Medical Grade Polypropylene Market Size, Share, Growth and Forecast (2026 - 2036)
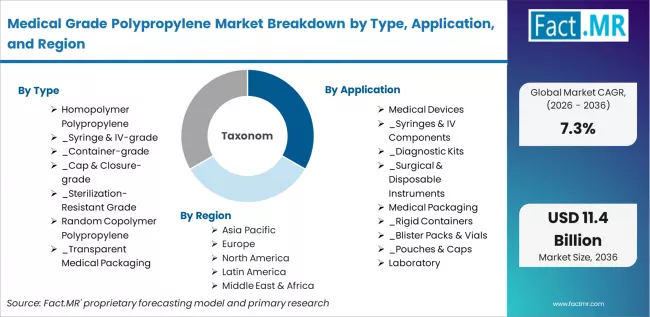
The Medical Grade Polypropylene Market is segmented by Type (Homopolymer Polypropylene (Syringe & IV-grade, Container-grade, Cap & Closure-grade, Sterilization-Resistant Grade), Random Copolymer Polypropylene (Transparent Medical Packaging Grade, Diagnostic Labware Grade, Flexible Medical Component Grade), Impact Copolymer Polypropylene, and Other Polypropylene Types), Application (Medical Devices (Syringes & IV Components, Diagnostic Kits, Surgical & Disposable Instruments), Medical Packaging (Rigid Containers, Blister Packs & Vials, Pouches & Caps), Laboratory Equipment, Nonwoven Medical Textiles, Drug Delivery Systems, and Other Applications) and Region. Forecast for 2026 to 2036.
Fact.MR opines the medical grade polypropylene market was valued at USD 5.25 billion in 2025. Sales are expected to reach USD 5.63 billion in 2026 and USD 11.40 billion by 2036. Homopolymer Polypropylene is poised to lead by Type with 58.5% share in 2026 and Medical Devices is estimated to command by Application in 2026.
Medical Grade Polypropylene Market Size, Market Forecast and Outlook By Fact.MR
The medical grade polypropylene market was valued at USD 5.25 billion in 2025, projected to reach USD 5.63 billion in 2026, and is forecast to expand to USD 11.40 billion by 2036 at a 7.3% CAGR. Medical device manufacturers transitioning from glass and metal components to injection-molded polypropylene parts are driving demand for sterilization-resistant, biocompatible polymer grades that meet pharmacopeial purity standards. Syringe and IV component producers that once managed resin procurement as a commodity plastics purchase now specify medical-grade polypropylene with documented biocompatibility, extractable and leachable profiles, and validated gamma and ethylene oxide sterilization compatibility.
Summary of Medical Grade Polypropylene Market
- Market Overview
- The medical grade polypropylene market is valued at USD 5.25 billion in 2025 and is projected to reach USD 11.40 billion by 2036.
- The industry is expected to grow at a 7.3% CAGR from 2026 to 2036, creating an incremental opportunity of USD 5.77 billion.
- The market is a specification-intensive, regulatory-compliance-driven polymer category where pharmacopeial purity standards, sterilization compatibility validation, and biocompatibility certification define procurement decisions.
- Demand and Growth Drivers
- Demand is accelerating as medical device manufacturers transition from glass and metal to injection-molded polypropylene components that meet sterilization-resistant biocompatibility standards.
- Single-use disposable medical product adoption and nonwoven surgical textile procurement are sustaining high-volume medical-grade polypropylene consumption.
- Government medical device production incentive schemes in India and localization mandates in China are expanding domestic medical-grade resin procurement volumes.
- Among key countries, India leads at 9.8% CAGR, followed by China at 9.0%, South Korea at 7.5%, the USA at 6.5%, Germany at 6.0%, the UK at 5.8%, and Japan at 5.5%.
- Product and Segment View
- Medical grade polypropylene is a high-purity, biocompatible thermoplastic polymer produced under controlled manufacturing conditions with documented extractable and leachable profiles, gamma and ethylene oxide sterilization compatibility, and pharmacopeial compliance for medical device and pharmaceutical packaging applications.
- Primary applications span syringe and IV components, diagnostic kits, surgical instruments, medical packaging, nonwoven medical textiles, and drug delivery systems.
- Homopolymer Polypropylene leads by Type with 58.5% share in 2026.
- Medical Devices leads by Application with 31.0% share in 2026.
- Scope includes homopolymer, random copolymer, and impact copolymer medical-grade polypropylene, excluding commodity polypropylene, polypropylene fiber for non-medical textiles, and polypropylene automotive components.
- Geography and Competitive Outlook
- India and China are the fastest-growing markets due to medical device manufacturing expansion under government incentive programs and localization mandates, while the USA and Germany represent mature high-specification markets driven by pre-filled syringe production and medical technology cluster demand.
- Competition is shaped by pharmacopeial compliance certification, sterilization compatibility validation data, and medical device OEM resin qualification relationships.
- Key companies include SABIC, Borealis AG, LyondellBasell Industries, ExxonMobil Chemical, Braskem, TotalEnergies Corbion, INEOS Olefins & Polymers, Trinseo, LG Chem, and Avient Corporation.
- Analyst Opinion at Fact.MR
- 'Shambhu Nath Jha', Principal Consultant for Chemicals and Materials, opines: 'In my analysis, I have observed that the true procurement barrier for medical grade polypropylene is not resin availability but the 12 to 18 month biocompatibility and extractable/leachable testing cycle required to qualify a new resin grade for medical device production. Device manufacturers locked into qualified resin suppliers face switching costs that extend beyond material pricing into regulatory filing amendments requiring agency review periods. Resin producers lacking USP and EP pharmacopeial compliance documentation face permanent exclusion from medical device customer procurement programs. Companies delaying medical-grade production line investment risk missing the qualification window as device OEMs finalize resin supply chains for next-generation single-use device platforms.' Strategic Implications / Executive Takeaways
- Polypropylene resin producers must invest in dedicated medical-grade production lines with full USP and EP pharmacopeial compliance documentation to qualify for medical device OEM procurement programs.
- Medical device procurement teams should initiate resin qualification programs at least 18 months before new device launch timelines to accommodate biocompatibility and extractable/leachable testing requirements.
- Nonwoven medical textile converters must establish multi-source medical-grade polypropylene resin supply agreements to maintain production continuity during resin allocation periods.
