Multiple Sclerosis Therapeutics Market Size, Share, Growth and Forecast (2026 - 2036)
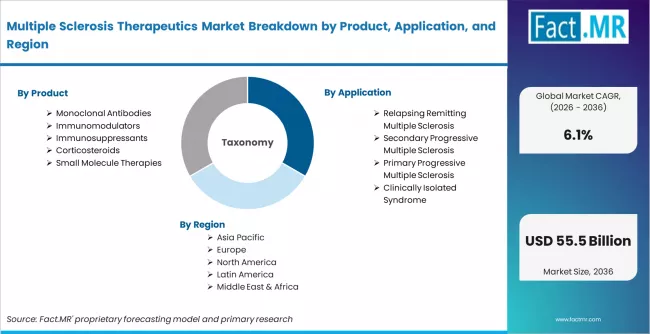
Multiple Sclerosis Therapeutics Market is segmented by Product (Monoclonal Antibodies, Immunomodulators, Immunosuppressants, Corticosteroids, Small Molecule Therapies), Application (Relapsing Remitting Multiple Sclerosis, Secondary Progressive Multiple Sclerosis, Primary Progressive Multiple Sclerosis, Clinically Isolated Syndrome), Route of Administration (Injectable, Oral, Intravenous), and Region, with forecasts covering the period from 2026 to 2036.
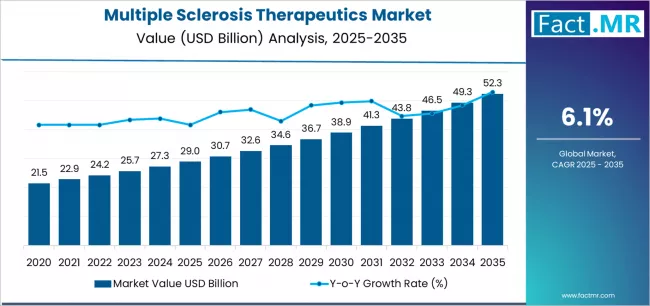
According to Fact.MR estimates, the global multiple sclerosis therapeutics Market was valued at USD 29.0 billion in 2025. The market is projected to reach USD 30.7 billion in 2026 and is expected to grow to USD 55.5 billion by 2036, expanding at a CAGR of 6.1%. Monoclonal Antibodies is anticipated to account for 41.6% of the product segment in 2026, while Relapsing Remitting Multiple Sclerosis is expected to remain the leading application with around 63.5% share.
Multiple Sclerosis Therapeutics Market Analysis and Forecast by Fact.MR
- The global multiple sclerosis therapeutics market is estimated at USD 30.7 billion in 2026 and has been forecast to expand at a CAGR of 6.1% to reach USD 55.5 billion by 2036.
- Multiple sclerosis therapeutics encompass disease-modifying therapies, immunomodulators, monoclonal antibodies, corticosteroids, and symptomatic treatments targeting immune-mediated neurological damage.
- The treatment landscape includes injectable biologics, oral small molecules, and intravenous infusions that aim to reduce relapse rates, slow disability progression, and manage acute exacerbations.
- Product development is shifting toward B-cell depleting agents, Bruton tyrosine kinase inhibitors, and subcutaneous formulations that offer improved convenience and adherence profiles across relapsing and progressive MS subtypes.
Summary of Multiple Sclerosis Therapeutics Market
- Market Snapshot
- In 2025, the global Multiple Sclerosis Therapeutics Market was valued at approximately USD 28.95 billion.
- The market is estimated to reach USD 30.7 billion in 2026 and is projected to attain USD 55.5 billion by 2036.
- The multiple sclerosis therapeutics market is likely to expand at a CAGR of 6.1% during the forecast period.
- The market is anticipated to create an absolute dollar opportunity of USD 24.8 billion between 2026 and 2036.
- Monoclonal antibodies account for 41.6% of product segment share in 2026, led by B-cell depleting agents and alpha integrin inhibitors.
- USA (6.2%) and Germany (6.1%) are the key growth markets during the forecast period.
- Demand and Growth Drivers
- Rising global MS prevalence, estimated at over 2.8 million diagnosed patients, is sustaining demand for long-term disease-modifying therapies across both relapsing and progressive forms.
- Expansion of high-efficacy treatment paradigms, including B-cell depletion and S1P receptor modulation, is shifting prescribing patterns toward earlier aggressive intervention in newly diagnosed patients.
- Growing adoption of subcutaneous self-administration formats is reducing dependence on infusion center visits, improving adherence and lowering per-patient treatment delivery costs.
- Increasing regulatory focus on progressive MS subtypes is creating new addressable patient pools, supported by late-stage pipeline candidates targeting smoldering neuroinflammation.
- Biosimilar entry into the interferon and natalizumab space is expanding access in cost-sensitive healthcare systems across Europe and Asia Pacific.
- Product and Segment View
- Monoclonal antibodies account for 41.6% of the product segment in 2026, led by B-cell depleting agents including ocrelizumab and ofatumumab that target CD20-expressing lymphocytes.
- Relapsing remitting multiple sclerosis represents 63.5% of the application segment in 2026, supported by the highest diagnosed patient volumes and broadest range of approved therapies.
- Injectable route of administration holds 46.2% share in 2026, driven by established biologic therapy dominance including subcutaneous and intravenous disease-modifying treatments.
