Enterprise Laboratory Informatics Market Size, Share, Growth and Forecast (2026 - 2036)
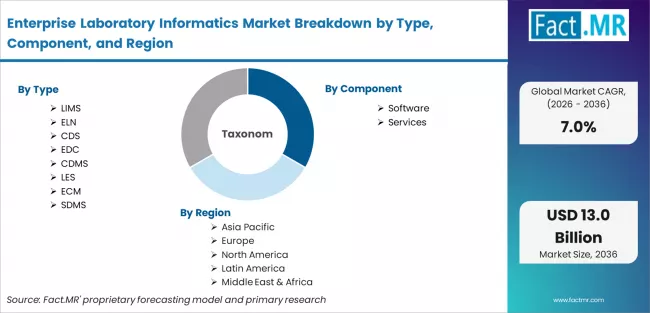
Enterprise Laboratory Informatics Market is segmented by Type (LIMS, ELN, CDS, EDC, CDMS, LES, ECM, and SDMS), Component (Software and Services), Delivery (On-Premise and Cloud), Industry (CRO, CMO, Pharma, Biotech, Chemical, Agriculture, Oil & Gas, and Others) and Region. Forecast for 2026 to 2036.
Fact.MR highlights a shift in the enterprise laboratory informatics market toward compliance-led platform integration. Traditional standalone systems like LIMS and ELN are being replaced by unified, cloud-based solutions that combine multiple functions in one environment. Vendors that cannot offer cloud-native platforms aligned with strict regulatory standards, such as FDA 21 CFR Part 11 and EU Annex 11, risk losing ground. New cloud-first providers are gaining traction by offering faster deployment, built-in compliance features, and lower overall costs.
Summary Of Enterprise Laboratory Informatics Market
- Market Definition
- The market covers LIMS, ELN, CDS, SDMS, LES, EDC, CDMS, and ECM platforms used to automate lab workflows, enforce data integrity, and maintain electronic records compliance across pharma, biotech, CRO, CMO, chemical, and industrial laboratory settings.
- Demand Drivers
- FDA Computer Software Assurance (2025) is easing validation and accelerating cloud LIMS adoption
- China NMPA rules are driving shift from paper to LIMS and ELN systems
- Thermo Fisher acquisitions are expanding LIMS use and increasing cross-sell revenue
- Key Segments Analyzed
- By Type: LIMS holds 20% share in 2026 as the core data platform
- By Component: Software leads with 65% share, driven by SaaS and licences
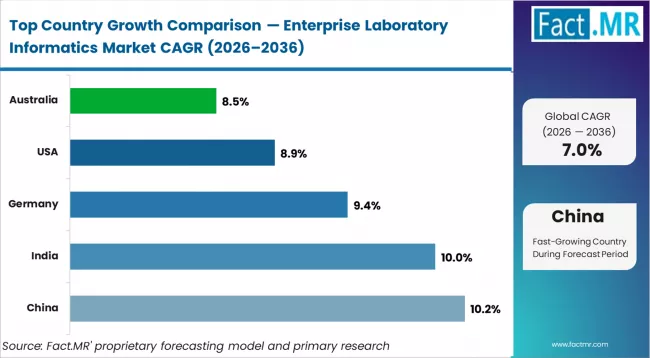
- By Geography: China leads at 10.2% CAGR, followed by India at 10.0%
- Analyst Opinion at FACT.MR
- Shambhu Nath Jha, Principal Consultant at Fact.MR, opines that CXOs will find this report essential for navigating the cloud LIMS migration decision, understanding how FDA's Computer Software Assurance guidance is reducing validation costs for cloud adoption, and identifying which industry verticals are generating the fastest enterprise informatics procurement cycles through 2036.