Medical Grade Polypropylene Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 5.63 billion |
| Industry Value (2036) | USD 11.40 billion |
| CAGR (2026 to 2036) | 7.3% |
Source: Future Market Insights, 2026
The absolute dollar opportunity of USD 5.77 billion between 2026 and 2036 reflects expanding medical device production volumes and a structural shift toward single-use disposable medical products that consume medical-grade polypropylene in higher per-unit quantities. Nonwoven medical textile applications including surgical gowns, face masks, and drape materials represent a high-volume consumption segment sustained by hospital infection control procurement standards. Drug delivery system applications for pre-filled syringe barrels and inhaler components require polypropylene grades with narrow melt flow index specifications to maintain dimensional consistency across high-volume injection molding production.
All major medical polypropylene specification corridors reflect accelerated adoption. India sets the pace with a 9.8% CAGR as domestic medical device manufacturing capacity expands under government production-linked incentive schemes. China follows at 9.0% with medical device localization mandates driving domestic medical-grade resin procurement. South Korea tracks at 7.5% through diagnostic device and medical packaging production growth. United States operations advance at 6.5%, supported by pre-filled syringe and single-use device production expansion. Germany registers a 6.0% pace anchored by medical technology cluster manufacturing demand. United Kingdom expands at 5.8% through NHS single-use device procurement volumes. Japan tracks at 5.5% through aging population-driven medical device consumption growth.
Why is the Medical Grade Polypropylene Market Growing?
Market expansion is being supported by the exponential increase in disposable medical device consumption and the corresponding need for sophisticated biocompatible polymer materials in healthcare applications across global medical manufacturing operations. Modern healthcare manufacturers are increasingly focused on advanced medical grade polypropylene systems that can improve product safety, enhance sterilization compatibility, and optimize manufacturing efficiency while meeting stringent regulatory requirements. The proven efficacy of medical grade polypropylene in various healthcare applications makes it an essential component of comprehensive medical device manufacturing strategies and pharmaceutical packaging programs.
The growing emphasis on patient safety standards and single-use device integration is driving demand for advanced medical grade polypropylene that meets stringent performance specifications and operational requirements for healthcare applications. Medical device manufacturers' preference for reliable, high-performance polymer materials that can ensure consistent product outcomes is creating opportunities for innovative resin formulations and customized grade solutions. The rising influence of healthcare quality initiatives and medical device regulations is also contributing to increased adoption of premium-grade medical polypropylene across different device categories and manufacturing systems requiring advanced polymer technology.
Opportunity Pathways - Medical Grade Polypropylene Market
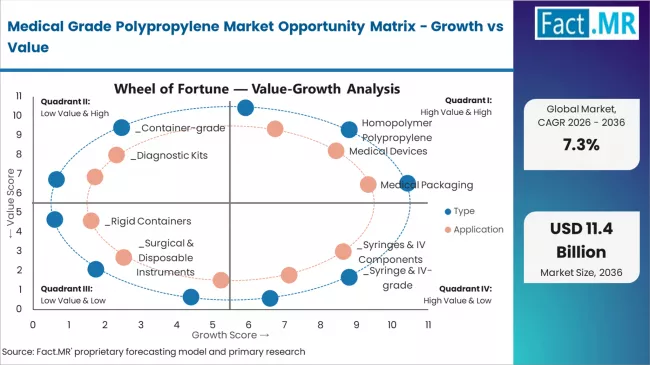
The medical grade polypropylene market represents a transformative growth opportunity, expanding from USD 5.25 billion in 2025 to USD 10.60 billion by 2035 at a 7.3% CAGR. As healthcare manufacturers prioritize patient safety optimization, sterile device production, and manufacturing excellence in complex medical environments, medical grade polypropylene has evolved from a commodity polymer to an essential healthcare component enabling precise medical device fabrication, comprehensive sterilization compatibility strategies, and multi-application material operations across hospital supply platforms and pharmaceutical packaging applications.
The convergence of disposable device adoption acceleration, increasing biocompatible polymer penetration, advanced resin formulation integration, and stringent regulatory compliance mandates creates momentum in demand. High-purity homopolymer grades offering superior autoclave resistance, cost-effective random copolymer solutions balancing transparency with economics, and specialized resins for pharmaceutical packaging applications will capture market premiums, while geographic expansion into high-growth Asian medical manufacturing hubs and emerging healthcare ecosystems will drive volume leadership. Healthcare manufacturer emphasis on material innovation and quality reliability provides structural support.
- Pathway A - Homopolymer Polypropylene Type Dominance: Leading with 58.5% market share, homopolymer applications drive primary demand through comprehensive medical device manufacturing workflows requiring sophisticated high-performance resin solutions for sterilization resistance and structural integrity optimization. Advanced syringe-grade and container-grade formulations enabling improved autoclave stability, enhanced chemical resistance, and superior mechanical property outcomes command premium pricing from medical device manufacturers requiring stringent performance specifications and regulatory compliance. Expected revenue pool: USD 3.07-6.20 billion.
- Pathway B - Medical Devices Application Leadership: Dominating with 31.0% market share through optimal balance of disposable device necessity and biocompatibility requirements, medical device applications serve the largest healthcare manufacturing segment while meeting diverse patient safety demands. This application addresses both single-use syringe manufacturing and diagnostic kit production requirements, making it the preferred category for healthcare manufacturers and medical suppliers seeking comprehensive material capabilities. Opportunity: USD 1.63-3.29 billion.
- Pathway C - Asian Market Acceleration: China (8.4% CAGR) and India (8.1% CAGR) lead global growth through medical device manufacturing expansion, healthcare infrastructure development, and pharmaceutical packaging capability advancement. Strategic partnerships with local healthcare manufacturers, resin localization expertise, and supply chain optimization enable the expansion of medical grade polypropylene applications in major medical device production hubs and pharmaceutical packaging centers. Geographic expansion upside: USD 1.18-2.68 billion.
- Pathway D - Random Copolymer Segment Growth: Random copolymer polypropylene with 27.0% market share serves critical transparent medical packaging applications requiring specialized clarity for diagnostic labware and pharmaceutical containers. Optimized transparent grades supporting multiple sterilization methods, regulatory compliance requirements, and proven optical performance maintain significant volumes from pharmaceutical manufacturers and laboratory equipment producers. Revenue potential: USD 1.42-2.86 billion.