- Key segmentation includes:
- Product: Monoclonal antibodies (B-cell depleting agents, alpha integrin inhibitors, CD52 targeting agents), Immunomodulators (beta interferons, glatiramer acetate, S1P receptor modulators, fumarates), Immunosuppressants, Corticosteroids, Small molecule therapies
- Application: Relapsing remitting MS, Secondary progressive MS, Primary progressive MS, Clinically isolated syndrome
- Route of Administration: Injectable (subcutaneous, intramuscular), Oral, Intravenous
- Distribution Channel: Hospital pharmacy, Retail pharmacy, Online pharmacy, Specialty pharmacy
- These systems enable:
- Targeted immune modulation through B-cell depletion, integrin blockade, and sphingosine receptor modulation
- Relapse rate reduction and disability progression delay across MS subtypes
- Flexible treatment delivery through hospital infusion, self-injection, and oral administration pathways
- Geography and Competitive Outlook
- North America remains the largest regional market, anchored by the USA where high disease prevalence, strong payer coverage for biologics, and early adoption of high-efficacy therapies sustain premium product demand.
- Europe shows steady growth led by Germany, UK, and France, supported by comprehensive MS center infrastructure, universal healthcare access, and growing biosimilar adoption.
- Asia Pacific is the fastest-growing region as countries like Japan expand access to approved disease-modifying therapies and emerging markets begin to adopt biologic treatment protocols.
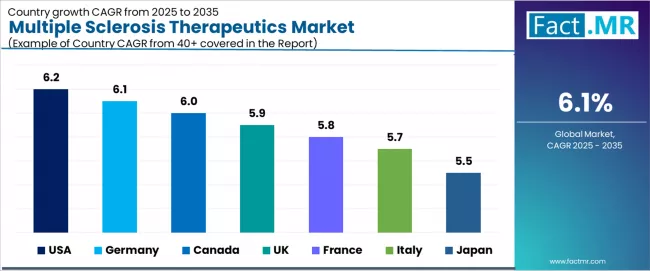
- USA (6.2%) and Germany (6.1%) lead country-level growth, while Canada (6.0%), UK (5.9%), and France (5.8%) maintain strong expansion trajectories.
- Leading companies in the market include Biogen, Teva Pharmaceutical Industries Ltd., Pfizer Inc., Bayer AG, Sanofi, F. Hoffmann-La Roche Ltd., Johnson & Johnson Services Inc., Merck & Co. Inc., Takeda Pharmaceutical Company Limited, and Horizon Therapeutics plc.
- Analyst Opinion
- Shambhu Nath Jha, Principal Consultant at Fact.MR, says 'The multiple sclerosis therapeutics market is entering a phase where treatment strategy, not just molecule availability, will define commercial outcomes. B-cell depleting monoclonal antibodies are consolidating share at the expense of older interferons, while BTK inhibitors represent the next competitive frontier if liver safety signals can be managed. The shift toward earlier high-efficacy intervention, combined with subcutaneous delivery convenience and biosimilar price pressure on legacy products, is reshaping how value flows across the treatment chain. Companies that pair clinical differentiation with strong patient support infrastructure will capture the most durable share gains through 2036.
Key Growth Drivers, Constraints, and Opportunities
Key Factors Driving Growth
- Rising global MS prevalence, particularly in North America and Europe, combined with improved diagnostic capabilities that identify patients earlier in the disease course, is expanding the treatable patient population.
- Shift toward high-efficacy treatment paradigms, including anti-CD20 monoclonal antibodies and S1P receptor modulators, is increasing per-patient treatment value and sustaining branded product demand.
- Growing demand for subcutaneous self-administration and oral formulations is creating new commercial opportunities as patients and providers prioritize convenience alongside efficacy.
Key Market Constraints
- High per-patient annual therapy costs, often exceeding USD 50,000 for branded biologics, continue to create payer resistance and access barriers, particularly in cost-constrained healthcare systems.
- Biosimilar entry into established biologic categories is compressing margins and requiring originator companies to invest in lifecycle management and next-generation product differentiation.
- Liver safety signals associated with emerging BTK inhibitor candidates have created regulatory uncertainty, delaying potential new treatment entries for progressive MS subtypes.
Key Opportunity Areas
- Progressive MS subtypes remain significantly undertreated, with limited approved therapies creating a large unmet need that could be addressed by brain-penetrant small molecules and targeted immune modulators.
- Expansion of MS treatment access in emerging markets across Asia Pacific and Latin America represents a growing revenue opportunity as healthcare infrastructure and reimbursement improve.
- Subscription-based and outcomes-linked pricing models for high-cost biologics could unlock new patient access while maintaining commercial viability for manufacturers.
Segment-wise Analysis of the Multiple Sclerosis Therapeutics Market
- Monoclonal antibodies hold 41.6% of the product segment in 2026, supported by the established clinical efficacy of B-cell depleting agents and growing use of subcutaneous monoclonal antibody formulations across relapsing MS management.
- Relapsing remitting multiple sclerosis represents 63.5% of the application segment in 2026, reflecting the highest diagnosed patient volumes and broadest range of approved disease-modifying therapies.
- Injectable administration holds 46.2% share in 2026, with subcutaneous and intravenous biologic therapies continuing to anchor the treatment landscape despite the rising convenience appeal of oral alternatives.
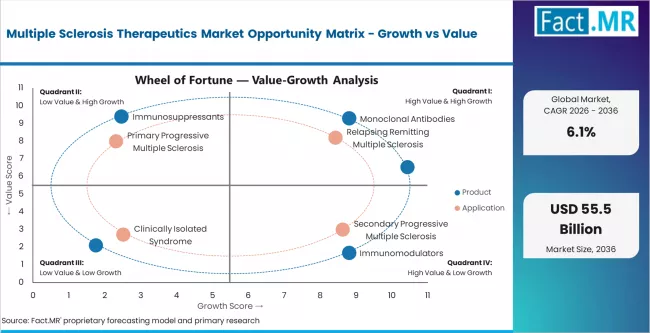
The multiple sclerosis therapeutics market is segmented by product type, application, route of administration, distribution channel, drug class, end use, pricing tier, and region. By product, the market covers monoclonal antibodies, immunomodulators, immunosuppressants, corticosteroids, and small molecule therapies. By application, coverage includes relapsing remitting MS, secondary progressive MS, primary progressive MS, and clinically isolated syndrome. Monoclonal antibodies lead by value in the near term because of their established efficacy in relapse reduction and disability progression delay, while oral therapies are expected to capture incremental share as newer agents demonstrate competitive outcomes with improved convenience.