- Strategic Implications
- Vendors should offer pre-validated cloud LIMS with built-in FDA and EU compliance to reduce validation cost and speed adoption
- Providers should invest in AI-based features like anomaly detection and predictive maintenance to stay competitive
- Companies targeting India and China should build local data infrastructure and partnerships to meet regulatory requirements
- Methodology
- Market sizing is based on LIMS licence and SaaS revenue, validated with vendor financial data
- Regulatory inputs include FDA CSA, EU Annex 11, NMPA China, and CDSCO India guidelines
- Forecasts consider cloud adoption, AI integration, pharma R&D growth, and expert interviews
Segmental Analysis
Enterprise Laboratory Informatics Market Analysis by Type
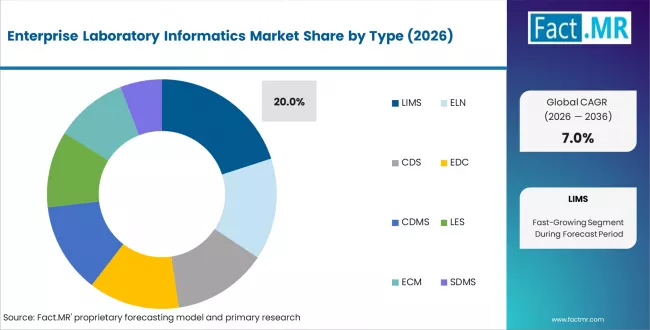
Based on FACT.MR analysis, LIMS holds 20% share in 2026. It remains the core system for sample tracking, compliance, and data management. Most labs adopt LIMS before adding ELN or SDMS modules.
- LabVantage 8.9 LIMS Launch: LabVantage 8.9 added AI features and better compliance tools. It supports integration with ELN and SDMS modules. The platform also expanded into forensic labs through partnerships [1].
- LabWare ASSURE SaaS Launch: LabWare launched ASSURE as a cloud LIMS for mid-sized labs. It targets biotech firms needing compliance without high complexity. This reflects growing SaaS adoption in the market [2].
- ELN and SDMS Adoption Growth Trend: ELN adoption is rising in biotech and research labs. Cloud platforms like Benchling reduce deployment time significantly. Regulatory scope expansion is also increasing ELN demand [3].
Enterprise Laboratory Informatics Market Analysis by Component
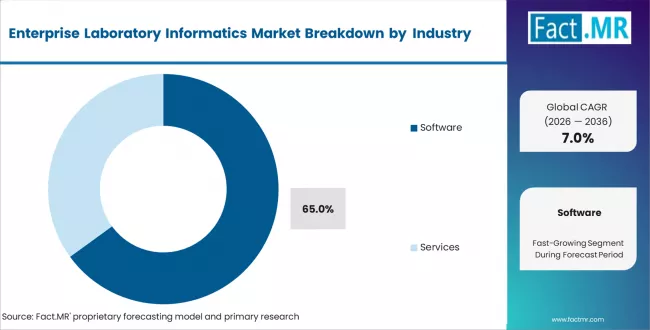
Software is expected to hold 65% share in 2026. Growth is driven by SaaS subscriptions and recurring licence revenue. Buyers prefer ongoing software updates and AI features over one-time services.
- Thermo Fisher Olink and Solventum Acquisitions: Thermo Fisher expanded SampleManager through major acquisitions. This increased its reach in proteomics and infection-control labs. It also strengthened cross-sell opportunities and customer lock-in [4].
- Agilent OpenLab Genohm Acquisition: Agilent extended OpenLab into genomics data management. The platform now includes AI-based analytics and workflow tools. This improves its position in biotech and research labs [5].
- Cloud Software Migration Trend: Hybrid cloud is becoming the standard model. Sensitive data stays on-premise while analytics move to cloud. FDA CSA guidance is reducing validation barriers and accelerating adoption [6].
Drivers, Restraints, and Opportunities
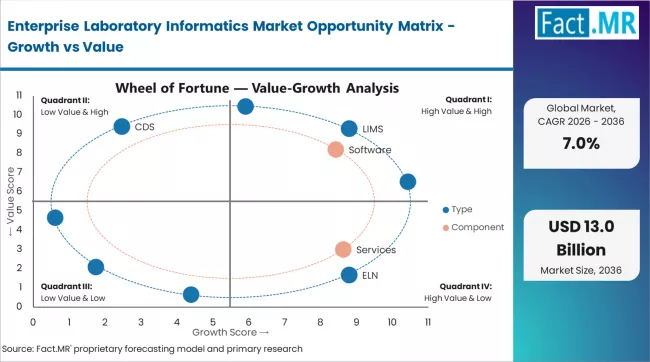
FACT.MR analysts observe that the enterprise laboratory informatics market has evolved from compliance-driven adoption since FDA 21 CFR Part 11. The current USD 6.0 billion market reflects long-term pharma and biotech demand. A new growth phase is emerging with cloud migration, AI workflows, and regulatory expansion in China and India. This is creating a second adoption wave in new markets.