- Pathway E - Advanced Resin Technologies & Ultra-High Purity Innovation: Companies investing in sophisticated pharmaceutical-grade formulations, advanced sterilization-resistant technologies, and ultra-high purity polymer systems gain competitive advantages through consistent quality delivery and regulatory approval. Advanced capabilities enabling customized molecular weight specifications and rapid grade development capture premium healthcare partnerships. Technology premium: USD 0.84-1.70 billion.
- Pathway F - Medical Packaging Applications: Medical packaging with 24.0% market share serves growing pharmaceutical and healthcare product packaging applications requiring specialized barrier properties for rigid containers, blister packs, and closure systems. Resins optimized for contact with drugs and medical products, FDA compliance specifications, and proven sterilization compatibility maintain substantial volumes from pharmaceutical packaging converters and medical product manufacturers. Application value: USD 1.26-2.54 billion.
- Pathway G - Emerging Applications & Market Development: Beyond traditional medical device and packaging applications, medical grade polypropylene in laboratory equipment manufacturing, nonwoven medical textile production, and advanced drug delivery systems represent growth opportunities. Companies developing novel formulations, supporting biocompatibility testing initiatives, and expanding into adjacent specialty medical and pharmaceutical markets capture incremental demand while diversifying revenue streams. Emerging opportunity: USD 0.84-1.70 billion.
Segmental Analysis
The market is segmented by type, application, and region. By type, the market is divided into homopolymer polypropylene (syringe & IV-grade, container-grade, cap & closure-grade, sterilization-resistant grade), random copolymer polypropylene (transparent medical packaging grade, diagnostic labware grade, flexible medical component grade), impact copolymer polypropylene, and other polypropylene types.
Based on application, the market is categorized into medical devices (syringes & IV components, diagnostic kits, surgical & disposable instruments), medical packaging (rigid containers, blister packs & vials, pouches & caps), laboratory equipment, nonwoven medical textiles, drug delivery systems, and other applications. Regionally, the market is divided into Asia Pacific, North America, Europe, Latin America, and Middle East & Africa.
Why Does Homopolymer Polypropylene Command Type Market Leadership?
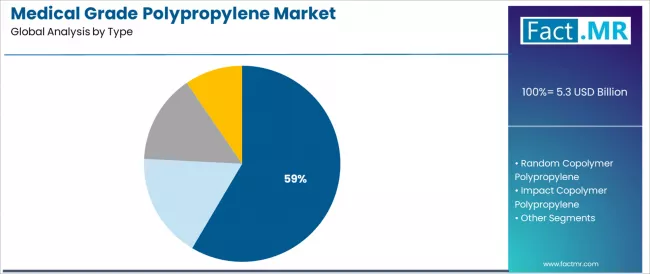
Homopolymer polypropylene is projected to account for 58.5% of the medical grade polypropylene market in 2025, reaffirming its position as the category's dominant material specification. Medical device manufacturers increasingly recognize the optimal balance of sterilization resistance and mechanical strength offered by homopolymer polypropylene for disposable syringe applications and rigid medical container environments.
This polymer type addresses both autoclave stability requirements and chemical resistance demands while providing reliable performance outcomes across diverse medical manufacturing operations. This segment forms the foundation of most medical device manufacturing protocols for injection molding and extrusion applications, as it represents the most technically proven and commercially established polymer category in the medical grade polypropylene industry.
FDA validation standards and extensive medical device application experience continue to strengthen confidence in homopolymer-based medical products among healthcare manufacturers and regulatory authorities. With increasing recognition of material consistency impact on device performance and patient safety requirements, homopolymer medical grade polypropylene aligns with both current manufacturing practices and quality assurance evolution goals, making them the central growth driver of comprehensive medical device strategies across multiple healthcare platforms.
Syringe & IV-grade represents 22.0% of homopolymer applications, driven by massive global demand for disposable injection devices and intravenous therapy components. Container-grade accounts for 18.0%, serving rigid medical packaging and pharmaceutical bottle applications.
Cap & closure-grade represents 10.0%, addressing medication bottle sealing and pharmaceutical closure systems, while sterilization-resistant grade comprises 8.5%, encompassing specialized applications requiring repeated autoclaving or gamma radiation sterilization.
Random copolymer polypropylene represents 27.0% of the market, reflecting significant utilization in transparent medical packaging and diagnostic labware requiring enhanced clarity. Transparent medical packaging grade accounts for 10.0%, diagnostic labware grade represents 9.0%, and flexible medical component grade comprises 8.0% of the segment.
What Drives Medical Devices Application Segment Prominence?
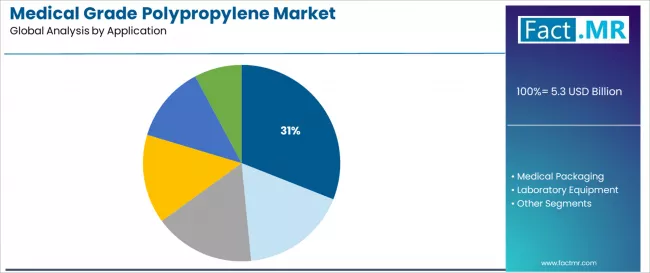
Medical devices are projected to account for 31.0% of the medical grade polypropylene market in 2025, establishing its position as the leading application segment. Healthcare manufacturers increasingly recognize that medical device production, encompassing disposable syringes, diagnostic kits, and surgical instruments, represents the most commercially significant and quality-critical category requiring specialized medical grade polymers due to direct patient contact and safety implications. This application addresses both single-use device manufacturing requirements and diagnostic equipment production demands while delivering critical patient safety outcomes across varied healthcare operations.
The segment is supported by the accelerating nature of disposable medical device adoption in global healthcare systems, driven by infection control protocols, safety regulations, and convenience preferences necessitating sterile single-use products. Additionally, medical device manufacturers are increasingly focusing on cost-effective polymer solutions that enhance production efficiency and maintain regulatory compliance while meeting stringent biocompatibility standards. As healthcare practices evolve and single-use device utilization expands, medical grade polypropylene for medical devices will continue to serve a crucial role in ensuring patient safety and healthcare quality within the global medical manufacturing market.