Which Product Segment Dominates the Multiple Sclerosis Therapeutics Market?
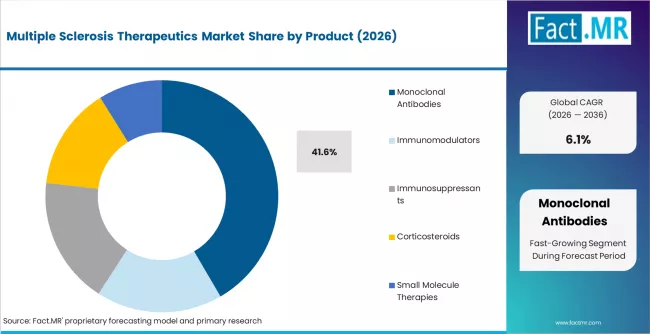
Monoclonal antibodies are expected to account for 41.6% of the product segment value in 2026. The segment leadership is supported by the established clinical profile of B-cell depleting agents such as ocrelizumab and ofatumumab, which have demonstrated strong relapse rate reduction and disability progression delay across relapsing and primary progressive MS. Anti-CD20 therapies are increasingly positioned as first-line options in aggressive treatment strategies, which is consolidating their share of new patient starts.
Alpha integrin inhibitors and CD52 targeting agents contribute additional share within the monoclonal antibody category, although their use is more restricted to specific patient populations due to safety monitoring requirements. The overall monoclonal antibody segment benefits from ongoing subcutaneous reformulation efforts that are improving administration convenience while maintaining the efficacy profiles that drive physician preference and payer acceptance.
Which Application Segment Dominates the Multiple Sclerosis Therapeutics Market?
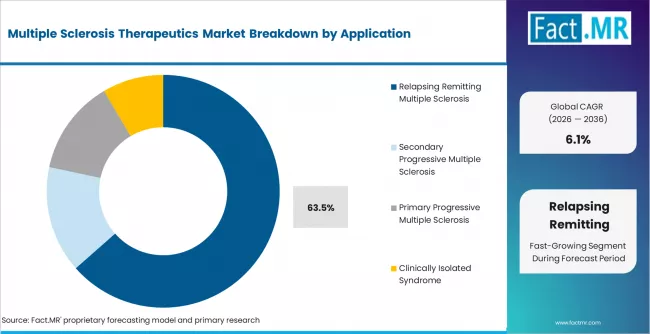
Relapsing remitting multiple sclerosis is expected to hold 63.5% of the application segment value in 2026. This dominance reflects the fact that RRMS accounts for approximately 85% of initial MS diagnoses globally, creating the largest treatable patient pool. The availability of multiple approved disease-modifying therapies across oral, injectable, and infusion categories provides physicians with a broad toolkit to manage this subtype.
Secondary progressive and primary progressive MS subtypes represent growing but still underpenetrated application areas. The limited number of approved therapies for these progressive forms creates both an unmet clinical need and a commercial opportunity for pipeline candidates that can demonstrate meaningful disability progression delay. As treatment options expand for progressive MS, the application distribution is likely to shift modestly, though RRMS will retain the majority share through 2036.
Which Product Trend is Shaping the Next Phase of Growth in the Multiple Sclerosis Therapeutics Market?
Bruton tyrosine kinase inhibitors are emerging as the most closely watched product development trend in the MS therapeutics market. These oral, brain-penetrant small molecules target smoldering neuroinflammation and microglia activation, mechanisms not adequately addressed by current approved therapies. Fenebrutinib from Roche and tolebrutinib from Sanofi have both advanced through late-stage clinical programs, with fenebrutinib demonstrating relapse rate reduction comparable to established anti-CD20 therapies in phase 3 trials reported in early 2026.
The development path for BTK inhibitors has not been without setbacks. Tolebrutinib failed to meet its primary endpoint in primary progressive MS in the PERSEUS trial in late 2025, and its US regulatory submission was rejected by the FDA in January 2026 due to liver safety concerns. The European Medicines Agency, in contrast, recommended approval for non-relapsing secondary progressive MS in April 2026. These mixed regulatory outcomes underscore the challenge of balancing efficacy gains against hepatotoxicity risk. If liver safety concerns can be managed through dose optimization and patient selection, BTK inhibitors could become a significant new treatment class, particularly for progressive MS subtypes where therapeutic options remain limited.
Regional Outlook Across Key Markets
.webp)
- USA leads country-level growth at 6.2%, supported by the highest global MS prevalence, strong payer coverage for branded biologics, and early adoption of high-efficacy treatment paradigms including anti-CD20 monoclonal antibodies.
- Germany maintains a 6.1% CAGR, anchored by comprehensive MS center infrastructure, universal healthcare coverage, and sophisticated clinical research networks that support rapid adoption of new therapies.
- Canada at 6.0% benefits from growing biologic access programs, while UK at 5.9% and France at 5.8% expand through NHS neurology pathways and national MS treatment registries respectively.
- Japan at 5.5% represents the leading Asia Pacific growth opportunity, supported by expanding regulatory approvals for disease-modifying therapies and growing disease awareness.
CAGR Table
| Country | CAGR (%) |
|---|---|
| USA | 6.2% |
| Germany | 6.1% |
| Canada | 6.0% |
| UK | 5.9% |
| France | 5.8% |
| Italy | 5.7% |
| Japan | 5.5% |
Source: Fact MR (FMR) analysis, based on proprietary forecasting model and primary research.