The key tension is between legacy systems and cloud efficiency. Large pharma still relies on validated on-premise LIMS with high switching costs. Cloud-native platforms offer faster deployment and lower costs. Startups prefer SaaS solutions, while big enterprises remain locked into long validation cycles.
- FDA Computer Software Assurance Guidance: FDA’s 2025 CSA guidance supports risk-based validation and reduces workload. It addresses the 61% validation burden reported by pharma firms. This lowers barriers for cloud LIMS adoption globally.
- China NMPA Electronic Records Expansion: China’s 2024 GMP updates mandate electronic records and audit trails. This is driving first-time LIMS and ELN adoption across 4,500+ pharma firms. Both domestic and global vendors are competing for deployments.
- AI-Powered LIMS Integration: AI features like predictive maintenance and anomaly detection are being added. LabVantage 8.9 and Agilent upgrades reflect this trend. AI is improving workflows and increasing platform value.
Regional Analysis
The enterprise laboratory informatics market is analysed across North America, Europe, Asia Pacific, Latin America, and Middle East and Africa, covering 40+ country-level demand shaped by pharmaceutical regulatory compliance frameworks, R&D investment levels, CRO sector size, and national electronic records mandates. The full report offers market attractiveness analysis based on validated site counts, pharma R&D spend, and cloud adoption benchmarks.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 10.2% |
| India | 10.0% |
| USA | 8.9% |
| Germany | 9.4% |
| Australia | 8.5% |
Source: Fact.MR (FACT.MR) analysis, based on proprietary forecasting model and primary research
Asia Pacific Enterprise Laboratory Informatics Market Analysis
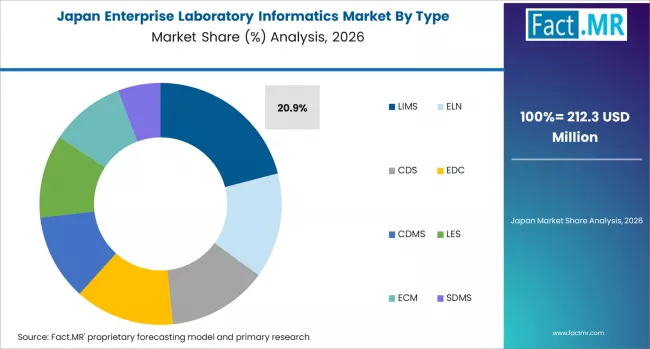
Asia Pacific is the fastest-growing region for laboratory informatics. Growth is supported by stricter compliance rules in China and India, along with rising biotech and CRO activity. Global vendors compete with local suppliers, while Australia remains a smaller but high-value market.
- China: China is leading regional growth with a CAGR of 10.2% through 2036. Updated NMPA rules are pushing pharma companies and CROs to adopt validated LIMS and ELN systems. The country’s large CRO base is increasing demand for multi-module platforms. International vendors such as Thermo Fisher and LabVantage are active, alongside local providers. Rising biotech investment is also expanding the market.
- India: India is showing strong growth at a CAGR of 10.0% through 2036. Updated Schedule M rules are driving LIMS and ELN adoption across pharma manufacturing sites. Large domestic pharma companies and CROs are upgrading informatics systems to support compliance. Government biotech initiatives are also funding new lab infrastructure. This is creating steady demand for enterprise platforms.
FACT.MR's analysis of the enterprise laboratory informatics market in Asia Pacific consists of country-wise assessment including China, India, Japan, South Korea, Australia and New Zealand, and ASEAN. Readers can find NMPA and CDSCO compliance demand data, CRO sector informatics adoption benchmarks, AI LIMS investment trends, and country-level CAGR forecasts by type and delivery mode.
North America Enterprise Laboratory Informatics Market Analysis
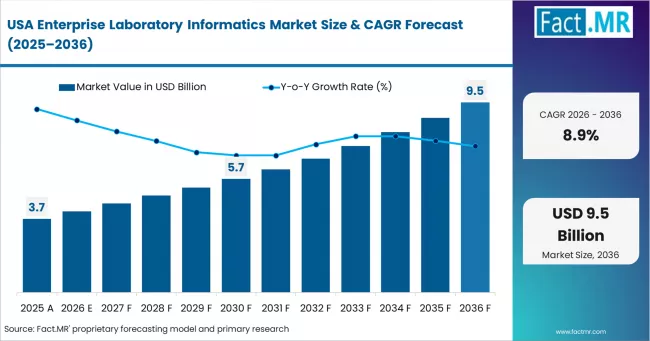
North America is the largest market for enterprise laboratory informatics. It is supported by strict FDA compliance requirements and a large base of pharma, biotech, and CRO labs. Established vendors lead in enterprise accounts, while cloud-native platforms are gaining traction in biotech.