Syringes & IV components dominate the application with 12.0% share, driven by massive global vaccination programs and chronic disease management requiring injectable therapies. Diagnostic kits represent 9.0%, serving growing molecular diagnostics and point-of-care testing applications. Surgical & disposable instruments account for 10.0%, encompassing single-use surgical tools and medical procedure equipment.
Medical packaging represents 24.0% of applications, reflecting substantial usage in pharmaceutical packaging, medical product containment, and sterile barrier systems. Rigid containers account for 9.0%, blister packs & vials represent 8.0%, and pouches & caps comprise 7.0% of the medical packaging segment.
What are the Drivers, Restraints, and Key Trends of the Medical Grade Polypropylene Market?
The medical grade polypropylene market is advancing rapidly due to increasing recognition of polymer biocompatibility importance and growing demand for specialized medical materials across the healthcare manufacturing sector.
However, the market faces challenges, including raw material price volatility affecting resin economics, limited availability of ultra-high purity grades in certain regions, and concerns about polymer sustainability in medical waste management. Innovation in bio-based polypropylene technologies and advanced sterilization-compatible formulations continues to influence resin development and market expansion patterns.
Proliferation of Single-Use Medical Devices and Disposable Healthcare Products
The accelerating adoption of single-use device principles is enabling the development of more comprehensive medical grade polypropylene applications and healthcare protocols that can meet stringent safety and infection control requirements.
Healthcare facilities demand complete disposable device integration for medical procedures, including sterile syringe systems and single-use diagnostic equipment formulations that are particularly important for achieving infection prevention requirements in complex clinical applications.
Single-use medical device technologies provide access to contamination-free healthcare delivery that can optimize patient safety strategies and enhance clinical outcomes while maintaining cost-effectiveness for diverse healthcare environments.
Integration of Advanced Sterilization Technologies and Gamma-Resistant Formulations
Modern medical device organizations are incorporating advanced technologies such as gamma radiation sterilization, ethylene oxide compatibility, and autoclave-resistant formulations to enhance polymer utility and manufacturing flexibility.
These material capabilities improve sterilization process options, enable seamless supply chain logistics, and provide better compatibility between polymer properties and sterilization methods throughout the manufacturing and distribution experience.
Advanced sterilization-resistant polypropylene grades also enable customized device specifications and enhanced shelf-life stability through superior material property retention, supporting improved product reliability and regulatory compliance outcomes.
Analysis of the Medical Grade Polypropylene Market by Key Countries
.webp)
| Country | CAGR (2025-2035) |
|---|---|
| China | 8.4% |
| India | 8.1% |
| South Korea | 7.8% |
| USA | 7.5% |
| Brazil | 7.2% |
| Germany | 6.9% |
| Japan | 6.7% |
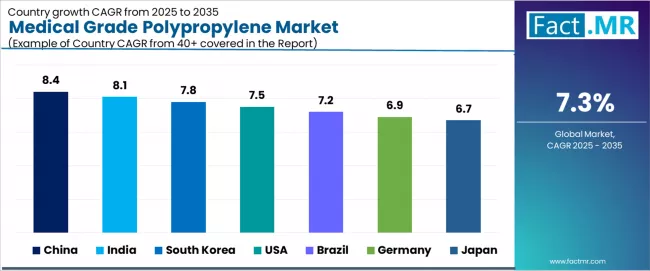
The medical grade polypropylene market is experiencing exceptional growth globally, with China leading at an 8.4% CAGR through 2035, driven by expanding medical device manufacturing scale, growing healthcare infrastructure, and increasing pharmaceutical packaging development across major industrial and metropolitan centers.
India follows at 8.1%, supported by massive generic pharmaceutical production, medical device manufacturing growth, and comprehensive healthcare expansion initiatives. South Korea records 7.8% growth, benefiting from advanced medical technology manufacturing and biotechnology sector development.
The USA demonstrates 7.5% growth, emphasizing advanced medical device innovation and pharmaceutical packaging sophistication. Brazil shows 7.2% growth with healthcare modernization initiatives and medical manufacturing expansion.
Germany records 6.9% growth, representing medical device manufacturing excellence and pharmaceutical industry leadership emphasis, while Japan shows 6.7% growth, representing established medical manufacturing standards and quality control excellence emphasis.
How Does China Demonstrate Exceptional Market Potential with Medical Manufacturing Expansion?
Revenue from medical grade polypropylene in China is projected to exhibit exceptional growth with a CAGR of 8.4% through 2035, driven by the country's rapidly expanding medical device manufacturing industry and increasing recognition of high-quality polymer materials as essential components for healthcare product quality and export competitiveness enhancement.
The country's massive medical manufacturing ecosystem and growing availability of specialized pharmaceutical-grade resin infrastructure are creating significant opportunities for medical grade polypropylene deployment across both disposable medical device production and pharmaceutical packaging manufacturing segments.
Major international polymer producers and domestic resin manufacturers are establishing comprehensive production facilities to serve the expanding population of medical device manufacturers and pharmaceutical companies requiring advanced medical grade materials across manufacturing parks, export processing zones, and pharmaceutical industrial clusters throughout China's diverse medical manufacturing regions.
The Chinese government's strategic emphasis on healthcare industry advancement and pharmaceutical manufacturing capability development is driving substantial investments in medical material quality capabilities and regulatory compliance infrastructure. This policy support, combined with the country's position as a global medical device manufacturing hub and growing domestic healthcare demand, creates a favorable environment for medical grade polypropylene market development. Chinese manufacturers are increasingly focusing on quality certification and international standard compliance to improve export competitiveness, with premium medical grade resins representing a key component in this manufacturing capability building.
- Government initiatives supporting medical device industry expansion and pharmaceutical manufacturing enhancement are driving demand for specialized medical grade polypropylene solutions across manufacturing segments
- Production capacity expansion and quality system advancement are supporting appropriate utilization of certified medical polymers among manufacturers and pharmaceutical facilities nationwide
- Medical device manufacturers and pharmaceutical companies are increasingly recognizing material quality importance in regulatory compliance, creating new resin demand capabilities
- Rising manufacturing investment and growing awareness of biocompatibility certification benefits are accelerating medical grade polypropylene adoption across healthcare categories
What Makes India Demonstrate Market Leadership with Pharmaceutical Manufacturing Excellence?