Market Outlook for Multiple Sclerosis Therapeutics in the United States
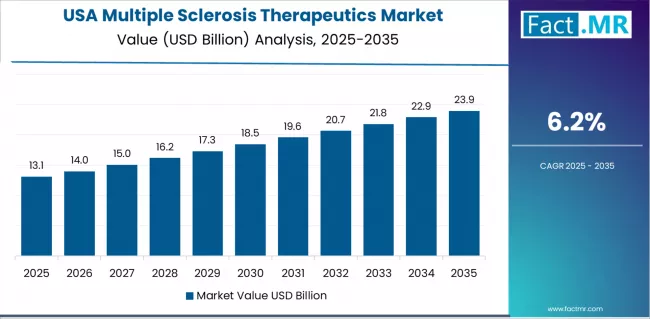
The US multiple sclerosis therapeutics market is projected to grow at a CAGR of 6.2% through 2036. Demand is supported by the highest global MS prevalence of approximately 1 million diagnosed patients, strong payer coverage for branded biologics, and early adoption of anti-CD20 therapies as first-line treatment options.
- High biologic adoption rates continue to support premium product demand.
- Biosimilar entry is beginning to reshape the competitive dynamics in established categories.
- Specialty pharmacy distribution remains the dominant access channel for high-cost MS therapies.
Multiple Sclerosis Therapeutics Demand Trends in Germany
The Germany multiple sclerosis therapeutics market is projected to grow at a CAGR of 6.1% through 2036. Demand is supported by a comprehensive MS center network, universal healthcare coverage, and a strong clinical research ecosystem that enables rapid adoption of newly approved disease-modifying therapies.
- MS center infrastructure supports high treatment initiation and compliance rates.
- Universal coverage enables broad access to branded biologics and oral therapies.
- Clinical trial participation strengthens early access to pipeline therapies.
Multiple Sclerosis Therapeutics Growth Assessment in Canada
The Canada multiple sclerosis therapeutics market is projected to grow at a CAGR of 6.0% through 2036. Demand is supported by strong MS prevalence rates, expanding biologic access programs, and growing adoption of subcutaneous self-administration formats that improve treatment adherence.
- Biologic access programs are expanding patient reach across provincial health systems.
- Subcutaneous formats are reducing infusion center dependency.
- Growing MS diagnosis rates support new patient treatment initiations.
United Kingdom Multiple Sclerosis Therapeutics Market Outlook
The UK multiple sclerosis therapeutics market is projected to grow at a CAGR of 5.9% through 2036. Demand is supported by NHS neurology service expansion, systematic MS care pathway implementation, and growing adoption of high-efficacy therapies in line with updated NICE treatment guidelines.
- NHS MS care pathways are improving treatment access and standardization.
- NICE guideline updates are expanding the approved use of high-efficacy therapies.
- Biosimilar adoption is improving cost-efficiency in hospital pharmacy settings.
Japan Multiple Sclerosis Therapeutics Market Demand Analysis
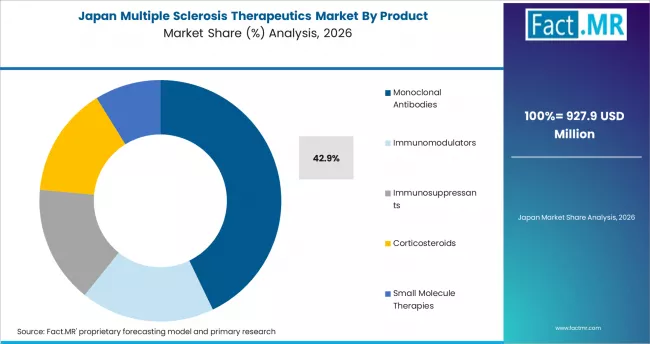
The Japan multiple sclerosis therapeutics market is projected to grow at a CAGR of 5.5% through 2036. Demand is supported by expanding regulatory approvals for disease-modifying therapies, growing disease awareness, and increasing use of biologics in specialty neurology centers.
- Regulatory approvals for newer biologics are expanding the available treatment toolkit.
- Disease awareness campaigns are supporting earlier diagnosis and treatment initiation.
- Specialty neurology center expansion is improving biologic therapy access.
Competitive Benchmarking and Company Positioning
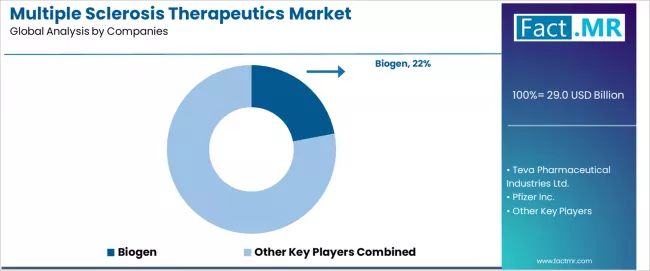
Multiple Sclerosis Therapeutics Market Analysis By Company
- Biogen, F. Hoffmann-La Roche Ltd., Sanofi, and Pfizer Inc. hold strong positions in the global multiple sclerosis therapeutics market through deep MS franchise expertise, broad product portfolios spanning injectable and oral therapies, and growing capabilities in next-generation B-cell targeting and BTK inhibitor development.
- Teva Pharmaceutical Industries Ltd., Merck & Co. Inc., Johnson & Johnson Services Inc., and Bayer AG participate meaningfully across specific drug classes, patient segments, and geographic markets, with competition shaped by efficacy data, safety profiles, and convenience of administration.
- Pipeline activity creates an important competitive dimension, with late-stage BTK inhibitor candidates from Roche and Sanofi, alongside biosimilar entries into established biologic categories, set to reshape competitive dynamics over the forecast period.
The competitive landscape is moderately concentrated, with a small number of large pharmaceutical companies controlling the majority of MS therapy revenue. Biogen remains the legacy market leader through its established franchise including TYSABRI, TECFIDERA, VUMERITY, and AVONEX, though its MS product revenue has been declining as newer entrants from competitors capture share. Roche has emerged as a formidable competitor through Ocrevus (ocrelizumab), which has become the leading single product in the MS market, and is now pursuing fenebrutinib as a potential follow-on BTK inhibitor.