- USA: The United States remains a major market with a CAGR of 8.9% through 2036. FDA guidance is reducing validation burden and supporting cloud migration. Updated Part 11 rules are also expanding compliance needs across ELN, EDC, and CDMS systems. Vendors are launching SaaS solutions to meet mid-sized lab demand. Recent acquisitions are also broadening informatics use beyond traditional pharma.
FACT.MR's analysis of the enterprise laboratory informatics market in North America consists of country-wise assessment including the United States, Canada, and Mexico. Readers can find FDA compliance policy impact data, cloud LIMS adoption benchmarks, CRO procurement trends, and country-level CAGR forecasts by type and industry vertical.
Europe Enterprise Laboratory Informatics Market Analysis
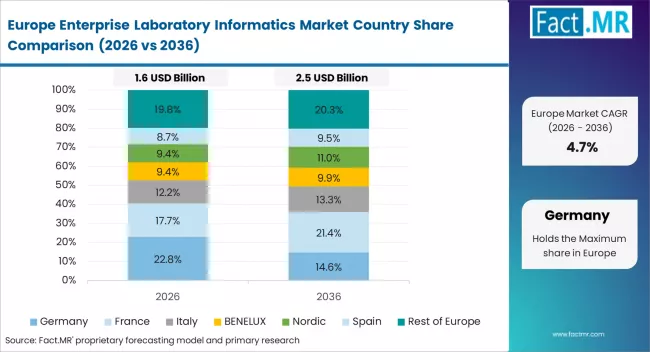
Europe is a compliance-led market for laboratory informatics. EU Annex 11 and EMA data integrity rules continue to shape software upgrades across the region. Germany is the largest market, while Australia is discussed here as a niche compliance-driven growth area.
- Germany: Germany is a key European market with a CAGR of 9.4% through 2036. Pharma companies are upgrading legacy LIMS and ELN systems to meet Annex 11 requirements. Chemical and testing labs are also increasing adoption due to ISO and REACH digitalisation needs. Large companies are investing in lab modernisation as part of broader efficiency programmes. Regulatory pressure will continue to support demand.
- Australia: Australia is growing at a CAGR of 8.5% through 2036. Updated TGA guidance is increasing demand for validated electronic lab records. Mining and food testing labs are also expanding informatics use for traceability and quality control. Research institutes are adopting cloud ELN platforms for collaboration. This is widening demand beyond pharma.
FACT.MR's analysis of the enterprise laboratory informatics market in Europe consists of country-wise assessment including Germany, France, the United Kingdom, Italy, Spain, Nordic countries, and BENELUX. Readers can find EU Annex 11 compliance benchmarks, pharma site upgrade trends, cloud LIMS adoption rates, and country-level CAGR forecasts.
Competitive Aligners for Market Players
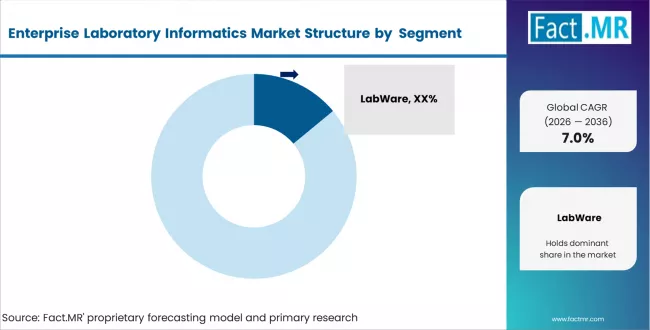
The enterprise laboratory informatics market is moderately concentrated, with a few major vendors controlling a large share of revenue. Companies like Thermo Fisher Scientific, LabWare, and LabVantage dominate core LIMS and ELN segments, while Agilent and PerkinElmer are strong in analytical informatics. Competition today is less about standalone tools and more about offering integrated platforms that combine multiple modules in a single compliant environment. Vendors that can deliver this integration tend to retain customers longer and generate higher revenue per account.
Thermo Fisher has a strong edge because its SampleManager platform is closely tied to its own laboratory instruments, making it difficult for customers to switch. LabWare is known for its high level of customisation and long-standing presence in large pharma companies. LabVantage competes with a flexible, cloud-ready platform and strong presence across healthcare, forensic, and CRO environments.