Revenue from medical grade polypropylene in India is expanding at a CAGR of 8.1%, supported by the world's largest generic pharmaceutical manufacturing industry, rapidly advancing medical device production capabilities, and comprehensive healthcare infrastructure integration across the country's developing pharmaceutical corridors.
The country's substantial pharmaceutical production scale and increasing sophistication of medical device manufacturing platforms are driving demand for specialized medical grade polypropylene solutions in both pharmaceutical packaging and medical device applications. International polymer suppliers and domestic compounders are establishing service capacity to serve the growing demand for certified medical materials while supporting the country's position as a global pharmaceutical manufacturing leader.
India's medical manufacturing sector continues to benefit from favorable healthcare policies, developing medical device infrastructure, and substantial pharmaceutical export operations. The country's focus on expanding generic drug production capabilities is driving investments in critical packaging materials including medical grade polypropylene for pharmaceutical containers and closures. This development is particularly important for medical polymer applications, as manufacturers seek reliable material sources for regulatory-compliant production to enhance global competitiveness and maintain pharmaceutical quality standards.
- Rising pharmaceutical export volumes and improving domestic medical manufacturing capabilities are creating opportunities for advanced medical grade polypropylene solutions
- Growing generic drug production and medical device assembly are supporting increased deployment of certified polymer materials across healthcare categories
- Expanding pharmaceutical quality expertise and emerging regulatory standards are driving innovative applications of medical polymers in drug packaging and device applications
- Manufacturing capability advancement is enabling delivery of internationally certified materials, supporting market growth and enhancing global pharmaceutical competitiveness
How Does South Korea Maintain Medical Technology Leadership?
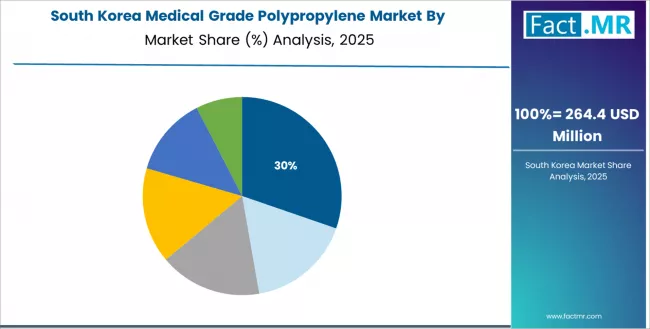
South Korea's advanced medical technology market demonstrates sophisticated medical grade polypropylene deployment with documented effectiveness in diagnostic device platforms and pharmaceutical packaging applications through integration with cutting-edge manufacturing technologies and quality control protocols.
The country leverages medical technology innovation leadership and precision manufacturing methodology to maintain a 7.8% CAGR through 2035. Leading medical device manufacturers and biotechnology companies, including major production facilities in industrial technology parks, showcase advanced polymer implementations where sophisticated medical grade polypropylene integrates with comprehensive quality assurance platforms and ongoing innovation initiatives to optimize device performance and regulatory compliance.
South Korean medical manufacturers prioritize technology excellence and quality innovation in healthcare product operations, creating demand for premium medical grade polypropylene solutions with advanced capabilities, including ultra-high purity specifications and integration with automated manufacturing systems. The market benefits from established medical technology infrastructure and commitment to research excellence that drives continuous material advancement.
Strategic Market Considerations:
- Medical device and diagnostic equipment segments leading growth with focus on advanced material specifications and comprehensive quality control applications
- High technology standards are driving sophisticated polymer portfolios from conventional medical grades to advanced pharmaceutical-certified platforms
- Medical technology excellence and biotechnology leadership supporting continued innovation in medical grade polypropylene applications
- Quality standards and regulatory requirements ensuring consistent material performance and device outcome optimization
What Drives USA’s Market Growth with Medical Device Innovation?
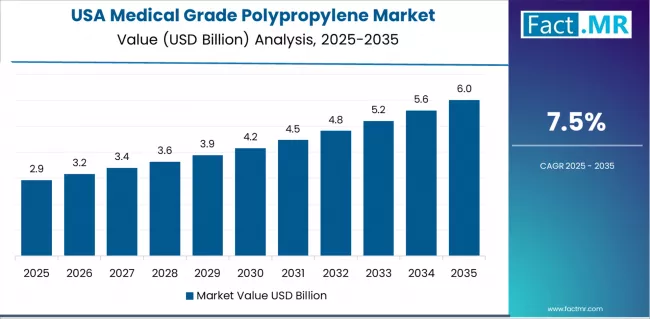
The USA's advanced healthcare market demonstrates sophisticated medical grade polypropylene deployment with documented effectiveness in medical device platforms and pharmaceutical packaging applications through integration with cutting-edge regulatory compliance and innovation protocols. The country leverages medical device development leadership and comprehensive FDA regulatory infrastructure to maintain a 7.5% CAGR through 2035.
Major medical device manufacturers and pharmaceutical companies, including leading healthcare product facilities in medical technology regions, showcase advanced polymer applications where sophisticated medical grade polypropylene integrates with comprehensive biocompatibility testing platforms and ongoing clinical research to optimize product safety and therapeutic effectiveness.
American healthcare manufacturers prioritize innovation excellence and regulatory compliance in medical product development, creating demand for premium medical grade polypropylene solutions with advanced capabilities, including comprehensive USP Class VI certification and integration with advanced manufacturing systems. The market benefits from established healthcare industry infrastructure and willingness to invest in premium materials that provide superior product outcomes and compliance with stringent FDA standards and healthcare quality expectations.
Strategic Market Considerations:
- Advanced medical device and pharmaceutical packaging segments leading growth with focus on innovation and comprehensive regulatory compliance applications
- High regulatory expectations are driving sophisticated material portfolios from standard medical grades to advanced biocompatibility-certified platforms
- Medical device innovation excellence and pharmaceutical research leadership supporting continued advancement in medical grade polypropylene applications
- FDA approval standards and quality requirements ensuring consistent material performance and patient safety optimization
Why Does Brazil Maintain Healthcare Manufacturing Growth?