Sanofi competes through its oral portfolio including Aubagio (teriflunomide) and has invested significantly in tolebrutinib, which received a positive CHMP opinion for non-relapsing secondary progressive MS in Europe in April 2026 despite its FDA rejection in the United States. This regulatory divergence illustrates the complexity of developing therapies for progressive MS and the different risk-benefit assessments applied across major markets.
Competition is also shaped by the emergence of biosimilars, particularly for natalizumab and interferon products, which are beginning to compress pricing in established therapy classes. Companies that combine clinical differentiation in underserved MS subtypes with strong patient support programs and flexible administration options are best positioned to sustain share through the forecast period.
Strategic priorities across the market include expanding subcutaneous delivery options, investing in brain-penetrant molecules for progressive MS, building digital patient monitoring capabilities, and securing early access to BTK inhibitor commercialization. Companies that pair clinical innovation with commercial execution will define the next phase of competitive leadership.
Key Companies in the Multiple Sclerosis Therapeutics Market
Biogen, F. Hoffmann-La Roche Ltd., Sanofi, and Pfizer Inc. are among the leading players in the multiple sclerosis therapeutics market globally, supported by deep MS franchise expertise, broad product portfolios, and established prescriber relationships across major markets.
Teva Pharmaceutical Industries Ltd., Merck & Co. Inc., Johnson & Johnson Services Inc., and Bayer AG hold established positions across specific drug classes and geographic markets, supported by branded and biosimilar offerings and strong distribution infrastructure.
Takeda Pharmaceutical Company Limited, Horizon Therapeutics plc, and emerging biotech companies continue to compete through focused pipeline programs, niche drug class expertise, and strategic partnerships that reinforce their relevance in an evolving market.
Recent Industry Developments
- Sanofi, EU Regulatory Recommendation for Tolebrutinib (April 2026)
- The European Medicines Agency's CHMP recommended approval of Cenrifki (tolebrutinib) for the treatment of secondary progressive multiple sclerosis without relapses. The recommendation is based on the HERCULES phase 3 study, which demonstrated significant delay in disability progression onset in non-relapsing SPMS patients.
- Roche, Phase 3 Data for Fenebrutinib (April 2026)
- Roche presented late-stage clinical data for fenebrutinib, its BTK inhibitor candidate for MS, showing efficacy in reducing relapse rates and slowing disability progression across both relapsing and primary progressive MS subtypes. The company initiated regulatory submissions based on these results.
- ScinoPharm Taiwan, FDA Approval of Generic Glatiramer Acetate (January 2026)
- The FDA approved ScinoPharm Taiwan's glatiramer acetate injection for the treatment of relapsing MS. This represents the first approval of a complex generic injectable for this therapy, expanding affordable treatment access for patients on established immunomodulatory regimens.
- Biogen, Pipeline Advancement and Portfolio Update (2025)
- Biogen reported continued execution across its MS franchise, with VUMERITY achieving year-over-year growth and the company advancing multiple late-stage pipeline programs. Biogen's 2025 full-year MS product revenue remained the largest single-company contribution to the global MS therapeutics market.
Leading Companies Shaping the Multiple Sclerosis Therapeutics Market
- Biogen
- Teva Pharmaceutical Industries Ltd.
- Pfizer Inc.
- Bayer AG
- Sanofi
- F. Hoffmann-La Roche Ltd.
- Johnson & Johnson Services, Inc.
- Merck & Co., Inc.
- Takeda Pharmaceutical Company Limited
- Horizon Therapeutics plc
Sources and Research References
- Biogen. MS product portfolio updates, annual financial filings, and clinical pipeline disclosures.
- F. Hoffmann-La Roche Ltd. Ocrevus and fenebrutinib clinical program data and regulatory submissions.
- Sanofi. Tolebrutinib (Cenrifki) HERCULES trial results and regulatory filing outcomes.
- World Health Organization. Multiple Sclerosis Atlas and global prevalence estimates.
- National Multiple Sclerosis Society. Prevalence data and treatment access publications.
- Primary interviews with neurologists, hospital pharmacists, payer organizations, patient advocacy groups, and pharmaceutical industry executives.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
Key Questions This Report Addresses
- What is the current and future size of the Multiple Sclerosis Therapeutics Market?
- How fast is the Multiple Sclerosis Therapeutics Market expected to grow between 2026 and 2036?
- Which product type is likely to lead the market by 2026?
- Which application segment is expected to account for the highest demand by 2026?
- What factors are driving demand for MS therapeutics globally?
- How is the shift toward high-efficacy B-cell depleting therapies influencing the market?
- Why are BTK inhibitors considered the next competitive frontier in MS treatment?
- How are biosimilars reshaping the competitive landscape in established therapy classes?
- Which countries are projected to show the fastest growth through 2036?
- What is driving market expansion in the USA, Germany, and Canada?
- Who are the key companies active in the Multiple Sclerosis Therapeutics Market?
- How does Fact.MR estimate and validate the market forecast?
Multiple Sclerosis Therapeutics Market Definition
The multiple sclerosis therapeutics market covers pharmaceutical products indicated for the treatment of immune-mediated demyelinating disorders of the central nervous system, including disease-modifying therapies, immunomodulators, immunosuppressants, monoclonal antibodies, corticosteroids, and small molecule treatments used across relapsing and progressive MS subtypes.
Multiple Sclerosis Therapeutics Market Inclusions
- The scope covers global and regional market size and forecasts for 2026 to 2036 across product type, application, route of administration, distribution channel, drug class, end use, pricing tier, and region.
- It includes regional demand analysis across major geographies, based on factors such as disease prevalence, treatment adoption rates, reimbursement frameworks, and biologic prescribing patterns.