Large pharma companies usually work with multiple vendors to reduce dependency and meet different lab needs. This limits pricing power but still allows vendors to expand through cross-selling. In contrast, biotech startups are more open to newer cloud-based platforms, where players like Benchling and Dotmatics compete on speed, ease of use, and faster deployment.
Key Players
- LabWare
- LabVantage Solutions, Inc.
- Agilent Technologies, Inc.
- Waters Corporation
- LabLynx, Inc.
- Dassault Systèmes SE
- LABWORKS LLC
- Autoscribe Informatics
- PerkinElmer, Inc.
- Accelerated Technology Laboratories
Bibliography
- [1] LabVantage Solutions Inc. Press Release: LabVantage Launches Version 8.9 LIMS with AI-Driven Productivity Enhancements and Partnership with Netherlands Forensic Institute for Forensic Laboratory Digitalisation. March 2025 and December 2024.
- [2] LabWare Inc. Press Release: LabWare Launches LabWare ASSURE SaaS Cloud LIMS for Mid-Sized Pharmaceutical and Biotech Laboratories Requiring FDA 21 CFR Part 11-Compliant Electronic Records. March 2025.
- [3] US Food and Drug Administration. Guidance for Industry: Part 11, Electronic Records and Signatures in Clinical Investigations Q&A, Clarification on ELN and EDC Scope, and Wearable Device Data Ingestion Compliance Boundaries. October 2024.
- [4] Thermo Fisher Scientific Inc. Annual Report and Press Releases: Acquisition of Olink Proteomics USD 3.1 Billion Completed July 2024, Acquisition of Solventum USD 4.1 Billion Completed February 2025, SampleManager LIMS Portfolio Expansion.
- [5] Agilent Technologies Inc. Press Release: Acquisition of Genohm to Expand OpenLab Informatics Software into Next-Generation Sequencing Data Management for Genomics and Biotech Laboratory Customers. April 2025.
- [6] FDA / CodePhusion. Computer Software Assurance Final Guidance: Risk-Based Least-Burdensome Software Testing for Cloud LIMS and ELN. September 2025. fda.gov/csa-guidance and 2024 State of Validation Report: 61% of Pharma Organisations Report Increased Validation Workload.
This Report Addresses
- Strategic intelligence on FDA Computer Software Assurance guidance, EU GMP Annex 11 requirements, China NMPA electronic records mandates, India CDSCO Schedule M GMP compliance deadlines, and Australia TGA digital records alignment driving enterprise laboratory informatics procurement through 2036.
- Market forecast projecting growth from USD 6.6 billion in 2026 to USD 19.8 billion by 2036 at a 7.0% CAGR, segmented by type, component, delivery mode, industry vertical, and region.
- Growth opportunity mapping across China's NMPA compliance first-time LIMS adoption wave, India's Schedule M implementation procurement, US FDA CSA-enabled cloud migration acceleration, Germany's EU Annex 11 upgrade cycle, and Australia's TGA and mining laboratory modernisation.
- Country CAGR outlook highlighting China 10.2%, India 10.0%, Germany 9.4%, Australia 8.5%, USA 8.9%, and Japan 8.2%, supported by pharma R&D spend data, validated site counts, and regulatory compliance timeline benchmarks.
- Competitive landscape analysis covering Thermo Fisher, LabWare, LabVantage, Agilent, PerkinElmer, Waters, Dassault Systèmes, Autoscribe, and cloud challengers Benchling and Dotmatics, focusing on platform integration, AI module roadmaps, and acquisition impact.
- Technology tracking including AI-powered anomaly detection in LIMS, cloud-native ELN deployment, hybrid on-premise and cloud informatics architectures, FAIR data principles implementation, and blockchain-secured audit trail adoption for GxP laboratories.
- Regulatory analysis covering FDA 21 CFR Part 11, Computer Software Assurance guidance, EU GMP Annex 11, PIC/S data integrity guidelines, NMPA China electronic records GMP requirements, India CDSCO Schedule M, and Australia TGA PIC/S alignment.
- Report deliverables include PDF report, Excel datasets, PowerPoint summary, and dashboards supported by Thermo Fisher, Agilent, LabVantage, and PerkinElmer financial disclosures, FDA regulatory publications, and primary research interviews.