Revenue from medical grade polypropylene in Brazil is projected to exhibit strong growth with a CAGR of 7.2% through 2035, driven by healthcare system expansion and growing medical device manufacturing infrastructure. The country's position as Latin America's largest pharmaceutical market and increasing medical manufacturing capabilities are creating significant opportunities for medical polymer integration across both pharmaceutical packaging and disposable device applications. Brazilian healthcare manufacturers are leveraging regional expertise to serve the growing demand for medical materials while supporting the country's position as a regional healthcare manufacturing leader.
The market benefits from established pharmaceutical manufacturing infrastructure supporting generic drug production, enabling optimized medical polymer deployment and comprehensive regulatory compliance. This development is particularly important for medical grade polypropylene applications, as manufacturers seek certified material solutions that maximize production efficiency while ensuring product quality in pharmaceutical and medical device environments.
Strategic Market Considerations:
- Pharmaceutical packaging and medical device segments leading growth with focus on production efficiency and quality enhancement applications
- Healthcare manufacturing economics and regulatory modernization are driving diverse polymer portfolios from standard medical resins to advanced pharmaceutical-grade platforms
- Pharmaceutical manufacturing excellence and regional healthcare leadership supporting competitive positioning in Latin American markets
- Quality standards and regulatory framework development influencing material specifications and certification adoption timelines
How does Germany Demonstrate Medical Device Manufacturing Excellence?
Germany's advanced healthcare market demonstrates sophisticated medical grade polypropylene deployment with documented effectiveness in medical device platforms and pharmaceutical packaging applications through integration with cutting-edge quality management systems and manufacturing precision protocols. The country leverages medical device manufacturing excellence and pharmaceutical industry leadership to maintain a 6.9% CAGR through 2035.
Major medical device manufacturers and pharmaceutical companies, including facilities in leading healthcare industrial centers, showcase advanced polymer implementations where sophisticated medical grade polypropylene integrates with comprehensive quality certification programs and ongoing research to optimize manufacturing precision and regulatory compliance.
German healthcare manufacturers prioritize engineering excellence and long-term quality assurance in medical product operations, creating demand for premium medical grade polypropylene solutions with advanced features, including comprehensive material traceability and integration with European medical device standards. The market benefits from established healthcare manufacturing infrastructure and commitment to technical innovation that drives continuous material advancement.
Strategic Market Considerations:
- Medical device and pharmaceutical packaging segments leading growth with focus on manufacturing precision and comprehensive quality validation applications
- Stringent regulatory requirements are driving sophisticated polymer portfolios from conventional medical grades to advanced European MDR-compliant platforms
- Manufacturing excellence and pharmaceutical leadership supporting competitive positioning in European healthcare markets
- Industry standards and technical specifications emphasizing rigorous approaches and continuous material quality improvement
What Drives Japan Market Growth with Manufacturing Quality Standards?
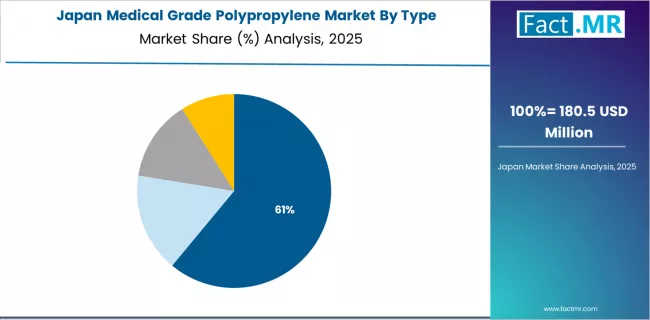
Japan's expanding medical grade polypropylene market demonstrates mature technology adoption with a 6.7% CAGR through 2035, driven by established medical manufacturing standards, precision production methodologies, and comprehensive quality control protocols across major healthcare industrial regions.
The country's emphasis on manufacturing excellence and meticulous quality practice is creating sustained demand for sophisticated medical grade polypropylene solutions across diverse medical device and pharmaceutical platforms. Japanese healthcare manufacturers are increasingly prioritizing advanced polymer materials that incorporate precision specifications for optimal product outcomes and manufacturing quality enhancement.
Market dynamics focus on high-quality medical grade polypropylene that balances established performance capabilities with reliability considerations important to Japanese healthcare practice standards and long-term product safety requirements. Mature domestic medical manufacturing expertise creates opportunities for next-generation polymer development and advanced processing technology deployment.
Strategic Market Considerations:
- Medical device and pharmaceutical packaging segments demonstrating steady growth with emphasis on material precision and comprehensive quality applications
- High-quality manufacturing requirements driving premium polymer specifications with advanced purity characteristics and performance optimization
- Manufacturing excellence supported by healthcare industry expertise and precision production delivery capabilities
- Healthcare manufacturer preferences emphasizing proven reliability and comprehensive regulatory validation in specialized medical grade polypropylene applications
Europe Market Split by Country
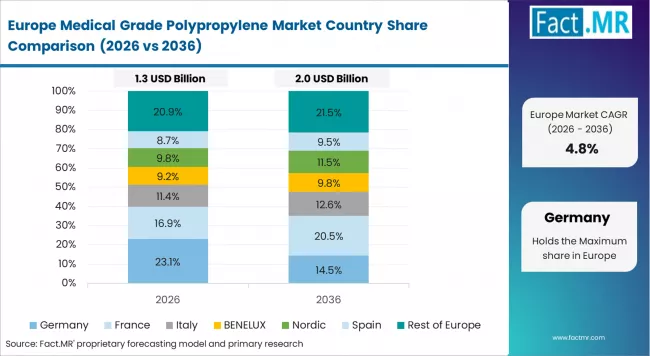
The medical grade polypropylene market in Europe is projected to grow from USD 1.26 billion in 2025 to USD 2.35 billion by 2035, registering a CAGR of 6.4% over the forecast period. Germany is expected to maintain its leadership position with a 34.8% market share in 2025, rising to 35.6% by 2035, supported by its dominant medical device manufacturing infrastructure, comprehensive pharmaceutical industry capabilities, and advanced polymer processing excellence throughout major healthcare manufacturing facilities and research institutes.
France follows with a 23.4% share in 2025, projected to reach 24.0% by 2035, driven by pharmaceutical manufacturing strength, medical device innovation, and expanding healthcare product sophistication serving both domestic and European markets. The UK holds a 18.6% share in 2025, expected to increase to 19.1% by 2035, supported by comprehensive pharmaceutical industry infrastructure and medical device manufacturing capabilities. Italy commands a 12.8% share in 2025, projected to reach 12.2% by 2035, while Spain accounts for 8.9% in 2025, expected to reach 8.3% by 2035.
The rest of Europe region, including Nordic countries with advanced healthcare standards, Eastern European emerging manufacturing sites, and smaller Western European medical production centers, is anticipated to hold 1.5% in 2025, declining slightly to 0.8% by 2035, attributed to market consolidation toward larger core markets with established medical manufacturing infrastructure and polymer certification expertise.
Competitive Landscape of the Medical Grade Polypropylene Market
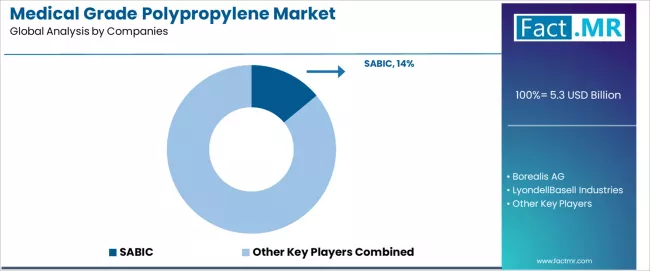
The medical grade polypropylene market is characterized by intense competition among established polymer manufacturers, specialized medical resin producers, and comprehensive healthcare material solution providers focused on delivering high-quality, reliable, and regulatory-compliant polymer products.
Companies are investing in resin purity enhancement programs, advanced grade development, strategic healthcare manufacturer partnerships, and comprehensive regulatory support initiatives to deliver effective, safe, and reliable medical polymer solutions that meet stringent FDA and MDR standards and healthcare manufacturer expectations. Material innovation, biocompatibility certification, and quality consistency strategies are central to strengthening polymer portfolios and market presence.
SABIC leads the market with a 14.0% market share, offering comprehensive medical grade polypropylene solutions with a focus on pharmaceutical-grade resin expertise and medical device material capabilities for healthcare applications. Borealis AG provides specialized medical polymer platforms with emphasis on homopolymer excellence and comprehensive sterilization-resistant capabilities across global medical manufacturing markets.
LyondellBasell Industries focuses on advanced polypropylene technologies and comprehensive medical grade solutions serving international healthcare communities. ExxonMobil Chemical delivers polymer manufacturing excellence with strong medical resin capabilities and quality assurance integration.
Braskem operates with a focus on Americas market medical polymer solutions and comprehensive biopolymer development for sustainable healthcare applications. TotalEnergies Corbion provides dedicated bio-based material expertise emphasizing sustainability and comprehensive renewable polymer capabilities.
INEOS Olefins & Polymers specializes in European market solutions and high-purity polymer technologies with emphasis on regulatory compliance. Trinseo delivers specialty polymer expertise to enhance medical material performance and provide comprehensive formulation solutions. LG Chem and Avient Corporation focus on specialized medical compounds and advanced additive technologies, emphasizing innovation leadership and comprehensive healthcare material support through dedicated research and development strategies.
Key Players in the Medical Grade Polypropylene Market
- SABIC
- Borealis AG
- LyondellBasell Industries
- ExxonMobil Chemical
- Braskem
- TotalEnergies Corbion
- INEOS Olefins & Polymers
- Trinseo
- LG Chem
- Avient Corporation
Bibliography
- U.S. Pharmacopeia. (2024). USP <88> Biological reactivity tests, in vivo: Updated polypropylene testing protocol. USP.
- European Pharmacopoeia Commission. (2025, January). EP 3.1.3 Polyolefins for pharmaceutical containers: Revised extractable limits. EDQM.
- U.S. Food and Drug Administration. (2024). Guidance for industry: Use of ISO 10993-1 for biocompatibility evaluation of medical device polymer components. FDA.
- International Organization for Standardization. (2024). ISO 11137-1:2024 Sterilization of health care products, radiation, requirements for development and validation. ISO.
- Ministry of Health and Family Welfare, India. (2025, February). Medical Devices Rules 2024: Updated polymer material compliance requirements. CDSCO, Government of India.
This bibliography is provided for reader reference. The full Fact.MR report contains the complete reference list with primary research documentation.
This Report Addresses
- Market sizing and quantitative forecast metrics detailing medical grade polypropylene expenditure across medical device, pharmaceutical packaging, and healthcare textile sectors through 2036.
- Segmentation analysis mapping adoption by polypropylene type and evaluating pharmacopeial compliance and sterilization compatibility specifications.
- Regional deployment intelligence comparing medical device manufacturing expansion in Asia against mature pre-filled syringe and diagnostic device markets in North America and Europe.
- Regulatory compliance assessment analyzing how USP, EP, and regional pharmacopeial standards shape medical-grade polypropylene resin qualification requirements.
- Competitive posture evaluation tracking dedicated medical-grade production line investments and medical device OEM resin qualification portfolios.
- Application engineering guidance defining the melt flow, purity, and sterilization compatibility specifications required for target medical device and packaging applications.
- Supply chain analysis identifying medical-grade resin allocation mechanisms and pharmacopeial testing capacity constraints.
- Custom data delivery formats encompassing interactive dashboards, raw Excel datasets, and comprehensive PDF narrative reports.
Medical Grade Polypropylene Market Definition
Medical grade polypropylene is a high-purity thermoplastic polymer produced under controlled manufacturing conditions specifically for medical device, pharmaceutical packaging, and healthcare product applications. The material meets pharmacopeial standards (USP Class VI, EP, JP) for biocompatibility, has documented extractable and leachable profiles, and is validated for gamma irradiation, ethylene oxide, and steam sterilization compatibility. Product types include homopolymer polypropylene for rigid medical devices, random copolymer for transparent packaging, and impact copolymer for durable equipment housings.
Medical Grade Polypropylene Market Inclusions
Market scope includes homopolymer, random copolymer, and impact copolymer medical-grade polypropylene resins with pharmacopeial compliance certification. The report covers global and regional market sizes, forecast period 2026 to 2036, and segment breakdowns by type and application.