- The report includes pricing and cost analysis across branded biologics, oral disease-modifying therapies, and biosimilar entries, including therapy initiation costs and annual treatment expenditure.
- It covers pipeline developments, regulatory approvals, market drivers and constraints, and the competitive landscape, including product portfolios, geographic reach, and commercial strategies of leading players.
Multiple Sclerosis Therapeutics Market Exclusions
- The scope excludes general symptomatic treatments not specifically indicated for MS, such as general pain management, physical therapy services, and rehabilitation programs without pharmaceutical components.
- It excludes diagnostic imaging services, biomarker testing platforms, and digital health monitoring tools sold independently of MS therapeutic products.
- The report excludes experimental gene therapy candidates in pre-clinical stages, off-label drug use not tracked in MS registries, and compassionate use programs outside commercial channels.
- It also excludes veterinary neurological treatments, nutraceutical supplements marketed for neurological support, and traditional medicine formulations without regulatory approval.
Multiple Sclerosis Therapeutics Market Research Methodology
- The methodology combines secondary research, primary interviews, and forecast modelling.
- It draws on 150+ secondary sources and benchmarks 70+ company product portfolios.
- Market sizing covers 30+ countries through a demand-side model, supported by top-down validation.
- Key inputs include MS prevalence data, treatment adoption rates, payer coverage frameworks, prescribing trends, and biologic market share dynamics.
- The model cross-checks demand with prescription volumes, pricing trends, channel mix, and product launch timelines.
- Primary validation includes 30+ interviews across manufacturers, neurologists, hospital pharmacists, payer organizations, and patient advocacy groups.
- Final estimates go through prescription-flow checks, sales-pattern review, and sensitivity testing.
Scope of Analysis
| Parameter | Details |
|---|---|
| Quantitative Units | USD 30.7 billion to USD 55.5 billion, at a CAGR of 6.1% |
| Market Definition | The Multiple Sclerosis Therapeutics Market covers disease-modifying therapies, immunomodulators, monoclonal antibodies, corticosteroids, and small molecule treatments indicated for relapsing and progressive MS subtypes across branded, generic, and biosimilar categories. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, Canada, UK, Germany, France, Italy, Japan, 30 plus countries |
| Key Companies | Biogen, Teva Pharmaceutical Industries Ltd., Pfizer Inc., Bayer AG, Sanofi, F. Hoffmann-La Roche Ltd., Johnson & Johnson Services Inc., Merck & Co. Inc., Takeda Pharmaceutical Company Limited, Horizon Therapeutics plc |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid demand-side and top-down methodology built on country-level disease prevalence, treatment adoption rates, pricing analysis, prescription validation, and primary interviews across manufacturers, neurologists, hospital pharmacists, and payer organizations |
Multiple Sclerosis Therapeutics Market Segmentation
-
Multiple Sclerosis Therapeutics Market Segmented by Product:
- Monoclonal Antibodies
- B Cell Depleting Agents
- Ocrelizumab
- Ofatumumab
- Alpha Integrin Inhibitors
- Natalizumab
- CD52 Targeting Agents
- Alemtuzumab
- B Cell Depleting Agents
- Immunomodulators
- Beta Interferons
- Interferon Beta-1a
- Interferon Beta-1b
- Glatiramer Acetate
- S1P Receptor Modulators
- Fingolimod
- Siponimod
- Ozanimod
- Fumarates
- Dimethyl Fumarate
- Diroximel Fumarate
- Beta Interferons
- Immunosuppressants
- Antineoplastic Agents
- Mitoxantrone
- Other Immunosuppressants
- Azathioprine
- Cyclophosphamide
- Antineoplastic Agents
- Corticosteroids
- Intravenous Corticosteroids
- Oral Corticosteroids
- Small Molecule Therapies
- Cladribine
- Teriflunomide
- Monoclonal Antibodies
-
Multiple Sclerosis Therapeutics Market Segmented by Application:
- Relapsing Remitting Multiple Sclerosis
- Secondary Progressive Multiple Sclerosis
- Primary Progressive Multiple Sclerosis
- Clinically Isolated Syndrome
-
Multiple Sclerosis Therapeutics Market Segmented by Route of Administration:
- Injectable
- Subcutaneous
- Intramuscular
- Oral
- Intravenous
- Injectable
-
Multiple Sclerosis Therapeutics Market Segmented by Distribution Channel:
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Specialty Pharmacy
-
Multiple Sclerosis Therapeutics Market Segmented by Drug Class:
- Monoclonal Antibodies
- Immunosuppressants
- Immunostimulants
- Immunomodulators
- S1P Receptor Modulators
-
Multiple Sclerosis Therapeutics Market Segmented by End Use:
- Hospitals
- Specialty Clinics
- Homecare Settings
-
Multiple Sclerosis Therapeutics Market Segmented by Pricing Tier:
- Branded Drugs
- Generic Drugs
- Biosimilars
- Branded Drugs
-
Multiple Sclerosis Therapeutics Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- North America
- Frequently Asked Questions -
How big is the multiple sclerosis therapeutics market in 2025?
The global multiple sclerosis therapeutics market is estimated to be valued at USD 28.95 billion in 2025.
What will be the size of the multiple sclerosis therapeutics market in 2036?
The market size for the multiple sclerosis therapeutics market is projected to reach USD 55.5 billion by 2036.
How much will the multiple sclerosis therapeutics market grow between 2026 and 2036?
The multiple sclerosis therapeutics market is expected to grow at a 6.1% CAGR between 2026 and 2036.
What are the key product types in the multiple sclerosis therapeutics market?
The key product types include monoclonal antibodies, immunomodulators, immunosuppressants, corticosteroids, and small molecule therapies.
Which application segment will contribute significant share in the multiple sclerosis therapeutics market in 2026?