Scope of Report
| Attribute | Details |
|---|---|
| Quantitative Units | USD 6.6 billion (2026) to USD 19.8 billion (2036), at a CAGR of 7.0% |
| Market Definition | Software platforms and services for laboratory workflow automation, electronic records management, and data integrity compliance across pharma, biotech, CRO, CMO, chemical, agriculture, oil and gas, and industrial laboratory sectors. |
| Type Segmentation | LIMS, ELN, CDS, EDC, CDMS, LES, ECM, SDMS |
| Component Segmentation | Software, Services |
| Delivery Segmentation | On-Premise, Cloud |
| Industry Segmentation | CRO, CMO, Pharma, Biotech, Chemical, Agriculture, Oil & Gas, Others |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Canada, Mexico, UK, Germany, France, Italy, Spain, Nordic, BENELUX, Japan, South Korea, China, India, Australia and New Zealand, ASEAN, Brazil, Argentina, Chile, Saudi Arabia, GCC, Turkey, South Africa, Rest of MEA |
| Key Companies Profiled | LabWare, LabVantage Solutions, Agilent Technologies, Waters Corporation, LabLynx, Dassault Systèmes, LABWORKS LLC, Autoscribe Informatics, PerkinElmer, Accelerated Technology Laboratories |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using LIMS software revenue by type and industry, SaaS adoption rates, pharma R&D spend, government compliance programme timelines, and primary interviews with lab informatics procurement and validation specialists. |
Enterprise Laboratory Informatics Market by Segments
-
By Type :
- LIMS
- ELN
- CDS
- EDC
- CDMS
- LES
- ECM
- SDMS
-
By Component :
- Software
- Services
-
By Delivery :
- On-Premise
- Cloud
-
By Industry :
- CRO
- CMO
- Pharma
- Biotech
- Chemical
- Agriculture
- Oil & Gas
- Others
-
By Region :
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Mexico
- Argentina
- Chile
- Rest of Latin America
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Rest of Middle East and Africa
- North America
- Frequently Asked Questions -
How large is the enterprise laboratory informatics market in 2025?
The global enterprise laboratory informatics market was valued at USD 6.0 billion in 2025.
What will the market size be in 2026?
Based on Fact.MR analysis, the market is estimated to reach USD 6.6 billion in 2026.
What is the projected market size by 2036?
The market is projected to reach USD 19.8 billion by 2036, generating USD 13.2 billion in absolute revenue opportunity over the forecast period from 2026 to 2036.
What is the expected CAGR?
The forecast CAGR from 2026 to 2036 is 7.0%.
Which type segment leads the enterprise laboratory informatics market?
LIMS holds approximately 20% share in 2026.
Which component segment dominates?
Software holds approximately 65% share in 2026.
Which country shows the fastest growth?
China leads at 10.2% CAGR through 2036.
What is enterprise laboratory informatics?
It refers to software systems that manage lab data, workflows, and compliance processes.
Why is LIMS important in laboratories?
LIMS ensures sample tracking, data integrity, and regulatory compliance.
What is driving cloud adoption in this market?
Reduced validation burden and faster deployment are key factors.
Which industries use laboratory informatics the most?
Pharma, biotech, CROs, chemicals, and food testing labs are major users.
What is the role of ELN in laboratories?
ELN replaces paper notebooks with digital records for experiments.
What is a key market trend?
Shift toward integrated, cloud-based multi-module platforms.
Why are regulations important in this market?
Compliance with FDA and EU rules is mandatory for lab operations.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type, 2026 to 2036
- LIMS
- ELN
- CDS
- EDC
- CDMS
- LES
- ECM
- SDMS
- LIMS
- Y to o to Y Growth Trend Analysis By Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Component
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Component, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Component, 2026 to 2036
- Software
- Services
- Software
- Y to o to Y Growth Trend Analysis By Component, 2021 to 2025
- Absolute $ Opportunity Analysis By Component, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Component
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Component
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Component
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Component
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Component
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Component
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Component
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Component
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Component
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Component
- Competition Analysis
- Competition Deep Dive
- LabWare
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- LabVantage Solutions, Inc.
- Agilent Technologies, Inc.
- Waters Corporation
- LabLynx, Inc.
- Dassault Systèmes SE
- LABWORKS LLC
- Autoscribe Informatics
- PerkinElmer, Inc.
- Accelerated Technology Laboratories
- LabWare
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Component, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Component, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Component
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Type
- Figure 23: North America Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Component
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Component
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Component
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Component
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Component
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Component
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Component, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Component, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Component
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis