Africa Market Volume (Units) Forecast by Application, 2020 to 2035
- Table 79: Middle East & Africa Market Value (USD Mn) Forecast by Sample Type, 2020 to 2035
- Table 80: Middle East & Africa Market Volume (Units) Forecast by Sample Type, 2020 to 2035
List Of Figures
- Figure 1: Global Market Volume (Units) Forecast 2020 to 2035
- Figure 2: Global Market Pricing Analysis
- Figure 3: Global Market Value (USD Mn) Forecast 2020 to 2035
- Figure 4: Global Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 5: Global Market Y-o-Y Growth Comparison by Product, 2025 to 2035
- Figure 6: Global Market Attractiveness Analysis by Product
- Figure 7: Global Market Value Share and BPS Analysis by Technology, 2025 and 2035
- Figure 8: Global Market Y-o-Y Growth Comparison by Technology, 2025 to 2035
- Figure 9: Global Market Attractiveness Analysis by Technology
- Figure 10: Global Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 11: Global Market Y-o-Y Growth Comparison by Application, 2025 to 2035
- Figure 12: Global Market Attractiveness Analysis by Application
- Figure 13: Global Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 14: Global Market Y-o-Y Growth Comparison by Sample Type, 2025 to 2035
- Figure 15: Global Market Attractiveness Analysis by Sample Type
- Figure 16: Global Market Value (USD Mn) Share and BPS Analysis by Region, 2025 and 2035
- Figure 17: Global Market Y-o-Y Growth Comparison by Region, 2025 to 2035
- Figure 18: Global Market Attractiveness Analysis by Region
- Figure 19: North America Market Incremental $ Opportunity, 2025 to 2035
- Figure 20: Latin America Market Incremental $ Opportunity, 2025 to 2035
- Figure 21: Western Europe Market Incremental $ Opportunity, 2025 to 2035
- Figure 22: East Asia Market Incremental $ Opportunity, 2025 to 2035
- Figure 23: South Asia Pacific Market Incremental $ Opportunity, 2025 to 2035
- Figure 24: Eastern Europe Market Incremental $ Opportunity, 2025 to 2035
- Figure 25: Middle East & Africa Market Incremental $ Opportunity, 2025 to 2035
- Figure 26: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 27: North America Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 28: North America Market Y-o-Y Growth Comparison by Product, 2025 to 2035
- Figure 29: North America Market Attractiveness Analysis by Product
- Figure 30: North America Market Value Share and BPS Analysis by Technology, 2025 and 2035
- Figure 31: North America Market Y-o-Y Growth Comparison by Technology, 2025 to 2035
- Figure 32: North America Market Attractiveness Analysis by Technology
- Figure 33: North America Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 34: North America Market Y-o-Y Growth Comparison by Application, 2025 to 2035
- Figure 35: North America Market Attractiveness Analysis by Application
- Figure 36: North America Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 37: North America Market Y-o-Y Growth Comparison by Sample Type, 2025 to 2035
- Figure 38: North America Market Attractiveness Analysis by Sample Type
- Figure 39: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 40: Latin America Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 41: Latin America Market Y-o-Y Growth Comparison by Product, 2025 to 2035
- Figure 42: Latin America Market Attractiveness Analysis by Product
- Figure 43: Latin America Market Value Share and BPS Analysis by Technology, 2025 and 2035
- Figure 44: Latin America Market Y-o-Y Growth Comparison by Technology, 2025 to 2035
- Figure 45: Latin America Market Attractiveness Analysis by Technology
- Figure 46: Latin America Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 47: Latin America Market Y-o-Y Growth Comparison by Application, 2025 to 2035
- Figure 48: Latin America Market Attractiveness Analysis by Application
- Figure 49: Latin America Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 50: Latin America Market Y-o-Y Growth Comparison by Sample Type, 2025 to 2035
- Figure 51: Latin America Market Attractiveness Analysis by Sample Type
- Figure 52: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 53: Western Europe Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 54: Western Europe Market Y-o-Y Growth Comparison by Product, 2025 to 2035
- Figure 55: Western Europe Market Attractiveness Analysis by Product
- Figure 56: Western Europe Market Value Share and BPS Analysis by Technology, 2025 and 2035
- Figure 57: Western Europe Market Y-o-Y Growth Comparison by Technology, 2025 to 2035
- Figure 58: Western Europe Market Attractiveness Analysis by Technology
- Figure 59: Western Europe Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 60: Western Europe Market Y-o-Y Growth Comparison by Application, 2025 to 2035
- Figure 61: Western Europe Market Attractiveness Analysis by Application
- Figure 62: Western Europe Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 63: Western Europe Market Y-o-Y Growth Comparison by Sample Type, 2025 to 2035
- Figure 64: Western Europe Market Attractiveness Analysis by Sample Type
- Figure 65: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 66: East Asia Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Product, 2025 to 2035
- Figure 68: East Asia Market Attractiveness Analysis by Product
- Figure 69: East Asia Market Value Share and BPS Analysis by Technology, 2025 and 2035
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Technology, 2025 to 2035
- Figure 71: East Asia Market Attractiveness Analysis by Technology
- Figure 72: East Asia Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 73: East Asia Market Y-o-Y Growth Comparison by Application, 2025 to 2035
- Figure 74: East Asia Market Attractiveness Analysis by Application
- Figure 75: East Asia Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 76: East Asia Market Y-o-Y Growth Comparison by Sample Type, 2025 to 2035
- Figure 77: East Asia Market Attractiveness Analysis by Sample Type
- Figure 78: South Asia Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 79: South Asia Pacific Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 80: South Asia Pacific Market Y-o-Y Growth Comparison by Product, 2025 to 2035
- Figure 81: South Asia Pacific Market Attractiveness Analysis by Product
- Figure 82: South Asia Pacific Market Value Share and BPS Analysis by Technology, 2025 and 2035
- Figure 83: South Asia Pacific Market Y-o-Y Growth Comparison by Technology, 2025 to 2035
- Figure 84: South Asia Pacific Market Attractiveness Analysis by Technology
- Figure 85: South Asia Pacific Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 86: South Asia Pacific Market Y-o-Y Growth Comparison by Application, 2025 to 2035
- Figure 87: South Asia Pacific Market Attractiveness Analysis by Application
- Figure 88: South Asia Pacific Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 89: South Asia Pacific Market Y-o-Y Growth Comparison by Sample Type, 2025 to 2035
- Figure 90: South Asia Pacific Market Attractiveness Analysis by Sample Type
- Figure 91: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2025 to 2035
- Figure 94: Eastern Europe Market Attractiveness Analysis by Product
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Technology, 2025 and 2035
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2025 to 2035
- Figure 97: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2025 to 2035
- Figure 100: Eastern Europe Market Attractiveness Analysis by Application
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Sample Type, 2025 to 2035
- Figure 103: Eastern Europe Market Attractiveness Analysis by Sample Type
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 105: Middle East & Africa Market Value Share and BPS Analysis by Product, 2025 and 2035
- Figure 106: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2025 to 2035
- Figure 107: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 108: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2025 and 2035
- Figure 109: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2025 to 2035
- Figure 110: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 111: Middle East & Africa Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 112: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2025 to 2035
- Figure 113: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 114: Middle East & Africa Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 115: Middle East & Africa Market Y-o-Y Growth Comparison by Sample Type, 2025 to 2035
- Figure 116: Middle East & Africa Market Attractiveness Analysis by Sample Type
- Figure 117: Global Market - Tier Structure Analysis
- Figure 118: Global Market - Company Share Analysis