In terms of application, relapsing remitting multiple sclerosis is expected to account for 63.5% share in the multiple sclerosis therapeutics market in 2026.
Which are the fastest-growing countries in the multiple sclerosis therapeutics market?
USA (6.2%) and Germany (6.1%) are among the fastest-growing countries in the multiple sclerosis therapeutics market during the forecast period.
Who are the key companies in the multiple sclerosis therapeutics market?
Key companies include Biogen, Teva Pharmaceutical Industries Ltd., Pfizer Inc., Bayer AG, Sanofi, F. Hoffmann-La Roche Ltd., Johnson & Johnson Services Inc., and Merck & Co. Inc.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Fact.MR Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Monoclonal Antibodies
- B Cell Depleting Agents
- Ocrelizumab
- Ofatumumab
- Alpha Integrin Inhibitors
- Natalizumab
- CD52 Targeting Agents
- Alemtuzumab
- Immunomodulators
- Beta Interferons
- Interferon Beta-1a
- Interferon Beta-1b
- Glatiramer Acetate
- S1P Receptor Modulators
- Immunosuppressants
- Antineoplastic Agents
- Mitoxantrone
- Other Immunosuppressants
- Azathioprine
- Cyclophosphamide
- Corticosteroids
- Intravenous Corticosteroids
- Oral Corticosteroids
- Small Molecule Therapies
- Cladribine
- Teriflunomide
- Monoclonal Antibodies
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Relapsing Remitting Multiple Sclerosis
- Secondary Progressive Multiple Sclerosis
- Primary Progressive Multiple Sclerosis
- Clinically Isolated Syndrome
- Relapsing Remitting Multiple Sclerosis
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Route of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Route of Administration, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Route of Administration, 2026 to 2036
- Injectable
- Subcutaneous
- Intramuscular
- Oral
- Intravenous
- Injectable
- Y to o to Y Growth Trend Analysis By Route of Administration, 2021 to 2025
- Absolute $ Opportunity Analysis By Route of Administration, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Specialty Pharmacy
- Hospital Pharmacy
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Class, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Class, 2026 to 2036
- Monoclonal Antibodies
- Immunosuppressants
- Immunostimulants
- Immunomodulators
- S1P Receptor Modulators
- Monoclonal Antibodies
- Y to o to Y Growth Trend Analysis By Drug Class, 2021 to 2025
- Absolute $ Opportunity Analysis By Drug Class, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Hospitals
- Specialty Clinics
- Homecare Settings
- Hospitals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Pricing Tier
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Pricing Tier, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Pricing Tier, 2026 to 2036
- Branded Drugs
- Generic Drugs
- Biosimilars
- Branded Drugs
- Y to o to Y Growth Trend Analysis By Pricing Tier, 2021 to 2025
- Absolute $ Opportunity Analysis By Pricing Tier, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- By Route of Administration
- By Distribution Channel
- By Drug Class
- By End Use
- By Pricing Tier
- Competition Analysis
- Competition Deep Dive
- Biogen
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Teva Pharmaceutical Industries Ltd.
- Pfizer Inc.
- Bayer AG
- Sanofi
- F. Hoffmann-La Roche Ltd.
- Johnson & Johnson Services, Inc.
- Merck & Co., Inc.
- Takeda Pharmaceutical Company Limited
- Horizon Therapeutics plc
- Biogen
- Competition Deep Dive
- Assumptions & Acronyms Used
List Of Table
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 8: Global Market Value (USD Million) Forecast by Pricing Tier, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 15: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: North America Market Value (USD Million) Forecast by Pricing Tier, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 22: Latin America Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 23: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Latin America Market Value (USD Million) Forecast by Pricing Tier, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 29: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 30: Western Europe Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 31: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 32: Western Europe Market Value (USD Million) Forecast by Pricing Tier, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 36: Eastern Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 37: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 38: Eastern Europe Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 39: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 40: Eastern Europe Market Value (USD Million) Forecast by Pricing Tier, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 43: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 44: East Asia Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 45: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 46: East Asia Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 47: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: East Asia Market Value (USD Million) Forecast by Pricing Tier, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 50: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 51: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 52: South Asia and Pacific Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 53: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 54: South Asia and Pacific Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 55: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 56: South Asia and Pacific Market Value (USD Million) Forecast by Pricing Tier, 2021 to 2036
- Table 57: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 58: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 59: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 60: Middle East & Africa Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 61: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 62: Middle East & Africa Market Value (USD Million) Forecast by Drug Class, 2021 to 2036
- Table 63: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 64: Middle East & Africa Market Value (USD Million) Forecast by Pricing Tier, 2021 to 2036
List Of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Route of Administration
- Figure 12: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Distribution Channel
- Figure 15: Global Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Drug Class, 2026 to 2036
- Figure 17: Global Market Attractiveness Analysis by Drug Class
- Figure 18: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 20: Global Market Attractiveness Analysis by End Use
- Figure 21: Global Market Value Share and BPS Analysis by Pricing Tier, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Pricing Tier, 2026 to 2036
- Figure 23: Global Market Attractiveness Analysis by Pricing Tier
- Figure 24: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 25: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 26: Global Market Attractiveness Analysis by Region
- Figure 27: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 28: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 29: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 30: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 31: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 32: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 33: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 34: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 35: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 37: North America Market Attractiveness Analysis by Product
- Figure 38: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 40: North America Market Attractiveness Analysis by Application
- Figure 41: North America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 43: North America Market Attractiveness Analysis by Route of Administration
- Figure 44: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026 to 2036
- Figure 46: North America Market Attractiveness Analysis by Distribution Channel
- Figure 47: North America Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Drug Class, 2026 to 2036
- Figure 49: North America Market Attractiveness Analysis by Drug Class
- Figure 50: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 51: North America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 52: North America Market Attractiveness Analysis by End Use
- Figure 53: North America Market Value Share and BPS Analysis by Pricing Tier, 2026 and 2036
- Figure 54: North America Market Y-o-Y Growth Comparison by Pricing Tier, 2026 to 2036
- Figure 55: North America Market Attractiveness Analysis by Pricing Tier
- Figure 56: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 57: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 59: Latin America Market Attractiveness Analysis by Product
- Figure 60: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 62: Latin America Market Attractiveness Analysis by Application
- Figure 63: Latin America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 65: Latin America Market Attractiveness Analysis by Route of Administration
- Figure 66: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026 to 2036
- Figure 68: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 69: Latin America Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 70: Latin America Market Y-o-Y Growth Comparison by Drug Class, 2026 to 2036
- Figure 71: Latin America Market Attractiveness Analysis by Drug Class
- Figure 72: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 73: Latin America Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 74: Latin America Market Attractiveness Analysis by End Use
- Figure 75: Latin America Market Value Share and BPS Analysis by Pricing Tier, 2026 and 2036
- Figure 76: Latin America Market Y-o-Y Growth Comparison by Pricing Tier, 2026 to 2036
- Figure 77: Latin America Market Attractiveness Analysis by Pricing Tier
- Figure 78: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 79: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 81: Western Europe Market Attractiveness Analysis by Product
- Figure 82: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 84: Western Europe Market Attractiveness Analysis by Application
- Figure 85: Western Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 87: Western Europe Market Attractiveness Analysis by Route of Administration
- Figure 88: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 89: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026 to 2036
- Figure 90: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 91: Western Europe Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 92: Western Europe Market Y-o-Y Growth Comparison by Drug Class, 2026 to 2036
- Figure 93: Western Europe Market Attractiveness Analysis by Drug Class
- Figure 94: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 95: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 96: Western Europe Market Attractiveness Analysis by End Use
- Figure 97: Western Europe Market Value Share and BPS Analysis by Pricing Tier, 2026 and 2036
- Figure 98: Western Europe Market Y-o-Y Growth Comparison by Pricing Tier, 2026 to 2036
- Figure 99: Western Europe Market Attractiveness Analysis by Pricing Tier
- Figure 100: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Product
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Application
- Figure 107: Eastern Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 108: Eastern Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 109: Eastern Europe Market Attractiveness Analysis by Route of Administration
- Figure 110: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 111: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026 to 2036
- Figure 112: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 113: Eastern Europe Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 114: Eastern Europe Market Y-o-Y Growth Comparison by Drug Class, 2026 to 2036
- Figure 115: Eastern Europe Market Attractiveness Analysis by Drug Class
- Figure 116: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 117: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 118: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 119: Eastern Europe Market Value Share and BPS Analysis by Pricing Tier, 2026 and 2036
- Figure 120: Eastern Europe Market Y-o-Y Growth Comparison by Pricing Tier, 2026 to 2036
- Figure 121: Eastern Europe Market Attractiveness Analysis by Pricing Tier
- Figure 122: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 123: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 125: East Asia Market Attractiveness Analysis by Product
- Figure 126: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 127: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 128: East Asia Market Attractiveness Analysis by Application
- Figure 129: East Asia Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 130: East Asia Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 131: East Asia Market Attractiveness Analysis by Route of Administration
- Figure 132: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 133: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026 to 2036
- Figure 134: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 135: East Asia Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 136: East Asia Market Y-o-Y Growth Comparison by Drug Class, 2026 to 2036
- Figure 137: East Asia Market Attractiveness Analysis by Drug Class
- Figure 138: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 139: East Asia Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 140: East Asia Market Attractiveness Analysis by End Use
- Figure 141: East Asia Market Value Share and BPS Analysis by Pricing Tier, 2026 and 2036
- Figure 142: East Asia Market Y-o-Y Growth Comparison by Pricing Tier, 2026 to 2036
- Figure 143: East Asia Market Attractiveness Analysis by Pricing Tier
- Figure 144: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 145: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 146: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 147: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 148: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 149: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 150: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 151: South Asia and Pacific Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 152: South Asia and Pacific Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 153: South Asia and Pacific Market Attractiveness Analysis by Route of Administration
- Figure 154: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 155: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026 to 2036
- Figure 156: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 157: South Asia and Pacific Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 158: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug Class, 2026 to 2036
- Figure 159: South Asia and Pacific Market Attractiveness Analysis by Drug Class
- Figure 160: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 161: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 162: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 163: South Asia and Pacific Market Value Share and BPS Analysis by Pricing Tier, 2026 and 2036
- Figure 164: South Asia and Pacific Market Y-o-Y Growth Comparison by Pricing Tier, 2026 to 2036
- Figure 165: South Asia and Pacific Market Attractiveness Analysis by Pricing Tier
- Figure 166: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 167: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 168: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026 to 2036
- Figure 169: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 170: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 171: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 172: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 173: Middle East & Africa Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 174: Middle East & Africa Market Y-o-Y Growth Comparison by Route of Administration, 2026 to 2036
- Figure 175: Middle East & Africa Market Attractiveness Analysis by Route of Administration
- Figure 176: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 177: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026 to 2036
- Figure 178: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 179: Middle East & Africa Market Value Share and BPS Analysis by Drug Class, 2026 and 2036
- Figure 180: Middle East & Africa Market Y-o-Y Growth Comparison by Drug Class, 2026 to 2036
- Figure 181: Middle East & Africa Market Attractiveness Analysis by Drug Class
- Figure 182: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 183: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026 to 2036
- Figure 184: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 185: Middle East & Africa Market Value Share and BPS Analysis by Pricing Tier, 2026 and 2036
- Figure 186: Middle East & Africa Market Y-o-Y Growth Comparison by Pricing Tier, 2026 to 2036
- Figure 187: Middle East & Africa Market Attractiveness Analysis by Pricing Tier
- Figure 188: Global Market - Tier Structure Analysis
- Figure 189: Global Market - Company Share Analysis