Medical Grade Polypropylene Market Exclusions
The scope excludes commodity-grade polypropylene for non-medical applications, polypropylene fiber for general textile use, recycled polypropylene, and polypropylene automotive components not classified under medical device regulations.
Medical Grade Polypropylene Market Research Methodology
Primary Research: Analysts engaged with medical-grade polypropylene production managers, medical device injection molding engineers, and pharmaceutical packaging procurement leads to map resin qualification requirements and supply chain formation timelines.
Desk Research: Data collection aggregated medical device production facility construction databases, pharmacopeial compliance certification records, and single-use device market penetration statistics.
Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of medical-grade polypropylene consumption per medical device manufactured, packaging unit produced, and nonwoven textile square meter converted, applying application-specific resin usage rates.
Data Validation and Update Cycle: Projections are tested against publicly reported medical-grade polymer segment revenues from major resin producers and medical device industry material procurement disclosures.
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 5.63 billion to USD 11.40 billion, at a CAGR of 7.3% |
| Market Definition | Medical grade polypropylene is a high-purity thermoplastic polymer produced under controlled manufacturing conditions specifically for medical device, pharmaceutical packaging, and healthcare product applications. The material meets pharmacopeial standards (USP Class VI, EP, JP) for biocompatibility, has documented extractable and leachable profiles, and is validated for gamma irradiation, ethylene oxide, and steam sterilization compatibility. Product types include homopolymer polypropylene for rigid medical devices, random copolymer for transparent packaging, and impact copolymer for durable equipment housings. |
| Type Segmentation | Homopolymer Polypropylene (Syringe & IV-grade, Container-grade, Cap & Closure-grade, Sterilization-Resistant Grade), Random Copolymer Polypropylene (Transparent Medical Packaging Grade, Diagnostic Labware Grade, Flexible Medical Component Grade), Impact Copolymer Polypropylene, Other Polypropylene Types |
| Application Segmentation | Medical Devices (Syringes & IV Components, Diagnostic Kits, Surgical & Disposable Instruments), Medical Packaging (Rigid Containers, Blister Packs & Vials, Pouches & Caps), Laboratory Equipment, Nonwoven Medical Textiles, Drug Delivery Systems, Other Applications |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | India, China, USA, Germany, Japan, South Korea, UK, and 40 plus countries |
| Key Companies Profiled | SABIC, Borealis AG, LyondellBasell Industries, ExxonMobil Chemical, Braskem, TotalEnergies Corbion, INEOS Olefins & Polymers, Trinseo, LG Chem, Avient Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Baseline values derive from a bottom-up aggregation of medical-grade polypropylene consumption per medical device manufactured, packaging unit produced, and nonwoven textile square meter converted, applying application-specific resin usage rates. |
Medical Grade Polypropylene Market by Segments
-
Type :
- Homopolymer Polypropylene
- Syringe & IV-grade
- Container-grade
- Cap & Closure-grade
- Sterilization-Resistant Grade
- Random Copolymer Polypropylene
- Transparent Medical Packaging Grade
- Diagnostic Labware Grade
- Flexible Medical Component Grade
- Impact Copolymer Polypropylene
- Other Polypropylene Types
- Homopolymer Polypropylene
-
Application :
- Medical Devices
- Syringes & IV Components
- Diagnostic Kits
- Surgical & Disposable Instruments
- Medical Packaging
- Rigid Containers
- Blister Packs & Vials
- Pouches & Caps
- Laboratory Equipment
- Nonwoven Medical Textiles
- Drug Delivery Systems
- Other Applications
- Medical Devices
-
Region :
-
Asia Pacific
- China
- India
- Japan
- South Korea
- ASEAN
- Australia & New Zealand
- Rest of Asia Pacific
-
North America
- United States
- Canada
- Mexico
-
Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
-
Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
-
Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Countries
- Rest of Middle East & Africa
-
- Frequently Asked Questions -
How large is the demand for medical grade polypropylene in the global market in 2026?
Demand for medical grade polypropylene in the global market is estimated to be valued at USD 5.63 billion in 2026.
What will be the market size of medical grade polypropylene by 2036?
Market size for medical grade polypropylene is projected to reach USD 11.40 billion by 2036.
What is the expected demand growth between 2026 and 2036?
Demand for medical grade polypropylene is expected to grow at a CAGR of 7.3% between 2026 and 2036.
Which Type is poised to lead global sales by 2026?
Homopolymer Polypropylene accounts for 58.5% in 2026 as syringe, IV component, and rigid medical container manufacturers specify homopolymer grades for their stiffness, chemical resistance, and sterilization compatibility.
How significant is Medical Devices in driving adoption in 2026?
Medical Devices represents 31.0% of application share as single-use syringe, diagnostic kit, and disposable instrument production consumes the largest volume of medical-grade polypropylene resin.
What is the India growth outlook in this report?
India is projected to grow at a CAGR of 9.8% during 2026 to 2036.
What is medical grade polypropylene and what is it mainly used for?
Medical grade polypropylene is a high-purity, biocompatible thermoplastic meeting pharmacopeial standards for medical applications. It is mainly used to manufacture syringes, IV components, diagnostic kits, medical packaging, and nonwoven surgical textiles.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type, 2026 to 2036
- Homopolymer Polypropylene
- Syringe & IV-grade
- Container-grade
- Cap & Closure-grade
- Sterilization-Resistant Grade
- Random Copolymer Polypropylene
- Transparent Medical Packaging Grade
- Diagnostic Labware Grade
- Flexible Medical Component Grade
- Impact Copolymer Polypropylene
- Other Polypropylene Types
- Homopolymer Polypropylene
- Y to o to Y Growth Trend Analysis By Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Medical Devices
- Syringes & IV Components
- Diagnostic Kits
- Surgical & Disposable Instruments
- Medical Packaging
- Rigid Containers
- Blister Packs & Vials
- Pouches & Caps
- Laboratory Equipment
- Nonwoven Medical Textiles
- Drug Delivery Systems
- Other Applications
- Medical Devices
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Application
- Competition Analysis
- Competition Deep Dive
- SABIC
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Borealis AG
- LyondellBasell Industries
- ExxonMobil Chemical
- Braskem
- TotalEnergies Corbion
- INEOS Olefins & Polymers
- Trinseo
- LG Chem
- Avient Corporation
- SABIC
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type, 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type, 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Type, 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis